Abstract
Boswellia resin has been used as a major anti-inflammatory agent and for the healing of wounds for centuries. Incensole acetate (IA), isolated from this resin, was shown to inhibit the activation of nuclear factor-κB, a key transcription factor in the inflammatory response. We now show that IA inhibits the production of inflammatory mediators in an in vitro model system of C6 glioma and human peripheral monocytes. Given the involvement of postinjury inflammation in the pathophysiology and outcome of traumatic brain injury, we examined the effect of IA on the inflammatory process and on the recovery of neurobehavioral and cognitive functions in a mouse model of closed head injury (CHI). In the brains of post-CHI mice, IA reduced glial activation, inhibited the expression of interleukin-1β, and tumor necrosis factor-α mRNAs, and induced cell death in macrophages at the area of trauma. A mild hypothermic effect was also noted. Subsequently, IA inhibited hippocampal neurodegeneration and exerted a beneficial effect on functional outcome after CHI, indicated by reduced neurological severity scores and improved cognitive ability in an object recognition test. This study attributes the anti-inflammatory activity of Boswellia resin to IA and related cembranoid diterpenes and suggests that they may serve as novel neuroprotective agents.
Introduction
Traumatic brain injury (TBI) is often associated with permanent cognitive disorders, learning disabilities, and various behavioral and emotional impairments. Most of the clinical trials conducted so far have failed to show any significant improvement in outcome measures (Marmarou et al, 2005; Narayan et al, 2002). Secondary brain damage, triggered by the initial impact, can increase mortality and worsen disability, but, unlike the primary lesion, may potentially be attenuated by appropriate treatment (Marmarou et al, 2005; Cernak, 2005). As it is now evident that secondary biochemical perturbations play an important role in the pathophysiology of TBI and may provide specific therapeutic targets for the treatment of the head injured, efforts are directed toward interference with the course of these events (Potts et al, 2006).
Growing evidence now indicates that inflammation is critical to both secondary injury and repair mechanisms in the central nervous system (Nortje and Menon, 2004; Potts et al, 2006). Studies on the production of cytokines after closed head injury (CHI) have shown that they may have a dual role such that high levels of proinflammatory cytokines within hours after injury are deleterious, yet their presence, at physiologic levels, is required for later recovery processes (Shohami et al, 1996, 1999). Postinjury expression of proinflammatory cytokines, namely tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β), within the initial hours after injury has been implicated in the development of secondary damage after CHI (Shohami et al, 1996; Bermpohl et al, 2007; Lu et al, 2005; Allan et al, 2005). The transcription of both IL-1β and TNF-α is regulated by nuclear factor-κB (NF-κB), an inducible transcription factor, which plays a central role in the mammalian innate immune response and chronic inflammation (Ben-Neriah and Schmitz, 2004). Closed head injury has been shown to induce prolonged activation of NF-κB (Beni et al, 2004; Panikashvili et al, 2005).
The use of plant extracts in the healing of inflammatory diseases is millennia old and continues to date. Boswellia species (Burseraceae) are native to Eastern Africa, where their resin, commonly known as ‘Frankincense’ or ‘Olibanum,’ is used and exported as incense and used for various medical purposes (Watt, 1962). The resin of Boswellia spp. has been used for the treatment of chronic inflammation and wounds in Europe, Asia, and Africa for many centuries (Watt, 1962; Poeckel et al, 2006). Boswellia extracts are currently used for the treatment of inflammatory conditions and marketed as food supplements in the United States and Europe. A considerable amount of work has been done on the anti-inflammatory properties of Boswellia. However, previous reports attribute the anti-inflammatory properties of Boswellia resin solely to boswellic acid and its derivatives (for example see Poeckel et al, 2006). In Oman, one of the world's largest frankincense exporters, it is also used to improve memory (Marshall, 2003).
We have previously isolated incensole acetate (IA; Figure 1) and its nonacetylated form, incensole, both cembrene-type diterpenes, from the resin of Boswellia carterii. The isolation protocol was based on the ability of these compounds to inhibit NF-κB activation (Moussaieff et al, 2007). Here we revisited the anti-inflammatory properties of Boswellia resin and examined the anti-inflammatory activity of IA in light of the inhibitory effect it exhibits on NF-κB activation. The centuries old medical use of Boswellia resin, taken together with our observations, suggesting an anti-inflammatory effect of IA, led us to examine the effects of IA on neurobehavioral and cognitive functions, as well as on the production of proinflammatory cytokines after CHI.
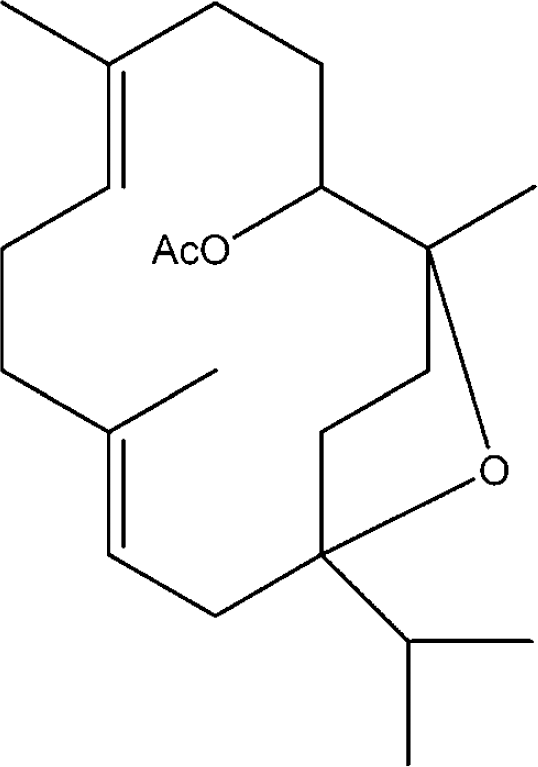
The chemical structure of incensole acetate (IA).
Materials and methods
Incensole Acetate Isolation
Incensole acetate isolation was performed as previously described (Moussaieff et al, 2007).
Cell Cultures
Rat C6 glioma cells and plasmid: Rat C6 glioma cell line was obtained from the American Type Culture Collection (ATCC) and was cultured in Dulbecco's modified Eagle's medium medium (Invitrogen, Karlsruhe, Germany) containing 10% heat-inactivated fetal bovine serum, 2 mmol/L
Isolation of human monocytes: Human peripheral monocytes from healthy human donors were prepared using a standardized protocol (Ficoll gradient preparation; Amersham Biosciences, Freiburg, Germany) with a completely endotoxin-free cultivation as previously described (Caballero et al, 2007). By using 50 mL tubes, 25 mL Ficoll was loaded with 25 mL blood of Buffy coats from healthy blood donors. The gradient was established by centrifugation at 1,800 r.p.m., 20°C for 40 mins, using slow acceleration and brakes. Peripheral blood mononuclear cells in the interphase were carefully removed and resuspended in 50 mL prewarmed phosphate buffered saline (PBS; Invitrogen) followed by centrifugation for 10 mins at 1,600 r.p.m. at 20°C. The supernatant was discarded, and the pellet was washed in 50 mL PBS and centrifuged as described above. The pellet was then resuspended in 50 mL RPMI-1640 low endotoxin medium (Invitrogen) supplemented with 10% human serum (PAA, Coelbe, Germany).
Animals
Male Sabra mice (Hebrew University strain), 8- to 10-week old, were used in this study. The study was carried out according to the regulations of the Animal Care Committee of the Hebrew University. Mice were kept under a 12-h light/dark reversed light cycle, and food and water were provided ad libitum.
Inhibitory κBα Degradation Assay
Cells were preincubated for 1 h with IA (100 μmol/L) or ethanol as vehicle control and then stimulated for 20 mins with lipopolysaccharide (LPS) (100 ng/mL). Cell extracts were tested by Western blotting for the phosphorylation and stability of inhibitory κBα (IκBα).
Western Blot Analysis
After separation by SDS-PAGE (sodium dodecyl sulfate-polyacrylamide gel electrophoresis), proteins were blotted to a polyvinylidene difluoride membrane (Millipore, Eschborn, Germany). The membrane was blocked in 5% (w/v) milk powder and then incubated in tris-buffered saline tween-20 (TBST) containing the primary antibody and 2% (w/v) milk powder. Inhibitory κBα antibody was purchased from Santa Cruz Inc. (Santa Cruz, CA, USA). After binding of a secondary antibody coupled to horseradish peroxidase, proteins were visualized by enhanced chemiluminescence according to the instructions of the manufacturer (Amersham Lifescience, Freiburg, Germany).
Luciferase assays: Twenty-four hours after transfection, the C6 cells were treated with IA in various concentrations for 30 mins, followed by stimulation with IL-1β (10 ng/mL) or LPS (1 μg/mL) as indicated for 6 h. The cells were washed twice in PBS and lysed in 25 mmol/L Trisphosphate, pH 7.8, 8 mmol/L MgCl2, 1 mmol/L DTT, 1% Triton X-100, and 7% glycerol for 15 mins at room temperature in a horizontal shaker. The lysates were then spun down, and the supernatant was used to measure luciferase activity using an Autolumat LB 953 luminometer (EG&G Berthold, Bad Wildbad, Germany) according to the instructions of the luciferase assay kit (Promega, Madison, WI, USA). After 12 h, the cells were washed twice with PBS and luciferase activity was measured. Results are presented as fold induction over untreated control.
Tumor Necrosis Factor-α, Interleukin-1β, Interleukin-6, and Prostaglandin E2 Levels in Monocytes
After counting the amount of human peripheral monocytes in a particle counter (Euro Diagnostics, Krefeld, Germany), cells were seeded in 24-well plates for ELISA (enzyme-linked immunosorbent assay) (1 to 2 times 106 cells per well) and incubated at 37°C/5% CO2. The medium and the nonadherent cells (lymphocytes) were removed, and fresh RPMI-1640 medium containing 1% human serum was added. Cells were preincubated with IA in various concentrations as indicated for 30 mins, followed by stimulation with LPS (10 ng/mL) for an additional 24 h. Culture supernatants were harvested and centrifuged 10 mins at 10,000g for the analysis of IL-1β, IL-6, TNF-α, and prostaglandin E2 (PGE2). Levels of the cytokines or PGE2 in the supernatant were measured by ELISA (IL-6, IL-1, TNF-α: Pelikine, distributed by ImmunoTools, Friesoythe, Germany) or EIA (PGE2: AssayDesign, distributed by Biotrend, Köln, Germany), according to the manufacturer's instructions, using a monoclonal antibody. The standards were used in the range of 0.6 to 450 pg/mL for IL-6 (sensitivity of 0.2 to 1 pg/mL), 1.4 to 1,000 pg/mL for TNF-α (sensitivity of 1 to 6 pg/mL), 0.4 to 300 pg/mL (sensitivity of 0.2 to 2 pg/mL) for IL-1β, and 39 to 2,500 pg/mL for PGE2 (sensitivity of 36.2 pg/mL). All experiments were performed with three Buffy coats from different blood donors in triplicates.
Closed Head Injury Model and Neurobehavioral Evaluation
Closed head injury was induced using a weight drop device, as previously described (Yatsiv et al, 2002). Briefly, after a sagittal scalp incision was performed under isoflurane anesthesia, a tipped Teflon cone was placed 1 mm lateral to the midline and 1 mm caudal to the left coronal suture, and a metal rod (95 g) was allowed a free fall on the cone from a precalibrated height. At 1 h after CHI, the functional status of the mice was evaluated according to a set of 10 neurobehavioral tasks, namely the neurological severity score (NSS). This score is based on the ability of the mice to perform 10 different tasks (Beni-Adani et al, 2001; see Table 1) that evaluate their motor ability, balance, and alertness. One point is given for failing to perform a task. The severity of injury is indicated by the initial NSS, which is evaluated 1 h after CHI and is also a reliable predictor of the later outcome. A score of 10 reflects maximal neurologic impairment, and a decrease of NSS during the recovery period indicates partial recovery of function. Neurological severity score was further evaluated at various time intervals up to 21 days, and recovery in IA-treated mice was compared with vehicle-treated mice.
NSS for head injured mice
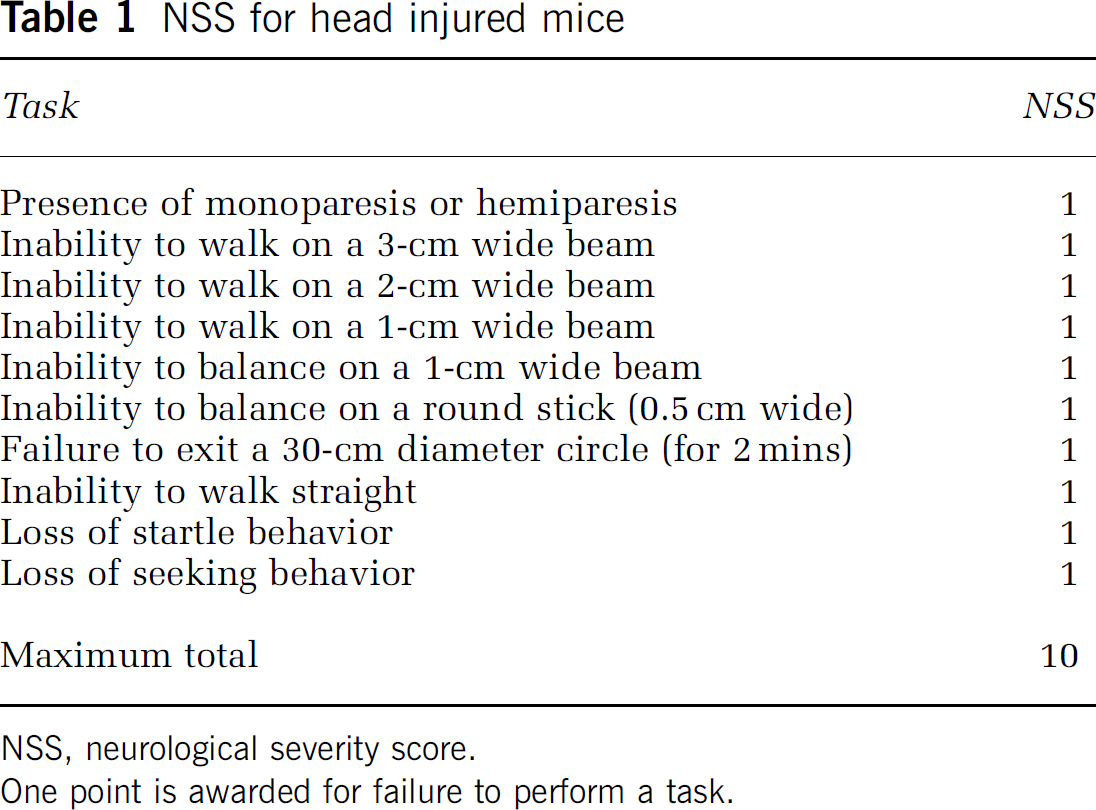
NSS, neurological severity score.
One point is awarded for failure to perform a task.
Evaluation of Cognitive Performance in the Object Recognition Test
The object-recognition test (ORT) was performed on groups of mice, treated with either IA or vehicle at various intervals during 21 days after CHI, as previously described (Ennaceur and Delacour, 1988; Biegon et al, 2004). One day before testing, mice were placed in the testing cage (a glass aquarium-like transparent box of 60 times 40 cm) for 1 h habituation. On the following day, they were put back into the same cage with two identical objects. The cumulative time spent by the mouse at each of the objects was recorded during a 5-min period by an observer blinded to the treatment received. Four hours later, the mice were reintroduced into the cage, where one of the two objects was replaced by a new one of similar size and complexion. The time (of 5 mins total) spent at each of the objects was recorded. The basic measure is the percent of the total time spent by mice in exploring an object during the testing period, whereby normal healthy rodents will spend relatively more time exploring a new object than a familiar, that is ‘memorized’ object.
Injection of Incensole Acetate or Vehicle
Immediately after NSS 1 h assessment, mice were assigned to the treatment groups such that both groups presented similar severity of injury (namely, similar NSS 1 h). Animals were then administered a single intraperitoneal injection of IA (50 mg/kg) in vehicle (isopropanol/Emulphor—a commercial emulsifier/saline = 1:1:18) or vehicle alone. The dose of IA was chosen according to our previous study on the effects of this compound in vivo (Moussaieff et al, 2007).
Tissue Preparation for Histology and Immunohistochemistry
Cerebral immunohistopathological parameters were evaluated at 24 h after CHI. Animals (n = 5 per group) were perfused via the ascending aorta with PBS, followed by cold 4% paraformaldehyde in PBS. Brain tissue was postfixed in the same fixative for 24 h at 4°C and embedded in paraffin thereafter. Coronal brain sections (6 μm thick) were serially taken at 200 μm intervals, throughout the neuraxis, between +3.92 and −2.30 mm from bregma. The limits of the area of trauma along the longitudinal axis as well as the penumbra area were first determined with Luxol Fast Blue staining. Sections for evaluation were randomly selected at various levels by investigators blinded to the treatment groups.
Histology—Immunohistochemistry
Sections were stained with Fluoro Jade B, a fluorescent chromofluor that selectively labels degenerating neurons (Anderson et al, 2005; Schmued and Hopkins, 2000). For all immunohistochemical stains, paraffin sections were deparaffinized and hydrated in alcohol solutions and xylene, and then rinsed in tris-buffered saline (TBS). Citrate buffer (pH 6.0) was used for antigen retrieval and the endogenous peroxidase was blocked with H2O2 (0.3% in PBS). Sections were then incubated in blocking buffer for 1 h. Astrocyte and microglia activation were initially identified. A series of preselected sections were then treated with primary antibody against glial fibrillary acidic protein (GFAP) (DakoCytomation, Glostrup, Denmark) followed by goat anti-rabbit IgG secondary antibody (Vector, Burlingame, CA, USA). Immunoreactions were visualized with the avidin—biotin complex (Vectastain; Vector), and the peroxidase reaction was visualized with DAB (diaminobenzidine) (Vector) as chromogen.
In adjacent sections, endogenous biotin blocking was performed by using the Avidin/Biotin Blocking Kit (Vector), and sections were incubated with Lectin (Lycopersicon esculentum tomato; Sigma, Missouri, St Louis, MO, USA), which stains microglia and macrophages. The immunoreactions were visualized with LSAB-2 System-HRP (DakoCytomation) with DAB as chromogen. Sections were finally counterstained with hematoxylin.
Caspase-3 immunohistochemistry was performed in an adjacent series of sections as an indicator of cellular death. Sections were incubated for 1 h with anti-caspase-3 rabbit polyclonal antibody (R&D Systems, Abingdon, UK), and visualized with Envision+System-HRP (DakoCytomation).
To identify the caspase-3-positive cells, double immunostaining for caspase-3 and microglia was performed in another series of sections. Briefly, sections were incubated overnight at 4°C with anti-caspase-3 rabbit polyclonal antibody and then with FITC-conjugated goat anti-rabbit IgG (Jackson ImmunoResearch, West Grove, PA, USA). The sections were then incubated with a rhodamine-conjugated rat anti-mouse antibody raised against the macrophage marker F4/80 (Serotec, Oxford, UK). For the detection of nonspecific binding, either the first or the second primary antibody was omitted in a number of sections.
Sections were examined under either a light or fluorescent microscope (Zeiss Axioplan 2) according to whether the sections were stained with DAB or fluorescence. Images were taken with a NIKON DS-5Mc-L1 Digital Camera System for Microscopy.
Assessment of Gliosis-Associated Immunoreactivity
Glial fibrillary acidic protein and lectin expression were evaluated at the area of the trauma and the hippocampus of the left or injured hemisphere of each animal. Two to three microscopic fields (0.37 mm2 each, as defined by an ocular morphometric grid adjusted onto the microscope lens) were evaluated per section (depending on the size of the hippocampus, which was captured within the image). Ultimately, a total of 10 randomly selected fields were evaluated per animal in each area. Section and field selection were conducted by blinded investigators. Fields were assigned scores representing assessments of staining intensity as follows: 0, no labeling observed; 1, few cells that are only weakly labeled; 2, moderate number of cells that are clearly labeled; 3, large number of intensely labeled cells present (as described by Simic et al, 2000). Evaluations were conducted by two independent raters who were blinded to the given treatments, and the procedure was repeated by a third rater in cases where significant discrepancies were obvious between the two observers. The raw data obtained by all raters were used to calculate the percent distribution of field scores per group.
Evaluation of Neurodegeneration
Neurodegeneration analysis was performed on hippocampal areas. For each animal, one slide, containing three sections, was randomly selected by a blinded investigator out of the nine groups of slides taken. This slide, together with its previous and following adjacent slides, was stained with Fluoro-Jade B. Fluoro-Jade-B-positive neurons were quantified by capturing images from two to three visual fields (depending on the size of each area) in each section onto an Intel Pentium computer using a digital camera (NIKON DS-5Mc-L1) mounted on the microscope. The images of CA1, CA3, CA4, and hilus areas were captured along the whole hippocampal area with a final magnification of × 480. In each image, a 305 times 122 μm rectangle with 40 square subdivisions (3.05 times 3.05 μm) was positioned over the captured hippocampal area. For all captured images, a square subdivision was counted if it contained at least one labeled element, defined as a labeled cell body and/or its processes. The scores of the studied areas for this stain represent assessments of fluorescence intensity as follows: 0, no labeling observed in any square subdivision; 1, 1 to 10 square subdivisions positive; 2, 11 to 20 square subdivisions positive; 3, >20 square subdivisions positive. Rating was carried out by observers as described for gliosis-associated immunoreactivity expression. The section that exhibited the most Fluoro-Jade-B-positive structures (highest mean of scores given by the raters) was subsequently selected from the stained serial sections (three groups of three sections adhered onto three separate slides) and used for statistical analysis (so as to reflect major pathology in the examined area and to avoid duplicate cell counting, as described by Sato et al, 2001). The raw data obtained by all raters were used to calculate the percent distribution of field scores per group.
Determination of Tumor Necrosis Factor-α and Interleukin-1β mRNA Expression
The expression of IL-1β and TNF-α in the brains of post-CHI mice was examined using quantitative real-time PCR (real-time PCR). On the basis of our earlier findings on cytokine expression in the same model (Panikashvili et al, 2005), vehicle- and IA-treated mice (n = 5) were killed 3 h after injury. Frontal cortical segments were taken and frozen at −70°C until use. Total RNA was isolated using TRI reagent (Molecular Research Center Inc., Cincinnati, OH, USA). All RNA samples were confirmed free of DNA contamination by 260/280 nm optical density measurement. Total RNA (1 μg) was reverse transcribed using a First Strand cDNA Synthesis Kit (Fermentas Inc., Hanover, MD, USA). Polymerase chain reaction was performed using Taqman® Gene Expression Assays-on-Demand (Applied Biosystems, Foster City, CA). The amplification reaction was carried out in an ABI 7700 Sequence Detection System (Applied Biosystems). The reaction was performed in a 20 μL reaction volume, which contained 90 ng of cDNA, 10 μL of Taqman Universal PCR Mix (Applied Biosystems), and 1 μL of the assay solution containing the specific primers and 6-FAM (6-carboxy-fluorescein)-labeled probe. The thermal profile for PCR was 95°C for 10 mins, followed by 45 cycles of 95°C for 15 secs and 60°C for 60 secs. Relative quantity values were analyzed using ABI 7700 Sequence Detection System Software (Applied Biosystems) according to the ACt method, which reflects the difference in threshold for each target gene relative to that of β-actin.
Effect of Incensole Acetate on Body Temperature after Closed Head Injury
Mice were subjected to CHI (n = 8 mice per group). One hour later, following NSS assessment, they were assigned to vehicle and IA treatment. Body (rectal) temperature was measured using a thermometer (Digi-sense Thermistor 400 Series; Eutech Instruments, Singapore, Singapore) 30 mins before (as baseline body temperatures) and 30, 60, 120, 180, and 240 mins after IA or vehicle injection. As a hypothermic effect was observed in the initial experiment, control groups receiving identical treatment with IA or vehicle, but maintained at a constant preinjection body temperature, were included in subsequent experiments so as to examine whether the lack of hypothermia disrupts the functional neuroprotective effect of IA.
Evaluation of Cerebral Edema
Cerebral edema was evaluated at 24 h after CHI by determining the tissue water content in frontal cortical brain segments, as previously described (Chen et al, 1996). Water content was calculated as %H2O = [(wet weight—dry weight)/wet weight] × 100. n = 10 mice per group.
Statistical Analysis
Data are presented as mean ± s.e.m., with the exception of the immunohistopathological data (see herein). The histologic and immunohistochemical data are presented as the percentages of distribution of field scores per group of animals. A commercial statistics package was used for determining statistical significance (GraphPad Prism version 4 or SPSS 11.5). Nonparametric NSS values were compared between the two groups (IA versus vehicle) at each time point. These data were analyzed for differences between groups at individual times (and not over time within the same group); hence, Mann—Whitney tests were used for comparisons. Object-recognition test data were analyzed for the difference between the performance in the test and in the baseline settings for each group. As these measurements do not constitute a repetition of the same experimental setting, comparisons were conducted using Student's t-tests. Because temperature data are generated from a continuous dependent variable and were also compared between the two different groups and not over time, they were analyzed by Student's t-tests. mRNA quantity results were also analyzed by Student's t-tests. Histologic and immunohistochemical data were categorical and Pearson's χ2 tests were thus used for comparisons. Significance level for all comparisons was set at P ≤ 0.05.
Results
To corroborate the anti-inflammatory activity of IA, we first examined its effect in vitro on inflammatory effectors using C6 glioma cells and human peripheral monocytes.
Incensole Acetate Inhibits Nuclear Factor-κB Activation and Gene Expression in Human Peripheral Monocytes and C6 Glioma Cells
LPS stimulation induced phosphorylation and degradation of IκBα in human peripheral monocytes. In cells treated with IA, IκBα phosphorylation and degradation was attenuated, suggesting that IA inhibits NF-κB activation in these cells (Supplementary Figure 1A). C6 glioma cells were stimulated by IL-1β or LPS, and NF-κB-dependent transactivation was measured using a Luciferase reporter assay. A three- to fourfold increase in NF-κB activation was noted in the LPS or IL-1β-treated cells. When IA was present in the incubation medium, a significant and dose-dependent inhibition of this response was found (Supplementary Figure 1B).
Incensole Acetate Impairs the Inflammatory Response in Human Peripheral Monocytes
To examine whether the above NF-κB inhibitory effect of IA confers an anti-inflammatory activity, human peripheral monocytes were exposed to LPS in the presence or absence of IA and the levels of TNF-α, IL-1β, IL-6, and PGE2 were determined. Incensole acetate attenuated the expression of all the inflammatory mediators studied in a dose-dependent manner: it inhibited TNF-α (F = 141, d.f. = 6, P ≤ 0.0001); IL-1β (F = 64, d.f. = 6, P ≤ 0.0001); IL-6 (F = 228, d.f. = 6, P ≤ 0.0001); and PGE2 (F = 27.6, d.f. = 6, P ≤ 0.0001), as depicted in Supplementary Figures 2A to 2D, respectively.
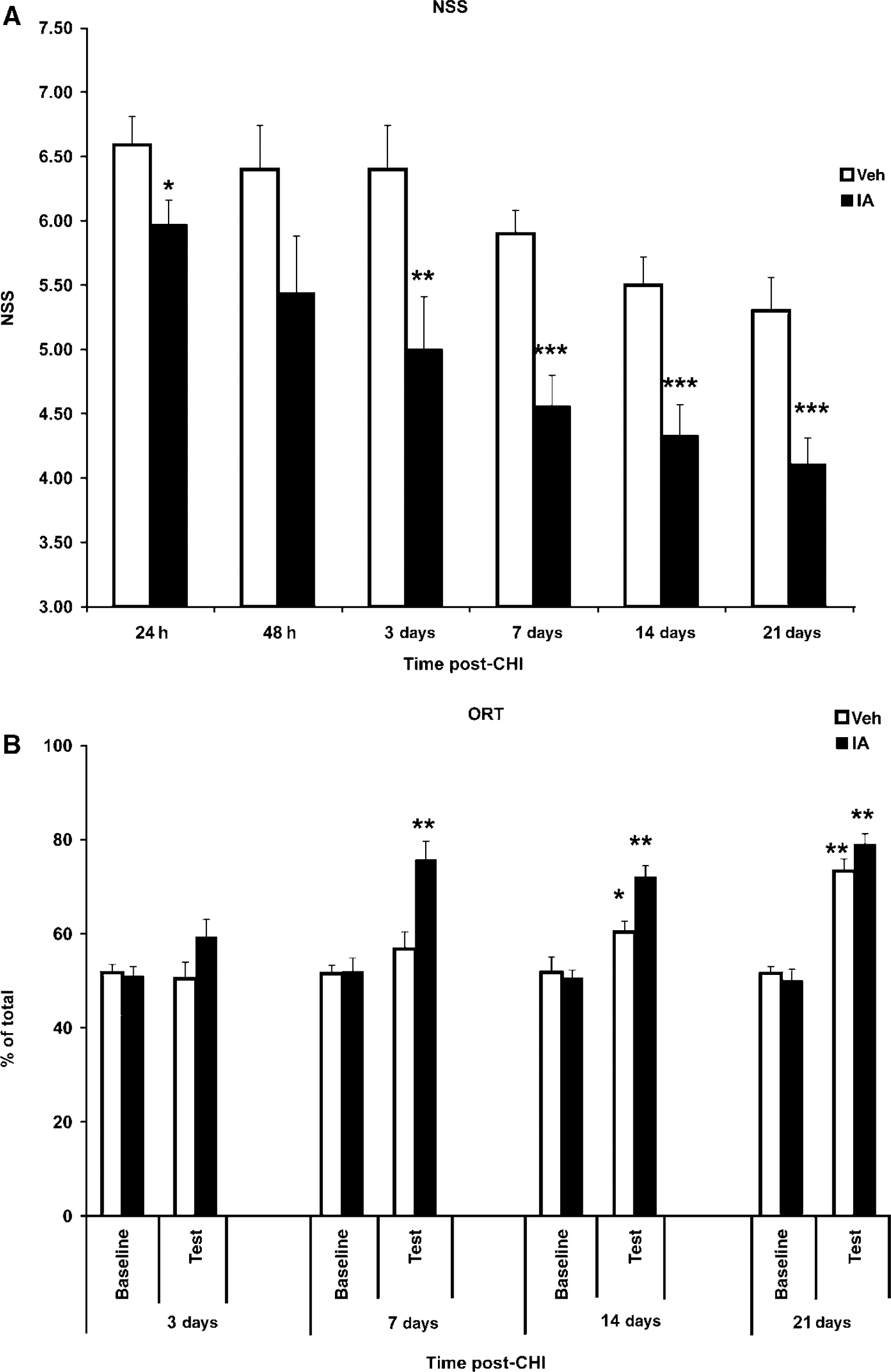
Incensole acetate (IA; 50 mg/kg at a single dose, given 1 h after trauma) exerts a beneficial effect of IA on neurobehavioral recovery and cognitive function after CHI. (
After establishing the anti-inflammatory effects of IA in vitro, we proceeded to investigate its effect on the functional outcome after CHI. Mice were hence treated with IA 1 h after induction of CHI; neurobehavioral and cognitive functions were examined during a 3-week period after injury.
Incensole Acetate Improves Post-Closed Head Injury Neurobehavioral Outcome
Functional recovery of injured mice treated with IA or vehicle was evaluated using the NSS scoring. Neurological severity score values at 1 h were similar in both groups (7.03 ± 0.19 and 7.03 ± 0.19), indicating moderate injury, with no difference in the initial severity of injury. Figure 2A depicts the spontaneous (vehicle-treated, open bars) and IA-induced (filled bars) recovery of motor ability during 21 days after injury. Comparison of NSS values of the two groups at each individual time point indicated significantly lower NSS in the IA group as early as 24 h after injury. Neurological severity score values continued to be significantly lower in IA-treated mice versus vehicle-treated mice at all subsequent time points, up to 3 weeks after injury (NSS (21 days) = 4.11 ± 0.20 versus 5.30 ± 0.26 in the IA and vehicle groups, respectively, P ≤ 0.001; Mann—Whitney tests n = 9 to 10 per group) (Figure 2A).
Incensole Acetate Improves Post-Closed Head Injury Cognitive Function
Although healthy rodents normally spend ~50% of the exploration time at each of the identical objects (baseline), after introducing a ‘novel’ object (test), they will spend relatively more time (70% to 80%) exploring the new object than a familiar, that is, a ‘memorized’ object.
Incensole acetate had a robust effect on postinjury ORT performance (‘test’; n = 9 to 11 per group; separate group than that used for NSS; Figure 2B), whereas naive mice were unaffected by the treatment (data not shown). In the baseline measurements, mice from both IA and vehicle groups spent ~50% of total exploration time at the two identical objects at all times post-CHI (Figure 2B). On day 7, after a familiar object was replaced by a novel one, IA-treated mice spent most of their exploration time at the new object, in contrast to the vehicle-treated animals, which could not distinguish between the two. This effect was noticed at day 7 and sustained up to 21 days (P < 0.001). At 21 days, exploration time of vehicle-treated mice reached a level similar to that of the IA-treated mice, as vehicle-treated mice regained their memory function, most likely due to spontaneous recovery.
Incensole Acetate Attenuates Inflammation and Neurodegeneration in the Hippocampus
To study the mechanism by which IA exerts its beneficial effect on functional recovery after CHI, we stained cortical (site of injury) and hippocampal slices, taken from IA- or vehicle-treated mice at 24 h after injury, with specific markers for inflammation and neurodegeneration. Treatment with IA attenuated the expression of GFAP within the hippocampus, indicating an inhibition of astroglial activation (Figures 3B versus 3A; P ≤ 0.0001; Pearson's χ2 test; Figure 3E). Activated ramified microglia were also significantly less evident in the brains of IA-treated mice (Figures 3D versus 3C; P = 0.023; Pearson's χ2 test; Figure 3F). Taken together, these data suggest that IA increases the percentage of the optical fields with moderate (+ +) while decreasing that with severe (+ + +) inflammation. Additionally, IA induced caspase-3 immunoreactivity in macrophages at the trauma area (Figures 4A and 4B; P ≤ 0.0001 as determined by Pearson's χ2 test; Figure 4G). Figures 4C to 4F depict double immunostaining for F4/80 (Figure 4C) and cleaved caspase-3 (Figure 4D), suggesting that IA elicits cell death in macrophages. Incensole acetate also reduced the amount of degenerating neurons within the hippocampus, as indicated by Fluoro-Jade-B staining (Figure 5). No optical field with severe or moderate degeneration was noted in the IA-treated mice, whereas the percentage of optical fields with mild degeneration (+) was increased (P ≤ 0.01; Pearson's χ2 test; Figure 5C).
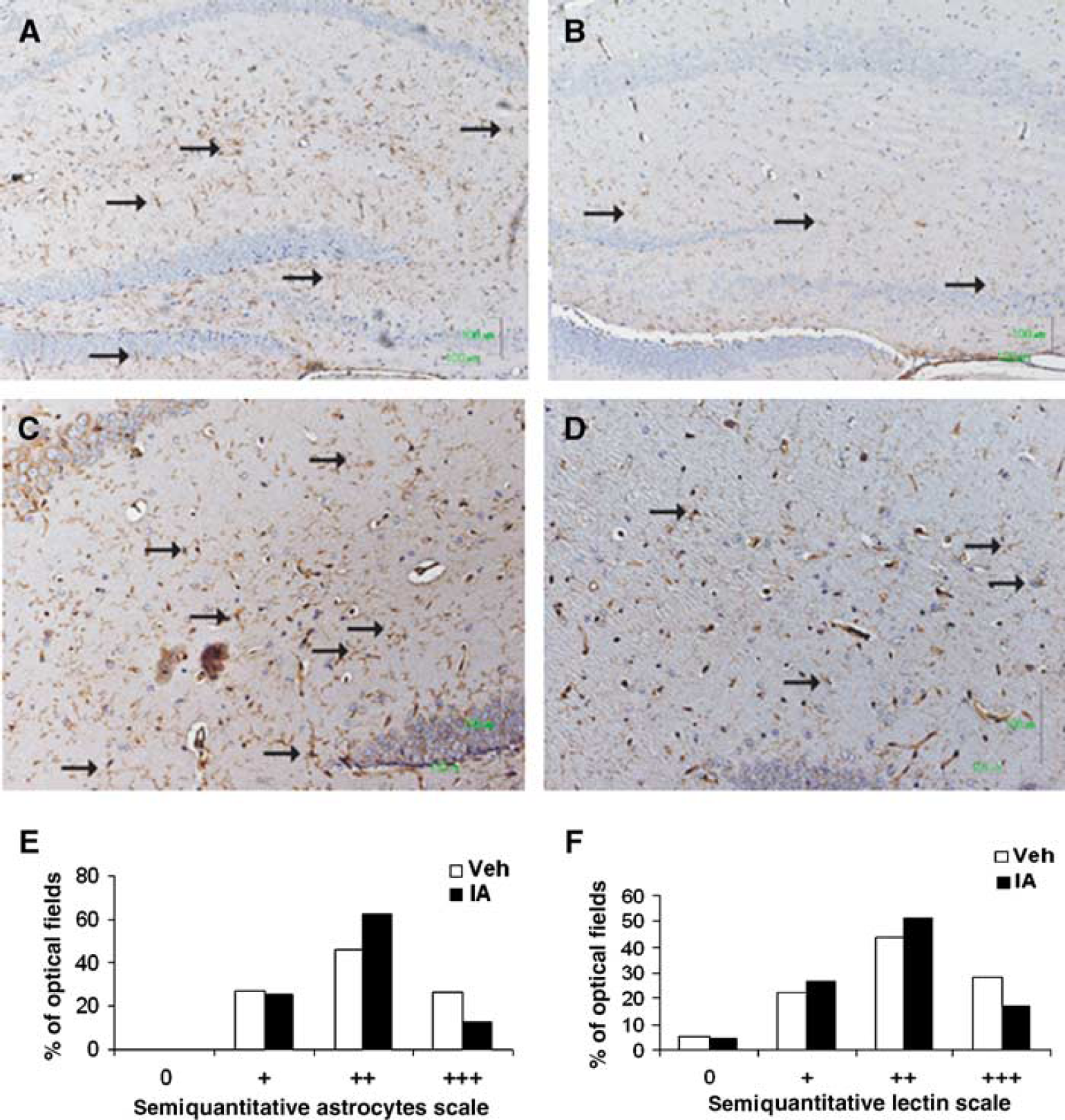
Incensole acetate (IA; 50 mg/kg) inhibits glial activation in the hippocampus of posttraumatic mice. Twenty-four hours after CHI, mice were killed and their brains processed for immunohistochemistry, as described in the Materials and methods section. Glial fibrillary acidic protein immunohistochemistry was then performed to study astroglial activation in IA-treated (
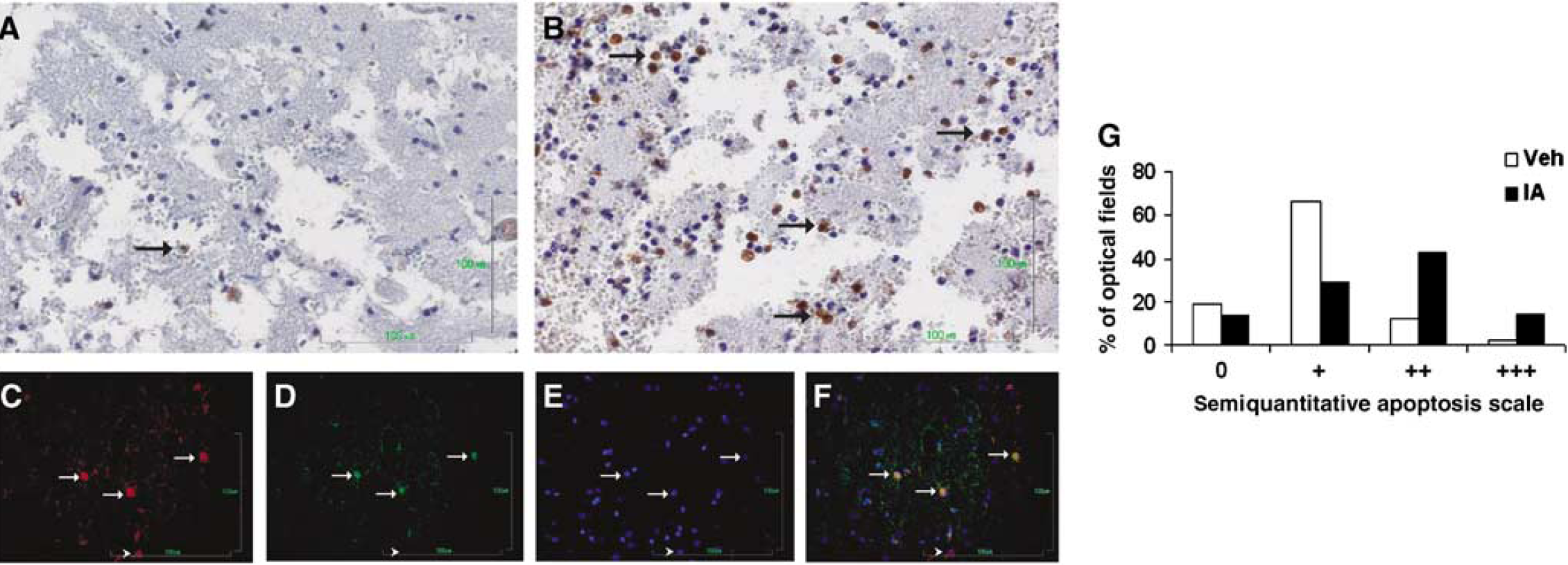
Incensole acetate (IA; 50 mg/kg) induces cell death in macrophages at the area of trauma. Twenty-four hours after CHI, mice were killed and their brains processed for immunohistochemistry, as described in the Materials and methods section. Caspase-3 immunoreactivity, indicative of cell death, was evident in IA-treated mouse brain sections (arrows,
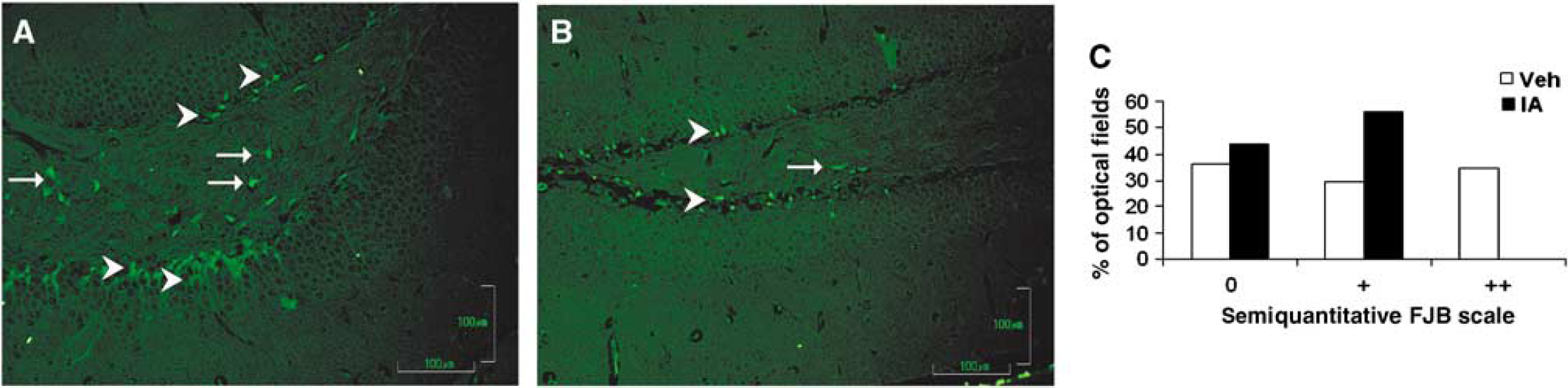
Incensole acetate (IA; 50 mg/kg) attenuates neurodegeneration in the hippocampus of posttraumatic mice. Twenty-four hours after CHI, mice were killed, and their brains processed for immunohistochemistry, as described in the Materials and methods section. Neurodegeneration was attenuated in posttraumatic IA-treated mice, as indicated by reduced Fluoro-Jade B staining. Degenerative cells in dentate gyrus (arrowheads,
Incensole Acetate Inhibits mRNA Expression of Tumor Necrosis Factor-α and Interleukin-1β mRNAs after Closed Head Injury
Inhibition of cytokines within hours after CHI, specifically TNF-α and IL-1β, was shown to benefit neurobehavioral outcome. Therefore, and as IA has shown inhibition of their production in vitro, we further examined its effect on the mRNA expression of these cytokines in mouse brains after CHI. At 3 h posttrauma, real-time PCR analysis was performed on the cortical tissue of IA- and vehicle-treated mice. The results are expressed as relative to β-actin in the same samples. Incensole acetate significantly inhibited mRNA expression of both cytokines (P ≤ 0.05 IA versus vehicle; Student's t-tests; n = 5 per group; Figure 6), corroborating the anti-inflammatory effect of IA after CHI in a mouse model.
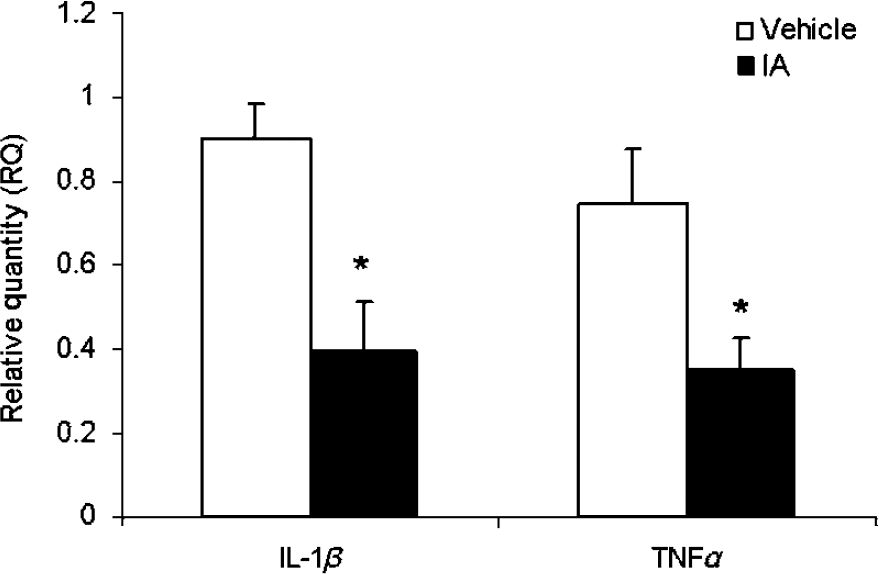
Incensole acetate (IA; 50 mg/kg) inhibits IL-1β and TNF-α mRNA expression after closed head injury. Interleukin-1β and TNF-α mRNA levels were quantified 3 h postinjury by real-time PCR. β-Actin was used as endogenous control. *P ≤ 0.05 versus vehicle; Student's t-tests.
Incensole Acetate Induces a Mild Hypothermic Effect on Closed Head Injury Mice
Immediately after CHI, the rectal temperature of all mice dropped from the preinjury temperature of 37.6 ± 0.1, by about 2°C. However, in mice treated with IA, a further decrease in body temperature was noted for 30 to 60 mins after injection, as compared with vehicle-treated mice (Figure 7, P ≤ 0.05; Student's t-test). This effect faded within the next 30 to 90 mins, and no significant hypothermic effect was then noted up to 4 h (data shown until 2 h). Observations of IA- or vehicle-treated mice that were maintained at constant preinjection temperature indicated that the lack of hypothermic effect did not hinder IA-induced neuroprotection. Thus, at day 7 after CHI, the NSS of vehicle-treated mice was 6.00 ± 0.06, whereas that of the IA-treated mice was 4.71 ± 0.52 (P = 0.04).
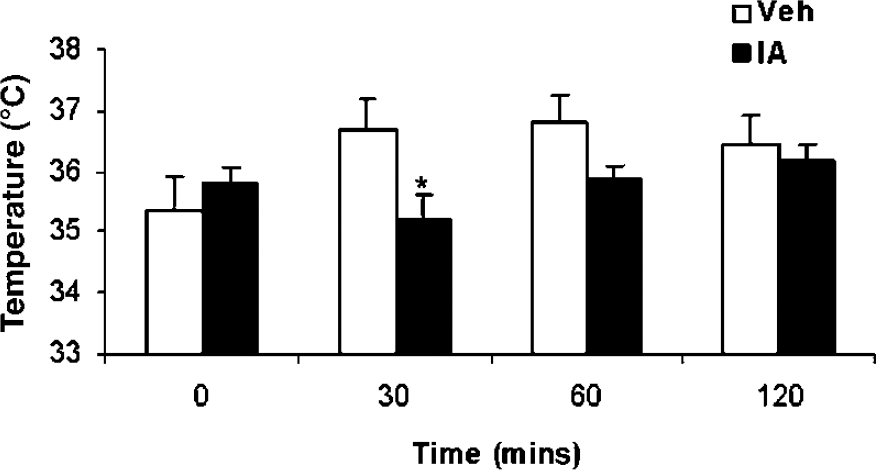
Incensole acetate (IA; 50 mg/kg) induces a mild, transient hypothermic effect on posttraumatic mice. Mice were subjected to CHI and treated with IA 1 h after trauma. *P ≤ 0.05 versus vehicle; Student's t-tests.
Incensole Acetate does not Attenuate Tissue Edema Formation
The development of local edema, expressed as an increase in tissue water content, is a notable feature of TBI. A pronounced edema was observed in the left (ipsilateral) hemisphere of all injured mice at 24 h after injury, indicating the effect of injury in both groups. Water accumulation tended to be smaller in IA mice (81.47% ± 0.35% in IA versus 82.16% ± 0.30% in vehicle), yet the difference did not reach statistical significance (P = 0.15; data not shown).
Discussion
This study allocates novel pharmacological activities to IA, a major constituent of the resin of Boswellia spp. Based on our findings, we attribute the anti-inflammatory effect of Boswellia resin, at least in part, to IA and its derivatives. Although first reported at 1967 (Corsano and Nicoletti, 1967), we are unaware of any report on the biologic activities of incensole derivatives before our studies.
In a recent study, we found that the anti-inflammatory activity of Boswellia resin involves the inhibition of the NF-κB pathway by diterpenoids with a macrocyclic cembrene skeleton, namely IA and its derivatives (Moussaieff et al, 2007). Given the long-lasting and widespread use of Boswellia resin as an anti-inflammatory agent, and that early upregulation of proinflammatory cytokines has a detrimental role in the pathophysiology of TBI and its outcome (Shohami et al, 1996, 1999; Lenzlinger et al, 2001; Truettner et al, 2005), we designed this study to further examine possible anti-inflammatory effects of IA in vitro and in vivo and to test whether IA may also improve functional recovery after CHI.
The current study shows that IA confers neuroprotection in an experimental model of TBI and emphasizes its beneficial long-term effect on motor and cognitive functions. As our preliminary study and our previously published results (Moussaieff et al, 2007) established that IA may well act as a novel anti-inflammatory agent, we proceeded to examine whether IA elicits an anti-inflammatory effect post-CHI. Our data suggest that the marked long-term neuroprotective effect of IA after CHI is associated with an early (3 h) inhibition of TNF-α and IL-1β mRNAs and with reduced glial activation and neuronal degeneration at 24 h postinjury. These findings, showing the inhibition of proinflammatory cytokines by IA and the beneficial effect conveyed by this treatment on motor and cognitive recovery, are in agreement with the notion that early inhibition of inflammatory cytokines after brain injury is beneficial for recovery (Shohami et al, 1996, 1999; Allan et al, 2005).
Reactive astrogliosis, characterized by increased GFAP content, is a hallmark associated with vigorous response of astrocytes to diverse insults including inflammation (Sharma et al, 2007). Thus, attenuated GFAP immunoreactivity, along with the reduced microglial labeling, suggests that IA exerts a neuroprotective effect, at least in part, via its anti-inflammatory activity. Given the NF-κB inhibitory effect of IA, shown in an earlier study (Moussaieff et al, 2007), and that IA-induced cell death in macrophages, it is plausible that the anti-inflammatory effect of IA after trauma is mediated by inhibition of NF-κB activation and by macrophage cell death.
The ability of IA to attenuate hippocampal neurodegeneration and to improve cognitive recovery, taken together with the established association between these two outcome measures, implies that improved cognitive ability may be mediated, at least partially, by reduced hippocampal neuronal death. This notion is substantiated by other studies in our injury model, which show cognitive deficits that are preceded by hippocampal neurodegeneration (Shein et al, 2008; Yaka et al, 2007). However, the existence of additional mechanisms underlying the functional benefit of IA cannot be excluded at this time, for example, the involvement of NMDA receptor modulation in cognitive deficits and recovery (Yaka et al, 2007). Further research is required to define the precise role of the anti-inflammatory effects of IA in the overall improved recovery and to examine other potential molecular mechanisms underlying restoration of cognitive ability, including the possible involvement of NMDA receptors.
Given the substantial amount of literature suggesting hypothermia as a further neuroprotective strategy in TBI (e.g., Clifton, 2004), and in light of previous work done in our model, which showed hypothermic effects together with altered cytokine expression in heat acclimation-induced neuroprotection (Shein et al, 2007), we examined the notion of a possible effect of IA on body temperature of mice after CHI. We found that it induces a mild hypothermia of short duration. It should be noted that previous reports, including work done in our own injury model, have suggested possible crosstalk between hypothermia and anti-inflammatory effects in central nervous system injury (Truettner et al, 2005; Shein et al, 2007); however, in this study, abolishing the hypothermic effect of IA did not hamper its neuroprotective effect. These data would indicate that the hypothermic effect does not play a central role in functional neuroprotection, which is induced by the compound, as can be expected, given the modest and transient nature of the observed decrease on temperature.
Incensole acetate and its derivatives are biomarkers of Boswellia species (Hamm et al, 2005). Our study hence concurs with the results of Schuhmann et al (2005) (presented as an abstract), who showed that the resin of B. carterii exhibits a neuroprotective effect after controlled cortical impact. Our findings suggest that the neuroprotective effect of B. carterii resin can be attributed, at least partially, to IA and its derivatives. However, unlike the data presented by Schuhmann et al, our present data suggest that IA has no significant inhibitory effect on the posttraumatic cerebral edema formation in mice. The discrepancy between the results may be due to the difference in species (rats versus mice) and models (controlled cortical impact versus CHI). It is also possible that Boswellia resin, as used by Schuhmann et al, contains other neuroprotective agents such as boswellic acids and their derivatives, which are also known for their anti-inflammatory effect (Poeckel et al, 2006).
This study may shed further light on the widespread use of Boswellia resin in traditional medicine in Europe, Asia, and Africa to treat inflammation and injuries for many centuries, given alone or as a component in a herbal mixture (for a prominent example of such a mixture, distributed for extensive use throughout Europe and Asia for hundreds of years, see Moussaieff et al, 2005). Moreover, as Boswellia resin is an important herbal remedy, it is also marketed as a food supplement for the treatment of inflammatory diseases in the United States and Europe. In view of our data on the anti-inflammatory activity of IA, we suggest that these products should be standardized for IA as well as boswellic acids. The possible synergistic effects of these compounds remain to be investigated.
Collectively, our data indicate that a single postinjury dose of IA, a novel anti-inflammatory agent, offers marked functional neuroprotection against TBI. As IA and its derivatives have been in extensive use for thousands of years as major components of Boswellia resin, they are probably safe as well as effective. From our in vivo work, we have also noted that IA is probably of very low toxicity, as we have not seen any signs of toxicity in animals even when treated with high and multiple doses of IA. We therefore propose IA and cembrene-type diterpenes as novel potential neuroprotective agents to be further explored for TBI as well as for neuroinflammatory and neurodegenerative diseases. Moreover, as cembranoids are present in tobacco blends in substantial amounts (El Sayed and Sylvester, 2007), the importance of their neuroprotective activity may well exceed the boundaries of theoretical research, and the possibility that this group of natural products poses neuroprotective activity needs to be further studied.
Footnotes
Acknowledgements
We thank Mrs Olga Touloumi for her technical support. E Shohami is the incumbent of the Dr Leon and Dr Mina Deutch Chair in Psychopharmacology, AHEPA University Hospital, Thessaloniki, Greece.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
