Abstract
It is not known if aging induces changes in nigral vascularization and nigral vascular endothelial growth factor (VEGF) levels similar to those previously reported for Parkinson's disease (PD). In this study nonexercised rats displayed age-dependent decreases in the density of nigral microvessels and VEGF mRNA expression, which were reversed by physical exercise. Such changes may enhance the vulnerability of dopaminergic neurons and the risk of developing PD, and may be reduced by exercise. Furthermore, the observed pattern is the opposite of that previously observed in PD, suggesting that the process underlying PD is not an accelerated age-dependent decline in the dopaminergic system.
Introduction
The progressive motor impairment that occurs during normal aging has been associated with nigrostriatal dysfunction and several studies have shown that the dopaminergic (DA) system is altered during normal aging (Schallert, 1988). Furthermore, aging is the most prominent risk factor for Parkinson's disease (PD). However, there is no consensus about how advancing age may affect PD or whether the changes in the DA system in PD differ from those induced by aging. It has been suggested that the normal decline in the DA system is accelerated in PD patients, or that exogenous insults that damage DA neurons exacerbate the progressive age-related loss of neurons. Recent studies suggest, however, that aging does not induce a significant loss of DA neurons but rather induces changes in soma size, tyrosine hydroxylase (TH) expression, and DA activity, which may increase the vulnerability of DA neurons to toxic damage and increase the risk of developing PD (Kubis et al, 2000; Collier et al, 2007).
Modifications of the vascular microenvironment of the DA neurons in the substantia nigra compacta (SNc) may alter the availability of nutrients and toxic compounds, and affect the vulnerability of DA neurons. Previous studies have reported changes in SNc vascularization in PD patients and animal models of PD (Barcia et al, 2005; Faucheux et al, 1999). Similarly, aging is known to induce changes in angiogenesis in the brain and other tissues (Iemitsu et al, 2006; Wang et al, 2004). However, it is not known if aging induces changes in nigral vascularization similar to those reported for PD. Vascular endothelial growth factor (VEGF) is a potent regulator of angiogenesis, and is thought to be involved in aging-induced deterioration of angiogenesis in several tissues (Iemitsu et al, 2006; Wang et al, 2004), as well as in vascular changes observed in PD and PD animal models (Barcia et al, 2005; Wada et al, 2006). Furthermore, several recent studies have shown that VEGF is a highly potent neuroprotective and neurorescue molecule for DA neurons (Yasuhara et al, 2004). However, there are no data on the effects of aging on nigral levels of VEGF. Finally, it has been observed that physical exercise ameliorates aging-induced effects on angiogenesis and VEGF levels in several tissues (Ding et al, 2004; Iemitsu et al, 2006), and also induces beneficial functional effects and neuroprotection in PD and PD models (Cohen et al, 2003; Chen et al, 2005). However, the effects of exercise on vascularization of aged SNc have not been investigated. In this study, we investigated aging-related changes in SNc vascularization and VEGF expression as well as the effect of exercise on these changes, and compared the results with previously reported findings for PD and PD models.
Methods
Experimental Design
Male adult Sprague—Dawley rats were used in the experiments. Male rats were used to minimize possible effects of changes in the endogenous hormonal environment, particularly differences in ovarian hormones in female rats during the menstrual cycle and between pre- and postmenopausal groups, which may affect the results of this study. All experiments were performed in accordance with the ‘
Motor Exercise
A straight two-lane treadmill (14cm wide, 50cm long) connected to a personal computer for system control and data management (CT-2 treadmill system; Columbus Instruments, OH, USA) was used. The rats were trained to run in the treadmill more than 28 consecutive days (one 30 mins session per day at 17 m/mins, with a previous training session of 5 mins at 5 to 10 m/mins).
Immunohistochemistry and Stereological Analysis of Nigral Microvessels
The rats were killed and perfused with 0.9% saline and then with cold 4% paraformaldehyde. The brains were removed and cut into 40-µm sections on a freezing microtome. Series of sections were processed for TH (as a marker of DA neurons) or rat endothelial cell antigen-1 (as a marker of microvessels) immunohistochemistry using a rabbit polyclonal antiserum to TH (Peel-Freez, Rogers, AR, USA; 1:500) or a mouse monoclonal antirat endothelial cell antigen-1; 1:2,000; Serotec, Kidlington, Oxford, UK). Serial sections were also processed for double immunolabeling against TH and rat endothelial cell antigen-1. The total number of microvessels in the SNc was estimated by stereological analysis with an Olympus CAST-Grid system. Uniform randomly chosen sections through the substantia nigra were analyzed for the total number of vessels by means of a stereological grid (fractionator), and the nigral volume was estimated according to Cavalieri's method. See Rodriguez-Pallares et al (2008) for details.
RNA Extraction and Real-Time Quantitative Reverse Transcriptase PCR
Total RNA from the ventral midbrain was extracted with Trizol (Invitrogen, Paisley, UK) according to the manufacturer's instructions. Total RNA (2 µg) was reverse transcribed to cDNA with deoxyribonucleotide triphosphates, random primers, and Moloney murine leukemia virus reverse transcriptase (200 U; Invitrogen).
Real-time PCR was used to examine relative levels of VEGF mRNA. VEGF primers sequences were: forward 5′-G GAGTACCCCGATGAGATAGAGTA-3′, reverse 5′-TATCTT TCTTTGGTCTGCATTCAC-3′. β-Actin was used to normalize the amount of cDNA samples (forward 5′-TCGTG CGTGACATTAAAGAG-3′, reverse 5′-TGCCACAGGATTCC ATACC-3′). Experiments were performed with a real-time iCyclerTM PCR platform (Bio-Rad, Hercules, CA, USA) using the SYBR Green SuperMix reagent (Bio-Rad). The comparative
Statistical Analysis
All data were expressed as mean±s.e.m. Multiple comparisons were analyzed by one-way analysis of variance followed by Bonferroni
Results
There was an age-dependent decrease in the density of nigral microvessels in sedentary (i.e., nonexercised) animals, which was statistically significant in middle aged and highly significant in the oldest rats (relative to young rats). Exercised rats showed a higher density of blood vessels in the SNc than nonexercised rats. This difference was not statistically significant in young rats, but was significant in the middle-aged and aged groups (Figure 1).
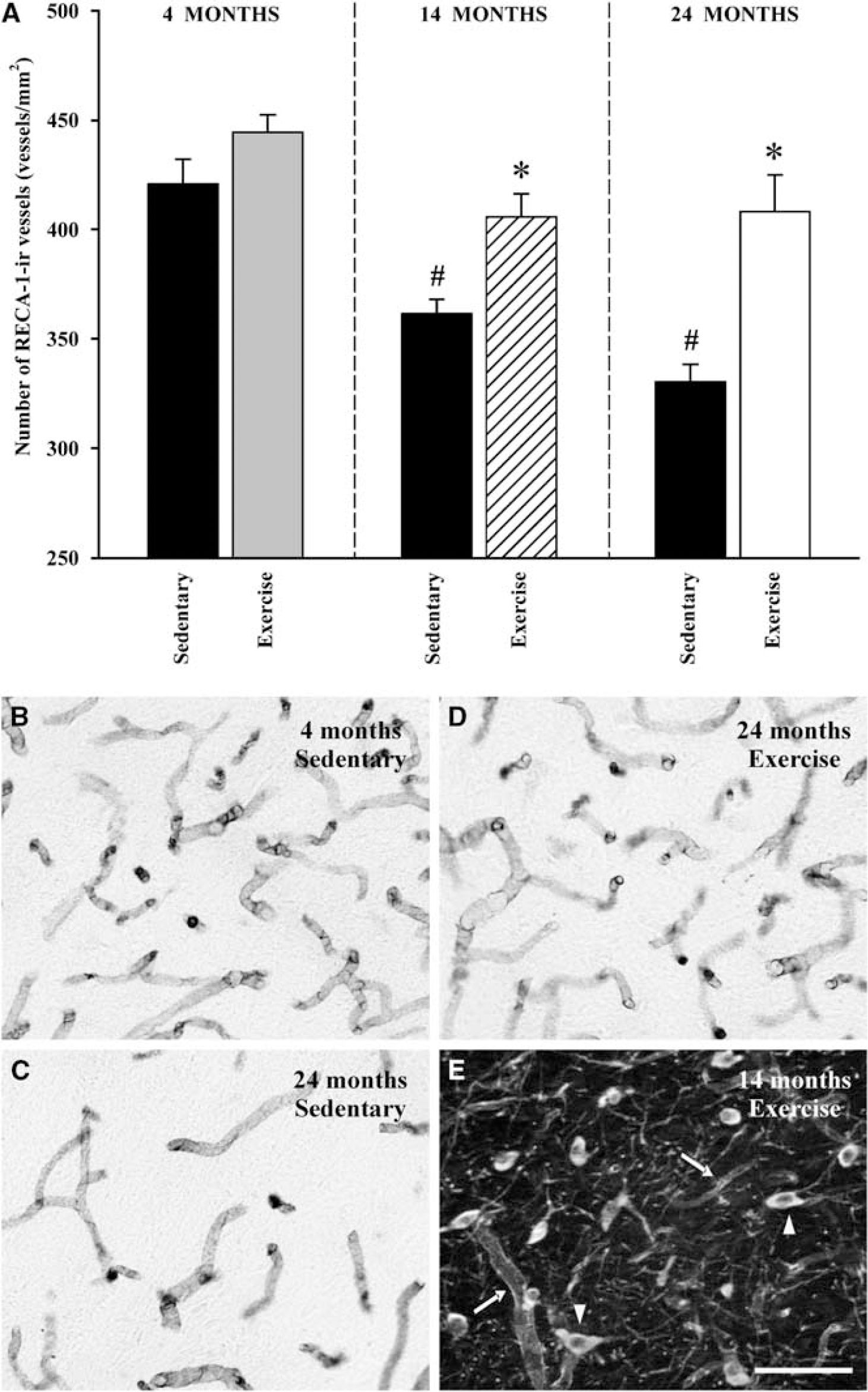
Density of microvessels in the substantia nigra of rats of different ages. (
The VEGF mRNA levels also decreased with increasing age in nonexercised rats, and the decrease was statistically significant in aged rats (relative to young rats). In the group of young rats, treadmill running did not induce any significant increase in VEGF mRNA expression. However, in middle-aged and aged rats treadmill running induced a significant increase in VEGF mRNA levels with respect to the sedentary animals of the same age.
Interestingly, motor exercise in middle-aged rats restored VEGF mRNA expression to levels similar to those observed in young adults. In the oldest age group, VEGF mRNA levels were significantly increased by motor exercise, although they did not reach the levels observed in young animals (Figure 2).
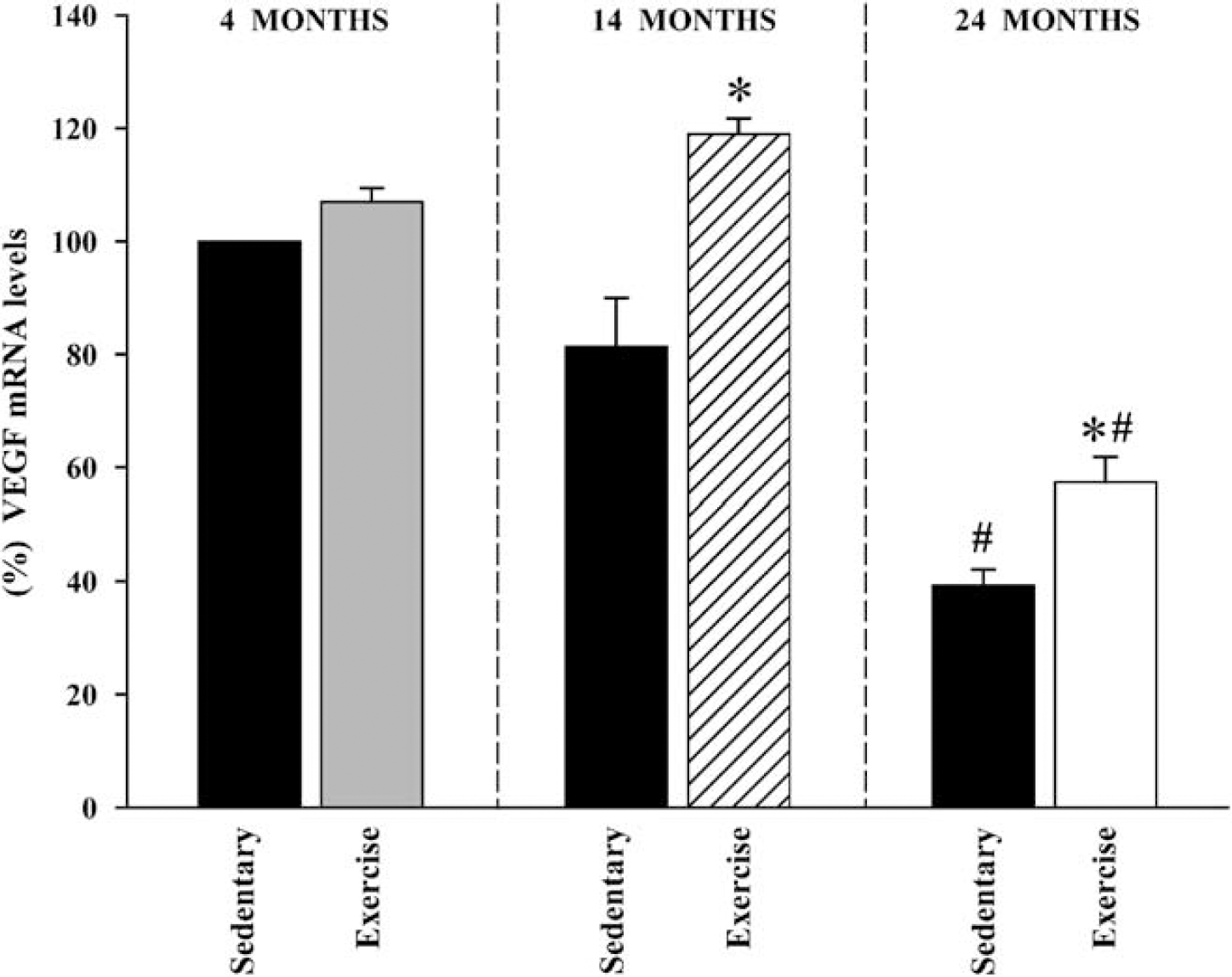
Expression of the VEGF mRNA in the ventral mesencephalon. Real-time RT-PCR analysis revealed that the expression of VEGF mRNA in sedentary (i.e., nonexercised) rats showed an age-dependent decrease, which was statistically significant in aged rats. In middle-aged and aged rats treadmill running induced a significant increase in VEGF mRNA levels, although in aged animals the levels did not reach those observed in young animals. Data represent mean±s.e.m. *
Discussion
In this study, we observed a significant decrease in the density of microvessels and a decrease in VEGF mRNA expression in the SNc of middle-aged and, particularly, aged sedentary animals. We also observed that continuous motor exercise significantly increased both nigral blood vessel density and VEGF mRNA expression. Angiogenesis has been reported to be impaired in aging in several tissues including brain (Iemitsu et al, 2006; Wang et al, 2004). Perfusion deficits have also been observed before the onset of clinical symptoms in other neurodegenerative disorders, which suggests that the deficits contribute to the pathogenesis of the disease (Storkebaum and Carmeliet, 2004). Conflicting results have been obtained in different studies, with either an age-dependent depletion or no change in DA neuron counts, which may be attributable to a number of methodological variables (Kubis et al, 2000; Collier et al, 2007). However, the present results support observations of a progressive functional decline in the SNc and increased vulnerability to injury with age.
The present results are consistent with previous findings that suggest that the mechanism responsible for impaired angiogenesis in different tissues of aged animals is related to reduced expression of VEGF (Iemitsu et al, 2006; Wang et al, 2004). Furthermore, recent studies have revealed that VEGF is also involved in neurodegeneration (Storkebaum and Carmeliet, 2004) and, particularly, that VEGF has neuroprotective and neurorescue effects on DA neurons (Yasuhara et al, 2004). The low levels of VEGF observed in aged animals in this study may contribute to the DA neuron vulnerability, in addition to possible effects of chronic ischemia. Interestingly, we observed that both the age-dependent decrease in nigral vascularization and nigral VEGF levels were reverted by locomotor exercise. Previous studies have reported that exercise has beneficial effects on neuroprotection against brain damage in ischemia/reperfusion models (Ding et al, 2004). The underlying mechanisms remain unclear. This study suggests the involvement of the exercise-induced increase in VEGF expression, possibly as a compensatory mechanism leading to increased capillary surface area in response to an increased demand for oxygen and energy. Several studies have reported neuroprotective effects of exercise in PD and animal models of PD (Chen et al, 2005), and an exercise-induced increase in striatal levels of glial cell-derived neurotrophic factor has been suggested as the underlying mechanism (Cohen et al, 2003). The present results suggest that an exercise-induced increase in nigral VEGF levels is also involved. It is possible that once cell loss begins a compensatory upregulation of trophic factors in SNc and other regions may help to slow the degeneration, a process that could be boosted by exercise. It is also possible DA neuron degeneration begins in a young brain and the neurologic deficits are minimized by compensatory adaptations, which may be enhanced by exercise in the SNc and possibly other regions, and during the aging process brain gradually loses compensatory capacity such that symptoms emerge (Schallert, 1988).
However, increased vascularization and upregulation of VEFG levels have been observed in the SNc of PD patients (Faucheux et al, 1999; Wada et al, 2006) and animal models (Barcia et al, 2005) in direct contrast to the pattern of changes observed in aged rats in this study. This clearly confirms that DA degeneration in PD cannot be considered as an acceleration of a progressive age-related decrease in DA function. The cause of the increased nigral vascularization and VEGF levels in PD has not been clarified. It may constitute a contraregulatory mechanism induced by DA cell death as suggested above, or may be secondary to the accompanying inflammatory process (see Rodriguez-Pallares et al, 2008), because upregulation of VEGF has been reported in other degenerative and inflammatory processes (see Storkebaum and Carmeliet, 2004 for review). In the case of PD, upregulation of VEGF may also be a consequence of a decreased inhibitory effect of DA on VEGF and VEGF-dependent angiogenesis (Basu et al, 2001), because of a reduction in basal levels of DA after the loss of a significant number of DA neurons. The abnormal regulation of VEGF may lead to loss of blood—brain barrier integrity and microvascular vascular leakage, enhanced inflammation, and may contribute to the vicious circle of DA cell death observed in PD.
In conclusion, this study revealed that aging induces a reduction in blood vessel density and VEGF levels in SNc, which may increase the vulnerability of DA neurons to additional insults and increase the risk of developing PD. Interestingly, these risk factors were reduced in middle-aged and aged animals by physical exercise. Furthermore, the pattern observed in aged animals was the opposite to that observed in PD, suggesting that the neurodegenerative process underlying PD is not an accelerated age-dependent decline in DA system function.
Footnotes
Acknowledgements
We thank Pilar Aldrey for her excellent technical assistance.
Disclosure/conflict of interest
The authors report no conflicts of interest.
