Abstract
Vascular endothelial growth factor (VEGF) is currently considered a potential pharmacologic agent for stroke therapy because of its strong neuroprotective and angiogenic capacities. Nonetheless, it is unclear how neuroprotection and angiogenesis by exogenous VEGF are related and whether they are concurrent events. In this study, the authors evaluated by stereology the effect of VEGF on neuronal and vascular volume densities of normal and ischemic brain cortices of adult male Sprague-Dawley rats. Ischemia was induced by a 4-hour occlusion of the middle cerebral artery. Low, intermediate, and high doses of VEGF165 were infused through the internal carotid artery for 7 days by an indwelling osmotic pump. The low and intermediate doses, which did not induce angiogenesis, significantly promoted neuroprotection of ischemic brains and did not damage neurons of normal brains. In contrast, the high dose that induced angiogenesis showed no neuroprotection of ischemic brains and damaged neurons of normal brains. These findings suggest that in vivo neuroprotection of ischemic brains by exogenous VEGF does not necessarily occur simultaneously with angiogenesis. Instead, neuroprotection may be greatly compromised by doses of VEGF capable of inducing angiogenesis. Stroke intervention efforts attempting to induce neuroprotection and angiogenesis concurrently through VEGF monotherapy should be approached with caution.
It is well documented that vascular endothelial growth factor (VEGF) is upregulated in ischemic brains after stroke (Beck, 2000; Hayashi et al., 1997; Lennmyr et al., 1998; Plate et al., 1999; Sun et al., 2003; Zhang et al., 2002). The increased expression of VEGF is believed to be involved with angiogenesis, vascular permeability, neurogenesis, chemotaxis of inflammatory cells, and inhibition of apoptosis (Bates and Harper, 2002; Carmeliet et al., 1996; Ferrara and Gerber, 2001; Forstreuter et al., 2002; Lafuente et al., 2002; Rosenstein et al., 1998; Sun et al., 2003; van Bruggen et al., 1999; Zhang et al., 2000). Recent reports affirm that VEGF also has a significant neuroprotective effect on neurons and glial cells and stimulates their growth and survival (Carmeliet and Storkebaum, 2002; Hayashi et al., 1998; Jin et al., 2000, 2002; Krum and Khaibullina, 2003; Krum et al., 2002; Sondell et al., 1999; Sun et al., 2003). Because of VEGF's potent and diverse effects on endothelial cells, neurons, and the nervous system, a challenge for current and future research is to clarify the usefulness of the growth factor as a therapeutic agent for stroke intervention, particularly with regards to its angiogenic and neuroprotective capacities (Bellomo et al., 2003; Carmeliet and Storkebaum, 2002; Ferrara, 2002; Krupinski et al., 2003; Sun et al., 2003; Zhang and Chopp, 2002).
However, the diverse activities of VEGF on the nervous system present a unique problem in applying the growth factor as a monotherapeutic agent for stroke treatment (Carmeliet and Storkebaum, 2002; Ferrara, 2002; Zhang and Chopp, 2002). Though VEGF promotes survival of neurons after injury, the blood vessels formed through angiogenesis tend to be immature and permeable (Bates et al., 2002; Dobrogowska et al., 1998; Roberts and Palade, 1995; Schoch et al., 2002; Thurston et al., 2000; Zhang et al., 2000; Zhao et al., 1998). VEGF is also a vasodilator (Laham et al., 2003). These biologic consequences stemming from the use of exogenous VEGF can lead to severe cerebrovascular pathology (Pettersson et al., 2000; Schoch et al., 2002; Sun et al., 2003; Thurston et al., 2000; van Bruggen et al., 1999; Zhang et al., 2002). Thus, the possibility that induced angiogenesis by exogenous VEGF could greatly compromise or even negate neuroprotection cannot be ignored.
Very few stereologic studies have been conducted to ascertain the morphometric relationship between neuronal density and vascular volume density of normal and ischemic brains after treatment with VEGF. Morphometric data are essential in elucidating the quantitative relationships between changes in the volume density of the blood vessels and the number of viable neurons immediately surrounding the restructured blood vessels. In this report, we present preliminary data quantifying the relationship between vascular volume density (VasV) and the density of viable neurons (NeuV) of normal and ischemic brains after treatment with low, intermediate, and high doses of VEGF.
MATERIALS AND METHODS
Animals
Male Sprague-Dawley rats were purchased from Harlan (San Diego, CA, U.S.A.) at the age of 6 weeks. The animals were kept in the Animal Research Facility of the Veterans Administration Medical Center (La Jolla, CA, U.S.A.). The rats were housed in pairs in the standard 48 × 27 × 20-cm3 shoe-box plastic cages with a floor of paper shavings, kept under a normal 12-hour light/dark cycle in a temperature-controlled room, and fed rat chow and water ad libitum. The weights of the animals ranged from 250 to 320 g at the start of the experiments.
Middle cerebral artery occlusion and implantation of the osmotic pump
The animal experiments were approved by the Animal Research Committee of the Department of Veterans Affairs Medical Center and were conducted according to the National Institute of Health's Guide for the Care and Use of Laboratory Animals. A modification of the method of Longa et al. (1989) for middle cerebral artery occlusion (MCAO) was used.
The rats were initially anesthetized by inhalation of 2% halo-thane in an airtight bell jar. Anesthesia was then maintained with 1% to 2% halothane and nitrous oxide:oxygen (60:40) by face mask. During surgery, the left temporalis muscle temperature was monitored with a temperature probe (Malinkrodt Anesthesia Products, Inc., St. Louis, MO, U.S.A.) linked to a controller (YSI Model 73 ATD; Yellow Springs Instrument Co., Yellow Springs, OH, U.S.A.) and a heat lamp to maintain normothermic brain temperature.
A midline neck incision was made exposing the left common carotid artery. The external carotid and pterygopalatine arteries of the left common carotid were then ligated with 5–0 silk ligatures. An incision was made in the arterial wall of the external carotid close to the bifurcation of the common carotid artery. A 4–0 heat-blunted nylon suture was inserted into the incision and advanced approximately 18 mm from the common carotid bifurcation into the internal carotid artery to block the origin of the middle cerebral artery. The suture was left in place for 4 hours, removed, and the tip of the osmotic minipump catheter was then inserted into the incision and advanced to the common carotid bifurcation.
Recombinant VEGF165 was obtained from Sigma (St. Louis, MO, U.S.A.). The osmotic minipumps (Alzet 2001; Alza Scientific Products, Mountain View, CA, U.S.A.) contained normal saline or recombinant VEGF165 at the concentrations of 10 μg/mL, 50 μg/mL, or 360 μg/mL in saline. The pumps were designed to deliver the contents at 1 μL/h for 7 days. The three VEGF concentrations would, therefore, deliver a total of 2 μg (low dose), 8 μg (intermediate dose), or 60 μg (high dose) of VEGF over the week. The filled pumps were incubated overnight in sterile saline at 37°C before use.
The osmotic minipump catheter was held in place in the artery with application of a biocompatible glue over the incision. After a skin incision over the left side of the skull, a pocket was formed over the neck and shoulder blades to hold the minipump. The neck and shoulder incisions were then permanently sutured. The animals were given 20 U of heparin in 0.2 mL normal saline intravenously at the conclusion of surgery. They were kept on a heated (37.5°C) water blanket (Gorman-Rupp Industries, Bellville, OH, U.S.A.) for 1 hour during recovery and then returned to their cage. All animals were maintained for 7 days after implantation of the osmotic pump and/or MCAO, and then perfused with histologic fixative as described below.
Experimental groups
The animals were classified into 10 groups, with three animals in each group, as follows.
Group 1: normal brain
The animals were perfusion fixed without undergoing 4 hours of MCAO or osmotic pump implantation. The results obtained from these animals served as the control data of VasV and NeuV in normal cortex.
Group 2: normal brain plus saline
The animals underwent osmotic pump implantation but no MCAO. The osmotic pump delivered normal saline for 7 days. The results obtained from these animals served as another source of control data of VasV and NeuV in normal cortex.
Group 3: normal brain plus 2 μg VEGF
The animals of this group, as with Groups 2, 4, and 5, underwent osmotic pump implantation but no MCAO. The osmotic pump contained VEGF at the concentration of 10 μg/mL in normal saline and delivered a total of 2 μg VEGF for the 7 days.
Group 4: normal brain plus 8 μg VEGF
The osmotic pump contained VEGF at the concentration of 50μg/mL and delivered a total of 8 μg VEGF for the 7 days.
Group 5: normal brain plus 60 μg VEGF
The osmotic pump contained VEGF at the concentration of 360 μg/mL and delivered a total of 60 μg VEGF for the 7 days.
Group 6: ischemic brain
The animals underwent MCAO without the osmotic pump implantation. After MCAO, the occlusion suture was removed and the animals were allowed regular reperfusion of their brains for 7 days.
Group 7: ischemic brain plus saline
The animals of this group, as with groups 8, 9, and 10, underwent MCAO and osmotic pump implantation. The osmotic pump delivered normal saline for 7 days after the MCAO.
Group 8: ischemic brain plus 2 μg VEGF
The osmotic pump contained VEGF at the concentration of 10μ g/mL and delivered a total of 2 μg VEGF for the 7 days.
Group 9: ischemic brain plus 8 μg VEGF
The osmotic pump contained VEGF at the concentration of 50μg/mL and delivered a total of 8 μg VEGF for the 7 days.
Group 10: ischemic brain plus 60 μg VEGF
The osmotic pump contained VEGF at the concentration of 360 μg/mL and delivered a total of 60 μg VEGF for the 7 days.
Neurological assessment
Neurologic examination to determine changes in the behavior of the animals was performed 24 hours after MCAO surgery. The examination followed a dichotomized version of the published rodent neurologic grading system (Bederson et al., 1986). The five criteria used were (1) reduced exploration when allowed to roam freely on a flat surface and in an open space, (2) forepaw retraction on tail lifting, (3) asymmetric forepaw grasp, (4) axial twist on tail lifting, and (5) forced circling when walking. Each behavior was rated as normal or abnormal. Animals that underwent MCAO and did not exhibit at least three of the five signs were excluded from the study and replaced, to maintain three animals per group.
These neurologic examinations were conducted to ensure that the brains of the animals that underwent the MCAO procedure would be ischemic. This information was gathered from our own experience during the early phases when we were designing our stroke model. On several occasions, we encountered situations where the brains of the animals that underwent the 4-hour MCAO procedure did not histologically show significant cortical infarcts. At worst, these brains only showed striatal infarction. Apparently, the procedure does not always produce complete occlusion of the middle cerebral artery. We later discovered that the animals that histologically showed no cortical infarction or only striatal injury were animals that exhibited no more than two of the five signs. In contrast, the animals that demonstrated at least any three of the five neurologic signs consistently showed cortical and striatal infarctions—particularly those animals that showed (1) forepaw retraction on tail lifting, (2) asymmetric forepaw grasp, and (3) axial twist on tail lifting. Based on this information, all animals included in the study were required to show at least any three of the five neurologic signs.
Fixation and tissue preparation
At 7 days after MCAO or start of VEGF infusion, the animals were anesthetized by an intraperitoneal injection with a 50% aqueous urethane (Sigma Chemical Company) solution at the dose of 0.003 mL/g body weight. The anesthetized animal was rapidly thoracotomized to expose the heart. The animal was not artificially ventilated and no artery was ligated.
Perfusion was initiated with the transcardial infusion of 15 mL saline (Baxter Healthcare, Corp., Deerfield, IL, U.S.A.) into the ascending aorta to rapidly clear blood from the vascular system. An incision was made in the right atrium to allow the blood to drain freely. The saline wash was immediately followed by 300 mL of fixative consisting of 2% paraformaldehyde and 3% glutaraldehyde in 0.1-mol/L sodium cacodylate buffer, pH 7.4. The Harvard perfusion pump (Harvard Apparatus, Boston, MA, U.S.A.) was used to provide pressure. The pressure was initially set to deliver the solution at 180 mL/min. After the initial 15 seconds of perfusion (which was 5 seconds of saline perfusion and 10 seconds of fixative), the perfusion rate was arbitrarily reduced to 30 mL/min to conserve the fixative and to avoid unnecessary exposure of the endothelial cells and brain tissue to the high rate flow, which could cause damage.
The formaldehyde solution was freshly prepared from paraformaldehyde (Fisher Scientific, Fair Lawn, NJ, U.S.A.) on the day of the fixation. Sodium cacodylate and 50% glutaraldehyde (electron microscopy grade) were purchased from Electron Microscopy Sciences (Fort Washington, PA, U.S.A.). The glutaraldehyde fixative was stored at 4°C before use. Both the saline and fixative solutions were used at room temperature.
After fixation, the brain was removed from the skull and stored overnight in the same fixative at 4°C. A 1-mm-thick coronal brain slice of the left and right hemispheres was cut with a razor blade 7 mm from the frontal pole of the brain using a rat brain slicer (Braintree Scientific Inc., Braintree, MA, U.S.A.) (Fig. 1). We used tissues from this region of the brain since this was approximately the middle portion of the infarcted brain after MCAO. It is known that the middle cerebral artery perfuses the brain region 2 to 11 mm caudal to the frontal pole of the adult rat brain (Cole et al., 1993). Cortical tissue could also be easily distinguished from the subcortex by the conspicuous arch of the corpus callosum in this region.
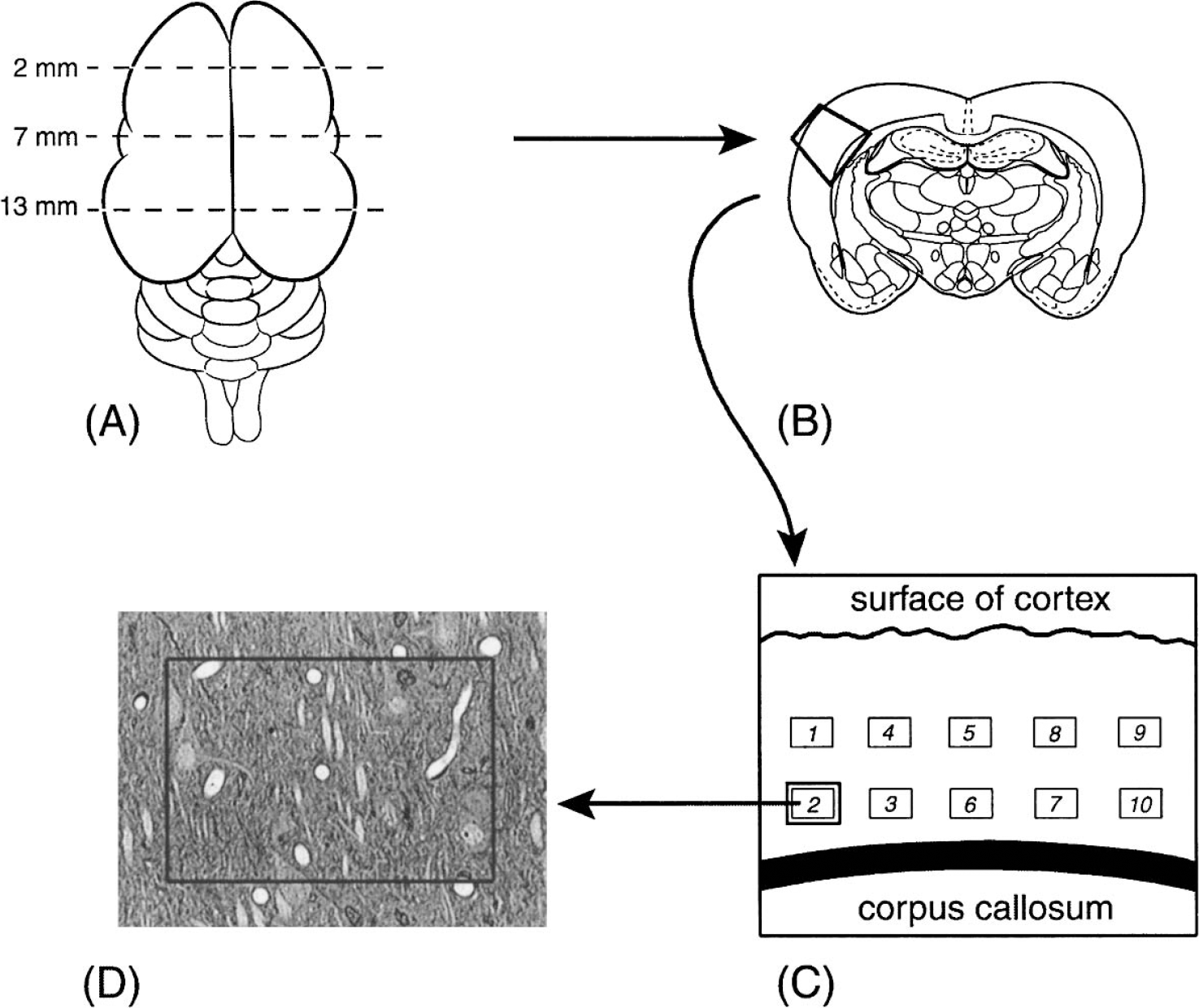
Schematic representation of the sequential steps used to obtain the cortical tissue for morphometric analyses of microvascular volume and neuronal densities. (
The 1 mm thick coronal brain slice was placed in 4 changes of 0.1-mol/L sodium cacodylate buffer, pH 7.4 (2 hours each change) to wash out the aldehyde fixatives and then postfixed in 0.1-mol/L sodium cacodylate buffered 1% osmium tetroxide (Electron Microscopy Sciences), pH 7.4, for 3 hours. The slice was dehydrated through 50%, 75%, and 95% reagent-grade ethanol (20 minutes each) followed by 2 changes (30 minutes each) of 100% ethanol. Infiltration of the slice was done with two changes (30 minutes each) of propylene oxide, followed by 1:1 and 1:2 propylene oxide: Epon mix (6 hours each) and pure Epon (6 hours). Polybed 812 was the Epon embedding medium (Electron Microscopy Sciences).
Selection of cortical region for evaluation
A 2 × 2-mm2 (length by width) piece of cortical tissue was cut from the parietal cortex of the left hemisphere with a razor blade (Fig. 1). For ischemic brains after MCAO, this cortical tissue would be within the infarcted area. Though the infarct area could be readily observed visually, ischemic injury was confirmed by cutting the adjacent 1 mm thick brain slice at 80 μm with a vibratome and staining the sections with hematoxylin. The infarcted area would appear pale in contrast to the surrounding purplish-staining tissue.
The 2 × 2-mm2 tissue block included all six layers of the cortex and the upper portion of the white matter (Fig. 1). The tissue was trimmed to a trapezoid with the pial surface of the cortex being the top edge of the trapezoid. The combination of the trapezoidal-shaped specimen together with the presence of the white matter provided easy tissue orientation, particularly when the tissue is blackened after osmication. The 2 × 2-mm2 piece of tissue was embedded in Epon to be cut in the coronal plane. Before cutting the sections, the Epon block was trimmed manually so the face of the block outlined the trapezoidal-shaped tissue. From each Epon block, a 1-μm-thick section was cut with a glass knife, stained with 1% toluidineblue/azure II, placed on a glass slide, and coverslipped with Permount (Electron Microscopy Sciences).
Image capture
The images of the cortex were captured using a Pulnix TMC-7 videocamera (CCTV Camera, VDC-2524; Sanyo, Chatsworth, CA, U.S.A.) attached to an Olympus CH2 microscope (Olympus CH2, Model BHT; Olympus Optical Co., Ltd., Tokyo, Japan). The image analysis software used was Image Pro Plus, version 4.0 (Media Cybernetics, Silver Spring, MD, U.S.A.), installed on a Pentium II PC. The digitized image was displayed at a final magnification of ×1,150 on the computer monitor.
Figure 1 diagrammatically shows the process of image selection. For each brain, one section was analyzed. From each section, 10 images from layers III and V of the cortex were captured. Five of the images were from layer III and the other five from layer V. The images were distributed evenly across the section.
Evaluation of VasV
Using features available in Image-Pro Plus, a rectangle covering a total tissue area of 0.0145 mm2 (0.145 mm × 0.1 mm) was positioned on each image (Fig. 1). All microvessels present within the sampling rectangle were included for the study. For those microvessels whose profile touched any of the borders of the rectangle, a stereologic exclusion rule was followed; any microvessel profile that touched the left or bottom border of the rectangle was excluded while those touching the right or upper borders were included. In each image, the luminal border of all the microvessels (endothelial surface facing the lumen of the vessel) included for the evaluation was manually outlined using the Image-Pro Plus tracing feature. The area of the microvessels was then automatically computed by Image-Pro Plus.
The total area of the microvessels for each rat was obtained by adding the microvessel area of the 10 images. VasV, defined as the percent total microvessel volume per unit volume of tissue, was obtained using the following stereologic formula:
Evaluation of NeuV
From each of the 10 images from each animal used for VasV analysis, the number of normal-appearing (viable) neurons was manually counted. Normal (viable) neurons were defined as neurons with pale nuclei, whether or not possessing darkened cytoplasm. Neurons showing darkened cytoplasm and nuclei, with or without pyknosis, were interpreted as damaged neurons and were excluded (Cammermeyer, 1960a, 1960b). Only neurons whose nuclei and cytoplasmic profiles were conspicuous were counted. This reduced the problem of differentiating neurons from astrocytes. As with the microvessel profiles, the exclusion rule was followed for neurons touching any of the borders of the rectangle.
NeuV is defined as the number of neurons per unit volume (cubic millimeter) of tissue. The unfolding method (Miki et al., 1997), a simple yet powerful stereologic method for estimating the NeuV was used. The NeuV of neurons was obtained by applying the stereological formula:
where NeuA is the total number of neurons counted per total area (0.145 mm2) of tissue. The total number of neurons was the sum of the number of neurons of the 10 images of each brain. The total tissue area of 0.145 mm2 was the sum of the tissue area of the 10 images (tissue area of 0.0145 mm2 per image). D is the mean nuclear diameter (in millimeters) of all the neuronal nuclei profiles obtained from the 10 images. T is the thickness of the section (in millimeters).
Since the total number of neuronal nuclei profiles counted in each brain (10 images) was less than 500, the Schwaltz-Saltikov diameter correction procedure (a mathematical correction method to obtain the true mean diameter) was not performed. Given the small number of the objects of interest counted, the use of the Schwaltz-Saltikov correction was unnecessarily arduous and the method generally requires at least 500 objects of interest to be counted. Furthermore, the Schwaltz-Saltikov correction would be necessary in the situation where the spherical objects counted were of different sizes. However, in our study, we assumed the nuclei of the neurons to be uniform (see the “Unfolding method for measuring NeuV” subsection of “Technical considerations” in the Discussion). To simplify the correction for the mean nuclear diameter, the mean diameter (that is, the mean nuclear diameter of all the neurons counted from the 10 images of each group) was multiplied by 4/π, an Abercrombie correction.
Statistical analysis
VasV and NeuV data of each of the 10 groups are expressed as mean ± SD (of the three animals in each group). For each of the density parameters (VasV and NeuV), comparisons were made among the 10 groups with one-way analysis of variance (ANOVA). When the ANOVA identified significant between-group differences, unpaired t-tests with Bonferroni post hoc corrections were used for intergroup comparisons. All reported P values were two tailed, and P value ≤0.05 was considered statistically significant.
RESULTS
The VasV and NeuV means ± SD of all the 10 groups are shown in Table 1. For the effect of VEGF on VasV, the ANOVA revealed significant difference between the 10 groups (F = 3.5; P = 0.01). For the effect of VEGF on NeuV, the ANOVA also showed significant differences (F = 48.6; P < 0.0001). Discussion of the results of the t-tests comparing differences between the groups with respect to VasV and NeuV is included in the caption of Table 1.
Vascular volume and neuronal densities
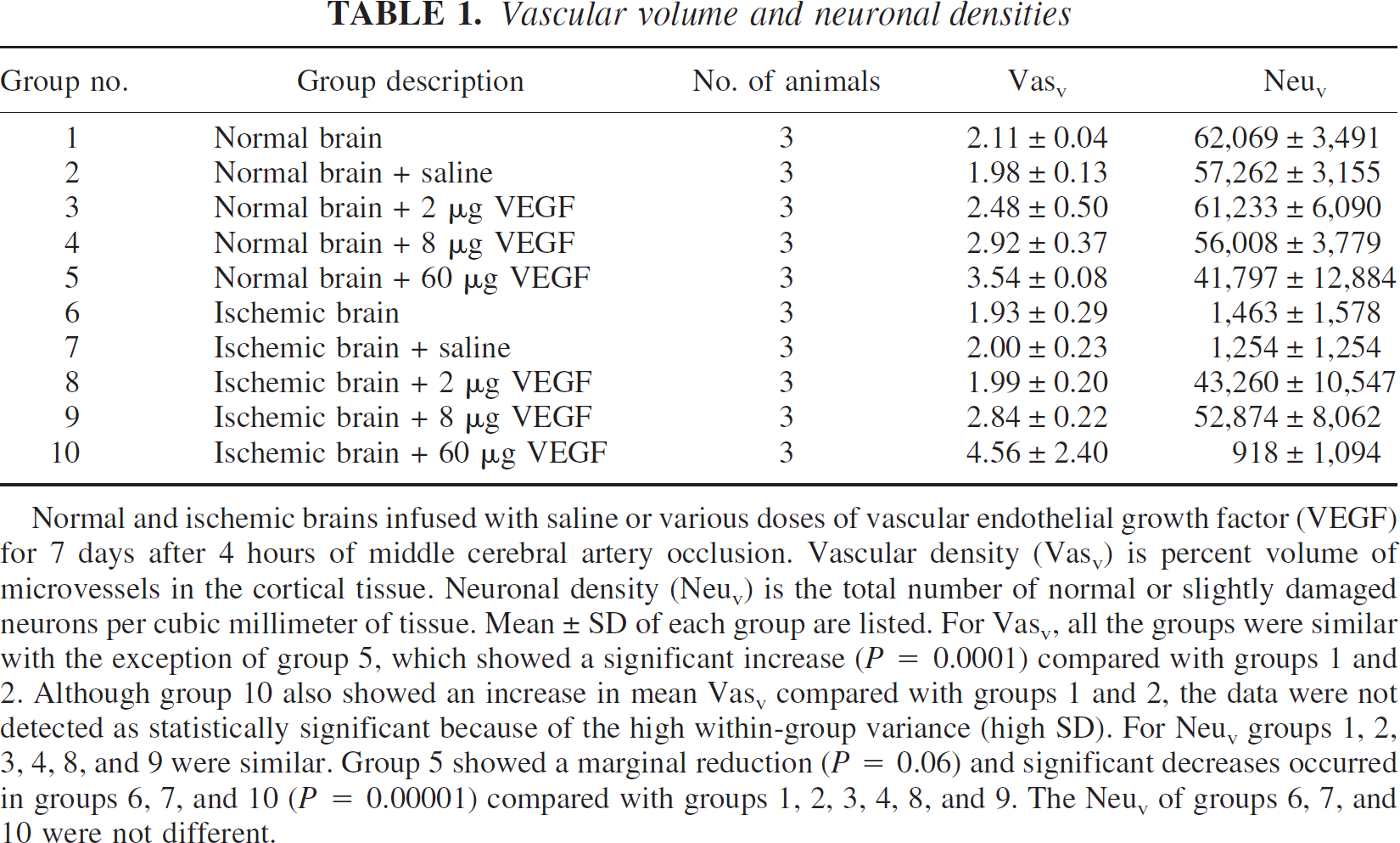
Normal and ischemic brains infused with saline or various doses of vascular endothelial growth factor (VEGF) for 7 days after 4 hours of middle cerebral artery occlusion. Vascular density (Vasv) is present volume of microvessels in the cortical tissue. Neuronal density (Neuv) is the total number of normal or slightly damaged neurons per cubic millimeter of tissue. Mean ± SD of each group are listed. For Vasv, all the groups were similar with the exception of group 5, which showed a significant increase (P = 0.0001) compared with groups 1 and 2. Although group 10 also showed an increase in mean Vasv compared with groups 1 and 2, the data were not detected as statistically significant because of the high within-group variance (high SD). For Neuv groups 1, 2, 3, 4, 8, and 9 were similar. Group 5 showed a marginal reduction (P = 0.06) and significant decreases occurred in groups 6, 7, and 10 (P = 0.00001) compared with groups 1, 2, 3, 4, 8, and 9. The Neuv of groups 6, 7, and 10 were not different.
Figure 2 shows the various morphologic manifestations of neurons as seen in the sections. Figure 2A shows the appearance of normal neurons whereas Figure 2B–D shows the various degenerating forms of the neurons, from slight damage (Fig. 2B) to more severe damage (Fig. 2C) to pyknosis (Fig. 2D). The neurons considered viable and counted in the study were neurons exhibiting morphology seen in Fig. 2A or Fig. 2B (normal to slight damage, respectively).
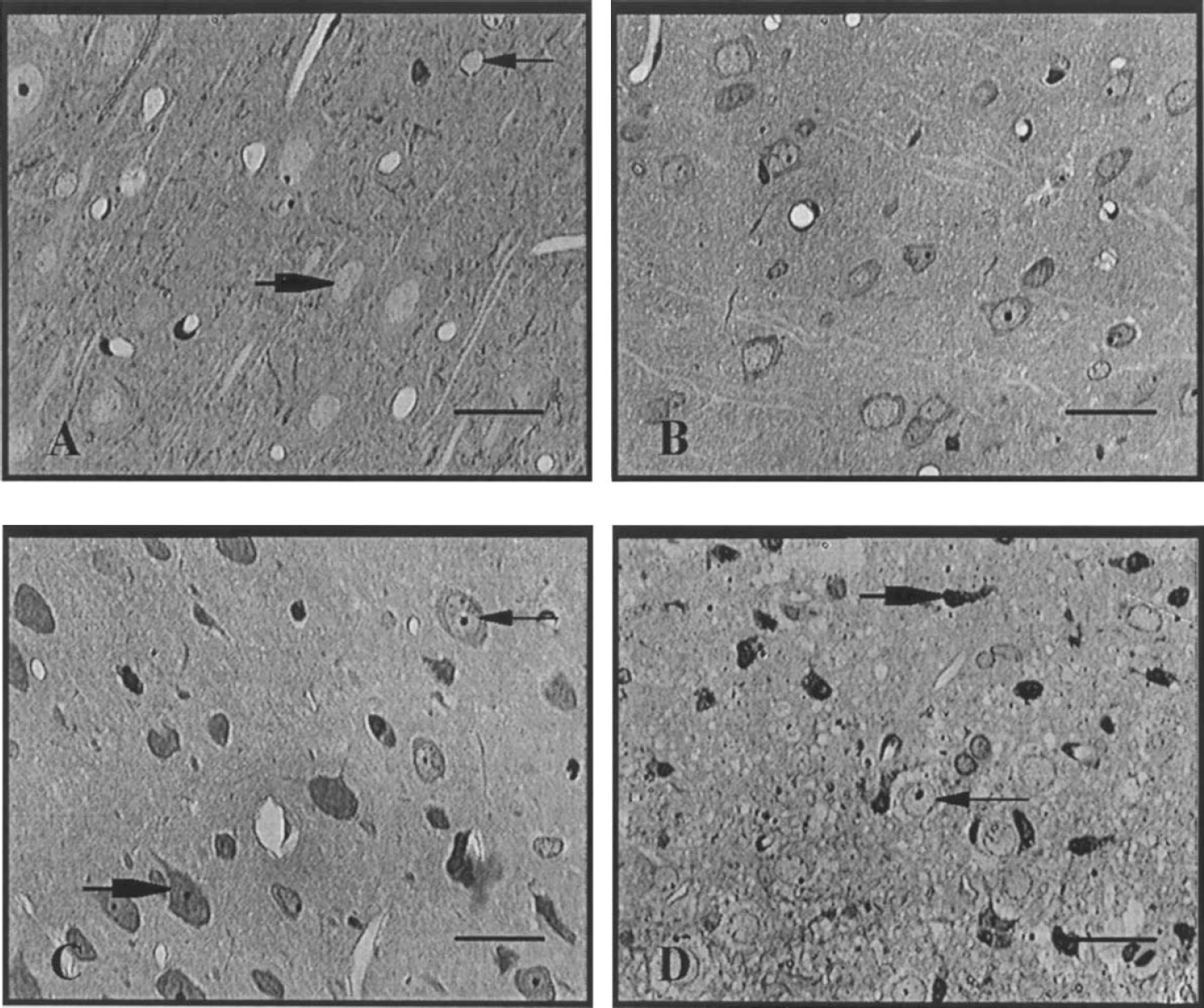
Morphology of normal and damaged neurons visualized in toluidine-blue stained, Epon-embedded, 1-μm semithin sections. (
DISCUSSION
Clinical implication of the study
The primary purpose of our study was to determine if in vivo neuroprotection and angiogenesis by exogenous VEGF might be unrelated. Given the well-established capacity of VEGF to cause vascular permeability, we sought to elucidate if neuroprotection by VEGF would occur whether angiogenesis is absent or present. To achieve this objective in a simple manner, we focused only on those doses that would and would not induce angiogenesis at a single time point after ischemia (i.e., after 7 days of VEGF infusion). The low dose was arbitrarily defined as the dose where no changes in VasV occurred; the intermediate dose was defined as the dose where VasV may occur; the high dose would be the dose where a significant increase in VasV would occur. Changes in NeuV were then examined in the brain tissue near the blood vessels evaluated.
At the doses and VEGF infusion duration (7 days) used in our study, the data suggest the following relationships between vascular density (VasV) and neuronal density (NeuV) of ischemic and normal cortical brains after VEGF165 arterial infusion:
NeuV values of ischemic brains were close to that of normal brains with the low and intermediate VEGF doses, where no changes in VasV were present. Thus, VEGF appears to possess a remarkable neuroprotective effect even at very low doses and in the absence of angiogenesis and after a prolonged period of complete ischemia (4 hours) The changes in NeuV were critically dependent on the dosage of VEGF. Most of the neuroprotection was achieved with the lowest dose. The nonangiogenic intermediate dose did not significantly promote neuroprotection over the low dose NeuV of ischemic brains infused with the high dose of VEGF (group 10) was similar to that of untreated ischemic brains or ischemic brains infused by saline. Increase in VasV also occurred with the high dose. These data suggest that neuroprotection and angiogenesis may not necessarily be concurrent or associated events. Rather, at the doses and duration of VEGF infusion used in our study, neuroprotection and angiogenesis may be unrelated The high dose of VEGF (group 5) applied to normal brain also showed a significant increase in VasV. Furthermore, the NeuV was slightly reduced. The low and intermediate doses of VEGF did not alter the VasV or NeuV in normal brains. These data suggest that a high level of induced angiogenesis may be associated with damage of neurons (though reexamination of the data in additional experiments with larger groups of animals needs to be pursued).
The clinical implication of our preliminary findings is that, until the mechanisms of neuroprotection in ischemic brain by exogenous VEGF are better understood, efforts to concurrently induce neovascularization and neuroprotection through VEGF monotherapy as a method for stroke treatment should be approached with caution. Until more information about the relationship between in vivo angiogenesis and neuroprotection by exogenous VEGF is available, the possibility that induced angiogenesis may be associated with reduction or loss of neuroprotection should not be ignored. Our preliminary data could serve as a vital foundation for further studies in this crucial stroke therapy research arena.
The data obtained from this study are critical in light of the fact that that several other investigators have previously called attention to the potential risk of VEGF-induced immature vessels, vascular permeability, and vasodilation (Bates et al., 2002; Dobrogowska et al., 1998; Laham et al., 2003; Roberts and Palade, 1995; Schoch et al., 2002; Thurston et al., 2000; Zhang et al., 2000; Zhao et al., 1998). These events can lead to severe cerebro-vascular pathology (Pettersson et al., 2000; Schoch et al., 2002; Sun et al., 2003; Thurston et al., 2000; van Bruggen et al., 1999; Zhang et al., 2002). Thus, it is possible that neuroprotection by VEGF may be comprised by angiogenesis and the two manifestations of VEGF activity may not be related in certain conditions.
Limitations of the study
We caution that the findings presented here are preliminary, with only low, intermediate, and high doses of VEGF examined. Also, the effects of these three doses on NeuV and VasV were studied only at a single time point (after 7 days of VEGF infusion) and at the single delivery rate of 1 μL/h. It is, therefore, possible that at other postischemic intervals or infusion rates, a given dose of VEGF might be both neuroprotective and angiogenic. That is, it is conceivable that the low or intermediate doses of VEGF that promoted neuroprotection without inducing angiogenesis, as seen in our study, could concurrently promote neuroprotection and angiogenesis if the VEGF infusion was allowed to progress beyond the 7 days or the infusion rate was altered. If induction of both neuroprotection and angiogenesis is to be a goal in the treatment of stroke, then there is no doubt that elucidating the relationship between VasV and NeuV under broader ranges of VEGF doses, infusion intervals, or infusion rates would be necessary.
In our experimental procedures, the common carotid artery had to be occluded for 10 minutes during implantation of the osmotic pump. This is to avoid hemorrhage when the tip of the pump is positioned in the external carotid artery. The brief blockage of the common carotid artery poses no problems in interpreting data of the 4-hour ischemic brains (Groups 7–10). However, our “normal brains” treated with saline or VEGF (Groups 2–5), in actuality, were 10-minute ischemic cortices. Nonetheless, since the VasV of Groups 1–4 is similar, it appears changes in VasV seen in Group 5 were due to the high dose of VEGF.
Relationship of neuroprotection and angiogenesis
It is unclear why the increase in VasV by exogenous VEGF, as observed in our study, had an inverse relationship with NeuV. Given the morphologic and morphometric approaches of our study, our experimental design provides no physiologic evidence to account for how angiogenesis could adversely influence neuroprotection. Nonetheless, it is possible that the reduction in NeuV could be due to the angiogenic vessels being immature or permeable, given that VEGF is a potent vascular permeability factor (Dobrogowska et al., 1998; Dvorak et al., 1995; Lopez et al., 1997; Proescholdt et al., 1999; Sellke et al., 1996; Sun et al., 2003; Zhang and Chopp, 2002). Furthermore, ultrastructural evaluation of the blood-brain barrier after VEGF treatment revealed that the vasculature was leaking blood proteins and albumin into the extracellular compartment of the brain (Dobrogowska et al., 1998). The endothelial cells showed interendothelial gaps, fragmentation of the endothelium with formation of segmental, fenestraelike narrowings, degenerative changes of the vascular basement membrane, and the appearance of fibrin gel in the vessel lumen. These changes, which were interpreted to be preparatory steps for angiogenesis, affected the structural integrity of the vascular segments (Dobrogowska et al., 1998). In light of the amount of data reported by several investigators regarding the permeability effect of VEGF (see Introduction), we hypothesize that the lack of neuroprotection seen with angiogenesis was due in major part to formation of leaky vessels.
VEGF combinational therapy for inducing both angiogenesis and neuroprotection
A method of stroke therapy that attempts to preserve neurons while concurrently increasing blood flow to the ischemic brain is, no doubt, an approach that deserves attention. Newly formed vessels could increase the amount of oxygen delivered to the ischemic tissue. However, if reduction in neuroprotection is associated with vascular leakiness, clinical intervention for stroke would need to minimize the vascular permeability. Thus, if both neuroprotection and angiogenesis by VEGF are to be simultaneously achieved in stroke patients, experiments targeted at elucidating the usefulness of combinational therapy rather than monotherapy need to be explored. With combinational therapy where other neurotrophic factors and agents effective in reducing vessel permeability are included with the VEGF, the neuroprotective effects of VEGF could be enhanced while the permeability of the angiogenic vessels could be minimized. For example, it is known that SSeCKS and angiopoietin-1 regulate endothelial tight junction formation and angiogenesis. These substrates reduce endothelial permeability (Lee et al., 2003; Thurston et al., 2000). Combinational therapy of VEGF with these agents could prove effective in concurrently promoting neuroprotection and proliferation of nonpermeable blood vessels.
Technical considerations
Unfolding method for measuring NeuV.
We chose the unfolding method for our evaluation of NeuV. The unfolding method is a less labor-intensive stereologic approach for counting particles in space than the traditional disector or other stereologic methods (Calverley et al., 1988; Miki et al., 1997) and is applicable to various regions of the brain (Raadsheer et al., 1994; Warren and Bedi, 1984). Though the strength of the traditional disector method is that the approach makes no assumptions about the objects of interest and is more accurate than the unfolding method, the requirements of the disector method cannot always be met for practical or technical reasons (Miki et al., 1997).
The unfolding method, on the other hand, rests on simplifying the assumptions regarding the shape of the particles to be counted (Cruz-Orive and Weibel, 1990). Each neuronal nucleus is assumed to represent a neuron and the neuronal nuclei are assumed to be spheres (Miki et al., 1997). Since cortical neurons of rats possess nuclei diameter of approximately 6 to 12 μm (Fukui et al., 1992; Miki et al., 1995, 1997), this size minimizes the effects of section thickness when using semithin sections of no greater than 1 μm. Our NeuV data of normal cortex are similar to the reports of other investigators who used the unfolding method for measuring cortical NeuV of adult rats (Miki et al., 1995, 1997).
To determine NeuV using the unfolding method, the following assumptions about the neurons were made: (1) the neurons were randomly oriented in space; (2) each neuron contained only one nucleus; (3) the nucleus was a spherical profile; and (4) the diameter of the nucleus was uniform. These reasons further account for why we had to exclude neurons with condensed nuclei (pyknotic neurons) from the study. Severely injured or pyknotic neurons, as commonly observed in ischemic brains, possess nuclei diameters smaller than normal range. Because NeuV calculated from the unfolding method assumes that the nuclei of all neurons are uniform, it would be difficult to compare the NeuV of ischemic brains to the NeuV of normal brains if the NeuV of ischemic brains included damaged (pyknotic) neurons.
As a method for evaluating neuroprotection, the stereologic unfolding technique was also preferable over measuring the total size or volume of infarction. No doubt, measurement of total infarct volume or size could serve as a quantitative approach for determining neuroprotection. We have, in our past works, used changes in infarct volumes as an indicator of neuroprotection (Lyden et al., 1997, 2000). The method is also widely used in stroke research for determining levels of neuroprotection.
However, the purpose of our work was to elucidate VasV and NeuV changes within the core area of the infarcted zone. Had we used total infarct volume as indicator of neuroprotection, we would inherently have to address the role of the penumbra to the reduction of the infarct size. This is because reduction of infarct size, by itself, does not necessarily indicate preservation of neurons in the core of the infarct. Instead, it could be argued that the reduction in infarct volume was due to increased neuroprotection in the penumbra of the ischemic zone, thus leading to reduction of the size of the infarct. This is a limitation of using total infarct volume as a measure of neuroprotection. Events occurring in the penumbra of the ischemic zone could play a great role in reduction of the infarct volume though the neurons deeper within the core of the infarct are killed or damaged. In contrast, the strength of the unfolding method is that the approach targets changes in neuronal density near the measured blood vessels within the core of the infarct itself, regardless of whether neuroprotection is occurring in the penumbra of the ischemic zone.
Arterial infusion as method for VEGF administration.
In developing methods for treating stroke, the use of clinically relevant experimental models is important. To enhance the practicality of the intervention procedure, the manner in which VEGF is administered to the brain should be compatible with current acute treatments for stroke. The animal studies reported in the literature have used several techniques for administering VEGF to the brain (Bao et al., 1999; Cheng et al., 1997; Dellian et al., 1996; Harrigan et al., 2003; Hayashi et al., 1998; Krum et al., 2002; Proescholdt et al., 1999; Rosenstein et al., 1998; Sun et al., 2003; van Bruggen et al., 1999). However, for our study, we chose the arterial infusion method over the intravenous, interstitial, intraventricular, topical, or cortical penetration approaches. VEGF was infused directly into the internal carotid artery rather than intravenously or intraperitoneally to better enable administration of VEGF to the brain without loss to the systemic tissues. The intraventricular, topical, and cortical penetration methods were avoided since the techniques are unnecessarily invasive because the skull would have to undergo some procedure to expose the brain. In addition, the intraventricular model was not used because VEGF receptors have been reported to be located primarily on the abluminal surface of the endothelial cells (Qu-Hong et al., 1995; Zhao et al., 1998). These studies showed that VEGF applied to the abluminal surface of the endothelial cells caused increased permeability much more rapidly than when VEGF was added to the luminal surface. Thus, it is possible that the level of permeability, or even angiogenesis, may vary markedly when VEGF is infused through the vasculature (as we did in our study) in comparison to applying VEGF to the brain from the ventricles (the intraventricular approach). With our arterial method, VEGF would presumably be traversing from the luminal to the abluminal surfaces of the endothelial cells to reach the brain. In contrast, with the intraventricular approach, VEGF would have a more direct access to the abluminal surface of the endothelial cells.
Footnotes
Acknowledgements
The authors thank Aloysius Kowalewski, biomedical technician in the Neural Engineering Program of the Huntington Medical Research Institutes (HMRI), for assistance with ![]() of the manuscript. The review of the manuscript by Dr. Albert S. Lossinsky, experimental neuropathologist of the HMRI, is appreciated.
of the manuscript. The review of the manuscript by Dr. Albert S. Lossinsky, experimental neuropathologist of the HMRI, is appreciated.


