Abstract
Background
Methods
Results
Conclusions
Introduction
Lung cancer is one of the malignant tumors with the highest incidence in humans and the leading cause of cancer-related death in the Western world (1). Non-small cell lung cancer (NSCLC) represents more than 80% of newly diagnosed lung cancer with different histological subtypes (2). The most common subtypes are squamous cell carcinoma, adenocarcinoma and large cell carcinoma. They are grouped together because of similar methods used for staging, prognosis and treatment (1). The standard treatment in advanced NSCLC is platinum-based, double-agent chemotherapy with similar efficacy and different toxicity profiles (3). The determination of histological subtypes in NSCLC has become the cornerstone for the choice of treatment (4, 5). The treatment of lung cancer depends on its stage at the time of diagnosis, and surgery may be indicated in stage I, II and selected cases of stage III. Otherwise, chemotherapy or targeted agents are the treatment of choice.
Angiogenesis is an important factor in tumor growth and dissemination (6). Progress in understanding the cancer biology and mechanisms of oncogenesis has led to the detection of specific molecular targets, such as epidermal growth factor receptor (EGFR), echinoderm microtubule-associated protein-like 4-anaplastic lymphoma kinase fusions (EML4-ALK), vascular endothelial growth factor (VEGF) and its receptor (VEGFR) (7, 8). When a tumor reaches 1-2 mm3 in size, tumor cell nutrition by diffusion becomes insufficient. So the expression of proangiogenic proteins should be upregulated in order to induce sprouting of pre-existing capillaries, which results in the formation of new vessels through endothelial cell migration and proliferation (9). VEGF is a key molecule in the upregulation of tumor angiogenesis (10). In addition, antiangiogenic agents have been demonstrated to be active in various malignant tumors including locally advanced and metastatic NSCLC (11). Therefore, an increase in progression-free survival (PFS) and overall survival (OS) time has been achieved by these targeted drugs when combined with chemotherapy in non-squamous non-small cell subtypes (12). However, most NSCLC patients died within 2 years after the diagnosis despite these new agents (13). Therefore, there is a need for biomarkers that allow identifying patients who are likely to respond to treatment.
Circulating endothelial cells (CECs) have been found to be an indicator of vascular damage with clinical relevance in cancer (14, 15). They are mature differentiated endothelial cells defined as CD146+ CD105+ CD45–. Several techniques have been described to count CECs. The optimal method for the quantification of CECs has yet to be determined. Immunomagnetic separation (IMS) using monoclonal antibodies and flow cytometry (FC), which takes into account multiparametric characterization of CECs, have been widely used for the quantification of these cells (16). Normal CEC counts derived by FC measurement were highly heterogeneous, ranging from 10 to 7900 cells/mL (17, 18). By contrast, CEC counts obtained by IMS were within the reasonable range of 1-100 cells/mL in normal individuals (19). In addition, IMS has been proposed as a preferable method for CEC quantification in a recent consensus guideline (20). However, FC is still the method of choice for the determination of required cell characteristics (e.g. viability and activation) (21).
Elevated levels of CECs in peripheral blood were observed in cancer patients compared with healthy controls (22, 23). The increased CEC count in cancer patients was found to be nearly normalized after tumor resection or chemotherapy (24). Therefore, CECs might reflect the extent of tumor angiogenesis. However, a change in CEC count depends on the response to antiangiogenic agents and on tumor type (25). High CEC levels have been correlated with tumor progression in breast cancer patients receiving metronomic chemotherapy (26). In addition, increased viable CECs were observed in cancer patients with progressive disease when compared to patients with stable disease or healthy volunteers (27). In the present study, we investigated the clinical significance of CECs in newly diagnosed NSCLC patients. In addition, we tried to determine the predictive value of CECs in NSCLC patients with clinical benefit after treatment.
Materials and Methods
Between March 2011 and September 2013, 151 newly diagnosed patients with histologically confirmed NSCLC and 25 healthy subjects were included in this study. All patients had been referred to Albairouni University Hospital in Damascus, Syria. After the approval of the hospital ethics committee, all patients and controls provided written informed consent. The nature of the study was explained in the informed consent form. This study was performed in accordance with the guidelines prescribed by the Helsinki Declaration of the World Medical Association.
The following criteria were required for newly diagnosed patients:
Age >18 years and histologically confirmed NSCLC with adequate renal, hepatic and bone marrow function
No concomitant disease such as ischemic heart disease, diabetes or uncontrolled hypertension
No prior treatment including chemotherapy, irradiation or fluid drainage.
The main clinical characteristics of the included patients are illustrated in Table I. The following 3 degrees of weight loss were considered: none, less and more than 10% of body weight during the last 3 months. According to body mass index (BMI) values, there were 4 groups of patients (underweight, healthy, overweight and obese). The degree of smoking was evaluated by number of packets per year (P/Y), resulting in 3 groups of patients (less than 30, 31-60 and more than 60 P/Y). Our population was classified according to histological grade into patients with well or moderately differentiated tumors (grade 1 and 2) and those with poorly differentiated tumors (grade 3). Staging was based on the TNM system and determined by chest and abdominal computed tomography (CT), resulting in patients with early stages (I-IIIA) and those with advanced stages (IIIB, IV). The major axis (a) and the minor axis (b) of the primary tumor were measured by a senior radiologist. Estimated tumor volume (ETV) was calculated using the following formula:
CLINICAL CHARACTERISTICS OF NEWLY DIAGNOSED NSCLC PATIENTS AND CONTROLS
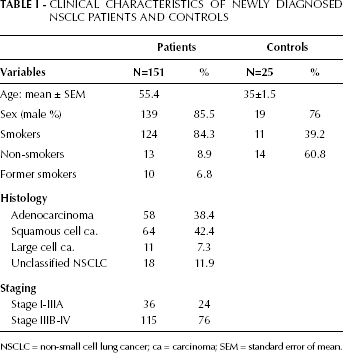
NSCLC = non-small cell lung cancer; ca = carcinoma; SEM = standard error of mean.
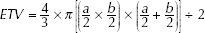
Twenty-five patients presenting with advanced-stage disease and showing clinical benefit after treatment were included for the evaluation of recurrence. They had been treated by double-agent platinum-based chemotherapy combined with radiotherapy if this was necessary according to tumor stage. Recurrence evaluation was based on the appearance of radiological signs of progression on CT during the follow-up period after treatment. Time to progression (TTP) and PFS duration were calculated in days for each patient. The change in CEC level (∆aCECs) at recurrence compared with the level after treatment was calculated as relative absolute quotient. It was considered significant when its value was ≥0.1. The main clinical and pathological findings are illustrated in Table II.
CLINICAL CHARACTERISTICS OF NSCLC PATIENTS AT RECURRENCE
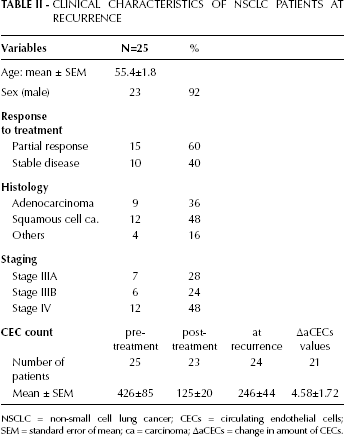
NSCLC = non-small cell lung cancer; CECs = circulating endothelial cells; SEM = standard error of mean; ca = carcinoma; ∆aCECs = change in amount of CECs.
Isolation and Quantification of CECs in the Peripheral Blood
Peripheral blood samples were collected in 3 mL vacuum tubes containing liquid tri-potassium EDTA (1 mg/mL) as an anticoagulant. The first 3-5 mL were discarded to prevent contamination with endothelial cells traumatically released from the punctured vessel wall. Samples were kept at 4-8°C and processed within 24 hours of collection. CECs were isolated from whole blood of NSCLC patients and counted using the previously validated IMS technique (28). After careful rotation of the tube, 1 mL of blood was diluted 1:3 with isolation buffer (PBS-BSA 0.1% and NaN3 0.1%) and incubated for 45 minutes with 15 µL (7×107 beads) of anti-CD146 coupled magnetic beads (Dynal M450 IgG1, Dynal AS, Oslo, Norway) at 4°C with gentle agitation in a head-over-head mixer. The unbound cells were washed out with buffer while bound cells were retained on the magnet. After additional wash cycles, the cells were incubated in 100 µL of acridine orange (5 µg/mL PBS, Sigma) for 10 minutes at 4-6°C in the dark. Isolated CECs were counted with a Nageotte hemocytometer (Hecht-Assistent, Sondheim, Germany) using a fluorescent microscope (Olympus, BX52, Japan) equipped with a 500/20 nm excitation filter. Isolated CECs were determined as CD146 rosetted cells bearing ≥5 beads and with a distinctive cell morphology, 15-50 µm diameter and distinct fluorescence due to expression of their specific endothelial marker (28, 29) (Fig. 1).

Circulating endothelial cells (CECs) under fluorescent microscopy. CECs were determined as CD146 fluorescent green cells due to their specific endothelial marker.
Statistical Analysis
All data were analyzed by means of the SPSS software, version 15 for Windows (SPSS Inc., Chicago, IL, USA). The Kruskal-Wallis test for all parameters was used to compare patient groups. In addition, the Mann-Whitney U test was applied for pairwise comparison. Bivariate correlation was performed using Spearman's correlation analysis. The correlation between CEC count and PFS was assessed by means of the Kaplan-Meier method. Survival curves of subgroups were compared using the log-rank test. A statistical difference was considered significant with a p value <0.05.
Results
A total of 151 patients with histologically confirmed NSCLC (36 at early stages, 115 at advanced stages) and 25 healthy volunteers were included in this prospective project. There was a significant difference in mean age between patients (55.4±8.68 years) and healthy controls (36±8 years) (p<0.0001). However, no significant change in CEC count was observed with increasing age (r=0.022, p=0.78). In addition, 25 patients with a partial response (PR, n=15) or stable disease (SD, n=10) were studied at recurrence with a mean follow-up of 117 days (range: 47-364) after treatment.
Quantification of CECs at Baseline
The median CEC count in NSCLC patients was 276 cells/mL (range: 36-1868), whereas the count in healthy controls ranged from 0 to 18 cells/mL (median: 5). A slightly increased CEC count at baseline (≤80 cells/mL) was observed in 24 patients, while a moderate increase (81-150 cells/mL) was found in 45 patients. High levels of CECs (151-300 and >300 cells/mL) were obtained in 34 and 43 patients, respectively. Significant differences in pre-treatment CEC counts were found between all histological NSCLC subtypes and healthy volunteers (p≤0.0001, Fig. 2). There was no significant correlation between baseline CEC count and smoking degrees (p=0.99). Furthermore, the baseline CEC count was not significantly correlated with ETV in the 101 assessable patients (p=0.96, Fig. 3). Although there was no significant correlation between CEC count and BMI (p=0.31), an increased amount of CECs at baseline was significantly correlated with more than 10% weight loss (p=0.03). There was also a significant difference in baseline CEC count between patients with advanced stages and those with early stages (p=0.026, Fig. 4). In addition, pre-treatment CEC levels were significantly higher in patients with poorly differentiated than those with well differentiated subtypes (p=0.02, Fig. 5). The clinical and pathological variables correlated with baseline CEC counts are summarized in Table III.
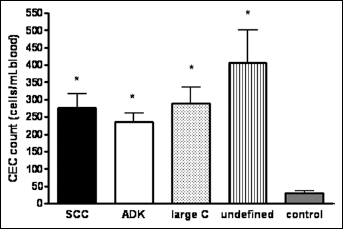
Comparison between pre-treatment values of circulating endothelial cells (CECs) in patients with NSCLC and healthy volunteers. Significant differences in CEC counts were found between all histological subtypes of NSCLC (squamous cell carcinoma [SCC]: n=64, adenocarcinoma [ADK]: n=58, large cell carcinoma: n=11, unclassified NSCLC: n=18) and healthy controls (n=25). Results are expressed as mean ± SD; * means p≤0.0001).
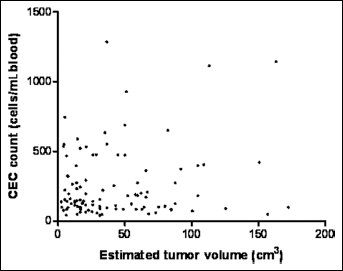
Scatter plot analysis to determine the correlation between circulating endothelial cells (CECs) at baseline and estimated tumor volume. Data analysis showed that there was no significant correlation between baseline CEC count and ETV (p=0.96).
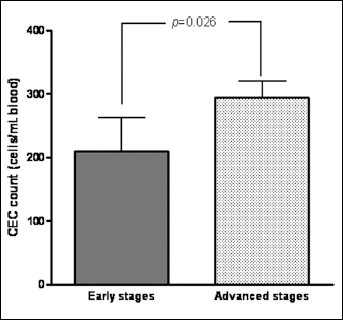
Pre-treatment values of circulating endothelial cells (CECs) according to clinical stage. There was a significant difference (p=0.026) in the baseline CEC amount between NSCLC patients with early stages (n=36) and those with advanced stages of disease (n=115).
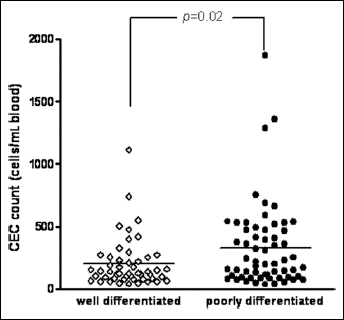
Relationship between circulating endothelial cells (CECs) count at baseline and histological grade in NSCLC patients. Pre-treatment CEC levels significantly increased in patients with poorly differentiated grade (n=61) compared with patients with well-differentiated grade (n=50) (p=0.02).
CLINICAL AND PATHOLOGICAL VARIABLES CORRELATED WITH CEC COUNT IN NEWLY DIAGNOSED NSCLC PATIENTS AND HEALTHY CONTROLS
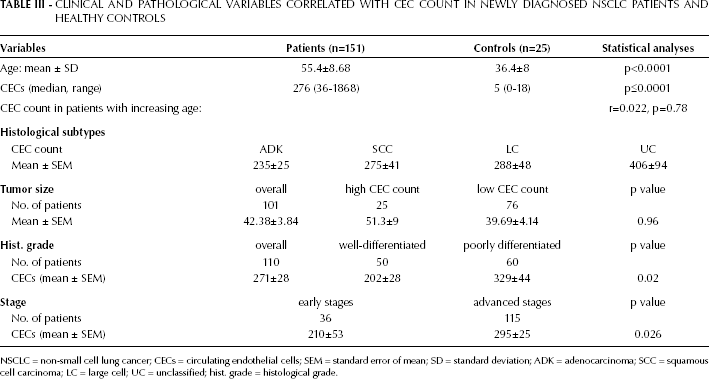
NSCLC = non-small cell lung cancer; CECs = circulating endothelial cells; SEM = standard error of mean; SD = standard deviation; ADK = adenocarcinoma; SCC = squamous cell carcinoma; LC = large cell; UC = unclassified; hist. grade = histological grade.
Quantification of CECs at Recurrence
In comparison with their levels after treatment, CECs increased significantly at recurrence in 20 of 21 assessable patients (95% sensitivity, p<0.0001, Fig. 6). However, no significant correlation was observed between ∆aCECs and TTP (p=0.55). In addition, there was no significant difference in the median PFS duration between patients whose baseline CEC count was less than 120 cells/mL and those whose baseline CEC count exceeded 120 cells/mL (75th percentile distribution value) (p=0.402, Fig. 7).
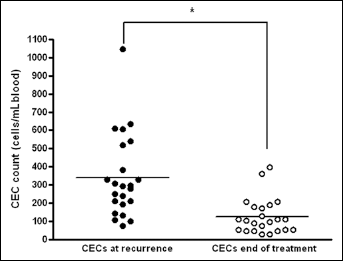
Comparison between the values of circulating endothelial cells (CECs) after treatment and at recurrence in NSCLC patients with clinical benefit after treatment. CEC levels at recurrence were significantly higher than those after treatment (p<0.0001).
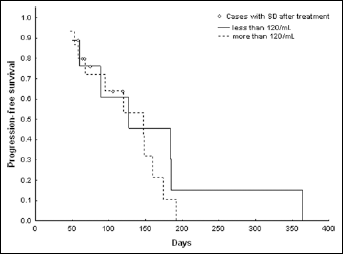
Correlation between pre-treatment circulating endothelial cells (CECs) and progression-free survival (PFS) in NSCLC patients with partial response (n=15) or stable disease (n=10) after treatment. The difference in median PFS duration was not significant (p=0.45) between patients whose CEC count was <120 cells/mL and patients whose CEC count exceeded 120 cells/mL (corresponding to the 75th percentile distribution value).
Discussion
An increased number of CECs has been previously described in multiple solid tumors, reflecting the perturbation of the vascular endothelium in malignancies (22, 24, 29). This increase in CEC count ranges from 1 to 10,000 cells per milliliter in the peripheral blood of cancer patients. As the patients were evaluated before treatment, these CECs may be mostly derived from the tumor site. The clinical significance of CECs in cancer is still poorly understood. Few papers are available in the literature regarding the clinical value of CECs in NSCLC. Kawaishi et al (30) observed an increased number of pre-treatment CECs compared with healthy individuals in only 31 NSCLC patients (30). Similar findings have been demonstrated in 151 newly diagnosed NSCLC patients in our study. No significant difference was observed in our patients in relation to age, smoking and BMI. So the increase in CEC counts seems likely to be due to the presence of the tumor. In vivo, the amount of CECs correlated strongly with tumor volume in an animal model (31). However, no significant correlation was observed between ETV and increased CEC count in a small number of NSCLC patients (30). Similar results were obtained in 101 assessable patients with NSCLC in our study. This is probably due to many factors related to tumor vasculature, neovascularization and tumor location (22). No significant difference in the amount of activated CECs has been observed between squamous and adenocarcinoma NSCLC subtypes (32). We found no significant difference in baseline CEC counts among all histological subtypes in NSCLC. These findings could be explained by the fact that factors other than primary tumor type determine the CEC count in cancer patients (27).
The clinical significance of CECs in cancer is still poorly understood (22). In the present study, increased pre-treatment CEC count was significantly correlated with weight loss of more than 10%. In addition, baseline CEC levels were significantly higher in patients with poorly differentiated NSCLC and patients with advanced stages. These findings support that NSCLC patients with high-risk factors have an increased CEC count at baseline. However, increased amounts of CECs were not found in all these patients. This is probably due to the many factors that determine the presence of CECs in the peripheral blood, such as the growth speed of the tumor vasculature and the location and size of metastases (27).
There were no conclusive data concerning the prognostic value of CECs in cancer. Concerning the potential role of CECs in predicting the clinical outcome of cancer patients, a wide difference among tumor types was observed regarding the techniques used for the quantification of CECs. Using automated IMS, an association between high pre-treatment levels of CECs and longer PFS was reported in NSCLC patients (29). With the same technique, a recent study with 53 assessable NSCLC patients undergoing surgery found that patients with a high CEC count at baseline had a significantly worse PFS and OS (33). Using a manual IMS technique, we demonstrated that baseline CEC count did not predict PFS in 25 NSCLC patients with clinical benefit after treatment. This discordance could be explained by the relatively small group of patients presenting with advanced stages in our study. On the other hand, a high CEC count at baseline was associated with a shorter PFS in a small number of patients with advanced colorectal cancer (ACC) in a study where 4-color FC was used for counting CECs (34). By contrast, a large study using an automated IMS technique found that baseline CEC count was not significantly correlated with median PFS in a subset of ACC patients (35). These controversial findings may be due to the difference in vascular turnover according to tumor origin, but could also result from differences in CEC counting methods and phenotypes (22).
An increased number of CECs in cancer patients has been previously reported when progression occurred (27). Furthermore, the amount of CECs significantly increased when the tumor progressed during or after various chemotherapy regimens (36). We found a significant change in CEC count at recurrence, which reflects progressive disease in NSCLC patients with a clinical response after treatment. However, no significant correlation between ∆aCECs and PFS was found in our small group of patients treated with double-agent platinum-based chemotherapy. Similar findings were obtained using FC in patients treated with navelbine and platinum (37). In this study, an inverse correlation between ∆aCECs and median PFS duration in patients treated with navelbine and platinum combined with rh-endostatin was observed. These findings suggest that CEC count could be a prognostic biomarker in NSCLC patients receiving antiangiogenic treatment.
In conclusion, the current study provides new data on the clinical significance of CECs in newly diagnosed NSCLC patients. Our findings demonstrate that high levels of CECs at baseline might be an indicator of more aggressive disease in NSCLC. In addition, an increased amount of CECs at recurrence seems to be a predictive biomarker in NSCLC patients with PR or SD after treatment. Further prospective studies are warranted to determine whether CECs may serve as a prognostic biomarker in NSCLC.
Footnotes
List of Abbreviations
Acknowledgment
The first author would like to thank Prof. I. Othman, Director General of AECS, for his cooperation especially at the beginning of this project. We also thank Mrs Nissren Almalla and Mr Moaz Altahan for technical assistance.
