Abstract
BACKGROUND:
Circulating endothelial cells (CECs) and microparticles (MPs) are proposed as useful biosensors for angiogenesis and membrane damage in cancer.
OBJECTIVE:
We investigated their predictive value for progression disease (PD) and clinical outcomes in advanced non-small cell lung cancer (NSCLC) patients treated with cytotoxic chemotherapy.
METHODS:
Peripheral blood samples were obtained from 60 patients. Immunomagnetic separation (IMS) and flow cytometry techniques were used to quantify CECs and MPs, respectively. Receiver operating characteristics (ROC) analysis was used to determine the optimal cutoff values for CECs and MPs counts according to their levels in patients with an objective response (OR) and non-responders after treatment. Baseline serum biomarkers levels and their kinetics after chemotherapy were correlated with tumor response and outcomes in advanced NSCLC patients.
RESULTS:
Forty-seven patients presented an OR after chemotherapy. Of these, 28 patients progressed within three months. Through an increase in their levels during or after chemotherapy, CECs and MPs correctly predicted PD in 57% and 61% of these patients, respectively. Regarding tumor stage, NSCLC patients with stage IV had significantly higher pretreatment CECs and MPs levels than stage III patients (
CONCLUSIONS:
Our findings suggest the increase in CECs and MPs number during or after chemotherapy as predictive biomarkers of tumor progression in advanced NSCLC patients. An association of basal CECs and MPs values with tumor stage was also shown in advanced NSCLC patients. However, baseline CECs levels and their kinetics after chemotherapy seem to be prognostic factors in advanced NSCLC.
Keywords
Abbreviations
LC, Lung cancer;
CECs, Circulating endothelial cells;
MPs, Microparticles;
EPCs, Endothelial progenitor cells;
NSCLC, Non-small cell lung cancer;
VEGF, Vascular endothelial growth factor;
EGFR, Epidermal growth factor receptor;
ALK, Anaplastic lymphoma kinase;
PFS, Progression-free survival;
TTP, Time to progression;
OS, Overall survival;
PR, Partial response;
SD, Stable disease;
OR, Objective response;
PD, Progression disease;
IMS, Immuno-magnitic separation;
CT, Computed tomography;
MRI, Magnetic resonance imaging
TNM, Tumor-node-metastasis;
RECIST, Response evaluation criteria in solid
tumors;
SEM, Standard error of mean;
ROC, Receiver operating characteristic;
PS, Performance status.
Introduction
Lung cancer (LC) is the third most common malignancy but remains the leading cause of cancer mortality in both males and females in the USA and throughout the world. It has resulted in rising death rates, with approximate 1.4 million deaths per year all over the world [8, 16]. Although advanced molecular biology techniques have greatly accelerated the understanding of cancer biology, still approximately 50% of the patients with lung cancer have advanced stage disease at the time of diagnosis [27]. However, the treatment of NSCLC is still given priority to chemotherapy [2]. The majority of patients presented in advanced stages of the disease with five-year overall survival rate less than 20% [7]. Because of high drug resistance in advanced NSCLC patients, progression disease (PD) is frequent during or after chemotherapy [9]. Cancer progression depends on sustained neo-angiogenesis that guarantees an adequate supply of nutrients, oxygen, and growth factors, such as vascular endothelial growth factor (VEGF), resulting in reduced tumor vascularity, which is a primary contributor to therapeutic resistance in cancer. However, the unfavorable outcome is probably due to the relatively inadequate understanding of the molecular mechanisms involved in carcinogenesis [4]. Nevertheless, some chemotherapy regimens might have an antiangiogenic activity by itself, mediated in part by a direct effect on tumor vessel endothelium [20]. It should be noted that the addition of antiangiogenic agents to cytotoxic chemotherapy can block tumor angiogenesis and tumor growth by inhibiting proliferation of vascular endothelial cells [13]. However, just a subset of patients will derive benefit from such treatment. So the development of molecular and cellular biomarkers for early detection of PD seems to be very important in NSCLC setting for improving therapeutic outcome. These biomarkers can be applied simply and easily as useful complementary tools in clinical practice.
Circulating endothelial cells (CECs) consist of endothelial progenitor cells (EPCs) that originate in the bone marrow, mature endothelial cells, apoptotic endothelial cells and some cells with endothelial function from cancerous cells [28]. Increased CECs count was observed in multiple tumor types when compared with healthy subjects. Otherwise, it has been recognized that CECs number was a promising tool for monitoring disease activity and tumor response after treatment through tumor angiogenesis in cancer patients [5, 23, 33]. Moreover, cancer patients with PD displayed increased levels of CECs when compared to those with stable disease [3].
Microparticles (MPs) are plasma membrane fragments of small size (0.1–1)
Advanced NSCLC is mainly treated by conventional chemotherapy and/or concurrent administration of chemotherapy and radiation. However, failure often occurs owing to chemo-resistance of neoplasm cells. The possibility to inhibit MPs production by pharmacological interventions represents a future challenge. In vitro, an evidence exists that MPs may have a role in angiogenesis [19]. In vivo, it has been found that platelet-derived MPs could mediate PD in breast cancer [14].
The objective of this study was to investigate the potential value of CECs and MPs levels and their kinetics during or after cytotoxic chemotherapy for the prediction of PD and outcomes in advanced NSCLC patients.
Materials and methods
Patients
This prospective study was conducted at Albairouni University Hospital from May 2012 to April 2015. Previously untreated patients with advanced NSCLC confirmed by histology or cytology as NSCLC according to the new Union for International Cancer Control criteria (version 7) were included in this study. Each patient was required to meet the following criteria: age over 18 years, an eastern cooperative oncology group (ECOG) performance status (PS)
All patients were treated with standard cytotoxic chemotherapy in combination with radiotherapy when it was indicated in stage III patients. Fourteen patients with locally advanced disease received chemo-radiotherapy. The remaining 8 patients with stage III received neoadjuvant chemotherapy before possible thoractomy. However, palliative radiotherapy was administrated in 8 patients with metastatic disease. NSCLC cases were staged according to radiological and pathological findings based on the American Joint Committee on Cancer/Union International Contra la Cancrum (International Union Against Cancer) tumor (T), node (N) and metastasis (M) (UICC/TNM) system of classification [31]. It was determined by chest and abdominal computed tomography (CT), magnetic resonance imaging (MRI) and bone scintigraphy. Mediastinoscopy was also used in stage III patients with suspected lymph nodes, which may affect therapeutic management. However, stage IV patients were systemically evaluated with clinical and CT findings, MRI and bone scintigraphy. Blood samples were collected before treatment and 21 days after the last cytotoxic infusion or 6 weeks after thoracotomy in order to avoid direct effects of chemotherapy or surgical wound healing on CECs and MPs number. Tumor response was assessed by the response evaluation criteria in solid tumors (RECIST), in which PD defined as radiologically documented (more than 25% increase in tumor size comparing with primitive tumor) or as clear clinical evidence of PD [10].
Response evaluation and survival
An objective response (OR) was considered for patients who achieved a partial response (PR) or stable disease (SD) after chemotherapy, while the remaining patients with PD were considered as non-responders. The percentage change in circulating biomarker levels was considered for the evaluation of their kinetics during or after chemotherapy. In patients with OR after chemotherapy, time to progression (TTP) was considered as the time between the moment of tumor response and the date on which patients had a progression. Progression-free survival (PFS) is defined as the time from baseline blood sampling to date of confirmed PD or death from any cause. Otherwise, it was censored at last follow-up. Overall survival (OS) duration was measured from the beginning of chemotherapy to the date of death or the date censored at the last follow-up.
Blood sampling
Whole blood samples were collected from patients by vena-puncture. The first (3–5) ml were discarded to avoid contamination with endothelial cells from the puncture wound of the vein. Peripheral blood for CECs quantification was collected in a siliconized tube containing (1.8 mg/ml) of ethylene-diamine-tetra-acetic acid (EDTA) as anti-coagulant. For the determination of total MPs levels, blood was collected in a tube containing sodium citrate. The ratio of anticoagulant (129 mM sodium citrate) to blood was 1:9 (v/v). Pre-analytic conditions were followed to analyze MPs count using 2 serial centrifugations (1500
Quantification of CECs
The isolation and quantification of CECs was performed using immuno-magnetic separation (IMS) technique with fluorescent microscopy following a consensus protocol [34]. Briefly, Samples were kept at (4–6)
Quantification of total MPs
Plasma samples containing MPs suspensions were thawed at room temperature immediately before assay. MPs were quantified by FACS Calibur™ flow cytometer controlled by Cell Quest Pro 4.0.2 software (BD Bioscience, San Jose, CA, USA) at high flow rate. As previously described, Annexin V-FITC conjugates (BioVision, Milpitas, CA, USA) were used to detect accessible phosphatidylserine on MPs membrane [21]. In brief, 10
Statistical analysis
The results were expressed as mean values
Clinical characteristics of patients with advanced non-small cell lung cancer
Clinical characteristics of patients with advanced non-small cell lung cancer
Relationship between treatment response and standard clinicopathological parameters (gender, age, smoking status, histopathology, staging) in advanced NSCLC patients
NSCLC non-small cell lung cancer, OR objective response, PD progression disease, SCC squamous cell carcinoma.
Patient characteristics
Sixty patients were prospectively enrolled in this study. As summarized in Table 1, there were 50 males and 10 females with a mean age of 54.72 (range 36–71) years. The histological and/or cytological diagnosis was adenocarcinoma in 23 (38.4%) patients, squamous-cell carcinoma in 29 (48.3%) patients, and unclassified NSCLC in 8 (13.3%) patients. On diagnosis, 38 (63.3%) patients were at stage IV and 22 (36.7%) patients were at stage III. A total of 48 (80%) patients were treated with standard chemotherapy. The therapeutic regimens included cisplatin in combination with vinorelbine, gemcitabine or docetaxel in 24 patients (40%), 8 patients (13.3%) and 3 patients (5%), respectively. By contrast, carboplatin was administrated in combination with vinorelbine, gemcitabine and docetaxel in 10 (16.7%), 2 (3.3%) and 1 (1.7%) patients, respectively. The remaining 12 (20%) patients were treated with docetaxel, vinorelbine or gemcitabine in 8, 2 and 2 patients, respectively. In addition, 14 patients with locally advanced disease received chemo-radiotherapy. The remaining 8 patients with stage III received neoadjuvant chemotherapy before possible thoractomy. However, palliative radiotherapy was administrated in 8 patients with metastatic disease.
Receiver operating characteristic (ROC) curves for the determination of optimal cutoff values for percentage changes in circulating endothelial cells (a) and microparticles (b) count after chemotherapy.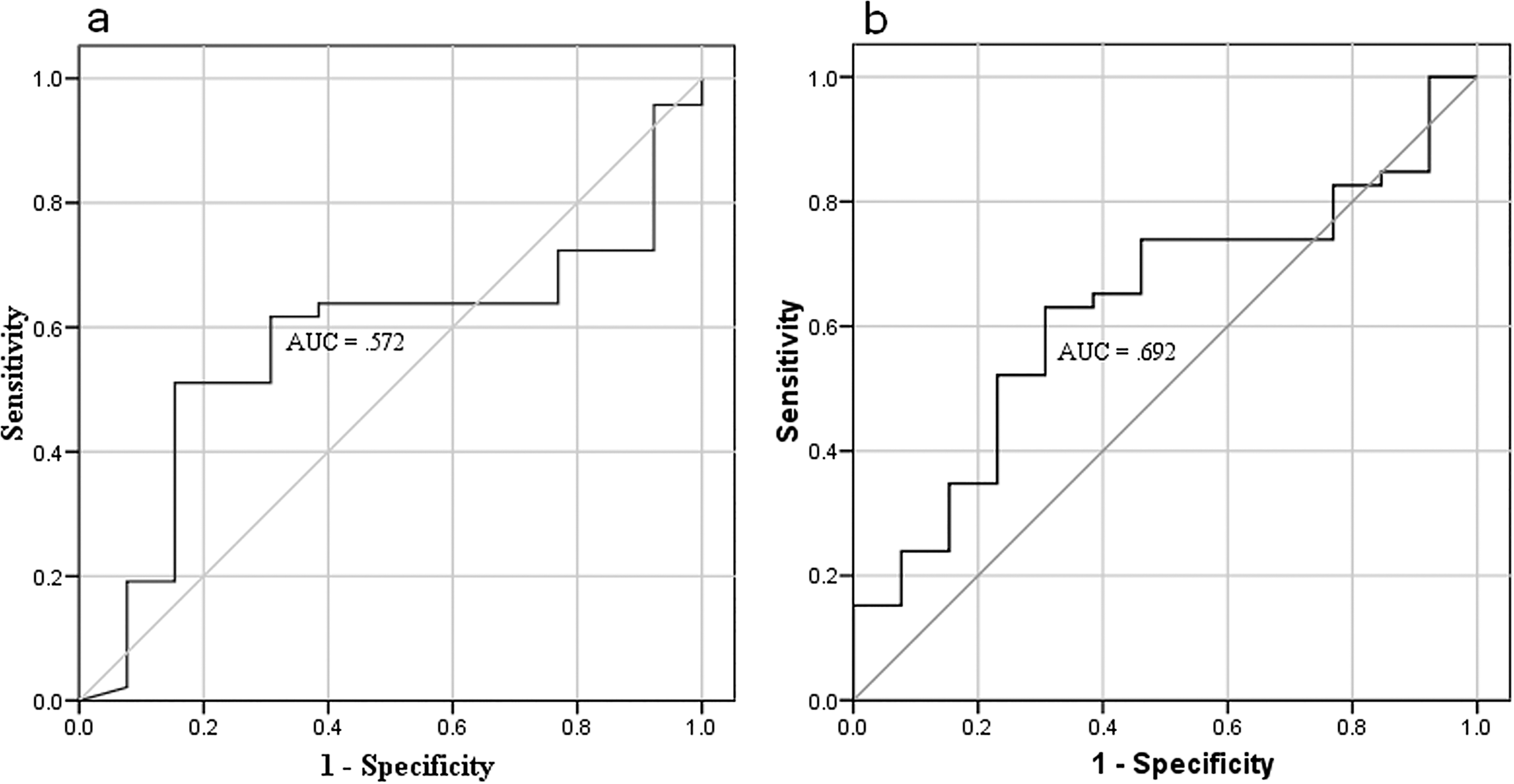
Kaplan-Meier plots showing time to progression (TTP) according to the percentage (%) changes in circulating endothelial cells (a) and microparticles (b) levels after chemotherapy.
Box plots for baseline circulating endothelial cells (a) and microparticles (b) values in patients with an objective response (OR) and those with progression disease (PD) after chemotherapy. Top and bottom of box represent 75
Box plots for pretreatment circulating endothelial cells (a) and microparticles (b) levels in stage III patients and those with stage IV. Significant differences have been found between NSCLC patients of stage III and those of stage IV for baseline CECs (
Receiver operating characteristic (ROC) curves for the determination of optimal cutoff values for baseline circulating endothelial cells (a) and microparticles (b) levels to discriminate patients with objective response (OR) from those with progression disease (PD) after chemotherapy.
Kaplan-Meier plots showing progression-free survival (PFS) according to measured baseline circulating endothelial cells (a) and microparticles (b) levels.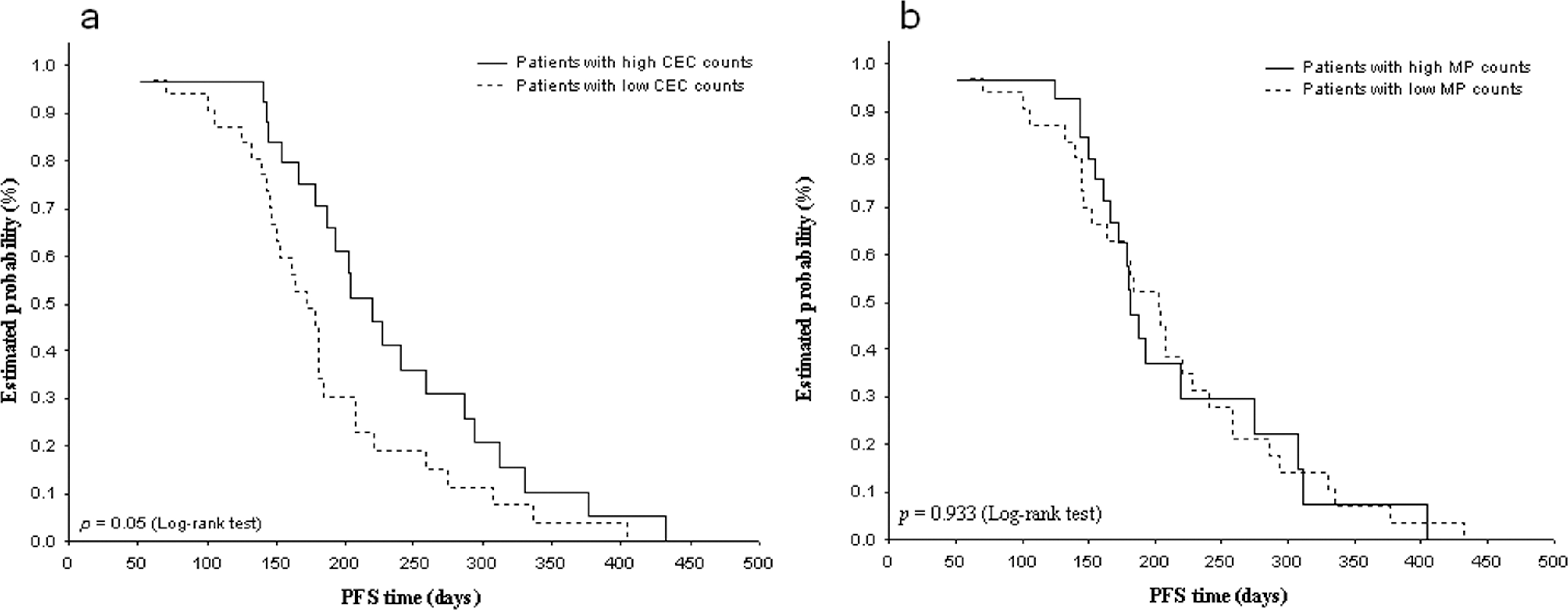
Kaplan-Meier plots showing overall survival (OS) according to basal circulating endothelial cells (a) and microparticles (b) values.
After chemotherapy, 47 (78.3%) patients achieved radiological OR, while 13 (21.7%) patients had experienced tumor progression. The association between treatment response and standard clinicopathological parameters was shown in Table 2. The tumor response was independent of age, gender, smoking status, histological sub-types and clinical stage (all
After chemotherapy, CECs and MPs count was available in 60 and 56 patients, respectively. Forty-seven patients showed an OR at the time of analysis. Of these, 28 (59.6%) progressed within three months. Confirmed radiological PD was correctly predicted by CECs and MPs through a significant increase in their levels after chemotherapy in 16/28 (57%) and 17/28 (61%) patients, respectively. Overall, the percentage change in CECs and MPs count predicted the response to treatment in 39 (65%) and 29 (51.8%) patients, respectively.
Using ROC analysis, the best efficacy for the percentage change in CECs after treatment was observed at 79.6% with a sensitivity of 70.8% and a specificity of 50% (Fig. 1a). Whereas the optimal percentage change in MPs number was 57.95% with a sensitivity of 54.2% and a specificity of 55.6% (Fig. 1b). Consequently, there were 37 and 25 patients with high percentage change in CECs and MPs, respectively. The median TTP duration was significantly longer in patients with high percentage change in CECs levels than that in patients with low percentage change after chemotherapy (
Baseline levels of circulating markers and clinical outcome
There was no significant difference in baseline CECs levels between patients with OR and those with PD after chemotherapy (median
Discussion
Locally advanced and metastatic NSCLC is mainly treated with chemotherapy. However, drug resistance often occurs resulting in high rate of PD in NSCLC. Actually, no biomarkers with high sensitivity and specificity for early detection of tumor progression in NSCLC have yet been identified. These potential biomarkers might avoid toxicities and reorient toward another treatment approach in patients with NSCLC.
It has been recently observed that the change in CECs levels was predictor of tumor response in patients with advanced non-squamous NSCLC treated with platinum and pemetrexed [32]. Wang et al. also found that CECs number increased when PD occurred in advanced NSCLC patients treated with antiangiogenic therapy [35]. It has been also revealed that CECs increased when diseases progressed both in NSCLC patients treated with chemotherapy alone or in combination with Endostar [22]. Moreover, previous studies suggest that an increase in circulating level of MPs may be associated with the activity of disease entity including lung cancer [24, 34]. To the best of our knowledge, this is the first study investigating whether increased levels of circulating CECs and MPs during or after chemotherapy could predict PD in cancer patients with clinical benefit after chemotherapy. Interestingly, we found that an increase in CECs and MPs amount during or after chemotherapy predicted PD in 57% and 61% patients with advanced NSCLC, respectively. These findings suggest these biomarkers as predictive tools of tumor progression in advanced NSCLC. However, not all progressed patients showed this increase in their numbers during or after chemotherapy. This may reflect that not all progressive cancer patients have an increased number of CECs or MPs in their peripheral blood. The speed of tumor growth, the number of actual metastases and the time of the residence of these biomarkers in the circulation may influence their presence in the peripheral blood of cancer patients including those with advanced NSCLC.
Our study implies that neither pretreatment CECs and MPs count nor clinicopathological parameters were predictive factors for treatment response in advanced NSCLC. However, it has been recently concluded that CEC count itself could not predict the response to chemotherapy with and without antiangiogenic agent in a homogenous group of patients with advanced non-squamous NSCLC treated with platinum and permetrexed [32]. Tseng et al. [34] found that baseline MPs levels were significantly less associated with squamous cell carcinoma subtype, which represents the majority of our population (48.3%). They also demonstrated that no type of circulating MPs was found to be predictive of diseases status in LC patients. These findings were in accordance with our data and may partially explain why baseline CECs and MPs levels were not significantly higher in patients with OR than those in patients with PD. By contrast, it has been previously demonstrated that baseline CECs number was significantly higher in PR patients than that in patients with SD/PD after chemotherapy [17, 25]. However, patients with PR and SD after chemotherapy were grouped together in our study because they were all without signs of clinical or radiological progression and they continued to receive the same chemotherapy regimen. On the other hand, it has been also shown that increased baseline CECs count between NSCLC patients in advanced stages compared with those in early stages [26]. Similar results was previously observed for VEGF levels in NSCLC patients [28]. Interestingly, we found that CECs and MPs values were higher in NSCLC patients with metastatic disease than those in patients with locally advanced disease. Therefore, the measurement of these biomarkers might aid in assessing the stage of the disease not only between early and advanced stage but also between locally advanced and metastatic NSCLC. This finding might be because of increased tumor size, but it could also be related to the tumor vasculature, the location and size of metastases [3].
With regard to long-term survival after chemotherapy, we found that patients with high baseline CECs levels but not MPs have significantly longer PFS duration. Accordingly, an association between increased baseline CECs levels and good prognosis was observed in patients with NSCLC [25]. By contrast, it has been previously shown that there was no significant correlation between CECs expression at baseline and PFS duration in advanced NSCLC [12, 32]. Nevertheless, high pretreatment CECs levels above 75
Regarding the variation in serum biomarkers for the prediction of prognostic outcome, Yuan et al. found that CECs reduction might be an estimation tool for treatment response in advanced NSCLC patients receiving platinum-based chemotherapy alone or in combination with endostatin [39]. They also demonstrated that CECs response could predict the efficacy of chemotherapy especially for patients treated in combination with endostatin. Furthermore, a longer TTP duration was observed in advanced NSCLC patients with a significant decrease in CECs count after angiogenesis therapy [35]. Although no antiangiogenic agents have been used in our population, we found that CECs variation was predictive of longer TTP in advanced NSCLC patients. Another essential finding of our study that the change in CECs and MPs levels after chemotherapy predicted treatment response in advanced NSCLC patients especially in those who progressed during or after chemotherapy. However, the percentage change in MPs levels was not correlated with TTP in our population. Could these results indicate that the emission of MPs has an important pathological role in tumor progression facilitating the release and the spread of cancer cells to generate metastases [1]?
This study has some limitations. First, the sample size was relatively small with a rather wide range of standard deviation of CECs and MPs values. However, the current study showed very attractive and promising results for the use of these biomarkers in NSCLC. Second, the predictive value of circulating MPs on long-term prognostic outcome was not determined in our population.
Conclusion
Our findings encourage the use of CECs and MPs as complementary biomarkers for early detection of tumor progression in advanced NSCLC patients treated with cytotoxic chemotherapy. Moreover, our study suggests that increased pretreatment CECs and MPs number was associated with metastatic disease in advanced NSCLC. Baseline CECs levels and their kinetics after chemotherapy might be also predictive of longer PFS and TTP durations in advanced NSCLC patients. Further prospective studies with a large cohort of patients are warranted to investigate the impact of MPs for the prediction of prognostic outcome in NSCLC.
Conflict of interest
None of the authors have any financial or personal relationships with people or organizations that could inappropriately influence this work.
Footnotes
Acknowledgments
The first author would like to thank Pr. I. Othman, the general director of AECS for his efforts in advancing the work in this project. Special thanks to Ms. Issraa Banat for technical assistance and Mr. Ali Mohammad for help in some statistical analyses.
