Abstract
Background:
This study was to investigate the influence of GSTP1 gene polymorphism on the clinical outcomes of patients with advanced non-small-cell lung cancer (NSCLC) receiving first-line bevacizumab plus chemotherapy regimen.
Methods:
A total of 128 patients with advanced NSCLC who were administered with bevacizumab-based first-line regimens were recruited in this study. Available blood specimen and peripheral blood mononuclear cells (PBMCs) of the patients were obtained for the analysis of polymorphism and GSTP1 gene mRNA expression, respectively. The association between genotype status and clinical outcomes and other variates was analyzed and presented.
Results:
The prevalence of rs1695 were in accordance with Hardy-Weinberg Equilibrium (P = .978). Patients with GG and AG genotypes were merged in a pattern of dominant inheritance to seek for the potentially clinical significance. Analysis of efficacy exhibited that the objective response rate (ORR) of patients with AA genotype and AG/GG genotypes were 62.1% (54/87) and 51.2% (21/41) (P = 0.245). Prognosis demonstrated that the median progression-free survival (PFS) of patients with AA genotype and AG/GG genotypes were 9.5 and 5.6 months, respectively (P = .007). Furthermore, the median overall survival (OS) of the two genotypes were 22.0 and 16.6 months, respectively (P = .003). In addition, adjusted in multivariate Cox analysis for OS, AG/GG genotype was an independent factor for OS. Interestingly, mRNA analysis suggested that the mRNA expression of GSTP1 in PBMC of the patients with AG/GG genotypes of rs1695 polymorphism was significantly higher than those of patients with AA genotype (P < .001).
Conclusion:
GSTP1 polymorphism rs1695 could be used for the prognostic evaluation of patients with advanced NSCLC receiving bevacizumab combined chemotherapy regimen.
Introduction
Lung cancer is reported to be the most common solid tumor with the highest morbidity and mortality all over the world. As the most common histological category, non-small cell lung cancer (NSCLC) accounts for approximately 85% in lung cancer. 1 Most patients were diagnosed with locally advanced or metastatic disease in China. 2 Recent years had witnessed remarkable achievements regarding the therapy of programmed death 1 (PD-1)/programmed death ligand-1 (PD-L1) inhibitors and targeted drugs for estimated glomerular filtration rate (EGFR), ALK, and ROS1, thus contributing to the significant survival benefits for the patients dramatically. 3 However, approximately 50% of patients with advanced NSCLC were still absent of useful driver targets and became a research hotspot currently. 4
Inhibition of angiogenesis could be used as a highly potent anticancer therapeutic approach and intensified the research for agents to hamper vessel formation in numerous cancers recently. 5 Consequently, the antiangiogenic targeted drug bevacizumab combined with chemotherapy in the first-line setting for patients with advanced NSCLC exhibited superior progression-free survival (PFS) and improved OS.6,7 However, the objective response rate (ORR) of the first-line platinum-based chemotherapy regimens for advanced NSCLC was less than 30%, 8 addition of bevacizumab could improve the ORR to approximately 50%. 9 Still and all, great individual difference was observed clinically. 10 And potential biomarkers that might predict the clinical outcomes of bevacizumab in patients with advanced NSCLC were needed currently.
Glutathione S-transferases (GSTs) family was composed of eight isoforms that were reported to detoxify the xenobiotic substrates by catalyzing their conjugation to reduced glutathione. Among them, glutathione S-transferase P 1 (GSTP1) was the most widely studied isoform in various disease. 11 Glutathione S-transferase P 1 is located at 11q13.2, contains 7 exons and is of 3.2 kb in length with the ability to protect cells from carcinogens and cytotoxins. Discrepancy regarding the mRNA expression of GSTP1 gene was observed among different populations. 12 Previous study demonstrated that GSTP1 gene played an important role in biodetoxification or biotransformation by binding to reduced glutathione. 13 As one of the phase-Ⅱ metabolizing enzymes, GSTP1 was reported to catalyze detoxifying endogenous reactions with GSH and protect cellular macromolecules from damage caused by a wide variety of endogenous and exogenous molecules, including chemotherapeutic agents. 14 Therefore, GSTP1 were crucial for the cell defense system. And these phase-Ⅱ detoxification enzymes were involved in the detoxification of a variety of chemotherapeutics including platinum and exogenous molecules such as bevacizumab. In vitro analysis revealed a significant association between high GSTP1 expression of tumor cells and decreased sensitivity to platinum agents. 15 And glutathione-conjugated exogenous molecules might be quickly excreted from tumor cell. Thus, it was plausible that high GST activity might result in more rapid metabolism that diminished the cytotoxic effects of chemotherapy on tumor cell. 16 Interestingly, a recent study investigated the correlation between genetic variations of GSTP1 and clinical outcome of platinum-based chemotherapy in advanced NSCLC and concluded that rs1695 polymorphisms might contribute to the platinum-induced clinical outcomes in terms of both toxicity and therapeutic response variations. 17 Furthermore, a previous meta-analysis study included a total of 23 published studies had indicated that GG genotype of GSTP1 IIe105Val polymorphism predicted superior therapeutic response to cisplatin-based chemotherapy among patients with NSCLC. 18 Collectively, the above researches suggested that GSTP1 polymorphism could be of potentially clinical significance to predict the efficacy of chemotherapy for patients with NSCLC clinically.
Therefore, the aim of this study was to investigate the implication of GSTP1 polymorphism on the clinical outcomes of patients with advanced NSCLC receiving bevacizumab-based regimens in first-line setting.
Patients and Methods
Study design and patient enrollment
Bevacizumab was approved in China more than 10 years and covered in the national medical insurance directory recently. Consequently, a large number of patients with advanced or recurrent NSCLC were able to receive bevacizumab plus chemotherapy regimen clinically. In addition, correlation analysis between GSTP1 polymorphism and clinical outcome of the bevacizumab-based regimen was also performed in this study, which indicated that biological specimens had to be collected from the patients to identify the genotyping results of the polymorphism accordingly. Therefore, this study was designed as a real-world retrospective study just as the previous polymorphism study regarding bevacizumab-based therapy did. 19
Patients with advanced or recurrent NSCLC who were administered with bevacizumab-based regimens as first-line therapy from January 2016 to October 2020 in the department of thoracic surgery of Shanxi Cancer Hospital were participated in this study. This study was undertaken in accordance with good clinical practice guidelines and the protocol was approved by the ethics committees of Shanxi Cancer Hospital (approved number: KY2020136). All the patients enrolled were provided with the informed consent in accordance with the recommendations of the Declaration of Helsinki.
The main inclusion criteria and exclusion criteria were adopted according to the previous retrospective study initiated by N Geng and colleagues. 20 Flow chart in Figure 1 illustrated the procedure in this study specifically. And 128 patients with advanced or recurrent NSCLC was included this study. The primary analysis of this study was to explore the association between GSTP1 polymorphism genotype status and clinical outcomes of patients receiving bevacizumab combined with chemotherapy regimen.

Flow chart of the retrospective study for influence of GSTP1 polymorphism on the efficacy and safety of patients with advanced non-small-cell lung cancer receiving bevacizumab plus chemotherapy regimens.
Treatment regimens and assessment of efficacy
All the patients included were administered with bevacizumab combined with platinum-based chemotherapy regimen in first-line setting. Chemotherapy regimens consisted of pemetrexed or paclitaxel or docetaxel in combination with platinum drugs (carboplatin or cisplatin). And the specific usage and dosage of chemotherapy and bevacizumab was similar with the previous study. 20
Tumor response among the 128 patients was performed using RECIST version 1.1 criteria by investigator assessment. 21 The drug response regarding the target lesions was assessed based on computed tomography (CT) scans every two cycles or depended on the actual situation when the clinical symptom of patients was getting worse. And the combination regimens could be terminated when potential life-threatening toxic reactions were observed.
Distinguish of genotyping for GSTP1 rs1695 polymorphism
Genomic DNA was extracted from the available biological specimens (mainly peripheral blood) using phenol-chloroform method and then stored at −20°C for subsequent usage. The polymorphisms of GSTP1 gene were selected according to the previous reports that the polymorphism of GSTP1 was of clinical significance, 22 which were rs4891, rs749174, and rs1695. However, it seemed that only rs1695 was significantly associated with PFS in the preliminary analysis. As a result, this study implemented the analysis between rs1695 and clinical outcomes accordingly. Rs1695 was genotyped using restriction fragment length polymorphisms polymerase chain reaction (PCR-RFLP). The size of PCR product that included rs1695 was 286 bp. Forward primer was 5’-CCCCAGTGACTGTGTGTTGA-3,’ the reverse primer was 5’-GCACCCTGACCCAAGAAG-3.’ A total of 2 μL PCR products were digested using the restriction enzyme BsmBI (Thermo Fisher Scientific, USA). Using the following band to distinguish the genotypes: AA genotype (one 286 bp band); AG genotype (three bands: one 286 bp band, one 150 bp band and one 136 bp band); GG genotype (two bands: one 150 bp band and one 136 bp band).
Collection of peripheral blood mononuclear cell specimen and GSTP1 gene mRNA expression analysis
Given that GSTP1 gene was related to the pharmacokinetics of chemotherapeutic drugs rather than the gene of tumor predisposition and development, we chose peripheral blood mononuclear cell (PBMC) specimen instead of tumor tissue to investigate the GSTP1 gene mRNA expression. Therefore, the matched PBMC specimens were selected from 86 samples originally. Nevertheless, as presented in Figure 1, extraction of PBMC specimens from 11 patients was not successful and 7 patients failed the RNA extraction. Therefore, a total of 68 specimens were available for GSTP1 mRNA expression analysis. The procedure of RNA extraction and mRNA expression experiment was performed according to the previous study. 20 Forward primer of GSTP1 gene was 5’-GTAGTTTGCCCAAGGTCAAG-3,’ the reverse primer was 5’-AGCCACCTGAGGGGTAAG-3.’ In addition, GSTP1 mRNA expression status was divided into GSTP1 high expression (GSTP1H) and GSTP1 low expression (GSTP1L) according to the median GSTP1 mRNA relative expression to identify the potential association with OS.
Statistical analysis
SPSS software version 25.0 was used to performed all the data analysis in this study. Chi-square test and Mann–Whitney U test nonparametric for the respective variables according to rs1695 genotype status was used appropriately. Definition of efficacy endpoints regarding ORR and disease control rate (DCR) was adopted according to the previous study. 23 Stata 14.0 was used to present the Kaplan–Meier survival curves for PFS and OS according to the genotype status of rs1695. Difference between the survival curve was calculated using the log-rank test. Definition of prognostic endpoints regarding PFS and OS was used on the basis of previous work. 20 Cox regression analysis was adopted for OS for multivariable analysis. P < .05 was accepted as statistical difference.
Results
Baseline characteristics
Baseline characteristics of the 128 patients with NSCLC enrolled were exhibited in Table 1. Patients included were the common NSCLC patients clinically. And it should be noted that majority of initial dosage of bevacizumab was 7.5 mg/kg (93.8%).
Baseline characteristics of the 128 patients with NSCLC according to GSTP1 rs1695 genotype status.

Abbreviations: ECOG, Eastern Cooperative Oncology Group; GSTP1, glutathione S-transferase P 1; NA, not available; NSCLC, non-small cell lung cancer.
The driver gene were EGFR mutation or ALK fusion.
Genotyping results of rs1695 polymorphism
rs1695 polymorphism was of clinical significance in the association analysis. The prevalence of rs1695 among the 128 patients with NSCLC were: AA genotype 87 cases (68.0%), AG genotype 37 cases (28.9%), GG genotype 4 cases (3.1%), minor allele frequency of rs1695 is 0.18. The distribution of three genotypes were in accordance with Hardy–Weinberg equilibrium (P = .978). Given that patients with GG genotype was rare, patients with GG and AG genotypes were merged in a pattern of dominant inheritance to seek for the potentially clinical significance. As illustrated in Table 1, the baseline characteristics of patients with AA and AG/GG genotypes were similar and comparable (P > .05).
Influence of GSTP1 rs1695 polymorphism on the efficacy and prognosis of the patients
Given that this study was designed as a retrospective study, the imaging evidence regarding the target lesions of the patients was collected and analyzed retrospectively. The best drug reaction assessment indicated that complete response (CR) was observed in 0 patients, partial response (PR) was noted in 75 patients, stable disease (SD) was seen in 42 patients, and progressive disease (PD) was reported in 11 patients. Consequently, ORR and DCR of the 128 patients with NSCLC was 58.6% (75/128) (95% confidence interval [CI]: 49.6%-67.2%] and 91.4% [117/128] [95% CI: 85.1%-95.6%], respectively. As illustrated in Figure 2, the ORR of patients with AG/GG genotype and AA genotype were 51.2% [95% CI: 35.1%-67.1%] and 62.1% [95% CI: 51.0%-72.3%], respectively [P = .245]. The DCR of patients with AG/GG genotype and AA genotype were 85.4% [95% CI: 70.8%-94.4] and 94.3% [95% CI: 87.1%-98.1%], respectively [P = .094].
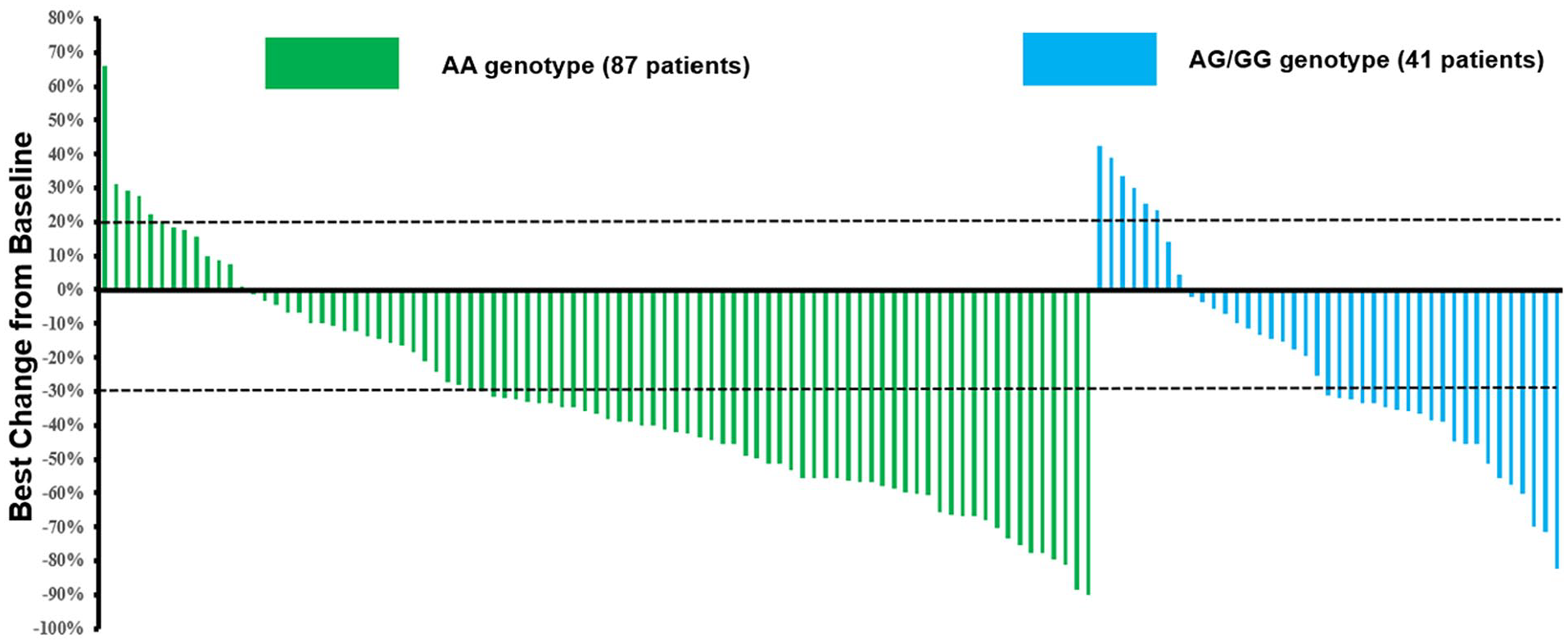
Waterfall plot for the best percentage change in target lesion size of the 128 patients with advanced non-small-cell lung cancer according to GSTP1 rs1695 genotype status.
Data cut-off date of this study was February 2021, and the median follow-up duration of the patients was 18.5 months (follow-up range: 0.5-36 months). Regarding PFS data, a total of 109 patients were observed the PFS events or death events when the data were cutoff. Therefore, the PFS data maturity was 85.2%. As illustrated in Figure 3, the median PFS of the 128 patients was 8.2 months (95% CI: 6.70-9.70). In addition, majority patients were available for the death status. And a total of 84 patients were noted the death events when the data were cutoff, and the OS data maturity was 65.6%. As exhibited in Figure 3, the median OS of the 128 patients with advanced NSCLC was 19.9 months (95% CI: 18.30-21.50).

Progression-free survival and overall survival of the 128 patients with advanced non-small-cell lung cancer.
Association analysis between rs1695 polymorphism status and prognosis was exhibited in Figure 4A, the median PFS of patients with AA and AG/GG genotype was 9.5 months (95% CI: 7.99-11.01) and 5.6 months (95% CI: 3.35-7.85), respectively, which was statistically significant (χ2 = 7.237, P = .007). Furthermore, as exhibited in Figure 4B, the median OS of patients with AA and AG/GG genotype was 22.0 months (95% CI: 19.74-24.26) and 16.6 months (95% CI: 10.92-22.28), respectively. And the difference was statistically significant (χ2 = 8.763, P = .003). Separately, median OS of patients with AA, AG, and GG genotype was 22.0 (95% CI: 19.74-24.26), 16.6 (95% CI: 9.95-23.25), and 10.0 (95% CI: 0.00-22.74) months, respectively. And the difference was statistically significant as well (χ2 = 10.47, P = .005).

Progression-free survival (A) and overall survival (B) of the 128 patients with advanced non-small cell lung cancer according to GSTP1 rs1695 genotype status.
Multivariate Cox regression analysis was introduced to for OS. Univariate analysis between baseline characteristics and OS was performed first. As illustrated in Table 2, age, Eastern Cooperative Oncology Group (ECOG) score and genotype of rs1695 were of positive correlation with OS in the univariate analysis. Therefore, these variables were included in the multivariate Cox regression analysis accordingly. Multivariate analysis results were presented in Table 2, rs1695 polymorphism status was an independent factor for OS (hazard ratio [HR] = 0.76 [95%CI: 0.55-0.85], P = .008) after multivariate adjustment. Besides, other statistically significant variables in the multivariate Cox analysis were age (HR = 0.82, P = .011) and ECOG score (HR = 0.73, P = .009).
Overall survival of the 128 patients with NSCLC according to baseline characteristics and polymorphism in univariate analysis and multivariate Cox analysis.

Abbreviations: CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; NSCLC, non-small cell lung cancer; OS, overall survival.
In addition, we further reanalyzed the therapeutic data regarding the second- and third-line therapy regimens among the available patients. A total of 75 patients (58.6%) had received second-line therapeutic regimens and a total of 49 patients (38.3%) had received third-line treatment. The most common second-line therapy was chemotherapy alone regimen and the most common third-line therapeutic treatment was anlotinib regimen. And the association analysis between GSTP1 rs1695 genotype status and therapeutic regimens failed to find significant difference (P > .05).
Association between rs1695 genotype status and mRNA expression of GSTP1
Expression of GSTP1 mRNA was determined by extracting RNA from 68 PBMC specimens, and the correlation between the genotype status of rs1695 and mRNA expression was analyzed. First, the prevalence of rs1695 polymorphism in the 68 PBMC specimens were AA genotype 46 cases (67.6%), AG genotype 19 cases (27.9%), and GG genotype 3 cases (4.4%). The MAF was 0.18 and distribution frequency of the three genotypes also accorded with the Hardy–Weinberg equilibrium (P = .570) and were comparable with that among the 128 patients. Similarly, AG and GG genotypes were merged in the subsequent analysis. As illustrated in Figure 5, the relative expression of GSTP1 mRNA in PBMC of patients with AG/GG genotype was significantly higher than that of patients with AA genotype of rs1695 polymorphism (3.96 ± 0.511 vs 2.81 ± 0.820), which was statistically significant (t = 6.033, P < .001).

Relative mRNA expression level of GSTP1 gene according to GSTP1 rs1695 genotype status.
Furthermore, patients with GSTP1H and GSTP1L were observed in 35 cases and 33 cases, respectively. As exhibited in Figure 6, the median OS of patients with GSTP1H and patients with GSTP1L was 18.0 months and 20.0 months, respectively. And the difference had a trend for marginally significant (χ2 = 3.445, P = .063).

Overall survival of the 68 patients with advanced non-small-cell lung cancer receiving bevacizumab plus chemotherapy regimens according to GSTP1 mRNA expression status.
Discussion
To our knowledge, target of bevacizumab was angiogenic signal pathway instead of tumor-driven signal pathways, which was a host-mediated process based on an individual’s unique inherited genomic profile. Consequently, it was necessary to use the germline DNA to identify potential biomarkers of bevacizumab-based regimen. 24
Furthermore, the heterogeneous nature of NSCLC had witnessed considerable discrepancy regarding the clinical outcomes of NSCLC therapy clinically. 25 Bevacizumab was proved to be of potential clinical activity for patients with NSCLC according to ECOG 4599 and AVAIL clinical trials.7,26 In addition, genetic variation of vascular endothelial growth factor (VEGF) pathway was proved to play an important role to predict the efficacy of antiangiogenic targeted drugs. 27 Fortunately, we found that Maria Gaibar and colleagues explored in 2021 that polymorphisms of angiogenesis pathway genes (ANGPT1, CD39, FGF2, and MMP9) were associated with clinical outcome of bevacizumab plus chemotherapy for metastatic colorectal cancer. 19 EJ Lewintre and colleagues reported recently that polymorphism of VEGFR-1 and VEGF-A and plasma levels of VEGF-A predicted the efficacy and prognosis of patients with nonsquamous NSCLC receiving chemotherapy combined with bevacizumab administration. 28 Collectively, polymorphism in the gene at the pathway single of bevacizumab disposition was proved to be of certain clinical significance for bevacizumab-based regimen.
With regard to the efficacy and prognosis of the total patients, ORR of the 128-patient cohort was 58.6%, DCR was 91.4%, the median PFS was 8.2 months, and the median OS was 19.9 months, which was inferior to the clinical outcome of the BEYOND clinical trial numerically. 6 One possible interpretation could be owing to the retrospective design of our study. Management and adherence of the patients in our research was poor compared with well-designed phase-Ⅲ clinical trial, which had been proved previously.20,29 Another reasonable explanation might be that patients with ECOG performance status 2 score were also included in our study and higher ECOG performance status score was correlated with worse prognosis. 30 Collectively, the real-world evidence among the 128 patients with NSCLC demonstrated that bevacizumab combined with platinum-based regimen exhibited satisfactory clinical outcomes.
Furthermore, it seemed that our study might be the first study that explored the relevance of efficacy of bevacizumab-based regimens to GSTP1 gene polymorphism in Chinese population. Interestingly, a previous exploratory study initiated by Celine P and colleagues investigated the association between VEGF polymorphisms and clinical outcomes of first-line bevacizumab plus chemotherapy for patients with nonsquamous NSCLC. 31 The conclusion indicated that four polymorphisms of VEGFA were associated with bevacizumab therapeutic outcomes. Therefore, these data exhibited the potential predictive significance of polymorphism in antiangiogenic single pathway for the efficacy and prognosis of patients receiving bevacizumab-based regimen. With regard to GSTP1 gene polymorphism, the previous study initiated by HY Lv and colleagues explored the clinical implication of genetic polymorphism of GSTP1 and ERCC1 in patients with NSCLC receiving platinum-based chemotherapy. 32 And the results indicated that patients with AG/GG genotype of GSTP1 were associated with superior clinical outcome, which was inconsistent with that in our study. We speculated that the discrepancy could be attributed to the heterogeneity of subjects included and the treatment regimens. However, another retrospective initiated by ZJ Yuan and colleagues investigated the association of GSTP1 and response of gemcitabine-cisplatin chemotherapy in Chinese patients with NSCLC. 33 The results suggested that A genotype carriers were significantly higher in sensitive group than in nonsensitive group, which was consistent with that in our study to some extent. Unfortunately, the two studies failed to investigate the association between the genotype status of the polymorphism and GSTP1 gene mRNA expression. Interestingly, it should be noted that patients with positive driver gene mutation (mainly EGFR mutation) conferred a longer OS (median OS: 20.9 vs 19.1 months) than those with other status of driver gene mutation numerically, even the difference was not statistically significant (P = 0.377). Previous study suggested that expression of VEGFA level was increased in EGFR mutation NSCLC compared with EGFR-wild type. 34 As a result, we noticed that recent study found patients with EGFR-positive mutation could benefit from bevacizumab combined with platinum-based chemotherapy in NSCLC with EGFR mutation, 35 which was consistent with the results in our study to some extent. Furthermore, to our knowledge, PD-1/PD-L1 blockades might be useless for patients harboring positive EGFR mutation previously. However, according to the IMpower150 clinical trial, bevacizumab combined with atezolizumab plus carboplatin plus paclitaxel (ABCP) regimen significantly improved PFS for patients with EGFR-mutated NSCLC, 36 which suggested that the addition of bevacizumab to other drugs could be beneficial for patients with positive EGFR mutation NSCLC. Collectively, NSCLC with positive EGFR mutation who harbored GSTP1 rs1695 AA genotype might benefit from bevacizumab combined with chemotherapy regimen significantly. And the conclusion should be confirmed in large-scale prospective clinical trials subsequently.
Interestingly, mRNA expression of GSTP1 disclosed that patients with AG/GG genotype of rs1695 were associated with higher GSTP1 mRNA expression. Previous work had elucidated that GSTP1 gene could contribute to the function of bio-interpretation or biotransformation by binding to reduced glutathione appropriately. 37 We speculated that the reason why higher GSTP1 mRNA expression conferred worse prognosis could be attributed that higher GSTP1 mRNA expression might reduce the level of drugs in tumor cells, thus compromising the efficacy of bevacizumab-based therapeutic regimen directly. In addition, located at coding area of GSTP1 gene, rs1695 polymorphism resulted in the alteration of amino acid from isoleucine to valine, which might compromise the expression and function of GSTP1 gene to some extent. 38 At present, the association between GSTP-1 gene expression level and prognosis of patients was still controversial. Previous study initiated by FM Yang and colleagues performed the research among patients with primary epithelial ovarian cancer (EOC) to investigate the prognostic significance of GSTP1 gene expression. 39 And the results supported that higher expression of GSTP1 predicted worse prognosis among patients with EOC, which was consistent with the mRNA expression results in our study. However, another exploratory study reported in 2017 initiated by XJ Liu and colleagues performed the analysis between GSTP1 expression and prognosis of patients with hepatocellular carcinoma. 40 The results indicated that higher GSTP1 levels could provide a superior PFS and OS through the suppression of tumorigenesis in HCC. This conclusion contradicted the results in our study, and the discrepancy could be related to the different significance of GSTP1 gene expression in different cancer. Collectively, patients with higher expression of GSTP1 conferred worse prognosis should be validated in large-scale prospective trials in the future.
From the objective view, some limitations could be unavoidable in our study. The small sample size and retrospective design of this study might compromise the conclusion in our study to some extent. And the prognostic data maturity regarding PFS and OS of the study was relatively low. Consequently, the conclusion in our study should be validated large-scale prospective trials further. Collectively, our study sufficiently performed the prognostic significance of rs1695 polymorphism and revealed part of the reasons for the prognostic difference caused by this polymorphism from the aspect of GSTP1 mRNA expression, which could be of clinical guiding significance for prognostic evaluation of bevacizumab-based first-line regimens in patients with advanced NSCLC.
Conclusion
Collectively, our retrospective study highlighted that GSTP1 polymorphism rs1695 could be used as a potential biomarker for the prognostic evaluation of patients with advanced NSCLC receiving bevacizumab-combined chemotherapy regimen.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to the patients and their families for participating in this study. They thank all the staff who took part in this study.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
F.H. and G.C. designed the study and prepared this manuscript. F.H. and H.J.T. collected the data and conducted the statistics. F.H. and B.L.J. participated in the experiments and follow-up. All authors participated in the reviewed of the manuscript and approved the final manuscript.
Ethical Approval
This study was approved by the ethics committees of Shanxi Cancer Hospital (approval no: KY2020136).
Informed Consent
All patients in this study provided written informed consent.
