Abstract
Background
Circulating endothelial cells (CECs) reflect the neovascularization in the tumor mass. We therefore investigated the potential role of CEC kinetics after first-line chemotherapy in advanced non-small cell lung cancer (NSCLC) patients.
Methods
Peripheral blood samples were obtained from 45 healthy subjects and 51 naïve patients with advanced NSCLC. Quantification of CD146+ CECs was performed using immunomagnetic separation (IMS).
Results
Pretreatment and posttreatment CEC levels in NSCLC patients were significantly higher than in healthy subjects (p<0.0001). An objective response was achieved after chemotherapy with partial response (PR) or stable disease (SD) in 26 patients, whereas the remaining 25 patients had progressive disease (PD). Baseline CEC levels were significantly higher in PR/SD patients than in PD patients (p = 0.039). After chemotherapy, CEC count significantly decreased in PR/SD patients (p = 0.014) and increased in patients with PD (p = 0.019). Moreover, there was a significant difference in the percentage change of CEC counts between the 2 groups (p = 0.0016). No significant difference in the median progression-free survival and overall survival (OS) was observed between patients with high baseline CEC counts and those with low baseline CEC levels. However, patients with high percentage change in CEC count had longer OS than those with low percentage change after chemotherapy (p = 0.05).
Conclusions
Changes in CEC counts after chemotherapy reflect tumor response in advanced NSCLC patients. Moreover, high percentage changes in CEC counts after chemotherapy may predict longer OS in advanced NSCLC. High baseline CEC levels might be an indicator of tumor response in advanced NSCLC patients after first-line chemotherapy.
Introduction
Non-small cell lung cancer (NSCLC) represents more than 80% of all lung cancer cases (1), and the 5-year survival rate is still below 15% across all stages (2). In addition, approximately 50% of patients with NSCLC are diagnosed at a locally advanced and metastatic stage (3). Platinum-based chemotherapy offers a small improvement in survival in patients with advanced NSCLC (4). The combination of platinum with new agents such as vinorelbine or gemcitabine has resulted in a high response rate and relative prolonged survival (5). Therefore, platinum-based double-agent chemotherapy, which has reached a plateau, has been considered as standard treatment for advanced NSCLC (6). Despite the application of targeted drugs in lung cancer in recent years, the response rates continue to be around 30%-40% making its treatment a challenge (7).
Tumor growth and dissemination depend on neovascularization (8), which involves a chaotic mixture of abnormal disorganized vessels (9). The treatment of NSCLC is now moving beyond conventional chemotherapy with the advent of molecular targeted agents (10). This kind of treatment depends especially on the intervention in cell cycle signal transduction and the tumor cell angiogenesis pathways. In this field, various agents have been developed, such as vascular endothelial growth factor (VEGF) and epidermal growth factor receptor (EGFR) inhibitors. So there is a need for developing biological markers in parallel with these targeted drugs. However, their use is restricted to non-squamous cell subtypes (11). In addition, a modest increase in survival rates of NSCLC patients has been achieved with approved targeted agents either used alone (erlotinib, gefitinib) or in combination with chemotherapy (cetuximab, bevacizumab) (12, 13).
Despite the different therapeutic strategies developed to date, NSCLC patients have poor outcomes (14, 15). Moreover, estimated deaths from lung cancer equals those of prostate, pancreas, breast and colon cancers combined according to surveillance, epidemiology and end results, cancer statistics reviews (2). In fact, many potential biomarkers are under evaluation in clinical trials, but no one has been able to carry out a close monitoring of the vascular structure of a particular cancer in relation to treatment undertaken (16, 17). Circulating endothelial cells (CECs) are considered as a relatively new biomarker that plays an important part in neovascularization. There is still controversy regarding the consensus definition of these endothelial cells (18). The term CECs is used with 2 distinct definitions: First, CECs in general are endothelial cells that circulate in the blood regardless of their function and origin. Actually, they may include the endothelial cells with proliferative potential, and may contain endothelial progenitor cells (EPCs). Second, mature CECs represent the endothelial cells in the circulation that have been detached from vessel walls elsewhere, which have limited growth capability and bear a mature phenotype. They may act as a surrogate marker of vascular damage (19). Increased amount of CECs, which has been reported in the peripheral blood of cancer patients (20), reflects tumor angiogenesis disorder related to tumor vascular volume (20, 21). Preclinical and clinical data suggest CECs quantification as a useful tool to determine the best treatment option for patients who are candidates for antiangiogenic therapy (22). It has been shown that the amount of CECs was correlated with plasma levels of VEGF, one of the pivotal factors promoting tumor angiogenesis (23). In contrast, baseline CEC levels seem to be correlated with tumor response in different types of cancer (23, 24). Furthermore, a strong correlation has been reported between change in CEC count during chemotherapy and tumor response, in multiple solid tumors (25, 26).
In this prospective study, we investigated the role of CEC levels and their kinetic modifications after first-line platinum-based chemotherapy, for the evaluation of treatment response and survival rates in advanced NSCLC patients.
Materials and Methods
Patients
The current prospective randomized study was conducted at Albairouni University Hospital, Damascus, Syria. The patients included were recruited at the Thoracic Oncology Department with histologically proven NSCLC in an advanced stage (III or IV). All patients were evaluated after first-line platinum-based chemotherapy. This study was authorized by the Ethics Committee of Albairouni Hospital at Damascus University. Written informed consent was provided for all patients after explaining the nature of the study. This study was performed according to the guidelines prescribed by the Declaration of Helsinki of the World Medical Association. The previously untreated patients included were required to be more than 18 years old with histological proven nonresectable NSCLC and an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of ≤1. Adequate renal, hepatic and bone marrow functions were also required for each patient. Patients who presented with pneumonia, pleural empyema, uncontrollable hypertension or any sign of infection were excluded from this study. Staging was determined by chest and abdominal computed tomography (CT) according to the seventh edition of the TNM classification (27). After first-line chemotherapy, CT-based clinical assessment for the determination of treatment efficacy was carried out using response evaluation criteria in solid tumors (RECIST) (28). Quantification of CECs was performed at baseline and at least 3 weeks after the end of the first-line chemotherapy. Patients were recruited between March 2011 and September 2013 at the Thoracic Oncology Department and followed until 30 June 2014. Progression-free survival (PFS) was defined from the first day of chemotherapy to the date on which the patient progressed or died from any cause. Overall survival (OS) was calculated from enrollment to death or to the last visit, at which the patient was referred to palliative therapy due to degradation in PS.
Quantification of Circulating Endothelial Cells
The isolation and quantification of CECs was performed using a previously validated manual immunomagnetic separation (IMS) technique (29). Briefly, peripheral blood was collected by venipuncture in a siliconized tube containing ethylenediaminetetraacetic acid (EDTA) as an anticoagulant. To prevent contamination of the sample with endothelial cells traumatically released from the punctured vessel wall, the first 2-3 mL were discarded. Samples were kept at 4°C-6°C and processed within 4-24 hours after collection. After carefully rotating the tube, 1 mL of the blood was diluted 1:3 with isolation buffer (phosphate-buffered saline [PBS]–bovine serum albumin [BSA] 0.1% and NaN3 0.1%). Cells were incubated for 45 minutes with 15 µL (7 × 107 beads) of anti-CD146 beads (Dynal M450 IgG1; Dynal AS, Oslo, Norway) at 4°C while gently agitating in a head-over-head mixer. To avoid nonspecific binding of leukocytes to the beads, the cell suspensions were flushed vigorously through the pipette tip during the washing step. The unbound cells were washed out with buffer, while the bound cells were retained on the magnet. After additional wash cycles, the sample was stained in 100 µL of acridine orange (5 µg/mL PBS, Sigma) for 10 minutes at 4°C-6°C in the dark. Isolated CECs were counted by a Nageotte hemocytometer (Hecht-Assistent, Sondheim, Germany) using a fluorescent microscope (Olympus, BX53, Japan) equipped with 500/20 nm excitation filter. The quality of images was improved by software Deltapix version 1.6 (Deltapix, Måløv, Denmark). As previously reported by Woywodt et al (29), the isolated CD146+ CECs were determined as nucleated cells in a well-defined cytoplasm and a diameter of 15-50 µm, bearing more than 5 immunomagnetic beads coupled to anti-CD146 with a well-preserved and recognizable morphology under microscope. The endothelial origin of CD146+ CECs was confirmed by staining with lectin UEA-1 (29, 30).
Statistical Analysis
The results were expressed as means ± standard deviation (SD) for continuous variables. The Mann-Whitney U test was used to correlate CEC count with tumor response, whereas the Wilcoxon signed-rank test was applied to analyze the change in CEC count after chemotherapy. Between-group comparisons were made using the chi-squared test. The univariate relationship between 2 independent categorical variables was examined using the chi-square test or Mann-Whitney U test. Receiver-operating characteristic (ROC) analysis was applied to determine the optimal cutoff value of the percentage change in CEC count after chemotherapy. The amount of CECs and their kinetics were correlated with PFS and OS using the Kaplan-Meier method and log-rank test. Statistical analysis was performed using the SPSS software program for Windows, version 15.0 (SPSS Inc., Chicago, IL, USA). All tests were 2-sided. A difference was considered statistically significant at a p value <0.05.
Results
Patients’ Characteristics, Treatment and Clinical Outcomes
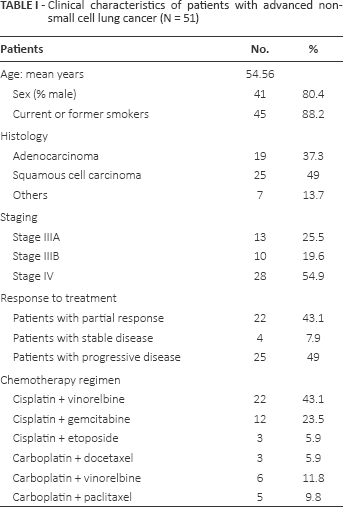
Among the 75 patients enrolled in this study, blood samples were available for CEC enumeration after first-line chemotherapy in 51 patients (68%) and 45 healthy volunteers. The included evaluable patients were 41 men (80.4%) and 10 women (19.6%) with a mean age of 54.56 years (range 36-70). Forty-four patients (86.3%) were former or current smokers, and 7 patients (13.7%) had never smoked. Twenty-three patients (45.1%) were classified as having locally advanced stage (stage IIIA and IIIB), while the remaining 28 patients (54.9%) were classified as having metastatic stage (stage IV). Histological subtypes included 19 patients with adenocarcinoma (37.3%), 25 patients with squamous cell carcinoma (49%), 2 patients with large cell carcinoma (3.9%) and 5 patients with unclassified NSCLC (9.8%). Chemotherapy was performed to 3 cycles in 9 patients (17.6%), 4 cycles in 5 patients (9.8%) and 6 cycles in 37 patients (72.6%). Regarding chemotherapy regimens combined with cisplatin, 22 patients received vinorelbine, 12 patients received gemcitabine and 3 patients received etoposide. In addition, carboplatin was administrated in combination with vinorelbine, paclitaxel and docetaxel in 6, 5 and 3 patients, respectively. After first-line chemotherapy, an objective response was obtained in 26 patients (51%) including 22 patients with partial response (PR) and 4 patients with stable disease (SD). However, the remaining 25 patients (49%) had progressive disease (PD). The clinical characteristics of patients are summarized in Table I.
Clinical characteristics of patients with advanced non-small cell lung cancer (N = 51)
CECs and Response to Chemotherapy
The number of CECs in healthy subjects ranged from 0 to 19 cells/mL (median 5), whereas the median basal CEC count in NSCLC patients was 137 cells/mL (range 40-1356). The mean levels of CECs were significantly higher in NSCLC patients before and after chemotherapy compared with healthy subjects (p<0.0001). Baseline CECs values were significantly higher in patients with PR/SD than those in patients with PD (mean ± SEM: 282 ± 50 vs. 174 ± 33, p = 0.039). By contrast, CEC values significantly decreased after first-line chemotherapy compared with those at baseline in PR/SD patients (p = 0.014). In patients with PD, a significant increase was observed in CEC levels after chemotherapy relative to those at diagnosis (p = 0.019) (Fig. 1).
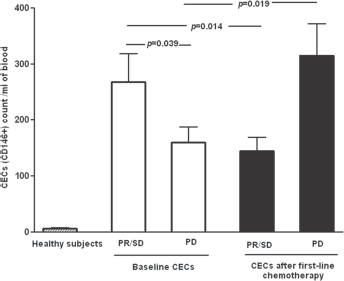
Circulating endothelial cell (CEC) count (cells/mL) in healthy subjects and patients with advanced non-small cell lung cancer (NSCLC). The levels were significantly higher in NSCLC patients before and after first-line chemotherapy than those in healthy subjects (p<0.0001). A significant decrease in CEC levels was found after chemotherapy compared with those at diagnosis in NSCLC patients with partial response or stable disease (PR/SD) (n = 26). CEC levels significantly increased after chemotherapy relative to those at baseline in patients with progressive disease (PD) (n = 25). Results are expressed as means ± SEM.
With the 75th percentile of the interquartile range of the median value (373 cells/mL) as cutoff, multivariable logistic regression model analysis showed that there was no significant difference in treatment response between patients with high pretreatment CEC levels and those with low baseline CEC count (odds ratio = 1.9; 95% confidence interval, 0.679-5.766; p = 0.16). However, there was a significant difference in the percentage change of CEC count between PR/SD patients and those with PD after chemotherapy (mean ± SD: 70.22 ± 88.59 vs. 132.22 ± 27.46, p = 0.0016). Regarding the relationship between clinicopathological characteristics of patients and treatment response, no significant difference was found according to smoking status, cell differentiation and histological subtypes. However, patients with locally advance stage had a more favorable response to chemotherapy than those with metastatic stage (p = 0.021). In this regard, there was a significant difference in treatment response between patients with high percentage change and those with low percentage change in CEC counts after chemotherapy (p = 0.018). The clinical and pathological variables of patients included are correlated with tumor response and summarized in Table II.
Relationship between clinicopathological variables and response to chemotherapy in patients with advanced non-small cell lung cancer
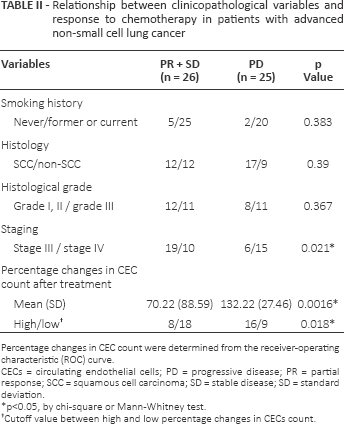
Percentage changes in CEC count were determined from the receiver-operating characteristic (ROC) curve.
CECs = circulating endothelial cells; PD = progressive disease; PR = partial response; SCC = squamous cell carcinoma; SD = stable disease; SD = standard deviation.
p<0.05, by chi-square or Mann-Whitney test.
Cutoff value between high and low percentage changes in CECs count.
CEC Counts and Survival
Median PFS was 170 (range 67-660) days. There was no significant difference in the median PFS between patients with high baseline CEC levels (n = 22) and those with low basal CEC count (n = 53) (202 vs. 151 days, log-rank test, p = 0.91; Fig. 2). With a median follow-up of 287 days (range 47-1066), no significant difference in the median OS duration was observed between patients with high pretreatment CEC levels and those with low CEC counts at baseline (287 vs. 284 days, log-rank test, p = 0.43; Fig. 3).
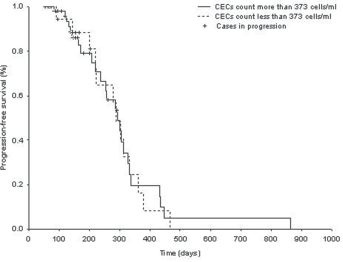
Progression-free survival (PFS) according to pretreatment circulating endothelial cell (CEC) count in non-small cell lung cancer (NSCLC) patients. No significant difference in the median PFS duration was observed between patients whose pretreatment CEC count was more than 373 cells/mL (n = 22) and those (n = 53) whose baseline CEC count was fewer than 373 cells/mL (corresponding to the 75th percentile of the patient group) (p = 0.91).
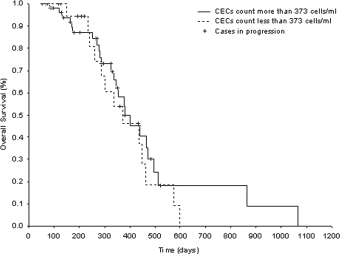
Overall survival (OS) according to basal circulating endothelial cells (CECs) count in non-small cell lung cancer (NSCLC) patients. No significant difference in the median OS duration was observed between patients whose pretreatment CECs count exceeded 373 cells/mL (corresponding to the 75th percentile of the patient group) and those whose basal CEC count was less than that (p = 0.43).
The percentage change in CECs count after first-line chemotherapy was calculated for each patient. The area under the curve (AUC) was 0.778, and the optimal cutoff value between high and low percentage changes in CEC counts after chemotherapy was 85.62%, providing a sensitivity of 73.1% and a specificity of 64% for the prediction of treatment efficacy (Fig. 4). Consequently, our population was divided into 2 subgroups: 24 patients above this cutoff value and 27 patients below it. Kaplan-Meier analysis with log-rank test revealed that there was no significant difference in the median PFS duration between the 2 subgroups (190 vs. 160 days, p = 0.36; Fig. 5). However, longer OS was obtained in patients with high percentage change in CEC counts than that in patients with low percentage change after chemotherapy (332 vs. 285 days, log-rank test, p = 0.05; Fig. 6).
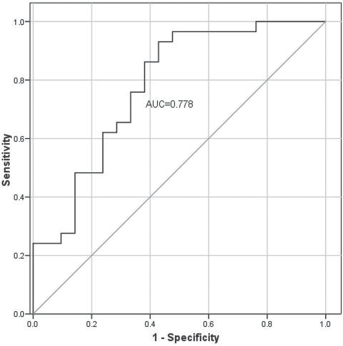
Receiver-operating characteristic curve generated to determine the cutoff value between higher and lower percentage changes in circulating endothelial cell (CEC) count after first-line chemotherapy. With the optimal cutoff value (85.62%), the sensitivity and the specificity for CEC percentage change to distinguish partial response + stable disease (PR + SD) from progressive disease (PD) were 73.1% and 64%, respectively. AUC = area under curve.
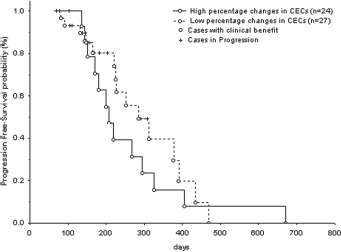
Progression-free survival (PFS) for non-small cell lung cancer patients according to percentage changes in circulating endothelial cell (CEC) counts after first-line chemotherapy. There was no significant difference in the median PFS duration between NSCLC patients with high percentage change (n = 24) and those with low percentage change (n = 27) after treatment, as determined by log-rank test (high vs. low, 160 vs. 190 days) (p = 0.36).
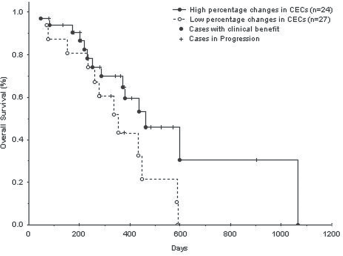
Overall survival (OS) for non-small cell lung cancer patients according to percentage change in circulating endothelial cell (CEC) counts after first-line chemotherapy. Significant longer median OS duration was found in NSCLC patients with high percentage changes (n = 24) than those with low percentage changes (n = 27) after treatment, as determined by log-rank test (high vs. low, 332 vs. 283 days) (p = 0.05).
Discussion
Systemic chemotherapy improves the survival rates and the quality of life in patients with advanced stage NSCLC (4). Several new therapeutic options have emerged including antiangiogenic and molecular-targeted agents. Despite the development of these agents, the median survival is still 7-12 months in lung cancer (31). It has become clear that the classical approach for cancer patients, based on changes in tumor size by radiological assessment, might not be sufficient to evaluate tumor response, especially when chemotherapy is used in combination with antiangiogenic or targeted agents. Therefore, conventional imaging evaluation needs to be integrated with other novel sensitive indicators for such treatment. As is known, the neovascularization in tumor vessels is generated by mature endothelial cells. Moreover, it has been found that CEC count may reflect tumor growth in cancer patients. So the change in CEC values after chemotherapy may represent a promising candidate biomarker that will reflect the effects of drug activity on the tumor mass. Consequently, the change in CECs count seems to be useful in evaluating the efficacy of chemotherapy.
In the current prospective study, we investigated the role of CECs and their kinetic modifications after first-line platinum-based chemotherapy for predicting tumor response and survival in advanced NSCLC. As previously reported (23, 32, 33), we found that CEC levels in NSCLC patients at baseline and after chemotherapy were much higher than in healthy subjects. This is probably due to the angiogenic switch associated with cancer growth and the related production of angiogenic growth factors and possibly others by cancer cells and/or various host cells (17).
We found that NSCLC patients with PR/SD had higher baseline CEC levels than patients with PD. It seems that systemic chemotherapy is probably more effective against tumors in PR/SD patients who have high baseline CEC values. Controversial published results have been obtained regarding the potential value of CECs at baseline for the prediction of tumor response during chemotherapy in NSCLC. Kawaishi et al (23) reported that higher pretreatment CEC values were observed in NSCLC patients with PR/SD than in those with PD in an early phase after 1 cycle of carboplatin and paclitaxel. The same findings were noted in a subset of advanced NSCLC patients with PR after 3 cycles of standard chemotherapy (26). Chu et al (34) retrospectively studied 107 patients with advanced NSCLC in early phases after 3 and 6 weeks of chemotherapy (paclitaxel plus carboplatin) combined with or without the antiangiogenic agent, endostatin. They found a significant relationship between higher median CEC levels at baseline and tumor response only in patients treated with chemotherapy combined with endostatin. However, the patients with PD included in this study presented lower baseline CEC levels after chemotherapy combined with or without endostatin. Based on these findings, baseline CEC values might be an indicator of tumor response at early and late phases of chemotherapy in advanced NSCLC patients.
According to the 75th percentile of CEC counts for the entire group, we found that baseline CEC count was not a predictive factor for either PFS or OS in advanced NSCLC patients. Our results were consistent with the findings of previous studies, in which more recent techniques have been used for CEC enumeration (35, 36). Using flow cytometry, no significant difference in the median PFS duration was obtained between NSCLC patients with high basal CEC levels and those with low CEC levels at diagnosis (35). Similar results were recorded for both PFS and OS in advanced nonsquamous subtypes according to the mean level of CECs quantified using the semiautomated IMS technique (36). However, no significant difference in baseline CEC levels was observed among histological subtypes in NSCLC (33, 37). The lack of a correlation between baseline CEC levels and PFS or OS may be due to the relationship between survival rates and the response to second-line chemotherapy in advanced NSCLC patients. It could be also due to subclinical aggressive disease in PR/SD patients with high baseline CEC levels. Other data reported in NSCLC were controversial. Fleitas et al (32) found that elevated baseline levels of CECs above the 75th percentile were correlated with poor survival in NSCLC patients after cytotoxic chemotherapy. Previous study has indicated that baseline CEC levels above the mean were correlated with longer PFS in NSCLC patients treated with carboplatin and paclitaxel (23). In other malignancies, the potential prognostic role of baseline CEC levels has also been investigated without definitive conclusions (24, 38, 39). These conflicting data could be explained by the difference in vascular turnover according to tumor origin, but could also have resulted from differences in phenotypes of CEC populations (40).
The different stages of disease, the anticancer agents being administrated and enrollment timing may have affected the number of CECs detected in cancer patients after treatment. We found that the kinetics of CEC levels were correlated with radiological response obtained after first-line chemotherapy in NSCLC patients with locally advanced and metastatic stages. The decrease in CEC counts in PR/SD patients is probably due to the agents used in chemotherapy that destroy some EPCs, which were in transition before their differentiation into CECs. However, CEC levels can be modified both positively and negatively depending on the agents used (41). In a recent study, no significant correlation was observed between changes in CEC levels and tumor response in NSCLC patients with clinical benefit after chemotherapy with platinum salt plus pemetrexed (36). Unlike in our work, the patients included in that study had nonsquamous subtypes of lung cancer in a metastatic stage. In addition, CEC changes were evaluated early before the second and the third cycle of chemotherapy. In a previous study, the number of CECs significantly decreased in PR patients, whereas a smaller change in CEC counts was observed in patients with PD after 1 cycle of carboplatin plus paclitaxel (23). Furthermore, a significant difference in CEC count was obtained between NSCLC patients with PR/SD and those with PD after antiangiogenesis therapy (35). By contrast, increased CEC counts were noted in NSCLC patients with PD after conventional chemotherapy (42). In our previous report (26), 89 patients with advanced NSCLC were studied after 3 cycles of platinum-based doublet chemotherapy. A significant decrease in CEC counts was observed in patients with PR, whereas no significant difference in CEC levels was obtained in patients with SD after chemotherapy. However, the number of CECs significantly increased in patients with PD after chemotherapy. Wang et al (37) studied 74 patients with advanced NSCLC treated with 2-4 cycles of chemotherapy in combination with or without antiangiogenic agent. The number of CECs decreased significantly in PR/SD patients after treatment in both groups. In addition, the increase in CEC counts was associated with PD in the patient group treated with chemotherapy alone. These findings suggest the change in CEC count as an indicator of chemotherapy efficacy.
In our study, the percentage changes in CEC counts were lower in patients who had an objective response than in those who progressed after first-line platinum-based chemotherapy. Chu et al (34) indicated that greater CEC reductions after 3-6 weeks of chemotherapy were observed only in PR/SD patients treated with endostatin. This disagreement with our findings may be due to the early evaluation of CEC changes and the low percentage of patients who progressed (only 14%) in their study. The larger number of patients with progression after treatment in our study also gives it much greater validity. In comparison with their baseline levels, it has recently been shown that an increase of more than 50% in the logarithm of CEC count after first and second cycle of chemotherapy is significantly correlated with PD in NSCLC (36). Similar findings were obtained in NSCLC patients with PD after first-line chemotherapy in our study. Liu et al (43) found that CEC counts increased progressively during chemotherapy compared with baseline counts in NSCLC patients who progressed after treatment. Moreover, CEC numbers significantly increased in patients with PD after chemotherapy with or without antiangiogenic agent. This increase was greater after the third cycle of chemotherapy in a statistically significant manner. These findings may be the potential explanation for why the percentage changes in CEC counts were significantly higher in patients with PD than in those with PR/SD after 3-6 cycles of chemotherapy in our study.
Finally, it was shown in NSCLC patients that high CEC reductions during chemotherapy combined with endostatin correlated with better PFS and OS. However, these changes in CEC levels failed to predict PFS and OS after chemotherapy only (34). Recently, it was found that the change in circulating EPCs during chemotherapy with or without paclitaxel did not predict PFS in advanced NSCLC (44). We found that OS but not PFS was significantly longer in NSCLC patients with high percentage changes in CEC counts than in those with low percentage changes in CEC counts, after first-line platinum-based chemotherapy. One of the potential explanations for these discrepant results lies in the biological mechanism of the selected therapeutic regimen. In reality, the inhibition of tumor angiogenesis suppresses the tumor growth factor resulting in a direct decrease of CEC numbers. By contrast, cytotoxic agents used in conventional chemotherapy reduce CEC counts in an indirect manner by causing cell death. The change in CEC levels, which was greater after 1-2 cycles than after 3-6 cycles of chemotherapy, may also explain this discrepancy.
Our study had certain limitations. Firstly, the number of patients involved was small. Secondly, our population received different treatment regimens and a heterogeneous number of chemotherapy cycles. In addition, the patients included in this study all came from 1 hospital. Multicenter studies are therefore required to confirm the predictive value of CECs for treatment responses in NSCLC.
In conclusion, our results suggest that changes in CEC counts after first-line platinum-based chemotherapy can be used as a predictive biomarker for tumor response and OS in advanced NSCLC patients. Moreover, high baseline CEC levels might be an indicator of treatment response in NSCLC after first-line chemotherapy. Given the biological mechanism of antiangiogenic agents to inhibit the pathway of VEGF, further prospective studies are warranted to determine the prognostic value of CECs in NSCLC patients treated with chemotherapy combined with or without antiangiogenic therapy.
Footnotes
Abbreviations
Acknowledgement
The first author thanks Pr. Othman I, the general director of AECS, for contributive comments in this work. The authors would like to thank Mrs. Nissreen Almalla for technical assistance and Mr. Ali Mohammad for help in some statistical analyses.
Financial support: None.
Conflict of interest: The authors have no conflicts of interest.
