Abstract
BACKGROUND:
Endostar (rh-endostatin) is a new recombinant human endostatin, which could inhibit cell proliferation, angiogenesis, and tumor growth.
OBJECTIVE:
To explore anti-angiogenesis short-term efficacy combined with neoadjuvant chemotherapy for stage IIIA (N2) non-small cell lung cancer (NSCLC), and identify the potential predictive factors.
METHODS:
We pathologically examined 26 patients diagnosed with stage IIIA (N2) NSCLC who received NP chemotherapy alone or combined with Endostar, respectively.
RESULTS:
Our results indicated that total clinical benefit rate (CBR) 87.5% and 64% (
CONCLUSION:
Our findings suggested that PMS and EPC may be used as a predictive factor for the short-term efficacy of the combined therapy in NSCLC.
Keywords
Introduction
Lung cancer is the leading cause of cancer-related human deaths and this cancer comprises less than 15% of the overall 5-year survival rate for cancers worldwide [1]. Non-small cell lung cancer (NSCLC) – which includes adenocarcinoma, squamous cell, and large cell carcinomas – comprises approximately 85% of lung cancers [2]. Poor prognosis is expected with mediastinal lymph node-positive stage IIIA NSCLC, confirmed either preoperatively or by postoperative pathology [3].
With advances in the treatment of tumors, neoadjuvant therapy of locally advanced NSCLC has developed from single treatment to multidisciplinary treatment. As chemotherapy provides limited choice and shows significant toxicity, side effects, and low efficacy for the treatment of NSCLC, evidence has suggested that the combination of anti-tumor angiogenesis therapy (which showed low toxicity with targeted treatment) and the combined treatment with anti-angiogenesis and neoadjuvant chemotherapy might provide a promising clinical therapeutic strategy for patients with lung cancer. However, there are still no molecular indicators or predictors that can accurately predict the efficacy of this treatment.
Endostatin, a 20-kDa C-terminal cleavage product of collagen XVIII, was originally identified by O’Reilly et al. as a tumor-derived, highly active, and endothelial-specific angiogenic inhibitor [4]. Endostatin is a specific inhibitor of endothelial proliferation and angiogenesis, and recombinant endostatin has been shown to inhibit the growth of a wide variety of tumors in vivo and in vitro, with no toxic side effects [5, 6]. Systemic administration of non-refolded precipitated protein expressed in Escherichia coli caused growth regression of Lewis lung carcinoma, T241 fibrosarcoma, B16 melanoma, and EOMA cells in a xenograft model, and no drug resistance was noted in these tumor cell lines [4, 7]. A recent study also indicated that endostatin can specific inhibit the migration of human umbilical vein endothelial cells (HUVECs) in response to vascular endothelial growth factor (VEGF), which result in the suppression of tumor growth [8]. In addition, more evidence shows that endostatin can interact with integrin on the surface of human umbilical vein endothelial cells, and, when used in a soluble form, endostatin functions as an antagonist of integrins to inhibit endothelial cell function [9, 10]. Moreover, several studies showed that endostatin can induce apoptotic effects in endothelial cells through the Bcl-2 pathway [11, 12, 13]. Until now, as one of the most broad spectrum and least toxic angiogenesis inhibitors, endostatin were reported to inhibit 65 different tumor types and modifies 12% of the human genome to downregulate pathological angiogenesis without side effects [14], thus suggesting it could be used in the combination of effective synergy in the control of tumor angiogenesis and growth.
Endostar is a novel recombinant human endostatin expressed and purified in Escherichia coli with an additional nine-amino acid sequence and forming another His-tag structure [15, 16]. To determine the short-term efficacy of Endostar combined with neoadjuvant chemotherapy for stage IIIA (N2) NSCLC, we compared its efficacy combined with NP (vinorelbine and cisplatin) chemotherapy versus the efficacy of NP chemotherapy alone. We used imaging, cell factors, morphological results, and other factors to identify the effective predictors for the short-term efficacy of the combined clinical therapy in NSCLC.
Materials and methods
Patients
Thirty patients with IIIA (N2) NSCLC hospitalized between April 2011 and December 2013 were selected and divided into a treatment group (20 patients) and a control group (10 patients). We used a random number table as the randomized method. The selected patients met the following inclusion criteria: (1) pathologically confirmed as NSCLC (histological confirmation); (2) evaluated as stage IIIA (N2) based on auxiliary examination; (3) imaging, laboratory examination, and other examination suggested the tumor could be completely resected; (4) received no chemotherapy or anti-angiogenesis treatment; (5) lesion could be measured; (6) signed the informed consent form.
The exclusion criteria were: pregnant women, women in lactation, or potentially pregnant women; serious infection; serious heart disease; uncontrollable nervous or mental disorders; serious diabetes mellitus; significant sign of hemorrhage; history of other tumors in past 5 years. The study was approved by the Clinical Research Ethics Committee of Tianjin Medical University Cancer Institute and Hospital. All of the patients signed the informed consent form after giving informed consent.
Twenty-six patients (16 in the treatment group and 10 in the control group) received a total of 52 cycles of treatment, and none experiencing any adverse events or died. Among those patients who were suitable for efficacy evaluation, differences in age, sex, and tumor characteristics were not statistically significant (
Treatments
This study adopted a randomized, controlled, and open clinical experiment design. The treatment group received NP combined with Endostar chemotherapy, whereas the control group received only NP.
Medications: Endostar 7.5 mg/m
Continuous IV administration from day 1 to day 14: vinorelbine 25 mg/m
Evaluation of the patients’ indicators
Main indicators for the efficacy evaluation included: response rate (RR), clinical benefit rate (CBR), and tumor regression rate (TRR). Efficacy was evaluated using the Response Evaluation Criteria Solid Tumors version 1.1 (RECIST1.1) guideline.
RR was defined as percent of patients with complete response (CR) or partial response (PR) in all the patients evaluable for efficacy; CBR was defined as the percent of patients with CR, PR, or stable disease (SD); TRR was defined as the proportion of reduction in the longest dimension of the target lesion.
CT perfusion imaging
A 64-slice CT scanner (Discovery CT750; General Electric Healthcare [GE], Fairfield, CT) was used at 100 kV and 100 mA. The center of the tumor received dynamic scanning. Patients received an antecubital intravenous injection of 50 ml iohexol contrast agent at 5 ml/second (s) before the scanning. The scanning began 5 s after the injection: one exposure each second for 60 s with the thickness of the scanning slice being 5 mm.
After the scanning, GE’s ADW4.0 workstation Perfusion software was used for data processing. The thoracic aorta was set as the input artery and brachiocephalic vein as the output vein. Images were processed and reconstructed via computer. The following parameters of the perfusion were measured: capillary permeability surfaces (PMS), mean transit time (MTT), blood volume (BV), and blood flow (BF). CT scanning and data measurement were conducted before and after the treatment.
Flow cytometry (FCM) analysis
After peripheral blood was taken from patients before and after the treatment, FCM analysis (BD FACSAria
According to the manufacturers’ protocols, peripheral blood mononuclear cells (PBMC) were isolated from whole blood using Ficoll-Paque
PBMC (100
The same procedure was performed in a test tube incubated for 30 min in the dark with 10
The samples were incubated with these desired antibodies at 4
ELISA assay
According to the protocol for serum preparation after collection of the patients’ whole blood, the blood was allowed to clot by leaving it undisturbed at room temperature; 15–30 min later, the clot was removed by centrifuging at 1,000–2,000 g for 10 min. Following centrifugation, the liquid component was immediately transferred into a fresh tube. The serum VEGF concentration was measured by a human VEGF ELISA kit according to the manufacturer’s protocol (Boster Biotechnology, Wuhan, Hubei Province, China).
Changes of predictors before and after the chemotherapy treatments
Changes of predictors before and after the chemotherapy treatments
In the treatment group, the percentages of PR, SD, and PD after two cycles of neoadjuvant chemotherapy combined with anti-angiogenesis therapy are 50%, 37%, and 13%, respectively. In the control group, the percentages of PR, SD, and PD after two cycles of neoadjuvant chemotherapy are 40%, 20%, and 40%, respectively.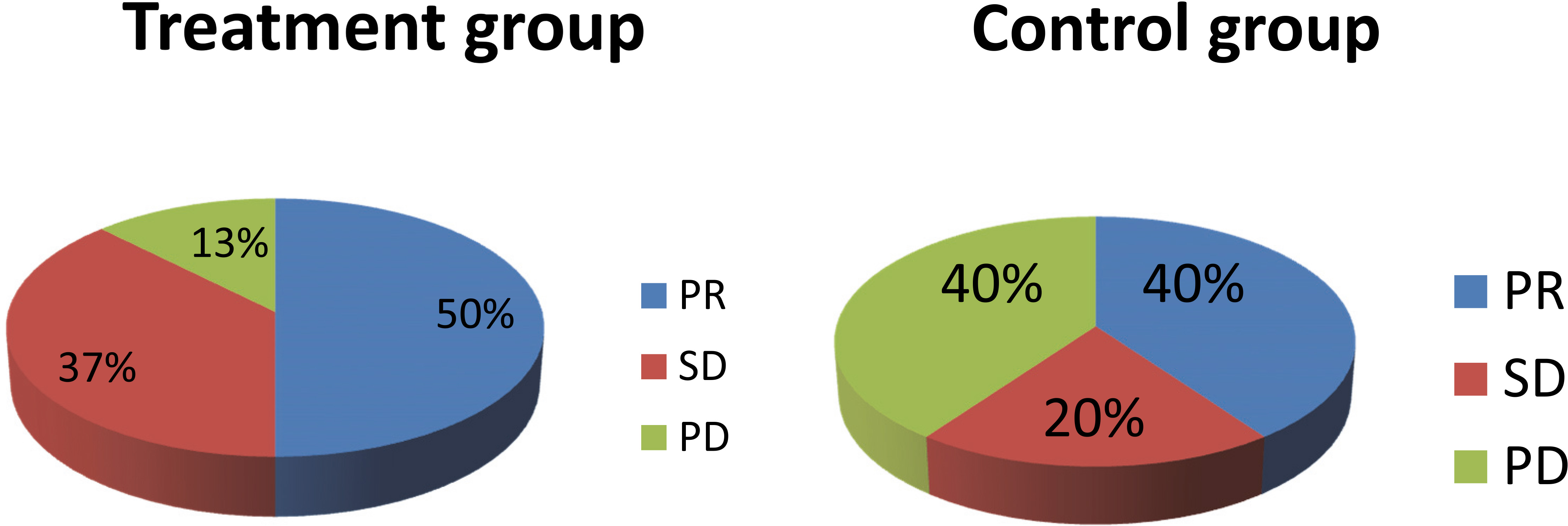
Fresh pathological specimens were fixed in formalin, and then embedded in paraffin. The sections were incubated and stained with anti-CD34 antibody (Golden Bridge Company, Beijing, China). After the specimens were dehydrated and mounted, the areas with most significant CD34 reaction (hot spots) were reviewed and scored under a light microscope, and the number of MVD in 10 random spots was counted at
Statistical analysis
All data were processed with SPSS v19.0 software (SPSS, Inc., Chicago, IL). Variables of normal distribution were represented with mean
Results
Efficacy evaluation of Endostar combined with NP chemotherapy
To evaluate the efficacy of Endostar combined with NP chemotherapy, we first compared the efficacy between the treatment group and the control group. Comparing the efficacies in the treatment group and the control group, respectively, total RR were 50% and 40% (
Notably, we did not find CR in any of the patients in either the treatment group or the control group. In the control group, the disease in two patients progressed to stage IV because of metastasis. Therefore, these results suggested that Endostar combined with NP exhibits better efficacy for patients than NP chemotherapy only.
Parameter analysis before and after the treatments
For the patients with NSCLC, to evaluate the changes of potential predictors in the anti-angiogenesis combined with neoadjuvant chemotherapy, we examined the levels of aCECs; EPC; serum VEGF; and CT perfusion parameters (BF, BV, PMS, and MTT); as well as some other parameters including MVD before and after the treatments (the NP chemotherapy alone versus anti-angiogenesis combined with neoadjuvant chemotherapy).
Our results indicated that anti-angiogenesis combined with neoadjuvant chemotherapy resulted in downregulation of the levels of serum VEGF, BF, PMS, and MVD, whereas reductions of the levels of VEGF and MVD only display statistical difference in the traditional NP chemotherapy for the patients with NSCLC (Table 1).
Changes of predictors for patients with clinical benefit in the treatment group
Changes of predictors for patients with clinical benefit in the treatment group
Changes of predictors for patients with clinical benefit in the treatment and the control group
These results suggested that these potential indicators might be involved in the evaluation of the treatment of anti-angiogenesis combined with neoadjuvant chemotherapy.
To further investigate the changes of the indicators of patients with CB in these two treatments, we next analyzed the levels of some indicators, including aCECs, EPC, VEGF, BF, BV, PMS, MTT, and MVD.
We found that the levels of EPC, VEGF, BF, PMS, and MVD were downregulated in the patients enjoying CB after anti-angiogenesis combined with neoadjuvant chemotherapy (
Correlations between variation of predictors TRR and variation of MVD
Correlations between variation of predictors TRR and variation of MVD
Next we analyzed the correlation between the variation of these predictors and TRR in the treatment group; our results revealed that the variation of EPC and PMS showed positive correlation with TRR (
Since the variation of MVD displays significant positive correlation with TRR, we therefore determined the correlation between the variation of other predictors and MVD level. Similarly, the variation of EPC and PMS also showed positive correlation with the variation of MVD (
These results suggested that PMS and EPC may be used as a convenient and effective predictive factor for the short-term efficacy of the combination treatment of anti-angiogenesis and neoadjuvant chemotherapy.
Discussion
Many clinical studies indicated that the efficacy of neoadjuvant chemotherapy for stage IIIA (N2) NSCLC before the surgery is not significant and the effect is still controversial [18, 19]. A recent study also demonstrated that anti-angiogenesis drugs combined with chemotherapy can improve CBR of patients with NSCLC, and that the combination treatment can prolong the total survival time as well as the disease-free survival time [20].
Because anti-angiogenesis therapy has been found to be involved in many pathways, independent anti-angiogenesis therapy does not exhibit effective efficacy and short-term improvement in imaging with morphological changes later than functional changes. Therefore, there are still no specific predictors for evaluating the short-term efficacy of anti-angiogenesis combined chemotherapy.
Since the most common method for quantitative analysis of tumor angiogenesis is the determination of the level of MVD with immunohistochemistry staining of tissue sections combined with vascular endothelial staining, we noted that among the patients with CB the level of MVD in the treatment group decreased more significantly than that of the control group using only chemotherapy thus suggesting that changes of MVD level can mainly reflect the efficacy of anti-angiogenesis therapy.
Moreover, the positive correlation between the variation of MVD and TRR (
Mononuclear cells developed from bone marrow can differentiate into mature endothelial cells CD34
A variety of cells and chemokines can promote EPC in stem cell nests to enter the cell proliferation cycle and then move into the peripheral blood circulation, or to mobilize EPC into new blood vessels to participate in the physiological or pathological process of angiogenesis.
A lack or inhibition of angiogenesis can suppress tumor growth; this might be helpful to predict the efficacy of tumor therapy. Schuch et al. found that anti-angiogenic drugs can inhibit the number of circulating EPC induced by VEGF and promote tumor regression [22]. In addition, the treatment with VEGFR2 inhibitor, which can specific inhibit membrane antigen VEGFR2 on the surface of EPC, will lead to a dose-dependent reduction in the number of EPC to exert its anti-tumor activity [23].
Rectal cancer and lung cancer clinical studies also showed that the EPC number and tumor vascular density were significantly decreased in patients who received anti-angiogenesis therapy [24]. To verify the influence of anti-angiogenesis therapy on EPC, we examined variation of EPC before and after the combination treatment to analyze the correlation between EPC and TRR/MVD (Table 4). Compared with the control group, the treatment group showed significantly different results before and after the combination treatment (
aCECs, mostly derived from endothelial progenitor cells (EPC) in bone marrow and activated by tumor secreted-VEGF, can be mobilized to surrounding areas of the tumor, develop into new blood vessels, and stay on the inner surfaces of these new blood vessels. Several studies indicated that dynamic changes of aCECs in patients with advanced tumors are positively correlated with tumor progression and treatment efficacy, thus suggesting that it might a useful biological marker for predicting the efficacy of chemotherapy [25, 26, 27]. Additionally, patients with CB showed significant reduction in the level of CEC, whereas patients with clinical progress showed the increment of the level of CEC [28]. However, our results demonstrated that the reduction of aCECs in the patients with CB of the treatment group did not display any statistical difference before and after either combination treatment of anti-angiogenesis and chemotherapy (or chemotherapy only) (Tables 2 and 3), which is not consistent with the previous reports [25, 26, 27, 28]. We surmise that the conflicting results obtained by these studies might be associated with different test methods (which caused the difference in measured CECs subsets), since the treatments increased CECs mainly in the stage of apoptosis.
In our study, Endostar may reduce fluid static pressure among tumor tissues – causing dilated, disordered, and distorted blood vessels to contract, thus reducing the area of tumor vascular bed and prompting activated neovascular endothelial cells to enter the blood circulation and increase CECs. When Endostar was applied via a long cycle, CECs would increase temporarily (in less than two cycles) and then decrease. Furthermore, chemotherapy may possibly increase aCECs through vascular normalization in a similar way.
VEGF – the strongest growth factor contributing to angiogenesis in tumor tissues – was produced and secreted by tumor cells, spliced, and expressed under the influence of mRNA, and then stored by platelets and monocytes. Endostatin can specifically influence vascular endothelial cells by blocking VEGFR and competitive binding downstream targets proliferating from vascular endothelial cells [7, 29, 30].
In this study, we expected that the reduction of VEGF level was mainly caused by the effective chemotherapy-induced inhibition of tumor growth, which can reduce tumor burden and subsequently downregulate tumor autocrine.
Our results also demonstrated that (in the patients with CB) regardless of whether patients received anti-angiogenesis therapy or not, VEGF values decreased significantly (
By observing perfusion changes in tumor tissues, CT perfusion imaging can be helpful for evaluating early treatment efficacy, particularly quantitatively evaluating microvascular physiology and tissue metabolism in tumor tissues. Our results showed that patients with CB in the treatment group exhibited significant changes in BF and PMS (
As we know, BF mainly represents the blood flow passing blood vessels of selected regions over a unit of time, and is influenced by blood volume, venous and lymphatic circulation, and oxygen consumption. PMS refers to the one-way transmission rate of a contrast agent entering the intercellular spaces from capillary endothelium, and reflects the integrity of the vascular endothelium and permeability of vessel walls. Therefore, the BF and PMS indicators might reflect the main targets of anti-angiogenesis therapy, and the variation of either BF or PMS should be the effective indicator for the short-term efficacy of the combined clinical therapy in NSCLC. Furthermore, PMS, a specific marker of vascular permeability, is subject to little interference of other factors and has a significant positive correlation with both TRR and MVD [32], direct morphological indicators of angiogenesis (
Additional evidence has shown that anti- angiogenesis therapy will be a promising anti-tumor clinical therapeutic strategy. Indeed, the application of anti-angiogenesis combined with chemotherapy in the treatment of IIIA (N2) NSCLC has been found to improve the clinical benefit rate, realize clinical downstaging, and increase the resection rate [16, 20].
After the analysis of the morphological and functional imaging of vascular endothelial cells, as well as the examination of the levels of VEGF and other regulators, our findings demonstrated that PMS and EPC may be used as a convenient and effective predictor for the short-term efficacy of the combined therapy.
Footnotes
Acknowledgments
This work was supported by the National Key Clinical Specialist Construction Programs of China and The National Key Research and Development Program of China (2016YFC0905501) and by grants from the National Nature Science Foundation of China (81470137) and the Tianjin Science and Technology Major Project, P.R. China (12ZCDZSY15400).
Conflict of interest
All authors declared that there were no conflicts of interest in this study.
