Abstract
The auditory system of locusts has high regeneration capacity following injury of the peripheral afferents. Regenerating auditory afferents can re-innervate their target areas even after changed neuronal pathways. Here, possible influences of contralateral deafferentation on regenerating afferents were investigated. Contralateral deafferentation was performed at different stages of the regeneration. Regeneration was triggered by crushing the tympanal nerve. The regenerated fibers showed aberrant fiber outgrowth, reduced density of terminations in the target area, the auditory neuropile and collateral sprouts crossing the midline. However, these results were not significantly influenced by the contralateral deafferentation. Therefore the bilateral symmetrical systems seem to be largely independent from each other.
Introduction
Nervous systems show a balance between the stability of neuronal connections and the necessity of adjusting to new requirements, where by simple nervous systems, like those of arthropod are often regarded as fixed systems. However, many dynamic processes like learning or injury related plasticity also occur in arthropods. Insects, like orthopterans show various regeneration and plasticity related processes in the nervous system. 1 9 A major sensory pathway with regeneration capabilities is the auditory afferent system.5,6,10–12
The locusts’ ear is located in the first abdominalsegment and the sensory cells are situated in the Müllers’ organ which is attached to the tympanal membrane. 13 The axons of these sensory cells run within the tympanal nerve into the metathoracic ganglion that represents a complex of the metathoracic neuromere and the first three abdominal neuromeres. 14 Within the ganglion, the auditory fibers project into the median ventral association centers (mVAC) of the respective neuromeres and thereby form two auditory neuropiles. 15 17 In the neuropiles, the afferent fibers connect to auditory interneurons. 16
These projection patterns can not only be restored but also be altered by regeneration processes. Crushing of the tympanal nerve in S. gregaria results in degeneration processes,5,18 and subsequent regrowth of fibers in normal or aberrant pathways. 12 A removal of the tympanal organ in locusts leads to a deafferentation of auditory interneurons within 3 to 5 days5,19 and a rearrangement of the auditory synaptic connections in the neuropiles. 20 During regeneration the original target regions are recognized and new synaptic connections are formed.5,12 Molecular mechanisms during degeneration and regeneration are not understood, although data on some molecules are available. For example, degeneration decreases the expression of acetylcholine esterase. 21 Furthermore, lectin binding indicates a rapid increase of glial cell related cell surface molecules in the neuropile. 22 In addition, regenerating fibers regrow along Fasciclin 1 immunoreactive tracts. 12
Thus, regeneration is related to up- and down-regulation of different molecules, which therefore might influence the growth and guidance processes. Such molecules could be either attractive or repulsive factors, and their amount might depend on the site and size of the injury. For example, a deafferentation of both auditory neuropiles in locusts might result in faster regenerative growth or more fibers growing into contralateral areas. Such contralateral growth could be either aberrant fiber growth or collateral sprouting. In the goldfish visual system regenerating fibers can innervate the deafferented visual structures of the other side. 23 These regenerating fibers could react to changes in attractive or repulsive influences due to the deafferentation. An influence of contralateral deafferentation onto regenerating fibers in locusts might indicate changes of such guidance molecules in the insect CNS. On the other hand, a lack of such an effect might reveal further mechanisms to maintain the stability of the system and show an independency of the two sides of a bilateral system.
This study investigates the effect of deafferentation on one side of the animal onto the regeneration of the contralateral tympanal nerve fibers. Therefore experiments with differently timed injuries were designed. The anatomical data should indicate whether regeneration can be influenced by the injuries of the contralateral side and thereby give indications for the search of molecular mechanisms.
Material and Methods
Animals
Adult animals and 4th instar larvae of the species Schistocerca gregaria were kept in cages at 28°C with a 12 h/12 h dark/light-rhythm. The animals were fed with fish food and wheat seedlings. For operations animals were cooled down at 4°C and fixed on a plasticine platform. Operated animals were marked individually and were kept together under the conditions mentioned above.
Experimental procedure
To investigate the regeneration processes within the CNS, four different experiments were designed. In this study nerve crushing was performed on the right side of the body while Müllers’ organ (MO) was removed from the left side of the body. 1) Removal of the MO and nerve crushing was accomplished simultaneously (R=C). 2) Removal of the MO took place 5 days prior to nerve crushing (R>C). 3) Removal of the MO followed 5 days after nerve crushing (C>R). 4) Only nerve crushing was performed (C). Controls were intact animals (Con). Backfills of the crushed nerve were always done 10 days after the crushing procedure.
The Müllers’ organ was removed from the left tympanal organ. After opening of the tympanal membrane, the MO was identified and pulled out using a fine forceps. To prevent haemolymphe loss, the injury of the tympanal membrane was covered with hot liquid wax. To crush the tympanal nerve, a small window was cut in the sternum ventrally of the caudal end of the metathoracic ganglion on the right side of the body. The cuticula was folded aside and the right tympanal nerve was crushed near the ganglion using a squeezing forceps. After that the cuticula was put back in place and sealed using a wax/colophonium mixture (1:2 wax:colophonium).
Anatomy
The cuticula ventral to the metathoracic ganglion was removed. For backfill into the CNS the tympanal nerve was cut distal to the crush and inserted into a glass capillary filled with neurobiotin (5% in Aqua bidest.) (Vector Laboratories Inc.) 24 (5% in Aqua bidest). After 48 hours, the metathoracic ganglion was removed and fixed in 4% formaldehyde in phosphate buffer (PB: 5.75gNa2HPO4, 1.48 g NaH2PO4 × 2H2O in 1l Aqua dest, pH 7.4) for 1 h. After washing three times in PB the ganglion was dehydrated, incubated in xylene (FlukaChemie AG) and rehydrated again. An enzyme reaction was carried out using 1 mg/ml Collagenase/Hyaluronidase (Sigma-Aldrich) in PB for 1 h at 37°C. Following washing three times in PB the ganglion was incubated in AB-complex (Vectastain ABC Kit, Vector Laboratories Inc.) overnight following Kit instructions, washed again and then incubated in DAB solution (2 drops DAB, 1 drop nickel solution in 3 ml phosphate buffer saline with Triton X-100 (PBST: 8 gNaCl, 0.2 g KCl, 1.44 g Na2HPO4, 0.24 g KH2PO4 in 1 l Aqua dest. with 0.1% Triton-X-100, pH 7.2), Vector Laboratories Inc.). A peroxidase reaction was performed using a H2O2 solution (1 drop in 3 ml PBST, Vector Laboratories Inc.), which was visually controlled and stopped by washing, followed by dehydration steps. The ganglion was covered in methyl salicylate (Merck KGaA).
To investigate the branching pattern, parasagittal sections of the stained ganglia were made. The ganglia were embedded into Agar 100 (Agar Scientific Ltd.) (15.4 g Agar 100, 9.5 g DDSA, 8.4 g MNA, 0.5 g BDMA) after incubation in methyl salicylate and agar 100 mixtures for 1 hour each in concentrations of 3:1, 1:1 and 1:3. Seven micrometer thin parasagittal sections were made using a microtome (Leica RM2165). The sections were counterstained with methylene blue in PB for several minutes, washed and covered with entellan (Merck KGaA).
Data analysis
The fibers growth was analyzed in respect to regeneration, aberrant growth and collateral sprouting. The marked ganglia and the sections were examined with a light microscope (BH2-RFCA, Olympus) and photographed (DFC 320, Leica; 2048 × 1536 Pixel). Drawings of the auditory projections were made using a light microscope and a drawing mirror (Leitz). The drawings were digitalized (Intuos PTZ-930, Wacom; CorelDraw 11, Corel Corporation).
The staining pattern of the auditory neuropile in the different experimental procedures was compared in whole mounts and sections. In whole mounts, the percentage of animals showing aberrant fibers outside of the neuropile, as well as the number of animals with contralateral fiber growth within the mVAC (“crossing fibers”) was compared in the different experiments and the controls. Statistical differences were calculated using a chi square test (Prism 4.0, Graph Pad Software Inc. Corporation). The percentage of stained area within the mVAC was measured in the digital images of the sections using the software ImageJ (Wayne Rasband, National Institute of Health, USA). 25 For that the complete mVAC area as well as the area with staining was measured as number of pixels. The percentage of stained area in the mVAC was calculated for each section. Of each examined ganglion, 5 sections near the ganglion midline were used and the mean of the percentage staining was calculated for each ganglion. At least 5 ganglia were examined per experiment and data were analyzed statistically using One-Way ANOVA with Tukey's Multiple Comparison posttest (Prism 4.0, Graph Pad Software Inc. Corporation).
Results
Influence of the different experimental procedures on the auditory projection on the right side of the metathoracic ganglion. Numbers represent the percentage of ganglia showing staining of the respective structure (indicated left) at the respective experimental procedure (indicated on top). The total number of experimental animals is indicated for each experimental procedure.

P < 0.05;
P < 0.01;
P < 0.001).
Projection pattern within the mVAC
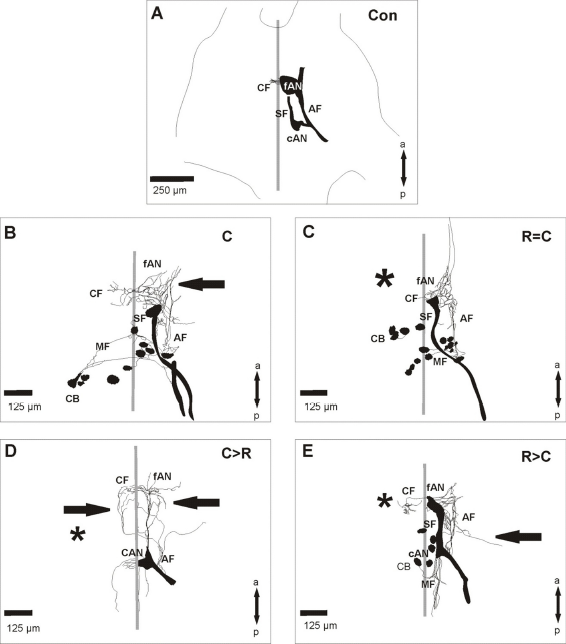
In intact animals, auditory fibers of the tympanal nerve projected within the metathoracic ganglion of S. gregaria through a fiber tract (AF) into two auditory neuropiles (cAN, fAN; Fig. 2A). About half of the ganglia had fibers crossing into the contralateral auditory neuropile (CF; Fig. 2A; Table 1). After crushing of the tympanal nerve the fibers regenerated into the metathoracic ganglion (Figs. 1A, 2B). The projection pattern usually differed from the control in a reduced density in the auditory neuropile and aberrantly growing fibers. To test whether a contralateral deafferentation might influence the regeneration process, a removal of the Müllers'organ as well as a nerve crush was performed in the same animal. However, the projection pattern in all three experimental procedures (Figs. 1B-D, 2C-E) were similar to those in animals with only a nerve crush (Figs. 1A, 2B). No significant changes could be found in the percentage of animals with marked auditory fibers in the different experiments compared to control animals (Table 1). But compared to the control situation all experiments had a significantly reduced percentage of animals with staining of the auditory neuropile (Table 1) and the terminations in the mVAC had a reduced density. In control animals auditory afferents terminated in most parts of the mVAC (Figs. 3A, 4). In most experiments the operated animals had a significantly decreased arborization area of fibers (Figs. 3B–E, 4). Animals with removal of the Müllers'organ following the crushing procedure (C>R) had similar arborization areas and fiber distribution in the mVAC as control animals (Figs. 4, 5). Within the complete neuropile, fibers were found in 60% of both control and C>R animals. The distribution of fibers in the ganglia with other experimental procedures was mostly limited to the posterior portion of the neuropile (Fig. 5).
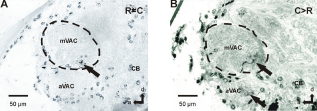
Projection of the auditory nerve within the metathoracic ganglion of S. gregaria in the different experimental procedures. Neurobiotin backfills were always performed on the crushed tympanal nerve. Schematic drawings of the auditory projection within the metathoracic ganglion of S. gregaria in the different experimental procedures. The focus is on the sensory structures; stained motoric structures were only partly included. Parasagittal sections of the metathoracic ganglion of S. gregaria in different experiments. Sections show the area of the mVAC after backfill staining of the crushed tympanal nerve. Percentage of staining within the area of the mVAC in the different experimental procedures. Numbers within the bars represent the number of animals. Distribution of the marked fibers within the area of the mVAC. “Complete” represents staining within most of the mVAC area; “Posterior” represents staining in the posterior-ventral portion of the mVAC; “Other” represents staining in the middle-ventral portion or within the anterior-ventral portion of the mVAC. Staining was never found dorsal-anterior in the mVAC area. Numbers within the bars represent the number of animals. Parasagittal sections of the metathoracic ganglion of S. gregaria from two different experiments indicating the terminations of the crossing fibers. Sections show the area of the median ventral association center (mVAC) contralateral to the backfill staining of the tympanal nerve. 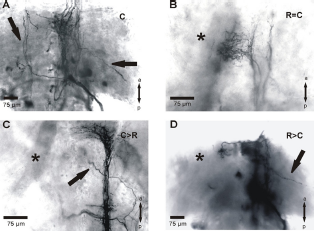



Contralateral sprouting
Fibers crossing the midline to the contralateral frontal auditory neuropile were not increased in operated animals compared to controls (Figs. 2A–E, Figs. 6A and B; Table 1). The crossing fibers usually terminated in the posterior-ventral area of the contralateral auditory neuropile (Figs. 6A and B). Usually only a small number of fibers crossed the midline and the fibers extended up to about 100 μm within the neuropile. Some of the fibers formed branches in the contralateral neuropile (Figs. 1D, 2B, E).
Aberrant fiber growth
In contrast to control ganglia, the projection of the tympanal nerve after crushing showed significant amounts of aberrant fibers (Table 1). Aberrant fiber growth was found in each experimental setup when crushing of the tympanal nerve occurred. These aberrant fibers had a wide range of morphological variation: they ranged from short processes on the ipsilateral side of the ganglion (Figs. 1D, 2E) to midline crossing fibers extending into the contralateral auditory neuropile (Figs. 1A, C, 2D). The crossing of the midline usually occurred near the caudal auditory neuropile (Figs. 1A, C, 2D). The percentage of animals with aberrant fibers was highest in experiments in which only the tympanal nerve was crushed (Table 1). The other experimental procedures had lower percentages although still more than the control.
Discussion
In this study we investigated whether contralateral deafferentation affects regeneration processes in the auditory system of locusts. It has been shown in the visual system of the goldfish, that regenerating optic fibers can innervate the deafferented structures of the other side. 23 Auditory afferents in orthopterans regenerate their ipsilateral projection after injury,5,6,10,11 although they may use different pathways. 12 In S. gregaria these auditory fibers run in a Fasciclin 1 positive area before reaching their original target area. 12 Therefore the pathfinding of regenerating fibers is influenced by molecular developmental factors, like axon guidance molecules. These molecular environments change after injury.21,22 Here it has been tested whether contralateral injuries and molecular changes can influence ipsilateral regeneration processes.
However, the regenerating fibers show no anatomical effects due to the removal of the contralateral tympanal organ and the regenerating fibers reinnervate their ipsilateral target areas. Major anatomical characteristics of regeneration (completeness of regeneration, collateral sprouting and aberrant fibers) were analyzed further.
The anatomic data indicate a less dense projection within the auditory neuropile 10 days after crushing. This finding can be explained by the time frame for restoration of the central projections in S. gregaria, which takes about 35 days 12 and that only 55% of the afferents regrow into the ganglion. 12 Therefore a decrease in the amount of fibers compared to the control situation was expected. Interestingly, in most cases the regenerating axons innervate the posterior area of the mVAC. In locusts, sensory type II cells detecting high frequency sounds project in that area.18,26 Furthermore, contralateral sprouting fibers are also found mainly in posterior regions. This might indicate different regeneration capacities of the fibers projecting into different neuropile areas. Similar results have been found in the auditory system of bush crickets, where also regenerating fibers have been found in posterior areas of the mVAC early after crushing. 11 These cells also detect high frequencies.27,28 Therefore the re-innervation of the posterior auditory neuropile by high-frequency detecting fibers seems to be a common characteristic in orthopterans. Interestingly, contralateral crossing fibers are frequently found in deafferented interneurons.4,5,11,20,29,30–32 These collateral sprouts even persist after reinnervation. 10
Regenerating fibers in the area of the mVAC crossing the midline into the contralateral neuropile were found to some extent in controls as well as in each experiment. The number of crossing fibers did not increase in the different experiments. Similar results have been obtained in crickets, where fiber crossing is found in intact animals and only the number of sprouts increased slightly in deafferentation experiments. 33 Therefore, contralateral deafferentation does not seem to promote contralateral sprouting and it can be expected that no growth-promoting factors are released. A third character of regeneration processes is aberrant fiber outgrowth. Such aberrant fibers are found after crushing and regenerative growth. This growth is independent from the removal of the contralateral Müllers’ organ.
In summary, we showed that the removal of the tympanal organ does not influence the regeneration properties of the contralateral auditory afferents. Therefore, the loss of afferents seems not to result from molecular changes like soluble attraction molecules. Additionally, regeneration seems to be restricted on one side of the bilateral nervous system. This restriction can be seen in the view of maintaining neuronal connection stability, rather than to allow an uncontrolled plasticity.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgement
We thank Casey S. Butler for correcting the English.
