Abstract
Noradrenaline has been shown to modulate sensory driven responses in the primary visual cortex (V1) of a number of vertebrate species. Moreover, this neurotransmitter has been postulated to bridge neuronal activation to genomic responses in order to instruct cells in long-lasting changes in neuronal performance. Here we show that local noradrenergic receptor activation in V1 is required for experience-regulated gene expression in the mouse V1. More specifically, we demonstrate that noradrenaline used locally within V1 mediates the light-driven gene expression of egr-1, an immediate early gene implicated as a mediator of neuronal plasticity. Visually-driven egr-1 expression largely depends on the α-adrenergic receptor subtype, with a lesser involvement of the β-subtype. Our findings suggest that noradrenergic transmission regulates plasticity associated gene expression in V1 of awake mice and is well positioned to broadly integrate experience-dependent changes at the cell's membrane and the genomic machinery in neurons.
Introduction
Noradrenergic transmission plays a central role in regulating attentional processing, sensory learning and the consolidation and recall of behaviorally-relevant memories. 1 4 Within the rodent primary visual cortex (V1), noradrenaline was shown to modulate neuronal firing properties 5 and various forms of experience-dependent synaptic plasticity. 6 9 Importantly, ablation of the projections from the locus coeruleus, the primary source of noradrenaline to the brain, was shown to significantly suppress experience-driven gene expression in V1. 10 Together, these data suggest that noradrenergic input modulates neuronal responses in V1 and may play a key role in coupling neuronal activation to genomic responses in visual cortical neurons, the latter of which are thought to underlie long-lasting changes in neuronal performance and, ultimately, forms of sensory learning.
In the present work we explored this hypothesis by investigating whether local noradrenergic receptor activation in V1 is necessary for experience-regulated gene expression in the mouse V1. Previous work has shown that visual experience drives a robust and transient expression of activity-dependent genes such as egr-1 and
Material and Methods
Animals, head-post implants and restraint adaptation
A total of 14 female Swiss Webster mice were used in this study. Animal protocols were approved by the University of Rochester Committee on Animal Resources and are in accordance with NIH guidelines. Animals were anesthetized with Equithesin (sodium pentobarbital (50 mg/kg) and chloral hydrate (200 mg/kg)) and placed in a stereotaxic device. Two small holes were drilled over the target area in V1 (AP -2.8; ML ± 2.5; DV 0.4 mm), and dental cement was used to form a well around the injection sites and to attach a metal head post to the skull. After a recovery period of a minimum of 48 hours, animals were subjected to a restraint adaptation protocol in the dark. Each restraint session consisted of gentle immobilization of the animal for 10 minutes, in the presence of the investigator, in a body tube and attachment of the head-post to an adaptor in the stereotaxic device. These restraint sessions were repeated every 2 hours, over a 10-hour period. Following the restraining period, animals were returned to their home cages, which were also maintained in complete darkness.
In-vivo injections in the awake mouse
After adaptation sessions, mice were restrained, as described above, for unilateral intracerebral injections of antagonists propranolol (300 mM in 200 ml) or phentolamine (200 mM in 200 μl). Vehicle was always injected contralaterally (Fig. 1). Injections were carried out with small glass pipettes and solutions were infused slowly (over 2 minutes) via a calibrated hydraulic pump. After injections, animals were returned to their home cages and stimulated with ambient light for 1 hour. Mice were then infused with an overdose of Equithesin and perfused transcardially with 20 ml of 0. 1M PBS (pH 7.4) and a cold solution of 4% paraformaldehyde. Brains were then extracted, cryoprotected in a 30% sucrose solution, embedded in Tissue-Tek, fast-frozen in a dry-ice/ethanol bath and cut at 20 mm on a cryostat for histological processing.
Schematic representation of a coronal section through the mouse brain (AP -2.8 mm), indicating the location of bilateral intra-cerebral injections. This stereotaxic level was used for all in-vivo injections carried out in this study.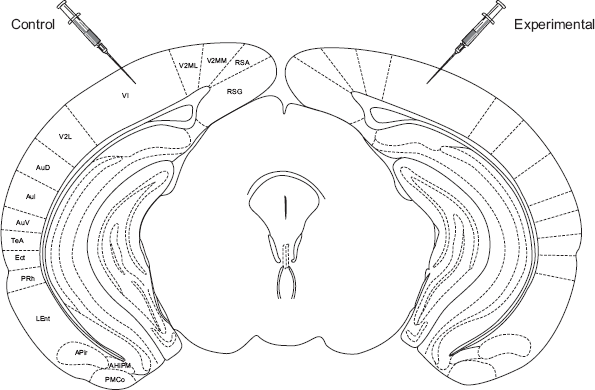
Immunocytochemistry (ICC)
Egr-1 protein expression was revealed by standard immunocytochemical procedures, as described previously (Pinaud et al, 2000). Briefly, sections containing V1 were sequentially incubated in 1) blocking buffer (0.5% albumin, 0.3% Triton in 0. 1M PB) for 30 min at room temperature; 2) anti-egr-1 primary antibody, raised in rabbit, in BB overnight at 4°C (dil 1:1000; Santa Cruz Biotechnology, USA); 3) biotinylated goat-anti-rabbit IgG, in BB, for 2 hr at RT (dil 1:200; Vector Labs, USA); 4) avidin-biotin solution in dH2O, for 2 hr at RT (dil 1:100; Vector Labs, USA). Each step detailed above was separated by 3 × 10 min washes in PB. Sections were subsequently developed by incubation in a solution containing 0.03% diaminobenzidine, 0.15% nickel ammonium sulfate in PB, to which 0.001% hydrogen peroxide had been added.
Quantification of Egr-1 expression and statistical analyses
Grids of 100 × 100 mm were placed either in the supragranular, granular and infragranular layers of V1, and egr-1 positive neurons were counted as profiles under brightfield microscopy and averaged across animals for each treatment. A one-way ANOVA tested for significant effects of drug treatment with an alpha value set for
Results
Egr-1 expression is regulated by visual experience in V1
To determine the extent to which Egr-1 is regulated by visual experience, we dark-reared a group of mice, individually, overnight. The following day, a subset of animals were either killed in the dark, or exposed to 1 hour of ambient light stimulation. Consistent with previous findings from other groups, we found that few Egr-1-positive cells could be detected in the V1 of dark-adapted animals (Fig. 2). In contrast, we observed a marked upregulation of Egr-1 protein in the V1 of light-stimulated animals. Egr-1-positive neurons could be detected across all cortical layers with the exception of layer I. To quantify this effect and determine the extent to which light stimulation differentially affect the expression of Egr-1, we quantified immunopositive neurons in the supragranular (II/III), granular (IV) and infragranular (V/VI) layers separately. In dark-adapted animals, we found that 4.1 ± 2.3, 3.2 ± 2.2 and 3.0 ± 2.6 Egr-1-positive cells could be detected in the supragranular, granular and infragranular layers, respectively. Light-stimulation led to a significant increase in the number of immunolabeled neurons across V1 cortical layers. Specifically, we found that 31.2 ± 2.1, 42.1 ± 2.5 and 35.9 ± 4.4 Egr-1 positive cells were found in the supragranular, granular and infragranular layers of V1, respectively. This difference amounted to a 8-fold increase in the number of immunopositive cells as a result of visual experience. These data indicate that dark adaptation suppresses Egr-1 expression levels, and that light stimulation drives a significant upregulation of this transcription factor in V1 neurons.
Light stimulation drives 
Blockade of β-adrenergic receptors partially suppresses light-induced Egr-1 expression
To determine the extent to which the local actions of noradrenergic input regulate the light-induced expression of Egr-1, we carried out bilateral, in-vivo pharmacological injections within the V1 of awake, restrained mice. Importantly, vehicle was injected unilaterally and noradrenergic antagonists were injected contralaterally; thus, each of our animals served as its own control.
Vehicle-injected hemispheres of light-stimulated animals underwent a marked light-induced upregulation of Egr-1 across all cortical layers. Layer- specific quantitative analyses revealed that 35.0 ± 1.9, 43.1 ± 1.8 and 34.6 ± 2.2 (mean ± S.E.) Egr-1-positive cells were detected in the supragranular, granular and infragranular layers, respectively (Fig. 3). Importantly, these cell densities were not statistically different from non-injected animals ( Blockade of β-adrenergic receptors partially suppress light-induced 
Blockade of α-adrenergic receptors suppresses light-induced Egr-1 expression
We next assessed whether α-adrenergic receptors also contribute to the light-induced expression of Egr-1. As with the experiment above, bilateral, in-vivo pharmacological infusions were carried out in the V1 of awake, restrained mice (vehicle was infused unilaterally, and phentolamine, a selective α-adrenergic receptor antagonist, was infused contralaterally).
We found that vehicle-injected hemispheres displayed a significant light-induced upregulation of Egr-1 across all cortical layers. In particular, we found 35.7 ± 1.5, 41.3 ± 1.2 and 35.7 ± 1.7 Egr-1-positive cells in the supragranular, granular and infragranular layers of V1, respectively; these cell densities did not differ from non-injected animals ( Blockade of α-adrenergic receptors largely suppresses light-induced 
Discussion
Here we show that local noradrenergic transmission is required for the expression of
Our findings also demonstrate that decreases in the number of light-induced egr-1 positive cells mediated by either α- or β-adrenergic receptor antagonism is more pronounced for the supragranular layers of V1. Based on the well known connectivity of V1, our findings suggest that noradrenergic transmission regulates plasticity-associated gene expression in neurons primarily engaged in intra-cortical connectivity. These findings are also congruent with the higher density of both α- and β-adrenergic receptor expression described for the mouse V1. In primary sensory cortices, including V1, neurons in the supragranular layers undergo marked levels of activity dependent plasticity, including synaptically-evoked long-term potentiation and depression. 16 In fact, noradrenergic transmission has recently been shown to act as a neurochemical “switch”, controlling both the polarity of synaptic plasticity changes in V1 9 and, based on the present findings, the engagement of molecular scripts putatively involved in experience-dependent neural rewiring.
Egr-1 expression has also been shown to require NMDA activation, a glutamatergic receptor subtype that carries a robust calcium conductance and that has been repeatedly implicated in synaptic plasticity, learning and memory formation. Notably, noradrenergic activation that is mediated by α-adrenergic receptors also controls the mobilization of intracellular calcium stores. Given the dependence of egr-1 on calcium for its expression, and the necessity of noradrenergic transmission for the expression of this immediate early gene, it is plausible that calcium regulation, either via NMDA or noradrenergic receptor activation (or a combination of both) may be a point of convergence in the intracellular cascades mediating egr-1 expression and the initiation of neuro-plasticity mediated changes in sensory cortex. Different adrenergic receptor subtypes (α versus β) exert disparate effects on the cellular biology of neurons. For example, whereas α-adrenergic receptors are coupled to stimulatory g-proteins, receptors of the β-subtype engage inhibitory g-proteins. Our findings indicate that α-adrenergic receptors contribute the brunt of egr-1's light-induced regulation; consequently, these observations indicate that enhancement of adenylyl cyclase activity putatively accounts for the induction of egr-1 in V1. Given however, that antagonism of α-receptors also partially inhibited the visually-driven
In summary, we show that noradrenergic modulation of V1 neurons is required for the light-induced expression of the plasticity-associated gene
Disclosure
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author and peer reviewers of this paper report no conficts of interest. The author confirms that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
We thank Lydia Schroeder for help with histological processing. Work supported by University of Rochester start-up funds, and NIH and University of Oklahoma Health Sciences Center
