Abstract
The disappointing results in bench-to-bedside translation of neuroprotective strategies caused a certain shift in stroke research towards enhancing the endogenous recovery potential of the brain. One reason for this focus on recovery is the much wider time window for therapeutic interventions which is open for at least several months. Since recently two large clinical studies using d-amphetamine or fluoxetine, respectively, to enhance post-stroke neurological outcome failed again it is a good time for a critical reflection on principles and requirements for stroke recovery science. In principal, stroke recovery science deals with all events from the molecular up to the functional and behavioral level occurring after brain ischemia eventually ending up with any measurable improvement of various clinical parameters. A detailed knowledge of the spontaneously occurring post-ischemic regeneration processes is the indispensable prerequisite for any therapeutic approaches aiming to modify these responses to enhance post-stroke recovery. This review will briefly illuminate the molecular mechanisms of post-ischemic regeneration and the principle possibilities to foster post-stroke recovery. In this context, recent translational approaches are analyzed. Finally, the principal and specific requirements and pitfalls in stroke recovery research as well as potential explanations for translational failures will be discussed.
Molecular basis of post-ischemic regeneration
Immediately after ischemic stroke a multitude of genes in the brain but also in peripheral organs are induced. This post-ischemic genetic response comprises both the activation of protective and adverse pathways with respect to the final functional outcome. The situation is complicated by the fact that timing of these events plays a critical role since the same gene product may exert protective effects in the acute phase but hinder regenerative effects in the subacute and chronic phase. For example, in experimental models matrix metalloproteinase-9 (MMP-9) has been shown to cause blood-brain barrier damage in the first days after stroke1,2 but later on it promotes angiogenesis and functional recovery. 3 MMP-9 is even indispensable for physiological plastic processes like hippocampal late-phase long-term potentiation and memory. 4 Similarly, in human stroke patients higher serum levels of MMP-9 are thought to be a predictor of worse outcome whereas at later time points MMP-9 levels could be positively correlated with clinical recovery. 5 The effects of MMP9 in the context of stroke become more complicated since tPA treatment additionaly elevates MMP9 levels and increases the risk of hemorrhagic transformation but on the other side it may potentially enhance regenerative processes in the long run (for an extensive review on matrix metalloproteinases and stroke see Rosell and Lo). 6 However, knowledge on the detailed temporal sequencing of MMP9 effects beyond the acute stroke phase is fragmentary yet. Nevertheless, delayed treatment with a virus mediated hypoxia-controlled MMP9 hyperexpression starting one week after transient middle cerebral artery occlusion (MCO) in mice demonstrated enhanced behavioral and structural recovery. 3 , 7 These two papers also highlight the need for a common definition of post stroke phases, since the two different terms “subacute” and “chronic” are used for the same postischemic timepoint of one week. The major players for successful post-stroke recovery induced by the ischemic event are neurogenesis, axonal sprouting, dendritic branching, synaptogenesis, oligodendrogenesis, angiogenesis, inflammation, neurotransmitter receptor regulation and white matter remodeling. About a quarter-century ago the post-ischemic regulation of single genes or gene families has been investigated to elucidate the molecular mechanisms of post-stroke pathophysiology.8,9 With the development of the microarray technology allowing a high throughput analysis of gene expression the transcriptional post-ischemic response or the post-ischemic “transcriptome” could be determined comprehensively.10–12 In the last decade the deciphering of the orchestration of the genomic response by epigenetic mechanisms like DNA-methylation, histone deacetylation, histone methylation and non-coding RNAs has become a major focus with promising translational options.13–16
Principle possibilities to foster post-stroke recovery
The rationale of all therapeutic interventions to improve post-stroke recovery is to either enhance spontaneously occurring regenerative processes and/or to eliminate or at least reduce processes impeding regeneration. Briefly, the following principal approaches are available (in more detailed discussed in recent reviews).17,18 The idea of
Apart from these approaches where any substances or molecules are applicated targeted to directly interfere at the transcriptional or translational level of regenerative pathways other therapeutic strategies are aimed to indirectly enhance brain repair. In this category fall the various classic
Examples for translation of therapies
Pharmacological
One of the classical examples for post-stroke recovery enhancement represents the use of neurotransmitters or neurotransmitter modulating drugs. Particularly
Another monoaminergic drug, the neuromodulator
The
Physical training
Another approach to enhance postroke recovery includes neurorehabilitative training paradigms. Training strategies can be in principle subdivided in task specific paradigms and unspecific ones such as general physical exercise or rehabilitative physiotherapy. Standard physiotherapy post-stroke either administered in rehabilitative units or in subsequent training session represents the current therapeutic standard. Although efficacy was not demonstrated in larger RCT, numerous smaller trials suggest a benefit of physical rehabilitation. Summarized evidence from these studies suggests physical rehabilitation to be more effective than usual care in improving motor function (12 studies, 887 participants), balance (five studies, 246 participants) and gait velocity (14 studies, 1126 participants). 61 Interestingly, there might even be an effect on dose of intervention, indicating that a dose of 30 to 60 minutes delivered five to seven days a week provides significant benefit compared to less intensive training. This finding correlates to experimental data indicating that intensity might be a key point, providing the basis for better long-term functional outcome compared to low intensity of voluntary training modalities. 31 One important example for such a controlled intensive training is the forced and specific training paradigm constraint induced movement therapy (CIMT), were the non-impaired extremity (typically the arm) is immobilized to foster mobility and training of the paretic arm. This approach was studied early in animals and has been successfully translated into patients, where constraint-induced therapy was associated with significant gains in motor outcome in 222 patients treated in the time window 3 to 9 months after stroke onset.62,63 Interestingly, parallel to animals,64–67 high doses of training too early after stroke proofed to be harmful: in the Very Early Constraint-Induced Movement during Stroke Rehabilitation (VECTORS) trial higher treatment intensity by CIMT applied within 1 month of stroke onset was associated with poorer motor outcome at 90 days. 68 Certainly, the great advantage of CIMT and its translational success is based on the fact, that only patients with very specific syndromes (moderate paresis of the arm), limited infarction in the respective motor area and an a priori defined readout such as motor function of the arm measured with the Action Research Arm test and Wolf Motor Function test qualify for this therapy.
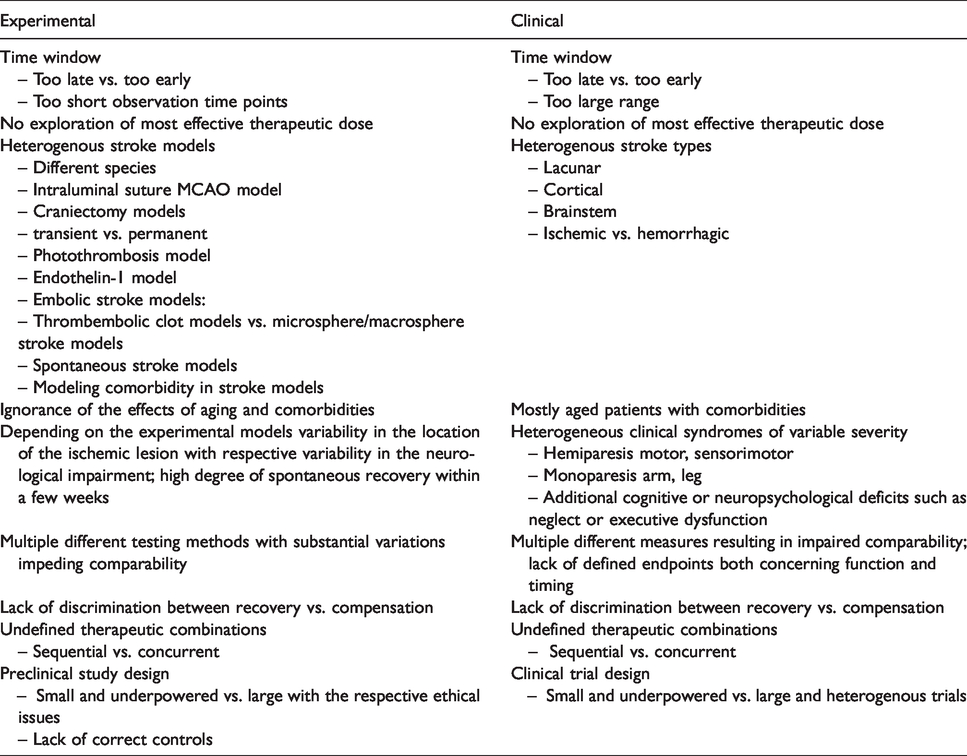
Basic requirements of recovery studies
Time window
While the time window for recovery enhancing therapies might be open in animals up to 6 weeks, in humans up to 6 months and potentially longer, 69 optimal timing appears to be crucial. Recent publications clearly show, early intensive training such as CIMT can impair functional outcome when initiated within minutes to hours after onset of cerebral ischemia both in rodents and humans.64–68 Importantly, such an early and intensive impulse on post-stroke recovery mechanisms impairs long-term functional outcome 6 weeks after cortical stroke independent of infarct evolution and final lesion volume.64,65 Clearly, the optimum time may be missed if the starting point is too late. 70 Interestingly, comprehensive experimental studies investigating the time window for neurorehabilitative therapies are very rare, in fact none of the translated therapies exhibits conclusive time window studies (Table 1). The situation in patients is even more complex due to the different etiology, size and location of infarctions as well as severity and heterogeneity of clinical syndromes. Indeed, no formal evidence is available in any of the translated rehabilitative drug therapies comparing different time windows of efficacy in humans (Table 1). Nevertheless, putting together current knowledge of post-stroke events Bernhardt et al. 71 suggested a framework of critical timepoints post stroke defining the temporal terms hyper-acute (0–24 hours), acute (1–7 days), early subacute (7 days–3 months), late subacute (3–6 months) and chronic (>6 months). This is a first step and considering these various phases together with neuroimaging biomarkers will potentially increase translational success. 72 In times of precision medicine defining post stroke periods by specific blood or genetic biomarkers which may indicate the most promising therapy should be the ultimate goal. This may be achieved in the future by aligning “timing of putative predictive biomarker levels with phases of physical recovery” 73 and with evaluation of therapeutic response. Comparing the biomarker levels between rodents and humans may eventually allow an adaption of the framework of post stroke recovery periods from mice to men and vice versa (Figure 1).
Reasons for translational failure.
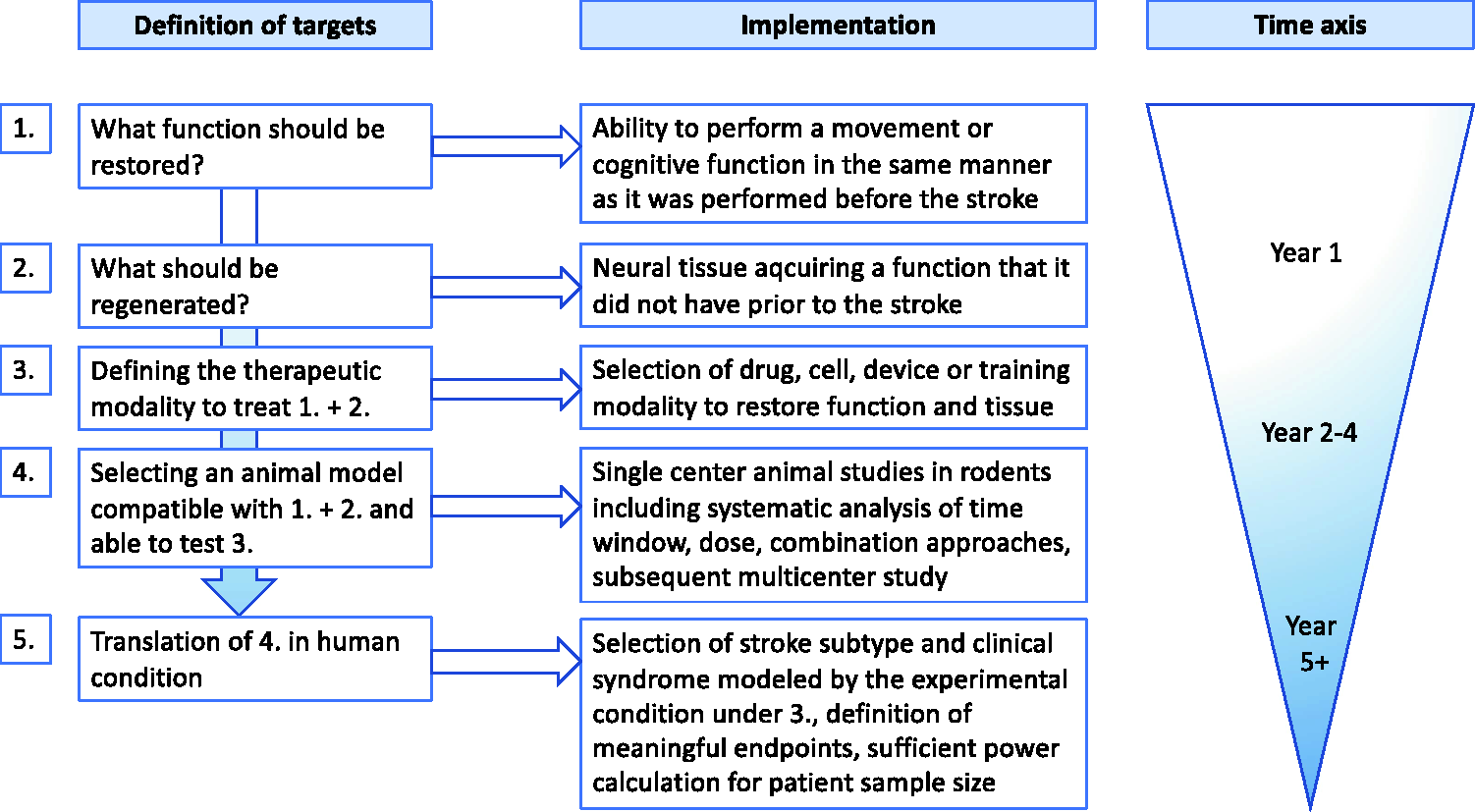
Algorithm of targeted interventions for development of a regenerative therapy. Definition of targets (1-5) should precede interventions. The process should be developed as integrative whole box approach from the definition of the targeted functional deficit to the translation of experimental findings into the human situation. In the latter one, a selected human condition should be defined and carefully selected for translation.
Optimal dosage
Currently, knowledge of the optimal dosage which is defined by frequency, duration and intensity of the respective therapy, is more or less based on try and error approaches. From the literature available it becomes clear, that there exists a dosage maximum which by going beyond will result in less benefit. This holds true not only for pharmacological treatment but has also been convincingly demonstrated for rehabilitative therapy (Table 1). In the VECTORS study stroke patients with high-intensity CIMT therapy did worse compared to the standard CIMT group and even compared to a standard treatment control group. 68 Theoretically infinite combinations are possible by varying the single parameters of the dosage of each single therapy. Furthermore, this variation is multiplied when combining priming and consolidating therapy (see below). In the future, stroke researchers should more involve biostatisticians and mathematicians to develop combination treatments with maximum benefit on a rational basis. However, also for single therapies such approaches are exceptionally rare. 74
Combinations
A major challenge is the combination of therapeutic approaches with the goal to potentiate beneficial effects, achieve synergistic effects and avoid neutralizing or even worsening ones. In principle, combination treatments are possible among all potential modalities e.g. synergistically acting drug combinations including bioactive molecules and antibodies, stem cell grafting or other biomaterials plus drug treatment. The available post-stroke recovery approaches can be distinguished in “priming” and “consolidating” therapies. 17 All therapies targeted to enhance spontaneously occurring recovery such as neurogenesis, angiogenesis, axonal sprouting, dendritic remodeling or synaptogenesis can be considered as “priming”. Therapies intended to exercise newly formed connections including all forms of training thereby stabilizing and strengthen them whereas non-used become pruned and eventually disappear would be classified as “consolidating”. Importantly, “consolidating” therapies can be combined with a pharmacotherapy e.g. with neuromodulators or neuro-enhancers to strengthen learning effects. All “priming” therapies build the biological basis for consolidation, may be started as early as possible after stroke and would be followed by “consolidating” approaches. 17 Indeed, the most common approach represents the combination of a drug treatment to a specific rehabilitative training. Although this has been practiced in numerous experimental and clinical studies, direct evidence for superiority of this approach is scarce (for detailed review of single studies see Ref. 17 ). For example, a sequential therapy with first neutralization of the growth inhibitory molecule Nogo-A for 2 weeks (“priming”) followed by subsequent skilled training paradigm (“consolidation”) resulted in nearly complete recovery while concurrent therapies failed. 25 Importantly, the concurrent treatment group performed worse than all other groups due to hyper-innervation and aberrant sprouting. 25 These findings correlate to combination studies with the growth factor G-CSF (“priming”) and forced training (CIMT, “consolidation”) in concurrent and sequential manner. 75 While all combinations with G-CSF resulted in improved sensorimotor outcome, CIMT alone did not. Importantly, starting with CIMT followed by G-CSF failed to enhance recovery. 75
Specific requirements and pitfalls in recovery studies
Principal problems of rodent models for stroke recovery
Recovery studies in animal models of stroke fall into two categories. In the first group, the spontaneous regeneration processes are characterized in a temporo-spatial manner to understand the biology underlying post-stroke brain plasticity. Manipulating these processes by knocking out some components of the repair machinery genetically, pharmacologically or functionally and correlating the resulting changes with the functional outcome is performed to get hints to the causal players in the recovery process. These experiments will be able to identify and characterize general principles and mechanisms of brain regeneration which may also play a role in humans. The results are also the basis for rational planning of therapeutic interventions aimed to enhance positive effects and inhibit adverse ones which can be tested in the respective animal models of stroke which represent the second group of stroke recovery studies. Major problems arise when trying to translate principally successful therapeutic approaches in rodents 1:1 into the patient. There are many publications problemizing the transfer of results from animals and especially from rodents into humans (for review see e.g. Ref.76,77). Differences concerning genetic and epigenetic background, size and anatomy of the brain, cerebral vascular anatomy, immune system, function and behavior are so fundamental that the translational road block is actually not really surprising. 76 In light of the well-known deficiencies of rodent models of ischemic stroke and post-stroke recovery the use of non-human primate models may provide more relevant information. However, there is no such thing as a free lunch. The more close the experimental animal to the human situation the higher the arising ethical issues (for review, c.f. Ref. 78 ).
Beside these principal issues which are reflected by nearly a complete lack of successful translation in post-stroke recovery 79 there are some specific requirements and pitfalls which will be briefly discussed.
Poorly correlative studies
As mentioned above the great majority of data is purely correlative and only minor attempts have been performed to demonstrate a causal link between plastic remodeling and functional outcomes (for review see Ref. 80 ). Nevertheless, there are sophisticated neuroimaging techniques to examine brain-wide remodeling, regional reorganization, neuronal circuit formation and activity. Furthermore, techniques to manipulate neuronal circuits and techniques to combine anatomical changes with molecular profiling to understand the causal underlying molecular changes are available.80,81
Long-term investigations and accompanying pitfalls in experimental studies
Apart from elegant experiments and the augmentation of scientific knowledge of the precise molecular mechanisms of regenerative post-stroke events, from the clinical perspective the only thing that matters for stroke patients is the amount of regained functional, cognitive as well as psychological and emotional health. Although these various parameters develop at different speeds all need a substantial amount of time. Concerning animal experiments the need for
There are some specific problems with “milder” ischemia models. In animals with smaller ischemic lesions, lesion size and functional outcome frequently do not correlate.75,82 Lack of correlation between lesion size and behavioral deficits was also seen in two permanent MCAO models in the rat. 89 Instead it is important to measure the volume of the remaining not directly damaged brain tissue.90,91 Shanina et al. 92 could demonstrate that quantification of the remaining brain tissue is superior to quantification of the total lesion size and represents a better predictor of functional impairment. Additionally, detecting subtle changes requires large group sizes, which deserves a thorough group size calculation. On the other hand, large group sizes although necessary for obtaining significant valid results may become an ethical problem (Table 1).
Imaging the spontaneous or therapy-induced post stroke regeneration processes longitudinally and correlating the results with neurobehavioral tests is another challenge in stroke recovery science. In principle, functional magnetic resonance imaging (fMRI) would be ideal for testing and analyzing the same animal overtime. However, due to the need of anesthesia several groups of animals at individual time points are frequently used leading to a high variability between time points. 93 Apart from the problem with anesthesia, resting-state fMRI technique is a step forward both towards reduction of variability in animals and improving the comparability between preclinical research and clinical studies due to reduced differences in the methodology used. 81 Other noninvasive in vivo techniques such as multi-parametric Optical Coherence Tomography (OCT) platforms allow for longitudinal imaging of ischemic stroke in mice both in the acute and chronic phase and may increase our understanding of recovery mechanisms. 94 In a rat photothrombotic stroke model with a chronic optical window, optical coherence tomography has been shown to allow 3D mapping of cerebral blood flow and cellular scattering offering an elegant technique to longitudinally evaluate treatment effects. 95 In a study with non-human primates, longitudinal evaluation of size and location of the ischemic lesion by T2 scans up to 30 days post transient MCAO showed a strong correlation between lesion volume and lesion localization to NHPSS scores. 96 This underlines the importance of correlating the exact lesion topography in the long run to more precisely predict the final behavioral outcome.
Longitudinal evaluation and endpoints in clinical studies
A general problem in clinical recovery trials is the uncertainty about endpoints and length of observation of the therapeutic effect (Table 1). In the acute stroke situation such key readouts are also not backed by primary evidence, but general agreement in the field build up over time resulting in the perception that an excellent or good functional outcome measured with the modified Rankin Scale is adequate when assessed 3 months after the stroke onset. These endpoints indeed proofed as measures for assessing and comparing the efficacy of recanalizing therapies.97,98 In the recovery field no such measures exist, nor is there an established agreement what kind of improvement would be functional meaningful. Clearly, a stroke patient would benefit from improvement of hand motor function restoring daily activities like writing or unbuttoning a shirt. Such functions were, however, not investigated in the recent large randomized trials. In the dopamine trial (DARS) assessment was focused on walking ability 8 weeks after stroke, 41 and in the fluoxetine trial (FOCUS) gross functional outcome was measured by the modified Ranking Scale 6 months after stroke. 24 The latter endpoint is even more incomprehensible with respect to the prior FLAME trial, where the fluoxetine treatment effect was assessed and effective on arm and leg motor function measured with the Fugl-Meyer Motor Scale 3 months after the stroke. 23 As indicated from these studies, the time interval to endpoint assessment is also variable and ranges from 2 – 6 months to even longer time points of 12 or 24 months assessed in previous studies.58,63
Altogether, these data suggest a lack of basic requirements in clinical recovery studies in defining a functionally meaningful endpoint and the appropriate timing of that endpoint assessment (Table 1).
Recovery versus compensation
Another important issue is the unprecise use of the term recovery. In the landmark paper by Levin et al. 99 clear definitions have been formulated to avoid any misunderstandings. At the neuronal level recovery is defined as “restoring function in neural tissue that was initially lost after injury” 99 whereas compensation means “neural tissue acquires a function that it did not have prior to injury”. 99 At the performance level recovery is defined as “restoring the ability to perform a movement in the same manner as it was performed before injury” 99 whereas compensation means “performing an old movement in a new manner.” 99 And at the functional level recovery is defined as “successful task accomplishment using limbs or end effectors typically used by nondisabled individuals” 99 whereas compensation means “successful task accomplishment using alternate limbs or end effectors.” 99 Taking this into consideration it is clear that most behavioral tests used are not able to reliably differentiate between true motor recovery and compensation (for thorough review c.f. Ref. 100 ). Only kinematic analyses,101,102 automized tests84,103 or tests in combination with kinematic analyses are able to discriminate compensation. 104 Apart from these problems many testing paradigms do no test for clinically relevant deficits or the tests are simply the wrong one for the respective ischemic lesion. Choosing the best experimental ischemia model for the respective scientific question seems to be a matter of course (Table 1). However, many experimental stroke studies suffer from carelessness concerning the choice of the experimental model.
Apart from the discrimination between true recovery and compensation differentiation between spontaneous and therapy-induced recovery is similarly difficult. In particular, in longitudinal investigations with multiple testing, the test procedure per se may act as an environmental enrichment or an interventional neurorehabilitative effect thus enhancing neurological outcome (for review see Ref. 105 ). Provided the real therapy is an add-on effect, using the adequate control groups would allow a clear distinction.
Proportional recovery
It was well known for long that the majority of stroke patients develop some degree of spontaneous neurological recovery. 106 A systematic analysis of patients with moderate post stroke hemiparesis revealed that most of them regained about 70% of their motor function of the upper limb within 3 months after stroke as assessed with Fugl-Meyer scale. 107 This phenomenon of the so called proportional recovery has some important implications, since in the first 3 months after stroke obviously neurorehabilitative therapies have little or no effect. 108 Whether this proportional recovery rule really holds true for other neurological systems is under discussion. 109 On the other hand, a subset of patients with severe impairment does not recover at all and thus does not fit to this rule. 107 Here, damage of the corticospinal tract seems to be causative for lack of spontaneous recovery. 110 Nevertheless, based on these findings the urgent obvious goal would be to find prognostic and predictive biomarkers that allow discriminating between recoverers and non-recoverers early after stroke enabling a “more effective triage and stratification for neurorehabilitation.” 108 However, since practically all patients with stroke obtain some form of neurorehabilitative treatment, it is very difficult to discriminate.
This proportional recovery rule has completely ignored for long in experimental models of stroke and thus may be a further component responsible for the lack of successful translation from mice to men. Most importantly, “preclinical models allow withholding of post-stroke rehabilitation, enabling the investigation of rehabilitation efficacy and the relationship between rehabilitative treatment and proportional recovery.” 111 In a systematic retrospective analysis of a cohort of 593 male Sprague-Dawley rats, Jeffers et al. 112 demonstrated that the proportional recovery holds also true for rats to a similar degree. Additionally, as for human stroke patients with severe impairments a subset of rats did not fit to the recovery rule. 112 Importantly, depending on the intensity of training treatments a subset of non-fitters benefited from therapy demonstrating that rehabilitation in fact plays a role for post-stroke recovery. Development of an algorithm was able to calculate the dose of rehabilitation necessary for each rat and thus translating this concept into the clinic may be a promising approach to a personalized stroke therapy. 112 Interesting and clinically so far not addressed is the finding, that periods of heightened responsiveness by a small cortical stroke itself but also by pharmacostimulation (Fluoxetine, see “III. Examples for translation of therapies, Pharmacological”, SSRIs) can enhance the response to training and mediate full recovery.51,113
Effects of aging
As for preclinical neuroprotection studies the effect of aging is frequently not investigated in recovery studies despite the fact the stroke mainly afflicts the elderly. Ignorance of this central issue may be one important factor for the frustrating lack of translational success. Preclinical studies in aged rodents on long-term post-stroke effects concerning structural and functional outcome are even more rare due to the higher mortality of aged stroke animals. There is accumulating evidence that the genomic post-stroke events differ substantially between young and aged individuals eventually resulting in impaired post-stroke structural regeneration and functional recovery in older rodents compared to younger ones (for review c.f. Ref. 114 ). Changes of the immune response with a shift towards a proinflammatory state and alterations of microglial differentiation and function are thought to play a key role for the reduced regenerative capacity of older brains.115–117 There is even evidence for changes in the gut microbiota of aged mice followed by increased levels of systemic proinflammatory cytokines resulting in worse outcome after stroke. 118 Not so surprisingly, there is proof that not only the endogenous regenerative capacity of the aged brain is reduced but also the response to various treatments when compared to younger animals. For example, post-stroke treatment with omega-3 polyunsaturated fatty acids after distal MCAO in 18 months vs. 2.5–3 months old mice attenuated brain damage and enhanced regenerative processes as well as sensorimotor function in both ages but aged mice benefitted less from this therapy. 119 A more thrilling question arises in the context of cell-based stroke therapy whether the age of the donor cells may also have an influence on the therapeutic result. Yamaguchi et al. 120 could demonstrate that age of the donor cell indeed matters. Transplantation of young vs. old human mesenchymal stem cells in a rat model of transient MCAO resulted in enhanced neurological outcome associated with increased production of trophic factors as well as enhanced anti-inflammatory effects, vessel maturation and neurogenesis. 120 Similarly, using an experimental mouse model of heterochronic bone marrow chimeras, Ritzel et al. 121 could show that aged animals reconstituted with young marrow developed reduced behavioral deficits compared to isochronic controls.
Finally, modelling risk factors and comorbidity such as diabetes mellitus, hypertension, atherosclerosis, hyperlipidemia, obesity or infection is of outmost importance to identify and disclose their impact on post-stroke recovery.76,122
Peripheral effects
Although it is not surprising that stroke recovery research focuses on the post-ischemic changes in the brain itself the importance of the peripheral aspect may have been underestimated yet and may contribute to the disappointing results in human stroke therapy. It has been shown that stroke induces substantial and prolonged changes in peripheral organs such as heart, kidney, liver, spleen or the microbiome of the gut which indirectly may modulate recovery processes.123,124
Conclusion and future directions
Actually, very ambitious goals have been formulated in the recovery field. The stroke recovery and rehabilitation roundtable (SRRR) taskforce e.g. claimed for a “radical new aim” for restitution and brain repair. 71 In this context the first thrombolysis trials are recalled to strongly motivate the stroke community. However, while it was relatively clear that any way to open the occluded vessel would be a promising approach a similar clear and defined target is not in sight for rehabilitation. The idea of neural tissue engineering to find “an effective curative, rather than symptomatic, therapy” 77 or reprogramming of the genome/epigenome to rebuild the brain 125 ignores one major problem. Regeneration of brain tissue per se may not be sufficient to regain the pre-stroke situation since there is also more or less substantial loss of information depending on the exact localization of the ischemic brain loss. It is a little bit the same dilemma as in Alzheimer’s disease where the idea that removing beta-amyloid would enhance cognitive state failed since too much neurons had already been gone. 126
Another major problem in stroke recovery research is a substantial incongruity between preclinical and clinical studies concerning the methods available to analyze the recovery mechanisms (Table 1). In particular the cellular level of regeneration processes in humans is largely a black box. Building up large stroke brain banks similar as we know it for neurodegenerative disorders would be a large step to be able to link findings from preclinical models to the human situation. “Better alignment of preclinical studies to clinical realities or constraints should be a priority for the field and funding bodies” as formulated by the Second Stroke Recovery and Rehabilitation Roundtable 127 may cover only one side of the medal. Even if well and thoroughly designed, preclinical and here in particular rodent models have their inherent specifics and limitations. The unquestioned power of preclinical stroke research is the elucidation of general endogenous regeneration processes and the possibility to look for checkpoint events by manipulating spontaneous recovery. Therefore, these experiments should not be pushed in the background.
Nevertheless, we must not fall in nihilism. Choosing the right model at the preclinical level and designing clinical trials strictly focusing of nearly identical stroke cases concerning size and location may improve the success rate calling for a shift of our recovery therapy concepts to a more mechanistically and personalized approach (Figure 1). This process should be developed as integrative whole box approach from the definition of the targeted functional deficit to the translation of experimental findings into the human situation - which must be defined and carefully selected for translation (see suggested algorithm, Figure 1). That would mean that recovery approaches would strictly focus on lesion size and location similar as in trauma and reconstructive surgery, where a femoral neck fracture is treated differently than a fracture of the tibial plateau. Taking all these points into consideration will hopefully break through the translational roadblock in the stroke recovery field.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CJS and WRS are inventors on the patent application “Hematopoietic factors for treatment of neurological condition” including stroke and other diseases. Recently a part of the application (ALS) was granted. CJS and WRS transferred their rights to Sygnis and received a minor financial compensation upfront. In case of efficacy CJS and WRS participate in form of royalties. CJS and WRS neither hold nor receive stocks of Sygnis nor have direct interest in the company. WRS received compensation in form of honoraria in his function as PI of the AXIS I study as indicated in this particular publication.128
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by German Research Foundation (Deutsche Forschungsgemeinschaft, DFG; SO 908/3-1, SCH 787/5-1).
