Abstract
Recently, we have described a novel approach to the treatment of cancer that employs a series of vectors that encode surface expressed chimeric proteins in which the cytoplasmic death domain of Fas is fused in-frame to the extracellular domain of one of a number of cell surface receptors that recognize and bind various ligands that are differentially expressed within the tumor microenvironment. Although the majority of tumor cells transduced with such vectors are killed in the presence of the corresponding cognate ligand, a small percentage survive and in vivo may go on to repopulate a treated tumor. In order to understand the mechanisms employed by tumors to escape the cytotoxic effects of pro-apoptotic signals triggered via Fas, we isolated a large number of 293 tumor cell clones that survive following transfection with a plasmid vector encoding Flk-1/Fas, a chimeric receptor that induces tumor cell death in the presence of the pro-angiogenic cytokine VEGF. Characterization of Flk-1/Fas-positive clones revealed that while survival can most often be attributed simply to the down-regulation of VEGF ligand expression, in cells that express both receptor and ligand, other proteins involved in the regulation of apoptosis may be targeted. Specifically, a Flk-1/Fas-positive, VEGF-positive clone was identified in which expression of APAF-1 was almost completely abrogated.
Introduction
A number of chemotherapeutic agents used in the treatment of cancer including doxorubicin, cisplatin, methotrexate, cytarabine and etoposide have been shown to mediate their cytotoxic effects, at least in part, through the induction of programed cell death or apoptosis.1,2 The Fas (CD95/TNFRSF6) pathway appears to play a key role in this process.1,2 Thus drug treatment enhances the expression of Fas and/or FasL on tumor cells, while subsequent death can be inhibited by the addition of antagonistic antibodies directed against the Fas receptor or its ligand.1,3 Additional evidence supporting a role for signals transduced via Fas in the response to chemotherapy comes from studies in which tumor cell lines selected for resistance to doxorubicin or mitoxantrone also acquired resistance to Fas-mediated apoptosis. 4 Importantly, in some tumor types, such resistance was associated with transcriptional down-regulation of Fas expression with levels returning to near normal upon removal of drug selection. 4
The Fas pathway has also been suggested to play a role in the response of normal and malignant cells to both UV and ionizing radiation.5–8 Clinically relevant doses of radiation can induce the expression of both Fas and FasL on certain cell types, particularly lymphoid cells, and interaction between these two molecules has been shown to be critical in determining outcome following exposure to radiation in some9–14 but not all,15–19 studies. Leveraging this finding, it has recently been demonstrated that APO010, a novel recombinant Fas/CD95 ligand, can enhance the cytotoxic activity of radiation against both lymphoid and solid tumor cell types in vitro, although disappointingly, little additional benefit was obtained in vivo when APO010 was administered at doses approaching MTD. 20
Although Fas is expressed by many tumor types, efforts at targeting the molecule using agonistic anti-Fas antibodies have met with only limited success. Human B cell lymphoma xenografts growing in nu/nu mice rapidly regressed in response to a single intravenous injection of anti-human Fas antibodies.21,22 However, in syngeneic systems normal tissue toxicity is a significant problem. Fas is also highly expressed in the liver and elsewhere and, in murine studies, intraperitoneal injection of antibodies directed against Fas produced extensive hepatic hemorrhage and necrosis. 23 Damage was also evident in a broad range of other organs including the spleen, thymus, lymph nodes, Payer's patch, skin, intestine, ovary, uterus, seminal vesicles, and so on. 24 A substantial proportion of treated animals died, usually within 6 hr of inoculation. 23 Fas-negative lpr mice were not similarly affected. 23
The therapeutic potential of systemically-administered FasL has also been evaluated and although more active and less toxic than anti-Fas antibodies in similar studies, 25 FasL also induced lethal hepatic necrosis following intravenous administration. 26
It is in an effort to overcome such toxicity that attention has turned to localized Fas-directed therapies. Of these, approaches involving gene therapy appear the most promising and practical. Specifically, several groups have shown that direct intratumoral injection of replication-deficient adenoviral vectors encoding FasL can induce the regression of Fas-positive tumors in vivo.27–30 Improved safety could be achieved by placing the transgene under the control of an appropriate tissue- or tumor-specific promoter/enhancer element.31,32 Inducible expression systems have also been explored. 33 A common finding in these studies is that FasL transduction could induce apoptosis even in cells that are resistant to the cytotoxic effects of anti-Fas antibodies. 30 Further, the reciprocal approach in which tumor cells expressing FasL are killed upon transduction with Fas was also effective. Although very encouraging, it goes without saying that strategies involving the transduction of Fas or FasL are limited to the subset of tumors that express the corresponding cognate ligand.
In order to broaden the range of tumors that can be targeted using pro-apoptotic gene therapy, while at the same time improving the safety of the approach, we have developed a series of vectors that encode surface expressed chimeric proteins in which the cytoplasmic death domain of Fas is fused in frame to the extracellular domain of one of a number of cell surface receptors that recognize and bind various multivalent ligands that are differentially expressed within the tumor microenvironment.34–36 We have confirmed that such chimeric proteins are functional and can induce the death of transduced cells upon the binding of various ligands including the pro-angiogenic cytokines vascular-endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF), and the glycosaminoglycan hyaluronan, an important component of the extracellular matrix that is often upregulated within the tumor microenvironment.34–36 Since the activating signals that have been selected for targeting have a restricted distribution, normal tissue toxicity is likely to be minimized as, even if the trans-gene is inappropriately expressed in non-targeted tissues, a death response will not be induced unless the required triggering signal is also present.34,35 Further, as a certain threshold level of ligand is needed for apoptosis to occur, 36 another inherent advantage of this particular approach is that it allows some “leakiness” in the activity of the transcriptional or other regulatory elements used to target gene expression to tumor cells, to be tolerated.
Future clinical development of this and other pro-apoptotic therapies involving signals transduced via the death domain of Fas would be facilitated if the mechanisms responsible for resistance were better understood. Such knowledge might help in the development of combination therapies designed to overcome such resistance and may allow patients that might not benefit from a particular approach to be identified and thus spared unnecessary treatment.
To address this issue we have isolated tumor cell clones that survive and grow following transduction with a vector encoding a Flk-1/Fas chimera and have employed microarray analysis to identify changes in gene expression that might explain such resistance. The results obtained clearly indicate that while loss of ligand expression is an important mechanism, a small proportion of clones appear to grow well despite expressing both the Flk-1/Fas transgene and the cognate ligand VEGF. Microarray analysis suggested that these cells survive by specifically down-regulating expression of key components involved in the apoptotic pathway including APAF-1. It is proposed that further analysis of these clones may reveal novel mechanisms that contribute to the resistance of tumor cells to pro-apoptotic signals.
Materials and Methods
Cell lines
The adenovirus 5 transformed human embryonic kidney cell line 293 was obtained from Q-BIOgene (Carlsbad, CA). The human carcinoid cell lines H727 and UMC-11 were purchased from the American Type Culture Collection (ATCC; Manassas, VA). All three lines were maintained at 37 ° C in an atmosphere containing 5% CO2 in Dulbecco's Minimal Essential Medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine, 50 units/ml penicillin, and 50 μg/ml streptomycin sulfate (DMEM + 10% FBS).
Antibodies
A purified polyclonal goat antibody directed against the extracellular domain of the human VEGF receptor Flk-1/KDR/VEGFR2 obtained from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA) was used to determine the level of Flk-1/Fas expression in transduced 293 cells by FACS analysis and to characterize the physiochemical nature of the molecule in Western blotting studies. Monoclonal antibody (mAb) 3D5, which defines an epitope encoded within the death domain of human Fas, and which thus recognizes both endogenous Fas and the Flk-1/Fas chimera on Western blots, was obtained from Alexis Biochemicals (San Diego, CA).
A rabbit polyclonal antibody to the human 150 kD Oxygen-Regulated Protein (ORP150) was purchased from ABCAM (Cambridge, MA).
Rabbit polyclonal antibodies directed against the apoptosis protease activating factor (Apaf-1) and the Bcl-2 binding protein Bnip-3 were obtained from Imgenex (San Diego, CA) along with a control rabbit polyclonal antibody against the “housekeeping” protein glyceraldehyde 3-phosphate dehydrogenase (GAPDH), which was used to confirm equal loading in Western blot studies. An affinity purified rabbit polyclonal antibody to the human DNA fragmentation factor, alpha polypeptide (DFFA) was purchased from Proteintech Group Inc. (Chicago, IL).
Fluorescein isothiocyanate (FITC)-conjugated rabbit anti-goat Ig secondary antibody for use in FACS analysis was purchased from Invitrogen (Carlsbad, CA). For Western blot studies, affinity-purified Alexa Fluor® 680-conjugated and IRDye800-conjugated donkey secondary antibodies were obtained respectively from Invitrogen (Carlsbad, CA) and Rockland Immunochemicals, Inc., (Gilbertsville, PA).
Construction of the Chimeric Flk-1/fas cDNA
The construction of the chimeric Flk-1/Fas cDNA has been described in detail previously. 34 Briefly, a cDNA encoding the extracellular ligand-binding domain of human Flk-1 was generated by PCR. cDNA prepared from mRNA isolated from primary human umbilical vein endothelial cells was used as a template and amplified using the following primer pair: 5′-Flk-1 (5′-GCGGGTACCGCCGCCGGTCGGCGCCCGGGC-3′) and 3′-Flk-1 (5′-GGCGGATCCCTTTTCCTGGGCACCTTCTATTATG-3′). After 40 cycles (95 °C for 30 s, 60 °C for 30 s, 72 °C for 2.5 min), the resultant PCR products were separated by gel electrophoresis and a 2.5 kb fragment that incorporates a 5′-Kpn1 site and a 3′-BamH1 site was isolated.
The full-length Fas cDNA was also generated by PCR. In this instance, Jurkat cDNA was used as a template together with the following primer pair: 5′-Fas (5′-GCGGAATTCAGGGGCGGGCACTGGCAC-3′) and 3′-Fas (5′-GGCTCGAGAATCTTTTCAAACACTAATTGC-3′). The reaction conditions were identical to those described above. A product of appropriate size (∼1.5 kb) was isolated following gel electrophoresis and sequenced to confirm its identity.
Digestion of the full-length Fas cDNA with BamH1 generated a ∼0.5kb fragment encoding the membrane-spanning and cytoplasmic death domains of the molecule. This was fused in frame to the cDNA encoding the extracellular ligand-binding domain of Flk-1, which had also been digested with BamH1. For expression studies, both the full-length Fas cDNA and the resultant chimeric Flk-1/Fas cDNA were cloned into the Epstein-Barr virus-based episomal expression vector pCEP4 (BD Biosciences).
Clonogenic Survival assay
The effect of Flk-1/Fas expression on the long-term survival and proliferation of tumor cells was determined using a standard clonogenic assay. Briefly, QIAfilter (QIAGEN, Valencia, CA) purified pCEP4, pCEP4.Fas, or pCEP4.Flk-1/Fas plasmid DNA was introduced into 293 cells using the TransIT®-293 Transfection Reagent (Mirus Bio, Madison, WI) and the 24 well-plate protocol provided by the manufacturer. Twenty four hours after transfection, cells were trypsinized and, after careful counting, aliquots of the resultant cell suspensions were distributed into 10 cm tissue culture dishes. DMEM + 10% FBS containing 200 μg/ml Hygromycin B (Sigma) was added to a final volume of 10 ml and the dishes incubated at 37 °C in an atmosphere containing 5% CO2 for approximately 14 days. For quantitative studies, the medium was carefully removed, the dishes washed once with Hank's balanced salt solution (HBSS) and the resultant hygromycin-resistant colonies fixed and stained with a solution of 1% methylene blue in methanol. The number of colonies per plate was determined using a ColCount colony counter (Oxford Optronix Ltd., Milton Park, Oxford, UK). In order to determine the mechanisms responsible for resistance to Flk-1/Fas-mediated killing, viable 293 colonies were picked and the cells expanded in DMEM + 10% FCS containing 200 μg/ml Hygromycin B.
FACS analysis
The expression of Flk-1/Fas on the surface of clones of cells that survive following transduction with pCEP4.Flk-1/Fas was determined by FACS analysis as previously described.34,35 Briefly, approximately 2 × 105 cells were incubated on ice for 30 min with goat anti-human Flk-1 primary antibody at a final concentration of 2 μg/ml. Following extensive washing in HBSS containing 2% FCS (HBSS + 2% FCS), the cells were incubated for a further 30 min on ice with FITC-conjugated rabbit anti-goat secondary antibody (2 μg/ml). After additional washing, the cells were resuspended in HBSS + 2% FCS containing 1 μg/ml propridium iodide to facilitate the identification and exclusion of dead cells and analyzed on a FACScan (Becton-Dickinson Immunocytometry Systems, San Jose, CA). When excited with the 488 nm laser in this instrument, FITC has a green emission that is collected at 530 nm using the FL1 detector. Propridium iodide in contrast has a broad emission spectrum with a peak around 620 nm that can be collected using the FL2 detector.
Western Blot analysis
Western blot analysis was carried out to ensure that the Flk-1/Fas proteins expressed by Flk-1/Fas-resistant 293 clones were of an appropriate size and had not therefore undergone substantial deletion or other changes that would lead to alterations in their molecular weight on SDS-PAGE. Briefly, Flk-1/Fas-resistant 293 cells were harvested by incubation with PBS containing 5 mM EDTA, washed in PBS and their membranes solubilized by incubating the pellets for 15 min on ice in 10 mM Tris pH 7.5, 150 mM NaCl, 2 mM EDTA, 1% NP-40 at ∼2 × 107 cells/ml lysis buffer. Nuclei and detergent insoluble material were removed by centrifugation and the lysates stored at −80 °C until required. Thawed samples were added to an equal volume of non-reducing sample buffer (125 mM Tris, 20% (v/v) glycerol, 4.6% (w/v) SDS, pH 6.8) and incubated at 100 °C for 5 min. Total cellular proteins were separated by SDS-PAGE and transferred onto nitrocellulose membranes. The filters were incubated overnight at 4 °C in PBS containing 5% (w/v) non-fat milk powder then probed with antibodies directed against either the Fas death domain specific mAb 3D5 (2 μg/ml in HBSS + 2% FCS), or a polyclonal anti-Flk-1 antibody (2 μg/ml in HBSS + 2% FCS). After extensive washing, filters were incubated for a further 1-2 hr with an appropriate fluorescent-labeled secondary antibody. After additional washing, protein products were visualized using a Odyssey Infrared Imager (LI-COR Biotechnology, Lincoln, NE). Densitometric analysis was carried out using GelEval v1.35 (FrogDance Software, Dundee, UK).
Quantitation of Vegf expression
The level of VEGF protein present in tissue culture supernatants conditioned by various tumor cell lines was determined using a commercial ELISA kit (R&D Systems).
A semi-quantitative RT-PCR approach was used to characterize both the expression and alternative splicing of VEGF mRNA in Flk-1/Fas-positive 293 cell clones and in the control tumor cell lines H727 and UMC-11. Single strand cDNAs prepared from oligo-dT-primed mRNA (5 μg) isolated from the various tumor cells were used as PCR templates. The primers employed correspond to sequences in VEGF exon 3 (5′-GAGACCCTGGTGGACATCTTCCAGGAGTACCC-3′) and exon 8 (5′-GGCTCCTTCCTCCTGCCCGGCTCACCGCCTCG-3′) permitting amplification of each of the major alternatively spliced VEGF isoforms. Primers corresponding to the actin sequence (5′-GCTGTGCTATCCCTGTACGCCTCTGGC-3′ and 5′-GATGGAGTTGAAGGTAGTTTCGTG-3′) were used as a control. As control studies had demonstrated that the amount of product produced using the conditions employed (94 °C for 30 s, 60 °C for 30 s, 72 °C for 1 min) increased in a linear fashion to a level dependent upon the concentration of template present in the starting material through 30 cycles, reactions were terminated after 25 cycles and the products separated on a 1% agarose gel, stained with ethidium bromide and photographed.
Microarray analysis
The relative expression in Flk-1/Fas resistant 293 clones of 112 genes involved in the induction and regulation of apoptosis was determined by microar-ray analysis using pathway-specific Oligo GEArray® Human Apoptosis “HybPlate” DNA Microarrays (SABiosciences, Frederick, MD). A comprehensive spectrum of functionally important genes are represented on the pre-prepared microarrays including members of the TNF receptor family along with their corresponding ligands, members of the bcl-2 family, caspases, IAPs, TRAFs, CARD family proteins, death domain-associated/activated proteins, CIDE domain proteins, and genes involved in the response to DNA damage. Genes involved in anti-apoptosis are also well represented. A full list can be found at the following URL http://saweb2.sabiosciences.com/gene_array_product/HTML/OHS-012.html. Targets that exhibited a two fold or more increase in expression or a greater than 50% reduction in expression were identified and Western blot analysis carried out as described above to determine whether such changes at the RNA level translate to changes in protein expression that could potentially affect function.
Results and Discussion
Tumor cells often evolve mechanisms to evade apop-tosis. They do so in order to survive in the face of the pro-apoptotic signals to which they are often exposed either naturally within the tumor microenvironment or upon treatment with ionizing radiation and/or certain chemotherapeutic agents. Such resistance may involve both down-regulation of key pro-apoptotic proteins or the up-regulation of molecules that have anti-apoptotic activity. Irrespective of the mechanism involved, tumor cells exhibiting reduced apoptotic responsiveness may flourish under hypoxic or other hostile conditions and may better resist the cytotoxic effects of therapy.
Although previous studies have demonstrated that the Flk-1/Fas chimeras developed in our laboratory are effective at killing malignant cells in the presence of VEGF,34,35 it is apparent that a small proportion (<10%) of cells that have been successfully transduced with a plasmid vector encoding the transgene (as evidenced by their growth in the presence of appropriate drug selection) nevertheless survive and give rise to colonies (Fig. 1). In an effort to define and ultimately counter the mechanisms responsible for this effect, a total of 100 Flk-1/Fas resistant 293 clones were isolated and expanded for further study.
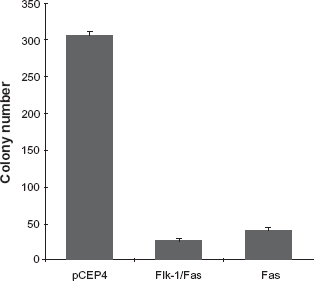
Effect of Flk-1/Fas expression on clonogenic survival. 293 cells were transfected with pCEP4, pCEP4.Flk-1/Fas, or pCEP4.Fas plasmid DNA and aliquots of the transduced cells plated in 10 cm tissue culture dishes. Cells that had taken up the corresponding plasmids were selected by the addition of Hygromycin B and their ability to survive and give rise to colonies determined on day 14 after the plates were fixed and stained in a solution of 1% methylene blue in methanol. The colony count data shown represents the mean ± SEM of three or more cultures.
Initial screening by FACS analysis using an antibody directed against the extracellular domain of Flk-1 was carried out to exclude clones in which the Flk-1/Fas transgene is simply not expressed. Not unexpectedly given the efficiency of Flk-1/Fas and other similar Fas chimeras in killing target cells when actually present, the majority of clones thus far examined appear to belong to this category. Although not without interest, such lines are not particularly useful with respect to defining the mechanisms responsible for development of resistance to Flk-1/Fas killing and were not further studied.
A number of Flk-1/Fas-resistant clones that did express the transgene were also identified. Although representing <10% of the total, the lines established from these clones appeared stable and maintained expression of Flk-1/Fas over many months in culture. The FACS profiles for three such clones are shown in Figure 2. Two of the clones, designated 1-1a and 1-1b, expressed similar high levels of Flk-1/Fas as determined by reactivity with a goat anti-human Flk-1 antibody, while the third, 1-1c, although strongly positive, was rather more heterogeneous, with a proportion of the cells expressing somewhat higher levels of the transgene. Parental 293 cells transfected with the “empty” pCEP4 plasmid vector were negative for Flk-1.
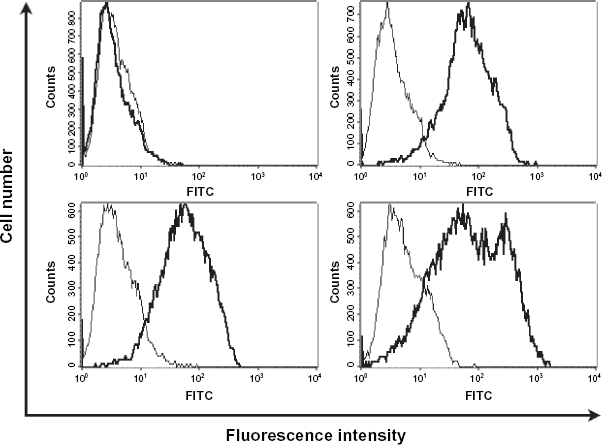
Expression of the chimeric Flk-1/Fas transgene on the surface of transduced tumor cell clones. Randomly picked clones of 293 cells transduced with pCeP4.Flk-1/Fas which had been selected and maintained in the presence of Hygromycin B were examined for surface expression of Flk-1/Fas by FACS analysis after staining with a purified polyclonal goat antibody directed against the extracellular domain of the human VEGF receptor Flk-1 and an appropriate FITC-conjugated secondary antibody.
Lysates prepared from these clones were also examined by Western blot. As shown in Figure 3, a protein species of an appropriate size that reacted with antibodies directed against Flk-1 and Fas was present in each of the Flk-1/Fas transduced lines but absent for 293 cells transfected with the control pCEP4 vector. These data argue against the possibility that the survival of these particular clones could be attributed to the cDNAs encoding Flk-1/Fas having undergone substantial deletions that might, for example, have resulted in the production of a truncated molecule that lacks the cytoplasmic death-domain of Fas but retains both the membrane spanning domain and extracellular domain derived from Flk-1. The presence of more subtle deletions and/or mutations that might affect the function of Fas cannot be excluded.
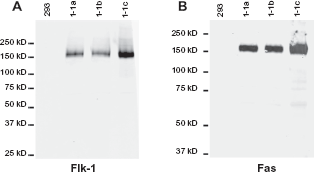
Expression and physiochemical characteristics of the chimeric Flk-1/Fas transgene in transduced tumor cell clones. The overall expression and basic physiochemical characteristics of the Flk-1/Fas transgene in clones of 293 cells transduced with pCEP4.Flk-1/Fas which had been selected and maintained in Hygromycin B was determined by Western blot analysis. Lysates were run on a 7.5% SDS-PAGE gel (∼2 × 105 cell equivalents per lane), transferred to nitrocellulose, and the membranes probed with either a goat polyclonal antibody directed against the extracellular domain of Flk-1 or a mAb against the death domain of Fas (mAb 3D5). After extensive washing, the blots were incubated with appropriate fluorescently-labelled secondary antibodies and the protein products visualized using a Odyssey Infrared Imager (LI-COR Biotechnology, Lincoln, NE).
Sequencing studies have revealed the presence of Fas mutations in both lymphoid and non-lymphoid malignancies with the highest frequencies being detected in thyroid lymphomas (65%) 37 and transitional bladder cancers (28%). 38 As might be expected if signals transduced via Fas are important in the development and pathogenesis of malignancy, the majority of amino acid changes occurred within the death domain. The valine at position 251 was a particular mutational hotspot and was changed to an isoleucine in 8 of 12 patients with transitional bladder cancer.2,38 It is noteworthy, that although fully capable of inducing apoptosis upon transduction into FasL-positive cells, the Fas cDNA employed in the present study to generate the chimeric Flk-1/Fas construct contained an A to G substitution at position 1117 that results in a single amino-acid change (Glutamic acid to Glycine) at position 297 located within the cytoplasmic domain of the mature protein. 35 Frame shift and splicing defects in Fas have also been described, most commonly in lymphoid malignancies. For example, almost half of Fas mutations occurring in Non-Hodgkins lymphoma involved deletion of exon 8 producing a frame shift that in turn affected expression of exon 9 which encodes the death domain.2,39
One obvious way in which cells expressing Flk-1/Fas could avoid being killed would be to down-regulate expression of the VEGF ligand that binds to and activates the chimeric receptor. To identify clones that exploit this potential mechanism of resistance, the presence and level of VEGF in media conditioned by each of the Flk-1/Fas-positive clones identified by FACS screening, was determined by ELISA. Unexpectedly, in approximately 2/3 of cases little if any VEGF could be detected in conditioned super-natants. The remaining 1/3 of clones did, however, express both the Flk-1/Fas transgene and VEGF. Secretion of VEGF by the three Flk-1/Fas-positive clones randomly picked for more detailed analysis as described above is shown Figure 4. Clone 1-1b is clearly positive for VEGF secretion, although the amounts produced are lower on a per cell basis than control 293 cells. In contrast, clones 1-1a and 1-1c appear to secrete only low levels VEGF that are presumably below the threshold required to trigger death via the Flk-1/Fas chimera.34,35
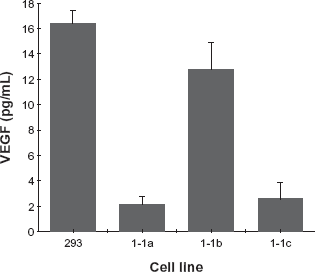
Production of VEGF by Flk-1/Fas resistant 293 tumor cell clones. The level of VEGF present in medium conditioned for 24 hr with equivalent numbers of the Flk-1/Fas-positive 293 cell clones 1-1a, 1-1b and 1-1c was determined by ELISA. For each cell line, the data shown represents the mean ± SEM of 4 independent determinations.
Although not the major focus of the present study, some efforts were made to define the mechanism(s) by which VEGF is down-regulated in non-secreting clones. Previous work on the control of VEGF secretion, mostly in response to hypoxic stimuli, emphasize the complexity of the process. Alterations in gene transcription, message stability and splicing, translation and protein transport have all been implicated in one experimental system or another.40–44 As shown in Figure 5, however, semi-quantitative RT-PCR analysis suggested that VEGF mRNA levels are similar in both VEGF-secreting and non-VEGF-secreting Flk-1/Fas-resistant clones. Confirming this finding, quantitative RT-PCR analysis using the same primer pairs demonstrated that irrespective of the clone from which RNA was derived, a similar number of cell cycles was required to produce a 50% maximal yield of VEGF product (data not shown). Alternative splicing of the VEGF pre-mRNA transcript also appears unaffected (Fig. 5). Taken together, these data suggest that the defect responsible for the down-regulation of VEGF secretion by Flk-1/Fas-resistant clones operates at a post-transcriptional level.
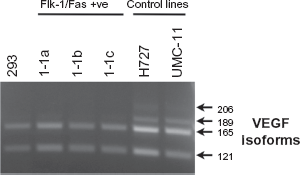
Expression and alternative splicing of VEGF mRNA in Flk-1/Fas resistant 293 tumor cell clones. A semi-quantitative RT-PCR approach was used to define both the expression and alternative splicing of VEGF mRNA in the Flk-1/Fas-positive 293 cell clones 1-1a, 1-1b and 1-1c and in the control tumor cell lines H727 and UMC-11. The primers used correspond to sequences present in VEGF exons 3 and 8 permitting each of the three major alternatively spliced VEGF isoforms to be amplified and independently characterized. After 25 cycles (94 °C for 30 s, 60 °C for 30 s, 72 °C for 1 min) products were separated on a 1% agarose gel, stained with ethidium bromide and photographed.
The oxygen-regulated protein 150 kD (ORP150) has been suggested to play a key role in regulating the secretion of VEGF in both normal 45 and malignant46,47 cells. ORP150 is a hypoxia-inducible chaperone protein “required” for intracellular transport of VEGF and other proteins from the endoplasmic reticulum (ER) to the Golgi apparatus prior to secretion. 48 Previous studies have shown that down-regulation of ORP150 caused VEGF to accumulate within ER while over-expression increased secretion of VEGF and accelerated tissue neovascularization in vivo.45,46 Although obviously important in some circumstances, Western blot analysis clearly demonstrated that ORP150 expression is not reduced in the two non-VEGF-secreting Flk-1/Fas-resistant tumor cell clones 1-1a and 1-1c identified above (Fig. 6). Indeed, densitometric analysis in which values were normalized against GAPDH expression indicated that the levels of ORP150 present in clones 1-1a and 1-1c were respectively 1.4 and 1.6 times higher than in control 293 cells. The level of ORP150 observed in the VEGF-secreting clone 1-1b was also elevated relative to control 293 cells (1.6 times higher) further arguing against a simple relationship between VEGF secretion and ORP150 levels. Why higher levels of ORP150 are generally seen in Flk-1/Fas resistant clones is unclear although it may be a consequence of the drug selection and/or cloning procedure used. Irrespective, these studies as a whole favor the involvement of post-transcriptional processes in the down-regulation of VEGF secretion by Flk-1/Fas resistant clones, although the precise mechanisms responsible for the effect remains elusive. Nevertheless, the availability of a sizable panel of isogenic Flk-1/Fas-resistant clones that differ in VEGF secretion may prove helpful in answering this question. It should be kept in mind, however, that in an in vivo setting down-regulation of VEGF may not be a viable means of escaping the cytotoxic effects of the Flk-1/Fas chimeras. Specifically, since VEGF is a key regulator of tumor angiogenesis, 49 tumor cells in which expression of the cytokine is lost or down-regulated may be unable to form the neovasculature necessary for the delivery of the oxygen and nutrients that are essential for continued tumor survival and growth.50,51 As such, they will remain localized and limited in size to the diffusion distance of oxygen, 52 unless another mediator can substitute for VEGF. 53
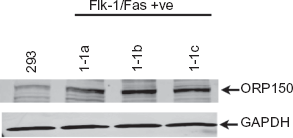
Expression of ORP150 in Flk-1/Fas resistant 293 tumor cell clones. Western blot analysis was used to determine the expression of ORP150 in the Flk-1/Fas-positive 293 cell clones 1-1a, 1-1b and 1-1c. Lysates were run on a 7.5% SDS-PAGE gel (∼2 × 105 cell equivalents per lane), transferred to nitrocellulose, and the membranes probed with rabbit polyclonal antibody directed against ORP150 or a control rabbit polyclonal antibody directed against GAPDH. After extensive washing, the blots were incubated with appropriate fluorescently-labelled secondary antibodies and the protein products visualized using a Odyssey Infrared Imager (LI-COR Biotechnology, Lincoln, NE).
The tumor clones that are most interesting from the point of view of future clinical studies are the small subset that co-express Flk-1/Fas and VEGF. Specifically, it was hypothesized that the cells from which these clones are derived may be defective in one or more of the proteins involved in the regulation of apoptosis. If so, it follows that if these proteins can be identified, then tumor cells could perhaps be screened prior to the initiation of therapy in order to identify those that might exhibit elevated resistance to the pro-apoptotic effects of Flk-1/Fas. Knowledge of the mechanism(s) by which cells escape killing by Flk-1/Fas also offers the prospect of improving the efficacy of the approach through the use of combination therapies that target the particular molecules involved. To this end, microarray analysis was carried out comparing the expression of key apoptotic genes in the three Flk-1/Fas-positive clones described above. Subsequently, Western blot analysis was used to confirm that alterations in gene expression evident at the RNA level translates to changes in the expression of the corresponding functionally important protein. An example of the data obtained in the course of these studies is shown in Figure 7. Thus, densitometric analysis confirmed near equal protein loading and suggested that clone 1-1b which co-expresses Flk-1/Fas and VEGF exhibited a >90% reduction in the expression of the pro-apoptotic protein Apaf-1. Other pro-apoptotic proteins including DFFA and Bnip3L are expressed at similar levels in all three lines. This particular finding was somewhat unexpected as Apaf-1 is involved in the Type 2 intrinsic/mitochondrial apoptotic pathway rather than the Type 1 extrinsic/Fas-mediated process (Fig. 8).54–56 Apaf-1 does, however, bind cytochrome-c, forming a large ternary protein structure known as the apoptosome that plays a key role in the activation of caspase 9 which in turn can activate various components involved in the control of extrinsic/Fas-mediated apoptosis including caspase 3 and caspase 7 (Fig. 8).57–60 Thus, there is compelling evidence that Apaf-1 can act via this mechanism to lower the signaling threshold needed to induce Fas-mediated death.61,62 It follows, then, that loss of Apaf-1 (as is the case in clone 1-1b) may increase the resistance of cells to Fas-induced death. Importantly, loss or down-regulation of Apaf-1 resulting in a disruption of normal apoptosome function has been documented in a number of tumor types suggesting that the molecule is a common target in development and pathogenesis of malignant disease.56,63,64
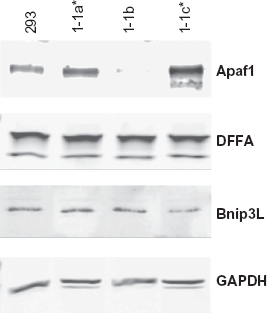
Expression of key regulators of apoptosis in Flk-1/Fas resistant 293 tumor cell clones. Western blot analysis was used to determine the expression of Apaf-1, DFFA and Bnip3 L in the Flk-1/Fas-positive 293 cell clones 1-1a, 1-1b and 1-1c. Lysates were run on 4%-20% (Apaf-1 and Bnip3 L) or 10%-20% (DFFA and GAPDH) SDS-PAGE gels (∼2 × 105 cell equivalents per lane), transferred to nitrocellulose, and the membranes probed with the appropriate rabbit polyclonal antibodies. After extensive washing, the blots were incubated with a fluorescently-labelled secondary antibody and the protein products visualized using a Odyssey Infrared Imager (LI-COR Biotechnology, Lincoln, NE).
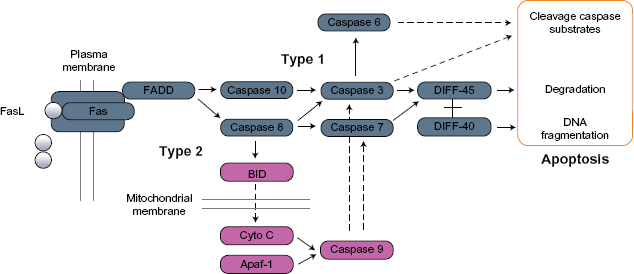
Role of Apaf-1 in the regulation of Type 1 and Type 2 apoptotic pathways.
Studies designed to investigate the impact of Apaf-1 loss on the growth of Flk-1/Fas transduced 293 cells and their response to therapy in vivo are currently underway.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributor-ship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
Footnotes
Acknowledgements
These studies were supported by grants from the University of Arizona, Women's Cancers Better Than Ever Program and the Arizona Biomedical Research Commission (Contract 0706). We gratefully acknowledge the technical support of Sean Walker and Kimiko Della Croce.
