Abstract
Psoriasis is a chronic inflammatory cell-mediated disease affecting skin and joints, histologically characterized by epidermal hyperplasia and abnormal differentiation of keratinocytes. Plaque psoriasis, which is the most prevalent form of the disease, is clinically characterized by raised, inflamed, erythematous lesions covered by white silvery scales, located on the elbows, knees, scalp, lower back, forearms, hands and feet. Several drugs are available for the management of moderate-to-severe plaque psoriasis, including topical and systemic treatments. Calcitriol (1,25 dihydroxyvitamin D3) is the most biologically active form of vitamin D3 and it can be used topically as both a primary and adjunctive therapy for psoriasis. Based on evidence that calcitriol inhibits proliferation and induces differentiation of epidermal cells, this hormone has been used successfully in the treatment of psoriasis. This article reviews the effectiveness and safety of calcitriol for the treatment of patients with psoriasis and recent findings related with the mechanism of action.
Introduction
Plaque-type psoriasis is a chronic skin disease, histologically characterized by epidermal hyperplasia and abnormal differentiation of keratinocytes. 1 Plaque psoriasis is clinically characterized by raised, inflamed, erythematous lesions covered by white silvery scales, located on the scalp and extensor surfaces.
Several studies have identified a dysregulated interplay between keratinocytes and infiltrating immune cells underlying cutaneous inflammation in psoriasis. 2 The current understanding of the molecular pathogenesis of psoriasis assigns central importance to an interaction between immune cells and keratinocytes. 3 Antimicrobial peptides (AMPs), dendritic cells, and T cells (Th1, Th17) secrete mediators that contribute to the vascular, epidermal changes, keratinocyte proliferation and abnormal differentiation of psoriasis through the activation of intracellular signal transduction pathways.3,4 In particular, recent publications highlight the role of dysregulated expression of AMPs in the pathogenesis of psoriasis. Human beta-defensins and cathelicidin, as well as psoriasin and other AMPs, are strongly increased in keratinocytes in psoriatic plaques.4–7 Peric et al 8 have recently demonstrated that treatment of lesional psoriasis skin with vitamin D3 derivatives normalizes the cutaneous phenotype and at the same time changes the AMP expression profile decreasing epidermal proliferation.
During the last decade the topical application of vitamin D3 derivatives has become one of the main therapies in the routine treatment of chronic plaque psoriasis. 9 Topical vitamin D analogues are used as both as a primary and adjunctive therapy for psoriasis and include: calcipotriol, maxacalcitol, tacalcitol and calcitriol.10–13 Calcitriol (1,25 dihydroxyvitamin D3) is the most biologically active form of vitamin D3. 14 It is known to inhibit proliferation and stimulate differentiation of keratinocytes and also to have immunomodulatory activity. 14
In patients affected by severe psoriasis the therapeutic effect and tolerability of vitamin D3 on psoriasis may be increased by its combination to phototherapy (e.g. UVA, psoralen plus UVA, narrow-band UVB, 308 nm monochromatic excimer light or laser) or systemic treatments (biologicals, methotrexate and cyclosporine). 15 Moreover in patients contraindicated to systemic therapies, topical treatments remain a valid option as a monotherapy. The effectiveness of topical vitamin D3 analogues in treating moderate psoriasis is well documented.10–17 This review article explores the characteristics of calcitriol in terms of effectiveness and safety for the treatment of patients with moderate to severe psoriasis.
Mechanism of Action and Metabolism Profile
Vitamin D3 or cholecalciferol, together with parathyroid hormone and calcitonin, is one of the major controllers of calcium and phosphorus homeostasis in the body, by regulating the uptake and release of calcium in the intestine, bone and kidney. Vitamin D is produced in the skin by UVB radiation (290-320 nm). The active metabolite 1,25-dihydroxyvitamin D3 [1,25(OH)2D3] or calcitriol results from hydroxylation of vitamin D in liver and kidney, but also locally in the epidermis, which suggests that 1,25(OH)2D3 may have important functions in the skin (Fig. 1). 18 Human epidermal keratinocytes are the only cells capable of performing the complete synthesis of 1,25(OH)2D3 from 7-dehydrocholesterol. 19 1,25(OH)2D3 is the active metabolite of vitamin D3 influencing the proliferation and differentiation processes. 20
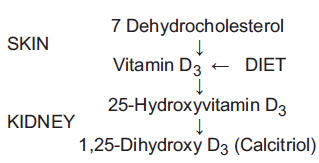
Metabolism of calcitriol.
Several studies demonstrated that tumour cells derived from a variety of different sources have shown to express the vitamin D3 receptor (VRD) and to be susceptible to growth arrest and/or cell death in response to vitamin D3 and its analogues.21–23 Calcitriol exerts a potent antitumoregenic effect by inducing apoptosis, differentiation and cell growth inhibition through VDR, which belongs to the super-family of steroid/thyroid hormone nuclear receptors.21–24 Physiological and pharmacological actions of calcitriol, along with the detection VDR in target cells, have indicated other potential applications of VDR ligands in inflammation, dermatological indications, osteoporosis, and autoimmune diseases. 24 In human primary keratinocytes several genes, growth factors and proteins involved in metabolism, components of the extracellular matrix, as well as proteins involved in DNA repair, apoptosis, and immune response are regulated by 1,25(OH)2D3. The majority of the target genes are involved in intracellular signaling, transcription, and cell cycle. 24 Calcitriol binds nuclear VDR in various cells involved in the pathogenesis of psoriasis such as keratinocytes, dermal fibroblasts, macrophages and T cells. The activated VDR then binds regions of DNA known as vitamin D response elements which are characterized by two direct repeated hexanucleotide half-sites. The activated VDR forms a heterodimer with another molecule, the retinoic X receptor (RXR), which is necessary for genomic vitamin D activity. This sequence of molecular events leads to changes in cell proliferation, differentiation, and production of inflammatory cytokines. 24
The use of vitamin D derivatives for the treatment of plaque psoriasis is well documented and shows discrete tolerability and effectiveness. The utility of vitamin D3 for the treatment of psoriasis was discovered by accident when a psoriatic patient who was receiving vitamin D as osteoporosis therapy showed clearing of her skin lesions.
25
The mechanism of action of calcitriol in the treatment of psoriasis has not yet been established while other vitamin D analogues have been shown to exert their primary action on keratinocytes, through inhibiting proliferation and stimulating differentiation.
23
Vitamin D derivatives exert a whole range of effects on the disease process such as regulation of epidermal hyperproliferation, enhancement of normal keratinization, inhibition of T helper 1 activities, induction of Th1 to Th2 switch, inhibition of neutrophil functioning, modulation of monocyte functioning, induction of apoptosis in inflammatory cells and angiogenesis inhibition, which may support the rationale of their utilization in the treatment of this immune-modulated skin disease
19
Moreover, it is well known that TNF-α plays a key role in the pathogenesis of psoriasis; calcitriol seems to prevent TNF-α induction of inflammatory cytokines through a process likely to be mediated by the vitamin D receptor. 30 Finally, Peric et al 8 showed that topical treatment with vitamin D3 derivatives significantly decreased cutaneous levels of human beta-defensins as well as IL-17 and IL-8.
Clinical and Safety Assessment
In the development and assessment of new therapeutical modalities for psoriasis, a clear definition of a patient's psoriasis severity is essential. The impact of a given therapy can only then be evaluated, based on the changes in the severity score during and after application of the treatment.
Clinical efficacy criteria, during trials conducted to evaluate the efficacy of vitamin D analogues, are psoriasis area severity index (PASI), body surface area (BSA), the psoriasis global assessment (PGA) which are usually documented at each visit. 31
BSA refers to the percentage of area affected by psoriasis. BSA is calculated using the rule of nine and the palmar side of the patient's hand as equivalent to 1% of BSA. In general, a BSA under 5% indicates mild psoriasis, moderate is between 5% and 10%, and over 10% is considered severe. 32
PASI scale is the clinical scoring system most commonly used to assess disease severity in clinical trials. To evaluate PASI score the body is divided into four sections and severity is measured by four different parameters: itching, erythema (redness), scaling and thickness. Again, each of these is used separately for each skin section. PASI score can vary from 0 to 72, with higher scores indicating more severe conditions. In chronic plaque psoriasis, a PASI under 8 is designated as mild, between 8 to 12 is moderate, and over 12 is considered severe. 33 Similarly, the target lesion score (TLS) is a prospectively identified psoriatic lesion assessed for plaque elevation, scale, erythema, and induration.
Beside PASI and BSA, the PGA is the system most often employed in clinical trials to measure patient's overall severity of disease; typically, a 7-point scale from clear to severe is used, although many variations have been employed. 31
Moreover, safety assessment includes general haematology and clinical chemistry. In particular, special focus on the parameters of calcium homeostasis as it follows: serum calcium; parathyroid hormone; spot urine: calcium creatinine and calcium/creatinine ratio; and, in some centres, urinary α1-microglobulines, serum calcitonin and serum calcitriol. 34
Safety was determined from reports of adverse events and blood chemistry parameters (including serum albumin, total and adjusted calcium, urea, phosphate, alkaline phosphatase and creatinine), measured at each assessment point.35–42
Clinical Studies
Several studies designed to assess the efficacy and safety of calcitriol 3 μg/g ointment are reviewed here (Table 1). Treatments were applied to all psoriatic lesions for the duration of the study or until clearing occurred. Some of these studies compared calcitriol with other topical treatments for psoriasis whereas other studies investigated the combination of calcitriol with other therapies.35–42
Published clinical trials on the treatment of plaque psoriasis with calcitriol.

Some reported clinical trials have also demonstrated the safety, tolerability and efficacy of calcitriol ointment for the treatment of plaque psoriasis on “sensitive” areas (face, hairline and retroauricular, axillary, inguinal, submammary, popliteal areas) compared to other formulations.35–42
Comparison with Short-Contact Dithranol
Hutchinson et al
35
compared the efficacy, safety and tolerance of a twice-daily application of calcitriol 3 μg/g ointment with dithranol cream in a 8-week, prospective, randomized, parallel-group trial. Anthralin or dithranol is a synthetic compound with a tricyclic structure, used for the treatment of psoriasis. Dithranol inhibits cell proliferation
Combination and Sequential Therapy of Calcitriol Ointment and Clobetasol Propionate Cream
In a two-phase parallel group study conducted by Lahfa et al, 36 calcitriol was compared with the vitamin D analogue calcipotriol in 125 patients with chronic plaque-type psoriasis. The proposed treatment regimen was an initial bitherapy for 2 or 4 weeks, with clobetasol propionate 0.05% cream (a super potent topical corticosteroid) applied in the morning and either calcitriol 3 μg/g ointment or calcipotriol 50 μg/g ointment applied in the evening, followed by monotherapy with either calcitriol or calcipotriol applied twice daily until week 12. Efficacy evaluations showed no significant differences between the two regimen groups at the primary endpoints (week 2 and week 12) or at any interim points. At week 2 the investigator's global assessment showed clinical success for more than 50% of the patients in both groups at week 12. There were no significant differences between the two groups with regards to cutaneous safety or to incidence of adverse events. This study showed that for the treatment of moderate plaque psoriasis calcitriol 3 μg/g ointment can provide a safe and effective alternative to calcipotriol 50 μg/g ointment while being administered within a regimen based on a combination therapy with corticosteroids followed by a vitamin D3 maintenance monotherapy.
Comparison with Calcipotriol
Ortonne et al 37 compared the safety and efficacy of calcitriol 3 μg/g ointment and calcipotriol 50 μg/g ointment in a multicentre, randomized, investigator-blinded, left-right comparison in mild to moderate chronic plaque psoriasis affecting sensitive areas: face, hairline, retroauricular and flexural areas. In the 75 subjects, both treatments led to clearing of at least one target lesion in 28% of the subjects each. Perilesional erythema, perilesional edema, stinging and burning were all significantly less severe with calcitriol than with calcipotriol. Similarly the subjective evaluation of local tolerability and global preference were in favor of calcitriol. Global assessment of improvement from baseline by the investigators was significantly greater for the calcitriol-treated lesions.
Zhu et al 38 conducted a multicenter, randomized, investigator-masked, and parallel comparison in subjects with mild to moderate chronic plaque-type psoriasis receiving either calcitriol or calcipotriol ointment twice daily for 12 weeks. A total of 250 subjects of both gender were recruited. At week 12, calcitriol demonstrated to be non-inferior to calcipotriol for global improvement. The mean worst score for the cutaneous safety assessment was higher in the calcipotriol group. These differences were statistically significant in favour of calcitriol. In conclusion, concerning this study calcitriol administered twice daily over a 12-week period demonstrated similar efficacy to calcipotriol, while showing a significantly better safety profile.
Comparison with Tacrolimus Ointment
A double-blind, parallel, 6 weeks-study conducted by Liao et al 39 compared the tolerability and efficacy of calcitriol 3 μg/g and tacrolimus 0.3 mg/g ointment in chronic plaque psoriasis affecting facial and genitofemoral regions. Fifty patients with moderate and severe psoriasis were included. The efficacy was measured using TLS and PGA. Both calcitriol and tacrolimus were well tolerated. Although calcitriol induced perilesional erythema in a statistically significant higher proportion of patients than tacrolimus, it did not necessitate treatment discontinuation. At the end of the study, tacrolimus was significantly more effective than calcitriol based on a significant reduction of mean TLS as well as more patients achieving complete or almost complete clearance by PGA.
Combination with Ultraviolet B Phototherapy
In a randomized and double-blind study Ring et al 40 compared calcitriol ointment with UVB vs with phototherapy alone for 8 weeks. One hundred and four patients with moderate psoriasis were included in the study: 49 and 53 patients in the calcitriol and vehicle groups, respectively. Efficacy was measured using global severity and PASI scores while safety was determined from reports of adverse events and blood chemistry analysis. At the end of the study, considerable improvement or clearance of lesions was seen in 45% of patients treated with calcitriol plus UVB and in 20% of those receiving UVB alone. The incidence of skin related adverse events was low and similar in the two treatment groups. No clinically significant changes in blood chemistry occurred.
Long-Term Studies of Topical Calcitriol
In an open-design and multicenter study, Gerritsen et al 41 investigated the topical and systemic safety and efficacy of twice-daily application of 3 μ/g calcitriol ointment in the long-term treatment of patients suffering from chronic plaque psoriasis. Efficacy was measured using PASI score while safety was determined from reports of adverse events and blood chemistry analysis. Two hundred and fifty-three patients treated all their psoriatic lesions, except for those on the head and scalp, for up to 78 weeks. No serious events were reported; 37/253 patients had a transient skin irritation reaction during the study that resulted in study withdrawal for seven of them. The baseline/endpoint analyses showed no clinically relevant changes in measures of calcium and phosphorus homeostasis and renal function. Slight hypercalcemia was observed in five patients and in four of those, serum albumin-adjusted total calcium levels normalized during treatment. In this study 88 patients withdrew: the 67% of those for reasons of “inefficacy”.
Personal Experience
In our experience 42 conducted at the dermatology department of “Tor Vergata” University of Rome, we confirmed the data above and we indicated that calcitriol 3 μg/g ointment, applied twice daily, is an effective alternative treatment for chronic psoriatic plaques, which involve also sensitive areas, resistant and/or intolerant to other topical therapies. In fact, 60 patients (39 males and 21 females, aged 10-74 years, median age 47.4) affected by mild-to-moderate plaque psoriasis with a PASI score ranging from 2.5 to 15 (Mean PASI: 8.3) were treated with calcitriol 3 μg/g ointment for 12 weeks. The results of this clinical trial showed 73.6% of complete remissions (PASI 75) and 22.6% of partial remission (PASI 50) in a 12 week time-frame without the occurrence of serious adverse events during the therapy. Mean PASI at week 4 was already reduced to 6.3, at week 8 to 4.5, and at week 12 it was progressively reduced to 2.4. During a follow-up period of 6 months, flares occurred gradually after 3 months (7/39 patients) and 6 months (31/39 patients). Indeed the ointment was discretely tolerated to the extent that the side effects (pruritus in approximately 20% of cases) disappeared spontaneously in 50% of the patients throughout the therapy. The baseline/endpoint analyses showed no significant changes in measures of calcium and phosphorus homeostasis. Exclusion criteria for the treatment with calcitriol 3 μg/g ointment were: body surface involvement >35%, pustular psoriasis, erythrodermic psoriasis, guttate psoriasis, history of hypercalcemia, renal dysfunction and calcium-based calculi, any condition that requires systemic intake of calcium or vitamin D supplements and pregnancy. In conclusion, according with our experience, calcitriol 3 μg/g ointment is a safe and effective choice worthy of consideration for the treatment of psoriasis vulgaris that involves <35% of the body surface and also sensitive sites. The low skin irritancy of this vitamin D3 analogue had suggested its use on sensitive areas (e.g. face, hairline, retroauricular area and folds) thus increasing patient's acceptability, quality of life and attitude towards such a life-altering chronic disease.
Calcitriol in Psoriatic Children
The available therapies for childhood psoriasis are similar to adult drugs, although in the pediatric age several adverse reactions occur and contraindications should be considered. Since most of the available systemic therapies, currently used for adult population, are not indicated for children, topical treatments remain the first choice in children affected with moderate to severe plaque type psoriasis. Among topical medications, steroids are considered the most useful treatment in clinical practise. Topical corticosteroids present a wide range of biological effects such as inhibition of the recruitment and migration of inflammatory cells, modulation of cytokines, chemokines release, and regulation of the DNA synthesis. 43 However, especially in childhood, corticosteroids have their own limitations including atrophy, telangiectases, and striae. Calcitriol represents a safer alternative for a long-term therapy. Its efficacy and safety is well-established in children especially in plaque type psoriasis and guttate psoriasis.44,45 Saggese et al 44 reported the effects of calcitriol ointment topically applied on skin lesions in children affected by psoriasis vulgaris. Each patient was instructed to apply 1 g of ointment once a day at bedtime followed by occlusive dressing with plastic wrap, on one-side lesion (treated) and petrolatum alone on the equivalent controlateral site (placebo). After 4 weeks of topical treatment all children showed a complete clearing of their skin lesions on the treated side, without appreciable changes on the placebo side. Serum ionised calcium, calcium and calcitriol levels or urinary calcium excretion did not vary significantly throughout the period of therapy. These findings suggest that topical calcitriol may be an effective and safe alternative therapy for psoriasis in children.
Nail Psoriasis Treated with Topical Calcitriol
Nail psoriasis may vary from slight to invalid forms, with functional and psychosocial impairments as well as severe impact on the patient's quality of life. The nail involvement is generally associated with pain, restrictions in daily life activities and the visibility of fingernail disease may hinder social and business interactions. Topical treatment is unsatisfactory and includes intralesional corticosteroids, tazarotene, 5-fluorouracil, and urea.
Usmani et al 46 reported a case of a 38-year-old woman with fingernail psoriasis. The patient was prescribed calcipotriol cream 50 μg/g to the nail fold twice daily. Within the first few days of application, the patient complained of skin irritation, and her prescription was changed to calcitriol ointment 3 μg/g to be used twice daily to the proximal and lateral aspects of the nail fold. Two months later the patient's fingernails showed dramatic clinical improvement. The nails were half grown out, and the new nail growth was considerably better than the previously irregular nails. Six months later a further improvement was observed. However, at 6 month-follow-up onycholysis, hyperkeratosis and pitting reoccurred.
Safety
The level of tolerance to calcitriol 3 μg/g ointment twice-daily was good in the reported studies. Mild to moderate skin irritation and asymptomatic hypercalcemia have been reported, but cases of discontinuation are uncommon. However, in all cases these changes were transient and serum calcium levels returned to normal during continued treatment. Hypercalcemia has also been reported with other topical vitamin D analogues such as tacalcitol or 22-oxacalcitriol.47,48 In the latter a severe acute pancreatitis was induced by hypercalcemia which developed as a reaction to ointment containing 22-oxacalcitriol, a vitamin D3 analogue. 47 Calcitriol has a higher affinity for the vitamin D-binding protein when compared to calcipotriol, resulting in its 100-fold higher effect on calcium metabolism.
There were no clinically relevant individual changes or statistically significant mean changes from baseline in the laboratory parameters recorded.35–42
The treatment of psoriasis with a corticosteroid alone is accompanied by several drawbacks and side effects, such as cutaneous atrophy, formation of telangiectases and striae. These side effects may be reduced while maximizing the beneficial effects in a concurrent administration with a vitamin D3 analogues. The study conducted by Lahfa et al 36 confirmed the efficacious and safe role of vitamin D3 and corticosteroids as a first line therapy and underlined the safe maintenance effect of vitamin D3 derivatives as a maintain monotherapy in the treatment of psoriasis. Lahfa et al 36 in their study also show that for the treatment of moderate plaque psoriasis calcitriol 3 μg/g ointment can provide a safe and effective alternative to calcipotriol 50 μg/g ointment while being administered within a regimen based on a bitherapy with corticosteroids followed by a vitamin D3 maintenance monotherapy.
In the study conducted by Hutchinson et al 35 calcitriol 3 μg/g ointment was found to be better tolerated than short-contact-dithranol ointment. In the two different studies conducted by Ortonne et al 37 and Zhu et al 38 perilesional erythema, perilesional edema and stinging/burning were all significantly less severe with calcitriol than with calcipotriol. In our experience 42 mild irritation and pruritus occurred in 12/60 subjects that disappeared spontaneously in 50% of subjects concerned.
Liao et al 39 confirmed the tolerability of calcitriol 3 μg/g ointment in the treatment of psoriasis on sensitive areas like the face and genitofemoral folds. Finally, concerning the long-term treatment Gerritsen et al 41 demonstrated that twice-daily calcitriol 3 μg/g ointment can be used long term without significant adverse effects.
Conclusions
This review highlights that topical calcitriol is an effective treatment for the management and control of psoriasis. The efficacy of topical calcitriol 3 μg/g ointment when used for short-term treatment of patients with chronic plaque psoriasis is now well-established. Many psoriatic patients have lesions on very sensitive skin (such as the face, hairline, retroauricolar areas and flexural areas). The use of topical steroids on these areas is then limited by their side-effects and synthetic vitamin D derivatives are not recommended because of their high irritancy potential. Several studies strongly suggest that calcitriol would be an adequate therapy for these sensitive areas affected by chronic plaque psoriasis and in which other vitamin D3 derivates could be irritant.
Topical corticosteroids and vitamin D3 analogues are the treatment of choice for mild to moderate psoriasis. Combination and sequential therapy is known to be superior to monotherapy and is commonly used. By combining medications with a different mechanism of action and safety profile, efficacy can be enhanced and/or safety improved. In our experience we observed that calcitriol seems to be essential to maintain the results achieved with topical steroid and to avoid rebound effects.
The effective management of psoriasis frequently necessitates combining therapies in order to achieve optimum response while minimizing any side-effects. Calcitriol can be safety and effectively used in combination with UVB phototherapy. The combination with calcitriol resulted in a significant reduction in the dose of UV radiation needed to produce an equivalent therapeutic response. In our experience we observed the role of adjunctive topical calcitriol treatment when used in combination with systemic therapies for the treatment of chronic plaque psoriasis.
Calcitriol can be used adjunctively for resistant lesions in patients with more extensive psoriasis undergoing therapy with either phototherapy, systemic or biological medications. The efficacy of a systemic treatment can be enhanced by them with vitamin D3 derivatives. This point is extremely important in patients affected by psoriatic arthritis. Although, several systemic therapies including methotrexate, leflunomide, and disease-modifying drugs are effective in the treatment of the joint component (psoriatic arthritis), only few patients experience a rapid and beneficial effect on skin psoriatic lesions. In this way, calcitriol can be combined to enhance efficacy in those patients who are improving but still have areas of active disease. This is especially important for the management of psoriasis on sensitive areas in which corticosteroids are contraindicated.
A treatment strategy that may be used for some patients with more extensive or severe psoriasis involves the use of topical therapy in combination with other treatments in an effort to achieve optimum control of disease.
Disclosures
The authors report no conflicts of interest.
