Abstract
Kambô is an Amazonian ritual which includes the application of the defensive secretion of the Phyllomedusa bicolor frog to superficial burns made on the skin of human participants. The secretion, which contains a range of biologically active linear peptides, induces a short purgative experience that is extensively reported by participants to leave them with positive physical, emotional and spiritual after-effects. Various peptides identified in the secretion exert analgesic, vascular, and gastric effects in vivo, and antimicrobial and anti-cancer effects, among others, in vitro. While there has been some investigation into the physiological effects of various individual peptides isolated from the P. bicolor secretion, very little is known about the putative synergistic effects of concurrent administration of the complete substance through the transdermal methods used traditionally in the Kambô ritual. In this review and commentary, the authors summarize the existing biological information from animal research on peptides from the P. bicolor secretion, then consider the evidence in the context of Kambô administration to humans. The presented information suggests that specific peptides are likely to contribute to analogous physiological effects of Kambô in humans. The possibility that beyond their physiological action, the experiential or phenomenological component of these effects may have therapeutic applications is discussed, concluding with a consideration of the feasibility of human clinical research.
Highlights
• Kambô-derived peptides exhibit analgesic, vascular, and gastric effects in vivo, and antimicrobial and anti-cancer effects in vitro. • The diverse peptide components of Kambô may have synergistic effects when administered concurrently in humans. • Kambô toxins have different structural properties compared to other animal venom peptides. Kambô peptides present certain health risks in some susceptible individuals, although they may offer significant therapeutic potential in appropriately screened recipients
Introduction
The numerous biologically active peptides contained in the skin secretions of the Amazonian Giant Monkey Tree frog, Phyllomedusa bicolor, exert diverse physiological effects on the human body when administered via small epidermal burn wounds as is standard practice in both traditional Amazonian Indigenous and Western neoshamanic Kambô practice (Figure 1).
1
(A) Kambô comes from the skin secretion of the Phyllomedusa bicolor or Giant Monkey Frog; (B) The frog is tied to harvest the secretion by gentle scraping; (C) The Kambô is prepared by reconstituting with water on a bamboo stick; (D) Kambô is applied to the superficial burns made on the skin; (E) “Dragon’s blood” sap from Croton lechleri is applied to burns post-Kambô as an antiseptic.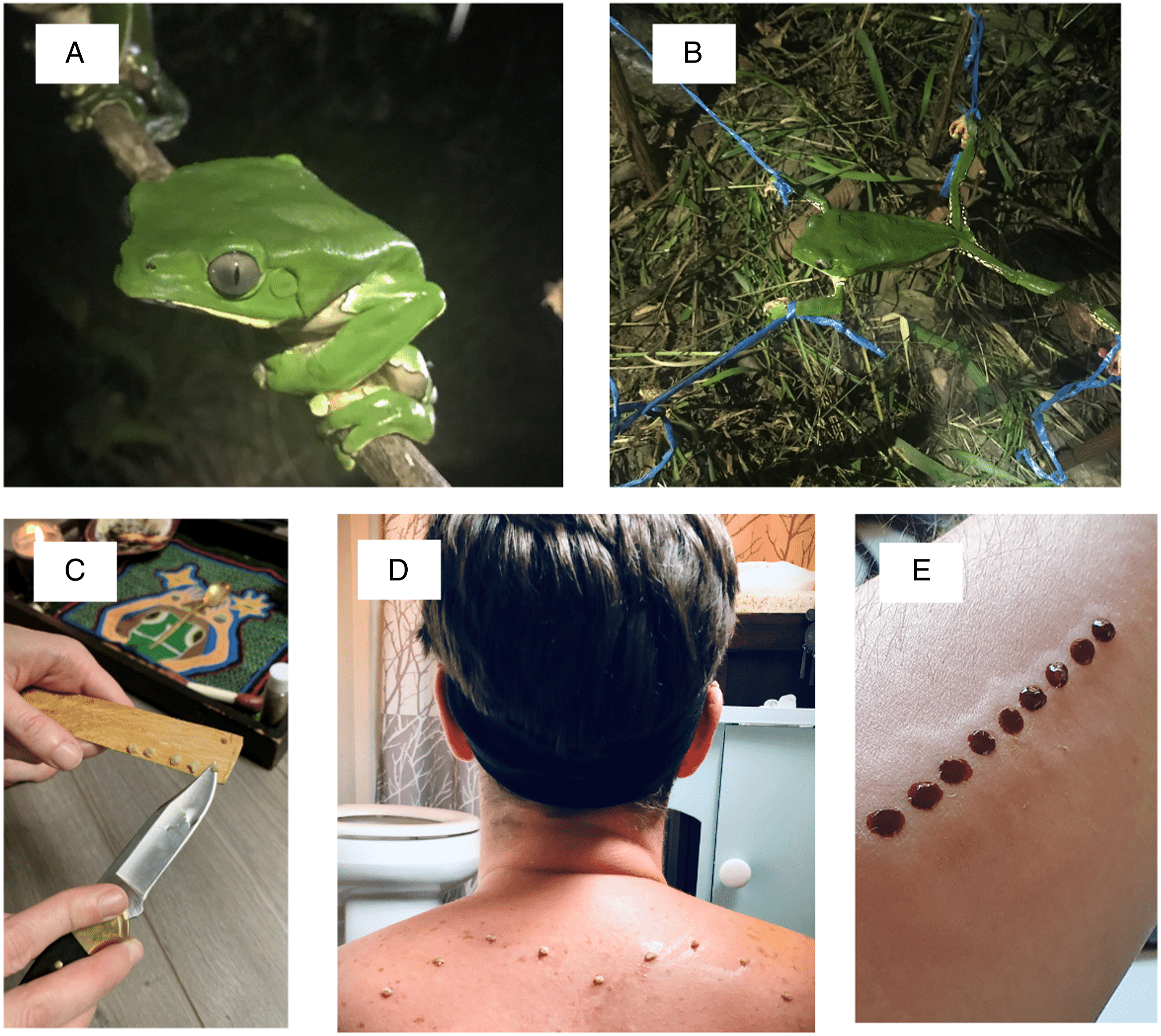
The amounts of bioactive peptides present in Kambô have been measured to be approximately 7.5% by dry weight, which hence enabled estimation of the amounts entering the body via transdermal application. 2 For one burn (dot or gate, as it is known by Kambô practitioners), the amount of Kambô applied has an approximate weight of 10 mg, meaning a standard dose of total peptides would be 30–50 mg via 3–5 gates. A single application of five points typically might contain phyllocaerulein (30–35 µg/mg): 1600 µg, phyllomedusin (22 µg/mg): 1100 µg, phyllokinin (18 µg/mg): 720 µg, sauvagine (3 µg/mg): 150 µg, deltorphins (5.3 µg/mg): 165 µg, and dermorphins (0.3 µg/mg): 15 µg, along with trace amounts of many other peptides. If Kambô is harvested from a P. bicolor frog that has already been milked in the recent past, lower concentrations of these peptides may be obtained. Furthermore, there might be differences in composition between Kambô harvested from a male or a female frog. 2
While there is a fair amount of literature on the biochemistry and physiological effects of various individual peptides isolated from the skin secretions of Phyllomedusa bicolor, there have been virtually no human or animal studies on the clinical outcomes of receiving Kambô in the traditionally applied way. Erspamer, one of the first researchers to isolate and categorize many of the peptides from P. bicolor, speculated that synergistic influence on physiology may be exerted by the combined administration of the peptides. 3 However, aside from a handful of case reports describing adverse effects, very little of the literature has explored the transdermal application of the whole Kambô secretion containing its diverse cocktail of peptides.
The Kambô experience
Since the introduction of Kambô to the western world in 1986 by anthropologist Peter Gorman, there has been increased use of Kambô as an alternative therapy for depression, anxiety, chronic pain, autoimmune disorders, PTSD (Post Traumatic Stress Disorder), Lyme disease, chronic and acute infections, drug addiction, and other physical and mental health conditions.4–6 There is a wide variety of styles of practice surrounding the administration of Kambô. Generally, the substance is applied to superficial burns made on the skin using a Titica vine (Heteropsis flexuosa) on the upper arm, spine, or lower leg. Traditionally, there is virtually no ceremonial practice amongst the Indigenous tribes that use Kambô. In Kambô rituals catering towards Westerners, the practice is often carried out in a ceremonial context, with the presence of songs, musical instruments, burning of incense, and prayers. Other traditional medicines such as Rapé (a tobacco snuff containing Nicotiana rustica) and Sananga (botanical eyedrops from Tabernaemontana undulata) are often administered in proximity to the Kambô.
Physiological effects occur rapidly following introduction of the peptides into the body. The effects are complex and sometimes paradoxical. Typically, the first symptoms reported are an initial rush of heat sensation and redness of the face. Nausea and vomiting are often experienced within several minutes, accompanied by general malaise, racing heart, dizziness, edema of the face, and sometimes an urge to defecate. In summary, the response after application generally consists of sudden feelings of warmth, palpitations, rapid pulse, flushed red skin or paleness, the feeling of a lump in the throat or difficulty swallowing, abdominal pain, nausea, vomiting, diarrhea, runny nose and tears, swollen lips, eyelids or face, and occasionally a swollen tongue or throat. The duration of the entire symptom complex is usually 5–30 min; very rarely, it may last for several hours. 7
In the medical literature, the responses listed above are sometimes referred to as “adverse events” of an anaphylactic nature. Other writers, however, note that this categorization as adverse events is inaccurate, and that the physiological effects are correctly described as the normal physiological responses of the human body to the Kambô peptides through the action of several receptor types to which the various peptides bind strongly and selectively. 7 Importantly, these physiological effects are generally expected and indeed sought by those performing and undergoing the Kambô procedure, which is also referred to in this review as an experience or ritual. It is equally important to note that physiological reactions that constitute true adverse events can, and occasionally do, occur. Due to lack of documentation, the prevalence of Kambô use worldwide is difficult to determine with any certainty, although estimates in the practitioner community range from 50,000 to 100,000 administrations per year. 8 Consequently, the statistical likelihood of an adverse event is difficult to calculate. A handful of documented cases of harms have been associated with Kambô participation; case reports describe hepatitis, psychosis, prolonged vomiting, hyponatremia, seizure, esophageal rupture, and cardiac arrest incidents associated with Kambô use.9–13
Despite these documented harms, many users of Kambô anecdotally report positive physical, emotional, and spiritual after-effects.
Peptide composition of Kambô
The broad range of small (700–4600 Da) 14 biogenic peptides identified in the secretions of various cutaneous glands in frogs of the Hylidae family, including Phyllomedusa bicolor are believed to provide the frogs with defenses against predators in their natural habitat.2,14–17
Much of the existing literature describes these amphibian peptides in reference to several frog species or families within the Phyllomedusae genus and other genera in the Hylidae frog family. Very few papers comprehensively present information that is species-specific to the Phyllomedusa bicolor frog in the context of peptide exposure from Kambô.
Gastrointestinal peptides
Phyllocaerulein is a 10-residue peptide with caerulein-like pharmacology. 21 Sauvagine is a peptide consisting of 40 amino acids 22 By dilating peripheral blood vessels, both caerulein and sauvagine peptide classes elicit hypotension and consequently reflex tachycardia.21–25 Additionally, through their action on smooth muscle, they cause contraction of the stomach, gall bladder, colon, and urinary bladder.21,23–25
Early gastrointestinal symptoms of nausea and vomiting during the Kambô experience are possibly predominantly attributed to phyllocaerulein and sauvagine, which are also known to be potent stimulators of gastric and pancreatic secretions and to have anorectic properties. 21 Caeruleins and sauvagines both appear to stimulate the adrenal cortex and pituitary gland, which may explain the reported sensation of increased strength and stamina experienced during the later phase of Kambô intoxication.21,22
Phyllocaerulein
Caerulein and phyllocaerulein, a caerulein-like peptide, are both found in Kambô. Phyllocaerulein is virtually indistinguishable from caerulein in bioassay and physiological effects, but appears to be somewhat more potent than caerulein when applied to tissue preparations and in vivo animal models. 21 In one study, in vivo treatment with phyllocaerulein resulted in a prolonged fall of blood pressure in the dog, contraction of the gall bladder of the guinea-pig and of the ileum of the dog, stimulated gastric acid secretion in the dog and the rat, and stimulated pancreatic secretion in the dog. 21
Caerulein has also been identified from other anuran (frog) skin extracts and found to elicit a number of effects including analgesia, changes in mood, sedation, satiety, and antipsychotic effects. 24 One study showed that caerulein was far more potent in reducing locomotor activity in rats compared to the antipsychotic, haloperidol, and morphine. 26 Some studies have suggested that caerulein may be useful in the treatment of schizophrenia, but there are conflicting results about the antipsychotic effects. 27
Caerulein’s stimulating effect on the gall bladder and gastrointestinal preparations appears to be mediated by acetylcholine and was inhibited by morphine and other opioid-like peptides. Caerulein has been found to upregulate I-CAM1 cells via the Nf-kB pathway, promoting neutrophil adhesion onto acinar cells and enhancing pancreatic inflammation. 25 Because of its pancreatitis-inducing effect, caerulein analogs have been used to develop pancreatitis models in research. 25 The pancreatic effects were only present in exaggerated doses significantly exceeding pharmacological doses, and appeared to be reversed upon discontinuation of the drug. 23 Despite its temporary and reversible inflammatory effects on the pancreas, caerulein shows extremely low acute and chronic toxicity in rats, mice, rabbits, and dogs. 23
Given its indistinguishable effects from caerulein, phyllocaerulein may promote similar physiological and behavioral effects. 21 This implies that, like caerulein, opioid-like peptides such as deltorphins and dermorphins may have allosteric effects on the activity of phyllocaerulein when an individual receives the entire peptide cocktail present in Kambô. It also implies that phyllocaerulein/caerulein may prompt temporary inflammation of the pancreas, making Kambô unsuitable for individuals with active pancreatic conditions.
Sauvagine
Like phyllocaerulein, sauvagine has been observed in rat studies to produce a prolonged drop in blood pressure. 22 Sauvagine also has been observed to have anti-diuretic effects in rats.22,28 In rats, sauvagine increased the plasma release of corticosterone, adrenocorticotropic hormone (ACTH), catecholamines, glucose, and β-endorphins in the brain. 28 The increased plasma corticosteroid levels and the β-endorphins may both contribute to its analgesic properties.
In in vitro and in vivo rat studies, sauvagine had effects on the anterior pituitary, which resulted in inhibition of release of thyroid stimulating hormone (TSH), prolactin (PLR) and Growth Hormone (GH). 28 Inhibition of the release of TSH was also observed in dog models. 28 10 µg sauvagine administered to rats resulted in marked inactivity and behavioral depressive effects. 1 Other behavioral observations in rats receiving intracerebroventricular injections of sauvagine included decreased motor response and emotionality, increase in grooming behavior, reduction in food intake, and decrease in rearing. 3
Like other peptides contained in Kambô, sauvagine is susceptible to degradation by chymotrypsin. However, unlike phyllocaerulein, phyllomedusin, phyllokinin, and bombesin-like peptides, it is also susceptible to inactivation by trypsin. 22 Sauvagine appears to be one of the few known natural active polypeptides that does not contain any aromatic amino acid residues. 28 It has significant similarity in structure and biological activity to the peptides, urotensin I and corticotropin-releasing factor (CRF). 3
The presence of sauvagine and its antidiuretic effects may protect against the risk of hyponatremia in Kambô participants by reducing the absorption of water by the stomach and intestines and increasing the reabsorption of Na+ and K+ in the kidneys. 22 More research is needed to determine the implications of antidiuretic effects on plasma electrolyte levels in humans. It is possible that sauvagine is also responsible for some of the behavioral changes such as reduced emotional reactivity, increased social connection, and suppression of appetite that one may experience after participating in a Kambô experience. 3
Bombesins
Bombesins stimulate the secretion of gastric acids and increase pancreatic secretion, intestinal myoelectric activity, and smooth muscle contractibility. Phyllolitorins are bombesin-like peptides found in the skin of Phyllomedusa bicolor species and include litorin, phyllolitorin, [Leu"]-phyllolitorin, and rohdei-litorin. 3 There is contradicting information about whether bombesins are present in the Kambô substance, but an analysis in 2017 of a Kambô sample identified bombesin dihydrochloride, bombesinnona peptide, and phyllolitorin, suggesting that bombesin and related peptides are present in trace amounts in the P. bicolor secretion. 15
Bombesin and bombesin-related peptides closely resemble two mammalian peptides, gastrin-releasing peptide (GRP), and neuromedin B (NMB), which are widely distributed in the central and peripheral nervous system and intestine of mammals. 29 These bombesin-related mammalian peptides appear to influence stimulation of smooth muscle contraction in the GI/urogenital tract, gut motility and GI peristalsis, potent effects on immune cells, stimulation of secretion, and hormone release including insulin in gastric, pancreatic, colon, and endocrine organs, potent growth effects on normal and neoplastic tissues, and potent CNS effects including regulating circadian rhythm thermoregulation, anxiety, satiety, and behavior. 29
Research on bombesin and phyllolitorins suggests that these peptides may elicit similar responses to the endogenous mammal bombesin-like peptides. In in vivo rat studies, bombesin and litorin were found to potently suppress food and ethanol intake. The suppression of feeding behavior has also been observed in chicks receiving intracerebroventricular injections of litorin. 30 Litorin has been observed in rats to decrease body temperature by 0.9°C in the short term. 31 It is possible that phyllolitorins are responsible for the shivering commonly observed during the latter stages of the Kambô experience, as well as appetite suppression.
Hypotensive and vascular peptides
Phyllokinin and phyllomedusin are two peptides present in Kambô that elicit dilation of peripheral blood vessels, decreased blood pressure, and consequent reflex tachycardia.3,32 The P. bicolor frog secretion contains significantly higher quantities of phyllokinin (2–5x) and phyllomedusin (10–750x) compared to secretions of other Phyllomedusid frogs. 33 Trace amounts of other tachykinin- and bradykinin-like peptides found in Kambô include neurokinin B, ranatachykinin A, preprotachykinin B (50–79), urechistachykinin II, and T-kinin. 15 However, these trace peptides are not discussed in detail in this review.
Bradykinin-related peptides
Phyllokinin is a bradykinin-related peptide (BRP) consisting of 11 amino acid residues. 32 Bradykinin occurs naturally in animals and has been found to play a role in allergic inflammation, reducing blood pressure, and increasing vascular permeability including permeability of the blood brain barrier (BBB). 34 Bradykinin appears to promote typical inflammatory responses such as vasodilation, redness, heat edema, hyperthermia, and pain. While increased vascular permeability and release of inflammatory mediators due to bradykinin can increase risk of edema, asthma, pain, and septic shock, bradykinin also appears to offer potential clinical benefits such has improved sensitivity to insulin, hypotensive effects, anti-ischemic effects, and potent antithrombogenic, antiproliferative, and antifibrogenic effects.34,35
It is unclear if phyllokinin elicits similar physiological effects to those of bradykinin. In one study administering phyllokinin intravenously to dogs and rabbits, an abrupt, short-lasting blood pressure fall was observed. 32 Phyllokinin appears to be much more susceptible to degradation by trypsin, unlike bradykinin which is predominantly trypsin-resistant. 32 The pharmacological actions of phyllokinin appear to result from activity of the intact molecule rather than a bradykinin-mediated activity, suggested by a 2.5–3.5-fold greater potency observed in physiological outcomes when compared to bradykinin. 32
Because bradykinins are typically pro-inflammatory, phyllokinin may induce an increase of capillary permeability locally at the site of the burns and increase absorption of the peptides from the Kambô application.32,33
Tachykinins
Phyllomedusin is a tachykinin peptide consisting of 10 amino acid residues that has been found to have a high selectivity for agonistic binding to the neurokinin 1 (NK1) receptor and appears to be important in facilitating ion transport across the frog’s skin.3,36,37 Tachykinins are a family of highly evolutionarily conserved peptides found in lower and advanced life forms that are actively involved in the central and peripheral nervous systems, as well as in cardiovascular and immune systems. 36 Tachykinins have been shown to elicit a wide array of biological activities, perhaps due to their short, flexible chains. 36 Tachykinins have been shown to prompt powerful vasodilation, hypotensive effects, and stimulation of extra vascular smooth muscle in mammals. 38 They are also potentially implicated in many clinical conditions including chronic pain, Parkinson’s disease, Alzheimer’s disease, depression, rheumatoid arthritis, irritable bowel syndrome, and asthma. 36
Like other tachykinins, phyllomedusin appears to be susceptible to degradation by trypsin and chymotrypsin. 39 Phyllomedusin also appears to act similarly to other tachykinins in eliciting a fall in blood pressure and spasmogenic effects on smooth muscle in dogs, rats, and rabbits.36,39,40 Phyllomedusin has strong effects on salivary glands, tear ducts, intestines, and bowels, contributing to smooth muscle contraction. 40 Interestingly, one study analyzing interactions of various tachykinins, including phyllomedusin, with α-adrenergic receptors in rat urinary bladder tissue found that the presence of adrenergic compounds such as the catecholamines, epinephrine, and norepinephrine, significantly altered the binding affinity of most of the tachykinins with the exception of phyllomedusin. 41 Curiously, phyllomedusin appeared to be spared from the modulating effects of the catecholamines. 41
While there is speculation that phyllokinin and phyllomedusin may mediate increased permeability of the BBB, facilitating the entrance of other Kambô peptides into the brain, there has been no research to support this conjecture.36,40,42 Phyllokinin in Kambô is possibly a key player in the onset of the heat-flushing experience, temporary redness of the skin, decrease in blood pressure, and potentially even the facial edema affectionately referred to as “frog face.” Phyllomedusin may play a role in the expulsion of tears and mucus from the eyes/nose, reduction of blood pressure, intestinal cramping, and the initiation of bowel movement.
Opioid-like peptides
Deltorphins and dermorphins share the N-terminal motif of Tyr-D-Xaa-Phe, where D is either D-alanine or D-methionine. With this shared N-terminal sequence enabling activity at both µ- and δ-opioid receptors, it is possible that C-terminal variations influence differences in receptor selectivity between the two peptide classes.
Dermorphin
Dermorphin, a 7-residue opioid peptide, has high affinity and selectivity for µ-opioid receptors and is calculated to have 40–1000 times the analgesic potency of morphine and endogenous enkephalins, depending on the tissue, measuring criteria, and specific analog.43,44 This potency of µ-opioid binding affinity and analgesic effects makes dermorphin peptides of great interest as potential clinical analgesic agents.44–46 While dermorphins do have some affinity for the κ and δ receptor sites, they appear to have high binding selectively that is 3–4 times greater for the µ sites. 44 When compared to morphine in rat studies, dermorphin produces significantly less tolerance and less severe withdrawal effects. 43
A handful of clinical trials of dermorphin took place in the 1980s, but did not continue despite its clear usefulness as an analgesic agent. In one clinical study administering to post-operative patients, dermorphin significantly out-performed morphine and control group outcomes for mean time of postoperative analgesia, the percentage of patients requiring additional analgesics, the mean analgesic consumption per patient, and mean postoperative pain intensity score. 46 50% of patients did experience nausea and vomiting; based on this observation, it is possible that the dermorphin peptides may contribute to the purgative experience of Kambô.45,46
At some point, dermorphin made its way into the illegal drugging of racehorses to increase performance. 47 This prompted the development of both urinary screening methods for doping control and in vivo pharmacological experiments using the horse as an animal model.47,48
The originally described dermorphin peptide contains the amino acid sequence H-Tyr-D-Ala-Phe-Gly-Tyr-Pro-Ser-NH2. 45 Since the discovery of the originally described dermorphin, two additional dermorphin-like analogs have been derived from the skin of Phyllomedusa bicolor, with shared features of a D-alanine at the second position, and the sequence Tyr-D-Ala-Phe.18,44,49 The primary distinguishing feature of the two analogs compared with the originally named dermorphin is the lack of the glycine residue required for a terminal amide, with instead an α-carboxyl group at the carboxy terminus.18,44,45 In P. bicolor, dermorphin and its variants occur in quantities of about 25 µg of [LysLOH]-dermorphin and 68 pg of [Trp4, AsnLOH]-dermorphin per gram of tissue. 49
In studies injecting male Wistar adult rats, the high-affinity µ-opioid ligand variant [Lys7-NH2]-dermorphin displayed potent analgesic effects, while the lower-affinity ligand variant, [Trp4,Asn7-NH2]-dermorphin, produced catalepsy and weak antinociceptive effects. 44
Despite having high and low relative binding affinities for the µ-opioid receptors, the potency of biological effects varied in different tissue samples between organ types and species. In one study analyzing the three dermorphin-like peptides derived from P. bicolor, all three peptides were several times more active in inhibiting electrically evoked contractions in guinea pig ileum than in mouse vas deferens. 44 Even though [Trp4,Asn7-NH2]-dermorphin had one-third the binding affinity for µ-opioid receptors, it displayed twice the potency when comparing the effects of [Lys7-NH2]-dermorphin on guinea pig ileum. Another analog, [Trp4,Asn7]-dermorphin had 1/20 the binding affinity for µ-opioid receptors compared to [Lys7-NH2]-dermorphin, yet displayed the same biological potency. 44 It is possible that this is due to multiple µ-opioid receptor subtypes exhibiting binding variation between the different analogs in different tissues.18,44
Deltorphins
Structurally similar to dermorphins, deltorphins are also 7-residue opioid peptides with a high affinity and selectivity for δ-opioid receptors in mammals.17,19 Deltorphin I was found in one study to be present in the dried Kambô secretion at a concentration of 5.31 µg/mg. 2
In rats, deltorphins have been observed to increase motor activity (locomotion, sniffing, and rearing) and induce behavioral changes (increased social contacts) in a dose-dependent manner. 50 Deltorphin II has also been found to increase extracellular dopamine concentrations and activity at D1 receptor sites in the rat nucleus accumbens (NA). 51
There have been two deltorphin variants isolated from the skin of Phyllomedusa bicolor with the shared amino acid sequence Tyr-Ala-Phe-Asp(or Glu)-Val-Val-Gly-NH2. 19 These two peptides called [D-Ala2]-deltorphin I and [D-Ala2]-deltorphin II contain a remarkable D-alanine in the 2nd position, resulting in higher affinity when compared to the originally described deltorphin extracted from Phyllomedusa sauvagei, which has a D-methionine in position 2. The flexible, linear structure of [3H][D-Ala2]-deltorphin I makes it a valuable tool for assisting in more precise mapping of opioid receptors.
In rat studies, deltorphin II prevented the appearance of ischemia-provoked ventricular arrhythmias while exerting no effect on heart rate or blood pressure in male Wistar rats subjected to coronary occlusions or reperfusions. 52 Based on these studies, deltorphin II shows promise as a prototype for an intervention to treat acute myocardial infarction.52,53 Another rat study found that supraspinal and spinal administration of [D-Ala2]-deltorphin II inhibited diarrhea and colonic propulsion in rats given castor oil and colonic glass beads. 54 Intrahypothalamic injection of deltorphin II produced hyperthermic reactions in rats. 55 It is possible that deltorphins play a role in the rise in temperature and heat-flushing experienced during Kambô sessions.
Deltorphin II also shows gastroprotective effects from ethanol-induced gastric mucosal damage in rat models. 56 Gastric mucosal protection resulting from deltorphin II administration appears to be mediated by supraspinal delta-2 opioid receptors, requires conservation of the integrity of the vagal nerve, and may also involve interaction with mucosal nitric oxide and prostaglandins.
Antimicrobial peptides
DRS display impressive antimicrobial, antiviral, and fungicidal properties, while having very low cytotoxicity to mammalian cells.57,65–69 DRS-B appear to be active against both Gram-positive and Gram-negative bacteria. 70 The DRS family and their derivatives have demonstrated activity against infectious agents such as malaria, sexually transmitted infections, antibiotic-resistant organisms such as E. coli and Staphylococcus aureus, HIV, Herpes Simplex Virus (HSV), Human papilloma virus (HPV), rabies, Candida albicans, and other common pathogens.57,65,67,69–71
Through cationic activity, antimicrobial peptides such as the DRS bind to the cell membrane of the target cell, permeating it and causing disruption to the lipid chains and transient pore formation, eventually causing the target cell membrane to collapse. 68 DRS were even able to reach and eradicate bacteria contained in biofilm. 57 The detrimental effects of this membrane lipid bilayer-destabilizing mechanism prevent an organism from the ability to develop resistance to the peptide, making compounds like DRS very appealing as novel antibiotic interventions. 68
Dermaseptin B2, also called adenoregulin, has also been found to have antitumorigenic and angiostatic properties at low micromolar concentrations in vitro.72,73 This study found that the anticancer activity appears to be via the process of cell necrosis rather than apoptosis. When in vitro PC3 prostate cancer cells were treated with DRS-B2 and DRS-B3 of the L-orientation and D-orientation amino acid sequences, there was no distinguishable difference in stereoselectivity, suggesting that receptors do not play a role in the interactions between PC3 cancer cell lines and DRS-B2/DRS-B3. 72
DRS-B2 (adenoregulin) has been found to enhance binding of agonists of A1 adenosine receptors, which are fundamental to the regulation of cellular metabolism.1,74 These receptors may offer a target for the treatment of depression, stroke, and cognitive loss diseases, such as Alzheimer’s disease and Parkinson’s disease, although it is currently unclear whether adenoregulin modulates the relevant receptor activities to a therapeutically useful degree.
Despite their potent cytotoxicity to bacteria and cancerous cells, DRS appear to have little interaction with or toxicity toward healthy animal cells. 68 One study using synthetic S4 DRS derivatives displayed stronger antimicrobial activity and reduced cytotoxicity when compared to conventional antibiotic treatments against sexually transmitted infectious organisms. 69 The specificity of antimicrobial peptides toward some pathogens and tumor cells may be due to the presence of a net negative charge of the membrane, the presence of glycosaminoglycans (GAG) on membrane surface, increased membrane fluidity due to pathology, or lack of cholesterol-supported integrity in the cell membranes of pathogens.57,72,75
The biotechnology industry has moved forward in leveraging the usefulness of these DRS-B. Genetic modification in plants to express a DRS-B1 derivative as an infection-resistant measure has been applied to tobacco, potatoes, and poplar trees in the willow family.76–78
Discussion
Interestingly, the pharmacological potency and mechanisms of action of some of the peptides in mammals appear to be substantially different from their effects in Phyllomedusa bicolor. P. bicolor has significant insensitivity to deltorphins in terms of opioid receptor binding affinity when compared to mammals. 79 P. bicolor also does not possess the same tachykinin/bradykinin system as mammals, alluding to an entirely different biological function of phyllokinin and phyllomedusin in the frog. 37
There are substantial structural differences between frog-based toxins and toxins found in other animals such as insects, molluscs, and reptiles. While the presence of cysteine residues in animal toxins is widespread, Kambô peptides lack cysteine residues, and hence are linear in structure.3,44,80 While disulfide bridges between cysteine residues contribute to thermal and enzymatic stability of other animal toxins, 80 the linear structure of Kambô peptides makes them relatively susceptible to enzymatic degradation. Kambô peptides also do not appear to act on voltage-gated ion channels, which are common binding sites for paralyzing and analgesic peptides present in snail, spider, and snake venoms. 81 In evolutionary terms, this is consistent with the role of frog peptides as defensive toxins rather than venoms used in foraging or predation. 81
The concentrations and proportions of the various biogenic peptides identified in Kambô may vary widely from batch to batch, depending on such factors as the age and sex of the frog(s) from which the secretions were obtained, the general health and degree of stress experienced by the frog(s) during collection of the skin secretions, the weather and other ambient conditions prevailing before and during harvest of the secretions, and the degree of drying and freshness of the Kambô secretions as influenced by conditions of preparation, transport, and storage. There is also significant variance in the surface area of the burns administered to participants during the Kambô ceremony, and consequently the quantity of peptides absorbed.
The actual amounts of the individual biogenic peptides administered to participants in a Kambô ritual, while clearly broadly physiologically active towards the whole human organism, may not be sufficient to elicit all (or any) of the effects observed by researchers in laboratory studies.
The potential for certain Kambô peptides to cross the blood–brain barrier from the peripheral bloodstream is open to conjecture and is not fully confirmed. Several putative neurological effects related to receptor binding and action within the central nervous system are therefore uncertain.
Based on the above summary of the predominant biogenic peptides in Kambô, the acute physiological effects may be summarized as follows.
The immediate effects through the combined action of the bradykinins, tachykinins, caeruleins, and sauvagines are dilation of peripheral blood vessels, resulting in a dramatic decrease in blood pressure along with a warm sensation within the body, a general flushing and burning sensation of the skin, and acute, dry discomfort in the eyes. The blood pressure drop will result in general weakness and may cause fainting—particularly due to orthostatic hypotension associated with standing from a seated position, or even sitting up from a prone position.
Subsequent to the decrease in blood pressure is a marked increase in heart rate, as the body’s homeostatic mechanisms endeavor to compensate for the blood pressure drop. The tachycardia may be accompanied by palpitations or arrhythmias, depending on the cardiac condition of the participant.7,9
The peripheral action of the caeruleins and sauvagines on smooth muscle of the gastrointestinal tract and on the gall bladder may cause nausea. The action of the vagus nerve, the tenth cranial nerve which connects the gastrointestinal tract to the brain, may also come into play. The dermorphin and deltorphin opioid peptides may contribute to nausea through the well-characterized effects of opioids on the area of the brain involved in nausea and emesis.
Further action of the caeruleins and sauvagines on the smooth muscle of the stomach, possibly along with the action of the opioid peptides on the mechanisms associated with emesis, results in activation of the vomiting reflex that will continue as long as the peptides remain in the body in active concentrations. Hence, this effect is likely to endure between 10 min and one or more hours and may have no correlation with the contents of the stomach.
Action of the caeruleins and sauvagines at the adrenal cortex is thought to release norepinephrine, leading to the characteristic “fight or flight” response, notably an increase in fear and anxiety. Action of these peptides on the pituitary gland may release a range of hormones and endogenous peptides associated with this gland, including corticosteroids, β-endorphins, antidiuretic hormone (vasopressin), oxytocin, and prolactin. The corticosteroids act as modulators of inflammatory pathways; β-endorphins mediate analgesia; vasopressin contributes to retention of fluid through modulation of kidney function leading to edema, and also increases blood pressure through arteriolar tone; prolactin is implicated in metabolism and modulation of the immune system; and oxytocin promotes social/bonding behavior but also induces uterine contractions via a positive feedback loop.
Longer-term physiological effects may result from the more subtle actions of the various Kambô peptides on a range of targets. Such effects include moderate anorexia through the action of the caeruleins and sauvagines; heightening of the visual and aural senses, along with enhanced energy for physical activity through the action of norepinephrine; and slightly enhanced activity of the immune system through the secondary immunomodulatory effects initiated by several of the peptides, including caeruleins and sauvagines. Further, it is conceivable that the demonstrated in vitro antimicrobial properties of the dermaseptins may contribute to the body’s response to a broad range of pathogens, although it must be noted that the Kambô peptides are present only briefly in the body and in very low systemic concentrations, making it unclear if Kambô elicits acute or longer-term antimicrobial properties in vivo. Likewise, the observed antitumorigenic properties of certain dermaseptins demonstrated in vitro may not translate to anticancer activity in vivo.
Aside from the range of physiological effects discussed above, there have been no observations of direct psychological effects directly attributable to psychoactive properties of any Kambô components. One study used the Altered States of Consciousness (ASC) rating scale and Mystical Experience Questionnaire (MEQ) to evaluate the psychoactive nature of Kambô, finding that the scores did not indicate an altered state, perceptual distortion or mystical experience. 82 Despite having no psychedelic effects, most users reported significant spiritual and personal benefits. Simply put, Kambô is not regarded as exerting any direct psychedelic or hallucinogenic effects, and nor is it known to be used by anyone for this purpose.
Future directions
Many of the individual peptides contained in Kambô may have useful potential for application as medicinal agents. It is also possible that combination of the peptides, as administered in Kambô rituals, may provide synergistic effects. Given rapid growth in the global popularity of Kambô as a healing ritual, we recommend further investigation of the potential physiological and psychotherapeutic effects, as well as characterization of the risks, of Kambô. Until clinical research has been conducted, the effects in humans of the crude peptides from the skin secretion of Phyllomedusa bicolor will remain speculative at best.
Although there are many anecdotal reports of benefits to individuals after receiving Kambô, no controlled studies have been conducted to evaluate its effects on physiological or psychological outcomes. In order for Kambô to be considered feasible for human research, more investigation into the profiles of the complex mixture of peptides, their ability to cross the blood brain barrier, dosage standardization challenges and the influence of the placebo-effect due to ritual should be addressed. With growing interest in Kambô as a ritual and alternative healing modality, the pressing need for research on Kambô has never been higher. Clinical research should be conducted to evaluate the safety and potential therapeutic efficacy of Kambô, in order to serve public safety and encourage medical innovation.
Footnotes
Author Contribution
Manuscript authorship by CT and MW. Figure created by CT. Tables created by CT and MW. All authors reviewed and approved manuscript prior to submission.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Caitlin Thompson provides Kambô rituals for participants via her practice at Medicine Frog Kambô. Other authors have no financial or otherwise relevant conflicts of interest to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


