Abstract
Recently, most coronary interventions rely on visual assessment of the coronary lesions using angiography with all known inherent limitations. Intravascular ultrasound (IVUS) allows for the evaluation of the coronary pathology to obtain information about both the extent and nature of atherosclerotic plaque; thus, planning the intervention strategy is based on objective data. Following the advent of gray-scale IVUS, several modes had been developed to study coronary plaque composition, thus determining the predominant component of the plaque, fibrous tissue, lipid-necrotic core, or calcium, and the intervention strategy. After intervention, IVUS plays an important role in optimizing the results after stent deployment regarding stent expansion and apposition to reduce the incidence of both stent thrombosis and in-stent restenosis. This review discusses the basic role of IVUS in evaluating plaque structure and parameters to optimize results after coronary intervention in light of recent evidence.
Introduction
Cardiovascular disease is the leading cause of death worldwide, with coronary artery disease (CAD) accounting for most of such mortalities, and atherosclerosis remains the most important and prevalent cause of CAD. 1 So far, invasive coronary angiography (CAG) is the gold standard method for diagnosing CAD, but since CAG is a two-dimensional tool for assessing the three-dimensional arterial structure, it has many limitations in clinical practice.
Intravascular ultrasound (IVUS) is the first widely applied catheter-based imaging modality in CAD. 2 IVUS is now one of the most important adjunctive tools in the cardiac catheterization laboratory, which has been used for more than two decades and in the assessment of lesions inside the coronaries, thus overcoming some of the limitations of CAG. Therefore, it can be of great help in diagnosing and optimizing treatment in atherosclerotic CAD.
IVUS and the Evaluation of Atherosclerotic Plaque Composition
Atherosclerosis is a disease in which atheroma forms, grows, and starts to cause serious problems in the vessel wall; in addition, the atheroma sometimes starts to grow outward, away from the lumen, long before it starts to cause lumen abnormalities (positive remodeling). 3 Most unstable plaques that cause acute coronary syndromes are borderline lesions with a degree of stenosis ranging from 50% to 70%, which do not cause much lumen compromise. 4 Since the advent of CAG in the year 1958 at the Cleveland Clinic, it has contributed much to both the diagnosis and treatment of CAD patients. 5 CAG draws a silhouette of the vessel lumen without any information about the vessel wall, which may have lesions with positive remodeling or diffuse atherosclerosis, either of which will not be detected by coronary lumenography. Hence, the need for a tool arises, which would allows us to evaluate the wall of the vessel and to have a closer look at the nature of the atherosclerotic involvement; thus, overcoming one of the major limitations of CAG.
The gray-scale IVUS image can be used to determine plaque composition by comparing the differences in echogenicity within the plaque, but this process is subject to many interfering factors, such as resolution, machine setting, and the operator, hence it is not considered accurate for tissue characterization of the coronary plaque. 6 However, recent advancements in IVUS analysis, such as analysis of the backscattered ultrasound waves using radio frequency, now called radio frequency IVUS (RF IVUS), have increased its potency in accurately determining the plaque composition. Currently, three different RF data analysis methods are available: autoregressive modeling, fast Fourier transform, and a newly designed pattern recognition algorithm and mathematically defined system. Three commercial software programs are also available for plaque tissue characterization: (1) virtual histology IVUS (VH–IVUS; Volcano Corp.); (2) iMAP (Boston Scientific); and (3) integrated backscatter IVUS (IB-IVUS; YD Co., Ltd.). 7 Using the previously mentioned analysis techniques, IVUS can distinguish four different plaque components: fibrotic plaque, fibrofatty plaque, necrotic core, and dense calcium, which are represented as green, light green, red, and white, respectively. 7
Pathological findings and IVUS plaque estimation
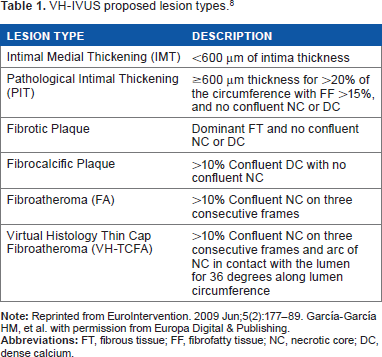
Pathological intimal thickening (PIT) is characterized by a plaque that does not encroach the lumen and consists of intimal thickening composed of fibrous plaque with extracellular lipid and proteoglycans and no true necrosis. The area overlying the lipid is rich in smooth muscle cells and may contain a number of macrophages and T-lymphocytes. A fibrous cap atheroma (fibroatheroma) is formed of a large lipid-necrotic core comprising large amounts of extracellular lipid, cholesterol crystals, and necrotic debris, which is surrounded by a fibrous cap consisting principally of smooth muscle cells in a collagenous proteoglycan matrix, with varying degrees of infiltration by macrophages and T-lymphocytes, and a variable number of inflammatory cells surrounding the lipid-necrotic core. A thin-capped fibroatheroma (TCFA) is characterized by a large necrotic core, containing numerous cholesterol clefts. The overlying cap is thin and rich in inflammatory cells, macrophages, and T-lymphocytes, with few smooth muscle cells. Plaques prone to rupture are those with decreased cap thickness, a large lipid-necrotic core, and severe inflammatory infiltrate (Table 1). 8
VH-IVUS proposed lesion types. 8
IVUS plaque assessment in clinical studies
VH-IVUS, as well as the two other RF IVUS modalities, has been validated with actual histology from coronary atherectomy specimens, explanted hearts, and carotid endarterectomy sections. It was found to have moderate predictive accuracies (80%–94%).8–11
In the ASTEROID trial, very high intensity statin therapy using rosuvastatin 40 mg/day in 507 patients achieved an average low-density lipoprotein (LDL-C) of 60.8 mg/dL and increased high-density lipoprotein (HDL-C) by 14.7% and caused significant regression of atherosclerosis for all three IVUS measures of disease burden at 24 months. 12 Seventy patients with ACS were enrolled in the ESTABLISH trial; they underwent emergency CAG and percutaneous coronary intervention (PCI) and were randomized to intensive lipid-lowering therapy (n = 35; atorvastatin 20 mg/day) or control (n = 35) groups after PCI. Volumetric IVUS analyses were performed at baseline and at six-month follow-up of a non-PCI site in 48 patients (atorvastatin, n = 24; control, n = 24). Percent change in plaque volume showed a significant positive correlation with the follow-up LDL-C level (R = 0.456, P = 0.0011) and percent LDL-C reduction (P < 0.0001). Thus, it was concluded that early aggressive lipid-lowering therapy with atorvastatin for six months significantly reduced plaque volume in patients with ACS. 13
In the PROSPECT trial, VH-IVUS was used to study the composition of nonculprit lesions in 697 patients with acute coronary syndrome, and patients with TCFAs with a plaque burden ≥70% and minimal lumen area (MLA) ≤4.0 mm2 had an 18% major adverse cardiac event (MACE) rate at three-year follow-up. Thus, it was concluded that the addition of radio frequency analysis to gray-scale IVUS provided important prognostic information. 14 In a subgroup analysis of the Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction (HORIZONS-AMI) study, RF IVUS was used to assess the changes that occurred in 99 untreated nonculprit lesions over a period of 13 months. At baseline, 41 were TCFA, but at 13-month follow-up, 32 of 41 lesions (78%) were still labeled as TCFA, with another additional 21 lesions newly classified as TCFA. TCFA lesions at baseline showed a decrease in minimal lumen area and an increase in percent necrotic core by the end of the follow-up period. 15 Kataoka et al found that calcified nodules, defined by IVUS as irregular protruding and convex calcification, were associated with a lower risk of ischemic events, whereas spotty calcification, defined by lesions of 1–4 mm in length with an arc of calcium <90°, was associated with greater percent and total atheroma volume and greater progression of percent atheroma volume on subsequent IVUS examination. 16
There are limitations to RF IVUS. It has poor axial resolution that is insufficient to accurately measure fibrous cap thickness, and RF IVUS defines TCFA on the basis of the presence of a necrotic core abutting the lumen, which is not accurate. Even proper acquisition and analysis of RF IVUS are also technically challenging, especially for serial imaging of specific lesions.17–19
So, from the aforementioned data, it is clear that IVUS with the new methods of analysis can provide an insight into not only the extent of CAD but also the structure of the atherosclerotic plaque, which influences prognosis and helps decide the most appropriate methods of intervention. Although many software modifications were applied to the gray-scale IVUS to improve its ability to differentiate between various tissue types within the atherosclerotic plaque, yet IVUS (due its low resolution) is not the optimal method for determination of the plaque type at least compared to optical coherence tomography, which has 10 times the resolution of IVUS.
IVUS and PCI
IVUS can be of much help during PCI; preintervention IVUS provides valuable information regarding the severity of stenosis, lesion length, vessel size, and plaque characteristics. It has been used to plan and guide PCI and also provides information on the extent of calcium, the need for vessel preparation, and the selection of device size and type, all of which have an influence on the PCI strategy to be used. Postintervention IVUS assessment aims at determining the degree of stent deployment, strut apposition, and the detection of complications such as dissection, thrombosis, and excessive plaque material prolapse. All of the previous elements affect the outcome after PCI, in both the short and long terms (Fig. 1).
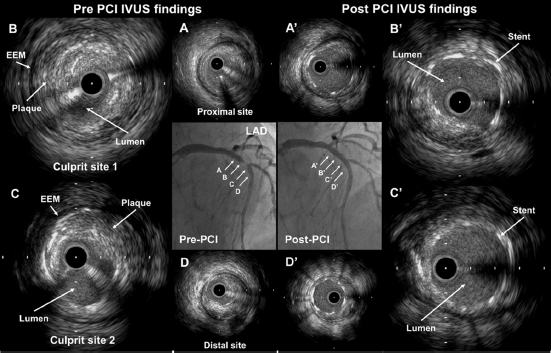
Case example of IVUS-guided PCI. CAG showing a significant lesion in the proximal left anterior descending (LAD) artery. Pre-PCI IVUS images show the proximal LAD with significant lumen stenosis and an eccentric plaque lesion (A–D). post-PCI IVUS images show good expansion and correct stent apposition (A'–D’).
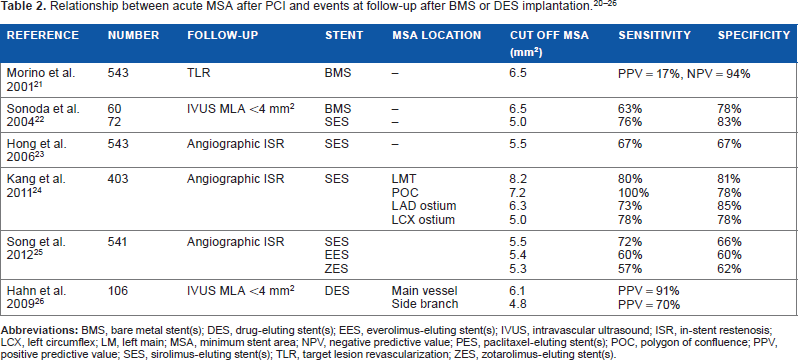
There are many studies that had been performed to assess the different IVUS parameters and its impact on the outcome of PCI after either bare metal stent (BMS) or drug-eluting stent (DES) implantation. Table 2 summarizes the results of some of these studies.20–26
IVUS in the BMS era
Since the advent of IVUS technology in coronary intervention in the early 1980s, many clinical studies and meta-analyses were reported in the BMS era to assess the influence of IVUS on the outcome of PCI. Different IVUS parameters were studied in these trials and were found to affect the outcome in PCI with BMS implantation, such as small minimal stent area (MSA), stent malapposition, underexpansion, and edge dissections.
The Multicenter Ultrasound Stenting in Coronaries (MUSIC) trial was the first study that established IVUS criteria for optimal stent implantation (Table 3). The data from the MUSIC trial showed that IVUS guidance improved immediate angiographic outcomes (post-minimal lumen diameter [MLD], 2.90 ± 0.36 mm), which had been translated into favorable clinical and angiographic outcomes at six months compared to only angiographic guidance (lowest target lesion revascularization [TLR] of 5.7% and restenosis rate of 9.7% with the largest MLD of 2.12 ± 0.67 mm). 27 The Restenosis after IVUS-guided Stenting (RESIST) study was a French study in which it was found that there was a significant increase in six-month stent lumen cross-sectional area (4.47 ± 2.59 mm2 vs. 5.36 ± 2.81 mm2, P = 0.03) in the IVUS-treated group, which indicates that IVUS guidance in stent deployment may be beneficial. On a multivariate logistic regression analysis, postprocedural stent cross-sectional area was the only predictor (inversely related) of six-month in-stent restenosis (odds ratio 0.7, 95% confidence interval [CI]: 0.47–0.93, P = 0.007). 28 In the Optimization with Intracoronary Ultrasound to Reduce Stent Restenosis (OPTICUS) study, 550 patients were randomized to stent implantation with IVUS or angiographic guidance. Six-month angiography showed no significant differences in restenosis, and the 12-month MACE rate was not different between the two groups. The authors concluded that the study did not support routine use of IVUS to guide coronary stenting. 29 The Thrombocyte Activity Evaluation and Effects of Ultrasound Guidance in Long Intracoronary Stent Placement (TULIP) study was a multicenter, randomized trial comparing IVUS-guided PCI to angiography-guided PCI in long lesions (>20 mm). IVUS optimization resulted in a larger final MLD (3.01 ± 0.40 mm vs. 2.80 ± 0.31 mm, P = 0.008) and acute gain (2.04 ± 0.62 mm vs. 1.81 ± 0.45 mm, P = 0.045), with a lower binary restenosis rate at six months. 30 The Angiography Versus Intravascular Ultrasound-Directed (AVID) trial was the largest multicenter randomized study; it enrolled 800 patients and showed significant clinical and angiographic benefits of IVUS-guided stenting over angiographic guidance alone. A significant reduction in TLR was observed in the IVUS group at 12-month follow-up (4.3% vs. 10.1%; P = 0.01). 31 An important meta-analysis by Praise et al, involving a total of 2,193 patients in five multicenter and two single-center studies, showed that IVUS guidance for BMS implantation improved the acute procedural results, thus reducing angiographic restenosis, repeat revascularization, and the MACE rate, with a neutral effect on death and myocardial infarction (MI) during a follow-up period of 6 months to 2.5 years. 32
MUSIC criteria for stent expansion. 27

Thus, from the above evidence, it is clear that conclusions from either clinical trials or meta-analyses support the use of IVUS in BMS implantation, since this approach is associated with better outcomes in both the short and long terms.
IVUS and DES era
From the aforementioned data, there is sufficient evidence that IVUS improved the outcomes after BMS implantation, but the evidence supporting the use of IVUS in DES implantation is lacking so far, and the available data are largely derived from observational studies and meta-analyses, and the few randomized controlled trials (RCTs) had small numbers, not sufficient to influence clinical practice.
In the long-term health outcome and mortality evaluation after invasive coronary treatment using drug-eluting stents with or without IVUS guidance (HOME DES) study, 210 patients were randomized to an IVUS-guided PCI strategy vs. an angiography-guided strategy. Although IVUS guidance led to the implantation of larger stents and higher postdilatation pressures, target vessel revascularization and TVR or MACE rates did not differ between the two groups. 33 The Angiography Versus IVUS Optimization (AVIO) study also compared IVUS-guided and angiography-guided PCI strategies with DES in 284 patients, but the AVIO population tended to have complex lesions (long lesions, bifurcations, chronic total occlusions, and small vessel disease). Although more aggressive criteria for optimal stent deployment were used in AVIO (Table 4), this, as in the HOME DES, was not translated into better outcomes in the IVUS group, and there was no significant difference in the MACE rates in the nine-month period. 34 Zhang et al reported in a meta-analysis that IVUS guidance was associated with significantly lower mortality (hazard ratio [HR]: 0.59; 95% CI: 0.48–0.73; P < 0.001), MACE (HR: 0.87; 95% CI: 0.78–0.96; P = 0.008), and stent thrombosis (HR: 0.58; 95% CI: 0.440.77; P < 0.001) rates compared with angiography-guided PCI, but with no change in MI, TLR, or TVR. 35
AVIO criteria for IVUS-guided stent deployment. 34

The Assessment of Dual AntiPlatelet Therapy with Drug-Eluting Stents (ADAPT DES) study found significantly less stent thrombosis (0.6% vs. 1.0%, P = 0.02), MI (2.5% vs. 3.7%, P = 0.002), and ischemia-driven TVR (2.4% vs. 4.0%, P < 0.001) in patients in whom IVUS was used to guide PCI compared with PCI without IVUS. 36 Jang et al, in another meta-analysis of three randomized trials and 12 observational studies consisting of 24,849 patients, found that IVUS-guided DES implantation was associated with a 21% reduction in the MACE rate and a 36% reduction in mortality compared with angiography-guided PCI. 37 In a recent study by Hong et al, the use of IVUS during PCI resulted in 2.9% absolute decrease in MACEs compared to only angiography-guided PCI but cardiac death and target lesion-related MI were not significantly different between the two groups one year after everolimus-eluting stent implantation. For cardiac death, there were three patients (0.4%) in the IVUS-guided group and five patients (0.7%) in the angiography-guided group (P = 0.48). Target lesion-related MI occurred in one patient (0.1%) in the angiography-guided stent implantation group (P = 0.32). 38
The evidence regarding IVUS-guided DES implantation is not as strong as the beneficial value in BMS implantation, and more large RCTs are needed to draw a final conclusion on this issue. This is largely because the evidence is derived mainly from meta-analyses, not from RCTs. Until evidence is clear on this issue, IVUS should still be used to guide DES implantations because malapposition with DES is a serious issue that may lead to both stent thrombosis and restenosis even more than BMS malapposition.
IVUS and left main stenting
Acute complications during left main (LM) PCI are of dire consequence, and late restenosis or thrombosis may lead to marked morbidity and mortality. Thus, optimizing the result of PCI is very important to prevent such complications. One of the early studies by Agostoni et al showed no long-term clinical benefit of IVUS-guided ULMCA in 58 patients. 39 On the other hand, Park et al, in another study, showed a reduced one-year MACE rate in 102 patients. 40
The Revascularization for Unprotected Left Main Coronary Artery Stenosis: Comparison of Percutaneous Coronary Angioplasty versus Surgical Revascularization (MAIN-COMPARE) registry is considered the largest analysis of IVUS-guided vs. angiography-guided LM PCI. In this registry, nonrandomized long-term clinical outcomes were evaluated in 975 patients. At three years, the results showed a general trend toward lower mortality in the IVUS-guided ULMCA PCI group compared to angiography-guided PCI (HR: 0.54, 95% CI: 0.28–1.03, P = 0.061), but subgroup analysis of DES patients showed a significant mortality benefit in the IVUS-guided PCI group compared to the angiography-guided group (4.7% vs. 16%, P = 0.048).40,41 Thus, studies showed at least a marginal benefit of using IVUS guidance during LM PCI for an important parameter, mortality. However, larger trials are needed to better clarify this benefit.
IVUS-guided PCI in guidelines
In the 2011 ACC/AHA/SCAI Guidelines on Coronary Revascularization, it was stated that IVUS may be used in coronary stent implantation, especially in LM stenting, and this was a class IIb recommendation/level of evidence B. 42 In the SCAI expert consensus document in 2013, IVUS used to optimize stent deployment and detect complications during PCI was stated as being definitely beneficial. 43 In the 2014 European Guidelines on Myocardial Revascularization, IVUS was recommended to guide stent implantation in selected patients, and this recommendation was a class IIa/level of evidence B. 44
Other uses of IVUS in CAD
Follow-up of cardiac transplantation
CAD is the major cause of death after the first year following cardiac transplantation and is usually clinically silent because the heart is denervated. Because of the diffuse nature of the disease, angiography has limited capability for detecting such changes and IVUS can detect early PIT of more than 0.5 mm before the disease is advanced. 45
Vein graft disease
IVUS can be used to assess early vein graft degeneration long before it becomes angiographically evident. The wall of the vein grafts is morphologically different from the native coronary arteries as the bypass graft wall is free from the surrounding tissue, has no side branches, and also veins do not have an external elastic membrane (EEM). However, vein grafts typically undergo arterialization with morphologic changes that include intimal fibrous thickening, medial hypertrophy, and lipid deposition. 46
Effect of radiation (brachytherapy) on the coronaries
Having the ability to visualize layers of the arterial wall, IVUS can be used to study the mechanisms and results of strategies for reducing restenosis, including brachytherapy. In non-stented lesions, serial (postradiation vs. follow-up) analysis of lesion site and reference segment EEM, lumen, and plaque plus media volume is the most accurate approach. In stented lesions, measurements should include serial (postradiation vs. follow-up) volumetric analysis of the stent, lumen, and intimal hyperplasia. 47
Limitations of IVUS
Although IVUS devices produce detailed views of the vessel wall, there are some limitations that may render the examinations subject to many errors. IVUS has the same problems found generally in any ultrasound examination as calcification produces high reflectivity that makes it difficult to visualize structures behind such shadow, also different structure may produce the ultrasonographic pattern similar to thrombus and high lipid content may have the same echolucent appearance.
There are also many artifacts that can occur during IVUS examination that may adversely affect ultrasound images, including ring-down artifacts produced by acoustic oscillations in the piezoelectric transducer that obscure the near field, resulting in an acoustic catheter size larger than its physical size. 43 Geometric distortion can result from imaging in an oblique plane (not perpendicular to the long axis of the vessel). An important artifact, nonuniform rotational distortion, arises from uneven drag on the drive cable of the mechanical style catheters.47,48
Conclusion
CAG is a feasible and practical tool for both the diagnosis and treatment of CAD, although it has limitations. IVUS gives us the ability to look inside the coronaries and has a reasonably accurate idea about the nature of atherosclerotic involvement for both extent and composition, which influences the plan of management. IVUS has been proven to positively influence the outcome after BMS implantation, but data supporting its role in optimizing the result after DES implantation is not as strong as for BMS. Finally, training and experience with IVUS help in knowing its limitations and confounders for it to be a good tool, when used appropriately at the right time.
Author Contributions
Wrote the first draft of the manuscript: AH, TD, HD. Contributed to the writing of the manuscript: All the authors reviewed and approved the final manuscript.
Footnotes
Acknowledgment
The authors thank the staff of the Department of Cardiovascular Medicine at Juntendo University.