Abstract
Shimoda and coworkers
1
published a paper on the pharmacological properties of glycosides of the peppery compound capsaicin (
Commentary to “Enzymatic synthesis and anti-obese properties of β-maltooligosaccharides of capsaicin and 8-nordihydrocapsaicin”, Shimoda et al, 2009.
Recently Shimoda and coworkers 1 published a paper on the pharmacological properties of glycosides of the peppery compound capsaicin (1) and its derivative 8-nordihydrocapsaicin (2). In their paper they conclude that the β-glucoside and β-maltoside of 1 and the β-glucoside of 2 still have the same potent anti-obese activity but further studies are needed to determine the physiological effect. This is indeed an interesting approach which should be pursued further and by many more.
It has been shown that activation of TRPV1 channels can prevent obesity and this is due to the effect on the calcium influx in cells. 4 In these studies it was clearly seen that wild type mice did not grow to be obese, whereas TRPV1 knockout mice became obese when both types were assigned to high fat diets. The effect of capsaicinoids on TRPV1 is crucial in understanding the pharmaceutical value of these chemical entities.
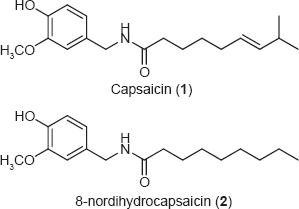
Structure of Capsaicin and 8-nordihydrocapsaicin.
To underline the importance of understanding the impact of the deactivation of TRPV1 channels it has recently been shown that TRPV1 has a role in the development of diabetes in non-obese diabetic mice. Again by the deactivation of the TRPV1 channels with 1, this development can be stopped. 5 These findings identify TRPV1 antagonists as a novel therapeutic intervention of pain relief, weight control and prevention of diabetes. High doses of 1 desensitize TRPV1 and thereby have antagonistic effect opposed to the agonistic effect of lower doses. 5 High doses of 1 desensitize TRPV1 and thereby have the opposite effect of lower doses. To sum up, the effect of capsaicinoids on TRPV1 is of importance to understand the development of new drugs against obesity and secondary diabetes. Shimoda et al 1 observed that the glycosylated capsaicinoids have an inhibitory effect on obesity–-this could indicate that the glycosylated compounds block the activation site on TRPV1 channels and thereby promote the effects previously described. 5 This of course, needs to be investigated further.
Shimoda et al 1 use the known fact that glycosylation will make a drug candidate more water soluble to promote the research for new drug candidates within the chemical group of capsaicinoids. Their findings support that this is a plausible way to enhance both bioavailability and “drug-likeliness” properties of drug candidates. This approach is already being investigated by the Swiss biotech company Evolva (www.evolva.com) in the pursuit to optimize nature wonder drugs. Evolva has discovered a number of wonder glycosyltransfereases (GT) that can glycosilate almost any compound with the desired sugar moieties. In a series of publications, Hansen and coworkers have shown that their line of 107 recombinant GT's from the GT plant family 1 can be used to modulate more than 100 different and very diverse natural products.6–8 Their results reveal that plant GTs are highly promiscuous towards small molecule drug compounds, being able to recognize a wide range of structurally diverse molecules. The promiscuity of the plant GTs emphasizes their potential as biocatalysts for drug development purposes as shown by Shimoda et al. 1 The use of even more GT's could provide further knowledge on what sugar is the most useful in the case of TRPV1 channels. This could also be studied by modeling the chemical entities on to the model of the TRPV1. Both the TRPV1 and the TRPV2 crystal structures are available for building pharmacorphore models. This could reveal further information on the pharmacological properties of these new and interesting glycosylated capsaicinoids and would aid the choice of sugar and relevant GT.
Footnotes
Disclosure
The author reports no conflicts of interest.
