Abstract
Reduction and glycosylation of benzophenone, which is an endocrine disrupting chemical, were investigated using immobilized marine microalga and plant cells from the viewpoint of bioremediation of benzophenone. Immobilized marine microalga of Chrysocampanulla spinifera reduced benzophenone to diphenylmethanol. Immobilized marine microalga of Amphidinium crassum glucosylated diphenylmethanol to the corresponding glucoside. The sequential biotransformation with C. spinifera and A. crassum effectively converted benzophenone into diphenylmethyl glucoside. On the other hand, immobilized plant cells of Catharanthus roseus transformed benzophenone to diphenylmethanol, diphenylmethyl glucoside, and diphenylmethyl primeveroside, which was a new compound, by one-step biotransformation.
Keywords
Introduction
Benzophenone is widely used as a photo initiator in UV-curing applications such as inks, imaging, and clear coatings in the printing industry. Benzophenone prevents ultraviolet light from damaging scents and colors in products such as perfumes and soaps. Benzophenone is released as a pollutant and toxic compound into rivers and seas. Recently, benzophenone has attracted considerable attention as it exhibited estrogenic activity in bioassays and has been listed among “chemicals suspected of having endocrine disrupting effects”. From the viewpoint of pollution control, many studies on the biological metabolites of aromatic compounds have been reported, eg, the benzene rings of aromatic compounds are degraded by some soil bacteria.1–5 However, little attention has been paid to the biological degradation of endocrine disrupting chemicals.
Recently, the biotransformation of exogenous substrates by cultured marine microalga has been reported.6,7 These cells have the abilities of hydroxylation, glycosylation, oxido-reduction, hydrogenation, and hydrolysis for various organic compounds. Particularly, glycosylation seems to be an efficient procedure for the bioremediation of environmental pollution, because the estrogenicity of endocrine disrupting compound, ie, bisphenol A, was eliminated by formation of its glycosides. 8 In addition, immobilization of microalga cells much improved the ability of cells to reduce and glycosylate exogenously added compounds. 9
This paper describes, for the first time, the reduction and glycosylation of benzophenone by the immobilized marine microalga of Chrysocampanulla spinifera and Amphidinium crassum, and immobilized plant cells of Catharanthus roseus. The sequential biotransformation with immobilized marine microalga of C. spinifera and A. crassum converted benzophenone into diphenylmethanol and diphenylmethyl glycoside. On the other hand, immobilized plant cells of C. roseus transformed benzophenone to diphenylmethanol, diphenylmethyl glycoside, and diphenylmethyl primeveroside, which has not been identified before, by one-step biotransformation.
Experimental Procedures
General
Benzophenone was purchased from Aldrich Chemical Co. The 1 H and 13C NMR, H-H COSY, C-H COSY, and HMBC spectra were recorded in CD3OD using a Varian XL-400 spectrometer (Varian Inc.). The chemical shifts were expressed in δ (ppm) referring to tetramethylsilane. The FABMS spectra were measured using a JEOL MStation JMS-700 spectrometer (JEOL Ltd.). HPLC was carried out on a YMC-Pack R&D ODS column (150 x 30 mm) at 25 °C [solvent: methanol-water (9:11, v/v); detection: UV (280 nm); flow rate: 1.0 ml/min].
Cell line and culture conditions
C. spinifera and A. crassum, gifts from Ehime Prefectural Fisheries Experimental Station, Japan, cells were cultivated in a synthetic seawater (500 ml) for 2 weeks at 20 °C with constant aeration by air (1 l/min) in 1 l flasks under illumination (1000 lx). The synthetic seawater contained 20.747 g NaCl, 0.8 μg MnCl2 · 4H2O, 9.474 g MgCl2 · 6H2O, 1.326 g CaCl2 · 6H2O, 3.505 g Na2SO4, 597 mg KCl, 171 mg NaHCO3, 85 mg KBr, 34 mg Na2B4O7 · 10H2O, 12 mg SrCl2, 3 mg NaF, 1 mg LiCl, 0.07 mg KI, 0.2 μg CoCl2 · 6H2O, 8 μg AlCl3 · 6H2O, 5 μg FeCl3 · 6H2O, 0.2 μg Na2 WO4 · 2H2O, 0.02 mg (NH4)6Mo7O24, 0.0045% Na2SiO3 and 1.07 ml of NM solution per 11 of distilled water. The NM solution (11) is a kind of vitamin solutions and composed of NaNO3 (150 g), Na2HO4 (10 g), EDTA-2 Na (0.9 g), Vitamin B12 (1.5 mg), thiamine · HCl (75 mg), biotin (1 mg), EDTA-Fe (2.5 g), and H2 NC(CH3OH)3 (5 g) in distilled water.
The cultured plant cells of C. roseus have been subcultured in 300 ml conical flasks containing Schenk and Hildebrand (SH) medium (100 ml, pH 5.7) on a rotary shaker (120 rpm) at 25 °C for every 3–5 weeks. Part of the callus tissues (fresh weight 30 g) was transplanted to freshly prepared SH medium (100 ml in a 500 ml conical flask, pH 5.7) containing 3% sucrose and was incubated for 3 weeks prior to use for this work.
Biotransformation of benzophenone by C. spinifera and A. crassum
Biotransformation experiments are repeated three times with essentially the same result, and one representative data set is shown. Cultured C. spinifera and A. crassum cells were harvested by centrifugation at 3000 rpm for 15 min and washed twice by adding 100 ml of synthetic seawater followed by centrifugation (3000 rpm for 15 min). To the 500 ml flask containing 9 g of cultured C. spinifera cells and 300 ml of a synthetic seawater was added 0.2 mmol of benzophenone. The cultures were incubated at 20 °C on a rotary shaker (120 rpm) for five days. After the incubation period, the cells and synthetic seawater were separated by centrifugation at 1000 g for 15 min. The synthetic seawater was extracted with EtOAc. The cells were extracted (three times) by homogenization with methanol, and the methanol fraction was concentrated and partitioned between water and EtOAc. The EtOAc fractions were analyzed by HPLC, combined, and concentrated. This EtOAc fraction was applied to a column chromatography on silica gel to give diphenylmethanol.
Diphenylmethanol (0.2 mmol) was administered to the 500 ml flask containing 300 ml of synthetic seawater and 9 g of cultured A. crassum cells, and the cultures were incubated at 20 °C for five days on a rotary shaker (120 rpm). After the incubation, the cells and medium were separated by centrifugation at 1000 g for 15 min. The filtered medium was extracted with EtOAc. The medium was further extracted with n-BuOH. The cells were extracted (x3) by homogenization with MeOH. The MeOH fraction was concentrated and partitioned between H2O and EtOAc. The EtOAc fractions were combined and concentrated. The H2O fraction was extracted with n-BuOH. The n-BuOH fractions were combined, concentrated, and applied to a Dianion HP-20 column, and the column was washed with H2O followed by elution with MeOH. The MeOH eluate was subjected to HPLC [column: CAPCELLPAK R&D C18 column (250 x 30 mm); solvent: MeOH2O (9:11, v/v); detection: UV (280 nm); flow rate: 1.0 ml/min] to give diphenylmethyl β-D-glucopyranoside.
Spectral data of diphenylmethyl β-D-glucopyranoside (3): FAB MS: m/z 369 [M + Na]+; 1 HNMR (400 MHz, CD3OD, δ in ppm): δ 3.66–4.15 (6H, m, H-2′, 3′, 4′, 5′, 6′), 4.45 (1H, d, J = 7.6 Hz, H-l′), 6.05 (1H, s, H-7), 7.18 (1H, t, J = 8.0 Hz, H-4), 7.25 (3H, t, J = 7.6 Hz, H-3, 5, 11), 7.33 (2H, t, J = 8.0 Hz, H-10, 12), 7.41 (2H, d, J = 7.6 Hz, H-2, 6), 7.51 (2H, d, J = 7.2 Hz, H-9,13); 13C NMR (100 MHz, CD3OD): δ 62.6 (C-6′), 71.5 (C-4′), 75.1 (C-2′), 77.8 (C-3′, C-5′), 81.1 (C-7), 101.1 (C-1′), 128.0 (C-2, C-4, C-6), 128.5 (C-11), 128.8 (C-3, C-5), 129.0 (C-9, C-13), 129.2 (C-10, C-12), 142.5 (C-1), 143.7 (C-8).
Biotransformation of benzophenone by C. roseus
Benzophenone (0.2 mmol) was administered to the 500 ml flask containing 300 ml of SH medium and 70 g of the suspension cultured cells of C. roseus, and the cultures were incubated at 25 °C for five days on a rotary shaker (120 rpm) under illumination (1000 lx). After the incubation, the cells and medium were separated by filtration with suction. The filtered medium was extracted with EtOAc. The medium was further extracted with n-BuOH. The cells were extracted (x3) by homogenization with MeOH. The MeOH fraction was concentrated and partitioned between H2O and EtOAc. The EtOAc fractions were combined and concentrated. The H2O fraction was extracted with n-BuOH. The n-BuOH fractions were combined, concentrated, and applied to a Dianion HP-20 column and the column was washed with H20 followed by elution with MeOH. The MeOH eluate was subjected to HPLC to give products.
Spectral data of a new compound, ie, diphenylmethyl β-primeveroside (4): FAB MS: m/z 501 [M+Na]+; 1 H NMR (400 MHz, CD3OD, δ in ppm): δ 3.51–4.19 (11H, m, H-2′, 2′, 3′, 3′, 4′, 4′, 5′, 5′, 6′), 4.55 (1H, d, J = 8.0 Hz, H-l′), 4.90 (1H, d, J = 7.6 Hz, H-l′), 6.05 (1H, s, H-7), 7.15 (1H, t, J = 8.0 Hz, H-4), 7.25 (3H, t, J = 7.6 Hz, H-3, 5, 11), 7.33 (2H, t, J = 8.0 Hz, H-10, 12), 7.41 (2H, d, J = 7.6 Hz, H-2, 6), 7.51 (2H, d, J = 7.2 Hz, H-9, 13); 13C NMR (100 MHz, CD3OD): δ 62.8 (C-5′), 67.7 (C-6′), 70.9, 71.5 (C-4′, C-4′), 74.5 (C-2′), 75.0 (C-2′), 77.6, 77.8 (C-3′, C-3′, C-5′), 81.0 (C-7), 100.1 (C-1′), 102.2 (C-1′),128.0 (C-2, C-4, C-6), 128.5 (C-11), 128.8 (C-3, C-5), 129.0 (C-9, C-13), 129.2 (C-10, C-12), 142.5 (C-1), 143.7 (C-8).
Preparation of immobilized marine microalga and plant cells in sodium alginate gel
Sodium alginate (2%) was suspended in water (500 ml), which was autoclaved at 120 °C for 30 min. The cultured cells in the stationary growth phase have been used for experiments. Cultured cells of marine microalga of C. spinifera and A. crassum (each 9 g), and plant cells of C. roseus were individually added to this solution and the mixture was stirred for 2 h until it became homogeneous. The suspension was added dropwise from a dropping funnel with a glass tube into a 5% CaCl2 solution (11) with stirring to form pieces of spherical sodium alginate gel with 5 mm diameter immediately. Washing with water gave each immobilized cells which were used for biotransformation of benzophenone.
Time course experiments
Time course experiments to examine the biotransformation of benzophenone were carried out using eight flasks containing cultured cells (9 g for marine microalga or 70 g for plant cells) or immobilized cells, which included 9 g cells for marine microalga or 70 g for plant cells. Substrate (0.2 mmol) was administered to each of flasks and the mixtures were incubated on a rotary shaker at 20 °C. At a day interval, one of the flasks was taken out from the rotary shaker, and the cells (or immobilized cells) and medium were separated by centrifugation at 1000 g for 15 min (or by filtration). The extraction and analysis procedures were same as described above. The yield of the products was determined on the basis of the peak area from HPLC and expressed as a relative percentage to the total amount of the whole reaction products extracted.
Results and Discussions
The biotransformation product was isolated from the culture medium of C. spinifera, which had been incubated with benzophenone (1) for five days, by silica gel column chromatography in 49% yield. After the incubation period, the cells looked healty. The reduction product 2 was detected by HPLC. No additional conversion products were observed in spite of careful analyses by HPLC. Incubation of the substrate in medium without cells gave no transformation products. The structure of the product 2 was determined as diphenylmethanol by FABMS, 1 H and 13C NMR analyses (Fig. 1). To investigate the biotransformation pathway, the time course in the conversion of 1 was followed. Figure 2 showed that the amount of product 2 increased with time during the reaction with cultured C. spinifera cells.
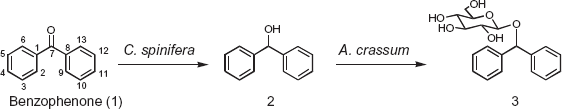
Glycosylation of benzophenone (1) by cultured cells of C. spinifera and A. crassum.
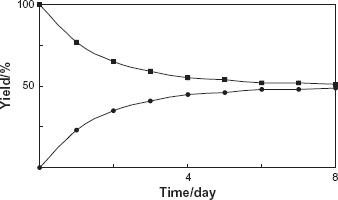
Time course of the reduction of benzophenone (1) by the cultured cells of C. spinifera. Yields of 1 (■) and 2 (•) are plotted.
C. spinifera cells were immobilized with sodium alginate at concentrations of 2%. The immobilized C. spinifera cells were incubated with benzophenole (1) for five days. The product 2 was obtained in 83% yield. The time course of the conversion of benzophenone (1) with immobilized C. spinifera cells was investigated. As shown in Figure 3, the reduction activity for benzophenone (1) was increased and the compound 2 was produced in higher yield in comparison with the case of the biotransformation using normal cells.
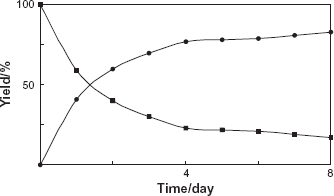
Time course of the reduction of benzophenone (1) by the immobilized cells of C. spinifera. Yields of 1 (■) and 2 (•) are plotted.
Next, incubation of cultured cells of A. crassum with diphenylmethanol (2) was carried out. A biotransformation product 3 was isolated by a combination of Diaion HP-20 column chromatography and preparative HPLC from MeOH extracts of the cell cultures of A. crassum. The yield of 3 was 2%. The structure of the product 3 was identifined as diphenylmethyl β-D-glucopyranoside (Fig. 1). To investigate the biotransformation pathway, the time course in the conversion of 2 by cultured A. crassum cells was examined and shown in Figure 4.
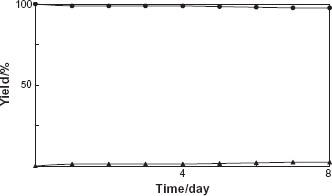
Time course of the glucosylation of diphenylmethanol (2) by the cultured cells of A. crassum. Yields of 2 (■) and 3 (▲) are plotted.
Biotransformation of diphenylmethanol (2) by immobilized A. crassum cells was investigated. The substrate, diphenylmethanol (2), was converted into product 3 at early stage of incubation period. The yield of 3 was 3%. The product 3 was obtained in higher yields in comparison with the case of the biotransformation using normal cells.
On the other hand, four biotransformation products 2–4 were isolated by a combination of Diaion HP-20 column chromatography and preparative HPLC after five days incubation of cultured plant cells of C. roseus with benzophenone. The yields of 2–4 were 20%, 11%, and 1%. The structure of the product 4 was identifined as diphenylmethyl primeveroside (Fig. 5). The product 4 was a new compound. To investigate the biotransformation pathway, the time course in the conversion of 1 by cultured C. roseus cells was examined. Products 2 and 3 were produced at an early stage of incubation. On the other hand, 4 was accumulated after 2 days of incubation (Fig. 6). These findings indicated that 1 was first converted to 2 and 3 and further glycosylation gave 4 as shown in Figure 5.
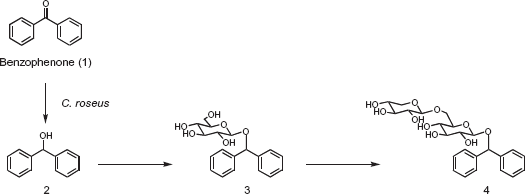
Glycosylation of benzophenone (1) by cultured cells of C. roseus.
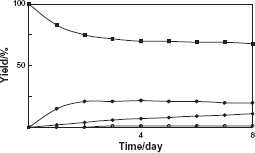
Time course of the biotransformation of benzophenone (1) by the cultured cells of C. roseus. Yields of 1 (■), 2 (•), 3 (◆), and 4 (○) are plotted.
Immobilized C. roseus cells were tested for their ability to convert benzophenone (1). The substrate, benzophenone (1), was converted into products 2–4 in 24%, 14%, and 2% yields by five days incubation. The products 2–4 were obtained in higher yields in comparison with the case of the biotransformation using normal cells.
The results of this experiment demonstrate that cultured marine microalga C. spinifera reduced benzophenone to diphenylmethanol and that cultured cells of A. crassum converted diphenylmethanol into its glucoside. The immobilized cells of both C. spinifera and A. crassum in sodium alginate gel are useful for bioremediation of benzophenone to be converted into diphenylmethyl β-D-glucopyranoside. On the other hand, cultured plant cells of C. roseus catalyzed reduction and glycosylation of benzophenone to give diphenylmethyl β-D-glucopyranoside and diphenylmethyl β-primeveroside. The immobilized cells of C. roseus would be useful biocatalysts to bioremediate freshwater contaminated by benzophenone.
Recently, it has been reported that freshwater microalga of Pseudokirchneriella subcapitata, Scenedesmusacutus, and Coelastrum reticulatum converted bisphenol A, which is an endocrine disrupting compound, into its glucoside. 10 On the other hand, recent paper revealed that estrogenicity of bisphenol A was eliminated by formation of the dig-lucoside and that reduced activity remained in the glucoside. 8 These studies demonstrate that metabolism of bisphenol A by freshwater microalga offers the possibility of bioremediation of contaminated water by endocrine disrupting chemicals. The present study showed, for the first time, that immobilized marine microalga of C. spinifera and A. crassum, and immobilized plant cells of C. roseus are useful bio-reactors for bioremediation of benzophenone, which is an environmental pollutant released into seas and rivers. The activity of plant cells C. roseus to glycosylate diphenyl methanol was much higher than that of microalga A. crassum. Further studies on sequential biotransformation of benzophenone using immobilized C. spinifera and C. roseus are now in progress.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributor-ship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
Footnotes
Acknowledgements
This work was supported by grant from The Salt Science Research Foundation, No. 1105.
