Abstract
Our previous study demonstrated that the apelin–APJ pathway contributed to myocardial regeneration and functional recovery after bone marrow-derived mesenchymal stem cell (BM-MSC) transplantation during the differentiation of BM-MSCs into cardiomyogenic cells in acute myocardial infarction (AMI) rat models. However, the underlying mechanisms by which apelin promotes cardiac repair and functional recovery have not been completely clarified. In the present study, we investigated whether apelin could mobilize and activate endogenous cardiac stem cells and progenitors, thereby mediating regeneration and repair of the myocardium after AMI in rat models. Six-week-old male Sprague–Dawley rats underwent AMI and received apelin-13 (200 ng, n = 10) or an equivalent volume of saline by intramyocardial injection (n = 10); there was also a sham operation group (n = 8). Proliferation of endogenous cardiac stem cells was analyzed by immunofluorescence staining in rat infarcted myocardium, and heart function was evaluated by echocardiography at 28 days after apelin-13 injection. Treatment with apelin-13 led to a significant increase of Ki-67+-c-kit+/Sca-1+/Flk-1+ endogenous cardiac stem or progenitor cells in the border zone and infarct zone of rat hearts at 28 days after myocardial infarction (MI). Significant increases in the expression of c-kit, Sca-1, and Flk-1 on both levels of transcription and translation were confirmed by real-time polymerase chain reaction (RT-PCR) and Western blot. Treatment of apelin-13 also resulted in a significant reduction of infarct size and improvement of cardiac function post-MI. We conclude that apelin-13 is able to enhance mobilization, survival, and proliferation of endogenous myocardial stem cells in the injured heart, providing a novel mechanistic explanation for how apelin-13 might repair the heart and improve cardiac function. Thus, apelin-13 or pharmacological agonists of the APJ receptor could act as novel therapies for heart regeneration.
Keywords
Introduction
Ischemic heart disease is still a leading cause of death throughout the world despite various therapeutic approaches that have been developed 1 . Recent studies have demonstrated that the adult mammalian heart is not a terminally differentiated organ and that there is a population of endogenous cardiac stem cells (eCSCs) or progenitors2,3. The presence of resident eCSCs in the adult mammalian heart of different species supports the notion that the adult myocardium has an intrinsic regenerative capability, which could be exploited to produce meaningful myocardial regeneration4,5. Malliaras and colleagues recently reported that autologous cardiosphere-derived cells from patients' endomyocardial biopsy specimens are capable of reducing scarring and reversing ventricular dysfunction after myocardial infarction (MI)6,7. Growing evidence has shown that adult CSCs and early committed cells express antigens c-kit, stem cell antigen 1 (Sca-1), and fetal liver kinase 1 (Flk-1)/kinase insert domain receptor (KDR)/vascular endothelial growth factor receptor 2 (VEGFR 2) and that c-kit+ or Sca-1+ cardiac-derived stem cells have been shown to be self-renewing, clonogenic, multipotent, and give rise to myocytes, smooth muscle cells (SMCs), and endothelial cells (ECs) in vitro and in vivo8–10. Although there are obvious advantages to using an allogenic product, the isolation and expansion of eCSCs for autologous cell transplantation is a lengthy and expensive process that is not always successful 11 . It is less likely that autologous eCSC transplantation will become widely available to treat acute and subacute MI12,13. Thus, it is critical to develop new strategies to specifically activate the intrinsic cardiac regenerative potential represented by the resident eCSCs in situ using soluble bioproteins 14 .
Apelin was discovered recently as an endogenous peptide associated with orphaned G protein-coupled receptor (APJ) in both humans and rodents 15 . Apelin-13, a natural peptide composed of 13 amino acids of apelin, is a potent inotropic agent and vasodilator. Treatment with apelin-13 demonstrates beneficial preclinical effects in MI models and shows significant improvement in heart function in patients with chronic heart failure16,17. The apelin–APJ pathway has only recently emerged as an important regulator of myocardial cell specification and heart development 18 . Ectopic expression of apelin and APJ results in the complete absence of cardiomyocyte development in zebrafish 18 . Apelin–APJ signaling has been proven to serve as a critical gradient for migration of mesodermal cells fated to the myocardial lineage, which plays a cell-autonomous role in myocardial specification and the formation of myocardial progenitors18,19. Our previous in vitro and in vivo studies demonstrated that apelin–APJ signaling was involved during the early development of the cardiomyogenic cells derived from mesenchymal stem cells (MSCs)20,21. However, the underlying mechanisms by which apelin promotes cardiac repair and functional recovery are not fully understood. The role of apelin in activation of endogenous myocardial progenitors or CSCs has yet to be established. In this study, we investigated whether apelin-13 could mobilize and activate CSCs and progenitors, thereby mediating regeneration and repairing the myocardium after acute myocardial infarction (AMI) in rat models.
Materials and Methods
MI Models and Apelin-13 Injection
Ligation of the left anterior descending coronary artery (LAD) was performed in 6-week-old male (200 ± 20 g body weight) Sprague–Dawley (SD) rats (Laboratory Animal Center of the Academy of Military Medical Science, Beijing, China). Animals were anesthetized using a combination of xylazine (4 mg/kg; Bayer Healthcare LLC, East Walpole MA, USA) and ketamine (80 mg/kg; Bayer Healthcare LLC) injected intramuscularly, and a left thoracotomy was performed under artificial ventilation with air. The heart was accessed through the fourth intercostal space. The pericardial sac was cut, and the heart exteriorized through the resulting space. The LAD was ligated with a 7-0 silk suture (Aesculap AG & Co. KG, Tuttlingen, Germany) approximately midway between the left atrium and the apex of the heart. Successful performance of coronary occlusion was verified by observing the development of a pale color in the distal myocardium after ligation and by recording the electrocardiogram (EKG; Nihon Kohden, Tokyo, Japan) evidence of infarction (ST segment elevation >1 mm in leads V1–V6). In the sham animals, the ligature was left loose. Animals were randomized into three groups: (A) sham-operated animals with no ligation performed (n = 8), (B) control animals that received only physiological saline by intramyocardial injection (n = 10), and (C) animals that received apelin-13 by intramyocardial injection (n = 10). Accordingly, apelin-13 (200 ng; Santa Cruz Biotechnology, Dallas, TX, USA) in 20 μl of saline, or an equivalent volume of saline without apelin-13, was injected at four different sites located in the infarct border zone for each rat. The investigators responsible for surgery and apelin-13 injection were blinded to the treatment groups. All procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals and the Animal Welfare Act in China and were approved by the Navy General Hospital Standing Committee.
Measurement of Infarct Size in the Myocardium
After echocardiographic measurements, the animals were anesthetized by a combination of xylazine and ketamine and euthanized, and the hearts were removed 28 days after MI and apelin-13 injection. Hearts were excised and sliced into 3-mm-thick cross sections below the ligature. Heart sections were incubated in 1% 2,3, 5-triphenyltetrazolium chloride (TTC; Sigma-Aldrich, St. Louis, MO, USA) and kept at 37°C for 30 min to stain the viable myocardium red and the infarction as pale areas. Infarcted and total left ventricular areas from both cut surfaces of each section were measured with Image Pro-Express software (Olympus, Tokyo, Japan).
Immunofluorescence Staining
The apical myocardium below the coronary artery ligation site was sectioned 28 days after apelin-13 treatments. Cryostat sections (5 μm) were cut from frozen tissue samples. Serial sections were immunolabeled with antibodies against c-kit (1:200; Biosynthesis Biotechnology Co. Ltd, Beijing, China), Flk-1 (1:200; Boster Biological Technology Ltd., Wuhan, China), and Sca-1 (1:200; Boster Biological Technology Ltd). For image analysis of myocyte proliferation, myocardial sections were stained with a rabbit monoclonal anti-Ki-67 antibody (1:200; Abcam, Cambridge, UK) followed by incubation with fluorescein isothiocyanate (FITC)-conjugated secondary antisera. Confocal images were obtained using a 40× objective on a Leica Microsystems TCS SP laser scanning confocal microscope (Leica, Solms, Germany). Digitized confocal images were processed by Leica confocal software and Adobe Photoshop (version 13.0; Adobe, San Jose, CA, USA). Myocardial Ki-67+/c-kit+, Ki-67+/Flk-1+, and Ki-67+/Sca-1+ cells were assessed by counting the numbers of positive cells per 100 nuclei of ischemic tissue.
Quantitative Real-Time PCR Analysis
Total RNA was extracted from peri-infarct areas of the apelin-13 (n = 10) and saline-treated (n = 10) myocardial tissues using an RNAqueous kit (Ambion, Austin, TX, USA), and cDNA was synthesized by reverse transcription using a QuantiTect Reverse Transcription kit (Applied Biosystems, Foster City, CA, USA). The quantitative real-time polymerase chain reaction (qRT-PCR) reactions were performed with Platinum SYBR Green qPCR SuperMix UDG (Invitrogen, Carlsbad, CA, USA) using gene-specific primers as follows: c-kit, 5′-AGCAAGAGTTAACGATTCCGGAG-3′ (forward) and 5′-CCAGAAAGGTGTAAGTGCCTCCT-3′ (reverse); Flk-1, 5′-TTTGGCAAATACAACCCTTCAGA-3′ (forward) and 5′-GCAGAAGATACTGTCACCACC-3′ (reverse); Sca-1, 5′-CTAGAGAACCCACGGGGAGA-3′ (forward) and 5′-TCTCACGTTTCTTGGGAG GC-3′ (reverse); glyceraldehyde 3-phosphate dehydrogenase (GAPDH), 5′-GACATGCCGCCTGGAGAAAC-3′ (forward) and 5′-AGCCCAGGATGCCCTTTA GT-3′ (reverse). PCR was performed using the Bio-Rad DNA Engine Opticon-2 (Bio-Rad, Hercules, CA, USA) under the following conditions: initial denaturation at 95°C for 10 min, followed by 40 cycles of 95°C for 15 s, and annealing of the primer template and extension at 60°C for 60 s. Target genes were assayed in triplicate on each plate. Ribosomal 18S RNA was used as a housekeeping gene for normalization. Expression of apelin-13 group relative to the saline group was quantified as described previously 9 .
Western Blot Analysis
Myocardial tissue lysates were extracted from peri-infarct and infarct areas in three groups: apelin-13 injection group (n = 10), saline treated (n = 10), and sham group (n = 8) for Western blot analysis of c-kit, Flk-1, and Sca-1 expression. Nitrocellulose membranes (Sigma-Aldrich) were blotted with rabbit monoclonal antibodies against c-kit, Flk-1, Sca-1 (1:1,000; Invitrogen), and polyclonal β-actin (1:1,000; Biosynthesis Biotechnology Co. Ltd). Membranes were then washed and incubated with a secondary antibody (1:5,000; Santa Cruz Biotechnology) coupled to horseradish peroxidase. The densitometric analysis was carried out using image acquisition and analysis software (Olympus).
Echocardiography
Rat heart function was assessed by transthoracic echocardiography, which was performed before surgery and at 28 days after MI and apelin-13 injection, using a 12-MHz transducer system designed for cardiac ultrasound (HP Sonos 5500; Philips, Eindhoven, The Netherlands). Two-dimensional images were obtained at midpapillary and apical levels. Left ventricular end-diastolic volume (LVEDV) and end-systolic volume (LVESV) were obtained by biplane area length method. Percent left ventricular ejection fraction (LVEF) and the percent fractional shortening (FS) were calculated:
LV%FS = LV internal diastolic diameter (LVIDd) – LV internal systolic diameter (LVIDs)/LVIDd × 100; LVEF = [(LVIDd)3 – (LVIDs)3]/(LVIDd)3 × 100.
All measurements were averaged on three consecutive cardiac cycles and were analyzed by two independent observers who were blinded to the treatment status of the animals.
Statistical Analysis
Data were presented as means ± standard error of the mean (SEM). Statistical comparisons were performed using either the Student's two-tailed t-test (paired) or the Wilcoxon matched pairs test as appropriate according to data distribution. All statistical tests were two sided and values of p < 0.05 were considered statistically significant. All analyses were performed with SAS software, version 9.3 (SAS Institute, Cary, NC, USA).
Results
Intramyocardial Injection of Apelin-13 Limits Infarct Size
To determine whether treatment with apelin-13 reduced the area of MI, we performed TTC staining analyses at 28 days after apelin-13 injection. Treatment with apelin-13 led to a significant decrease in the area of MI, as assessed by the ratio of the infarcted area to the LV area (Fig. 1). Injection of apelin-13 significantly limited infarct size (15.89 ± 3.14% of the LV, p < 0.0001) compared with the saline injection group (27.81 ± 5.32%) at 28 days after surgery.

The myocardial infarct area was significantly reduced in apelin-13-treated rats. Quantification of infarct size as assessed by the ratio of the infarcted area to LV area in the left ventricle of saline-injected and apelin-13-treated rats 28 days after AMI. **p < 0.0001.
Apelin-13 Activates eCSCs
To identify whether apelin-13 treatment can activate and mobilize resident eCSCs, we performed immunofluorescence of Ki-67 combined with c-kit, or Flk-1, or Sca-1 antibodies. The number of c-kit+, Flk-1+, and Sca-1+ cells was examined in the border and infarct zones of rat hearts 28 days after AMI and apelin-13 treatment. As shown in Figure 2A–C, apelin-13-treated infarcted hearts showed a significant increase of c-kit+, Sca-1+, and Flk-1+ cells, which coexpressed Ki-67 in the border and infarct regions. Quantitative analysis by double immunofluorescence staining showed that there were higher numbers of c-kit+/Ki-67+ cells in the infarcted area of rat hearts treated with apelin-13 compared with saline injection rat hearts (p = 0.033). Notably, both Sca-1+/Ki-67+ and Flk-1+/Ki-67+ cells were consistently significantly increased after apelin-13 injection compared with rats from either the sham or saline group (p = 0.022 and p = 0.019, respectively) as shown in Figure 2D.

Representative images showing the colocalization of Ki-67+ with c-kit+(A), Flk-1+ (B), and Sca-1+ (C) cells in the border and infarct zone of rat hearts at 28 days post-myocardial infarction (post-MI) and quantification of c-kit+, Sca-1+, and Flk-1+ cells (D). Nuclei were stained by DAPI (blue). (A) Ki-67+ cells (green) and c-Kit+ cells (red). (B) Ki-67+cells (green) and Flk-1+cells (red). (C) Ki-67+ cells (green) and Sca-1+ cells (red). Magnification: 40×. Ki-67+ and c-kit+, Flk-1+, and Sca-1+ cells were recruited into the ischemic area of mouse hearts at day 28 post-MI. Merged images show that Ki-67+ cells colocalized with c-kit+, Flk-1+, and Sca-1+ cells (yellow) in post-MI rats and that the increase was more striking in post-MI rats that received apelin-13 compared with saline-injected rats. Scale bars: 50 μm. (D) Comparison of quantification of c-kit+, Sca-1+, and Flk-1+ cells from 10 sections by counting the numbers of positive cells per 100 nuclei of ischemic tissue per rat 28 days after coronary ligation and administration of apelin-13 or saline. Bars indicate 95% confidence intervals (CIs). *p < 0.05, **p < 0.01.
Gene Expression of eCSC Markers
Next, we examined the expression of c-Kit, Sca-1, and Flk-1 mRNA in infarcted myocardial tissue as above at the same time points. Comparison of the RT-PCR quantitative analysis of the myocardial tissue in the sham, saline, and apelin-13 groups demonstrated that the infarct area in the apelin-13 injection group showed significant upregulation of the expression of c-kit, Sca-1, and Flk-1 mRNA in comparison to the saline and sham group (Fig. 3).

Quantitative reverse transcription polymerase chain reaction (PCR) analysis at 28 days after surgery. mRNA values are expressed as the ratio of Δ-ΔCT for c-kit, Flk-1, and Sca-1 mRNA relative to 18S rRNA. Data are presented as mean ± SE (n = 10). *p < 0.05, **p < 0.01.
Protein Expression of eCSC Markers
Western blot analysis showed that protein levels of c-kit, Sca-1, and Flk-1 were significantly increased in the border and infarct zones of rat hearts in apelin-13-treated rats compared with sham and saline-injected controls, which further confirmed that apelin-13 administration may activate eCSCs (Fig. 4).

Western blot analysis showed significantly upregulated expression of c-kit, Sca-1, and Flk-1 protein in rat infarcted myocardium treated with apelin-13 compared with saline-treated and sham rats. *p < 0.05, **p < 0.01.
Apelin-13 Treatment Halts LV Remodeling and Improves LV Function
At baseline (before coronary occlusion or sham operation), echocardiographic parameters of ventricular performance were similar in untreated and apelin-13-treated rats. At 28 days after AMI and apelin-13 injection, we evaluated the ventricular function by using the same method. In untreated rats, there was further deterioration of LV systolic function, additional LV dilation, and a further decrease in LV wall thickness. In contrast, in rats injected with apelin-13, the cardiac performance and anatomy remained essentially unchanged up to 28 days. As a consequence, apelin-13-injected rats exhibited a significantly higher LV FS (33.59 ± 2.36% vs. 20.65 ± 7.63%, p = 0.017), ejection fraction (53.80 ± 5.73% vs. 44.28 ± 5.21%, p < 0.023), and a significantly lower LVESV and LVEDV compared with saline-injected rats (Table 1).
Quantitative Measures of Left Ventricular Function
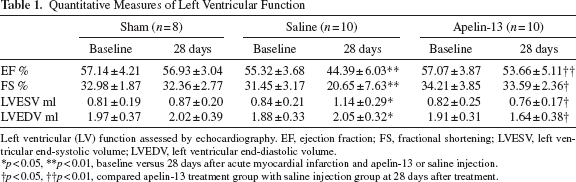
Left ventricular (LV) function assessed by echocardiography. EF, ejection fraction; FS, fractional shortening; LVESV, left ventricular end-systolic volume; LVEDV, left ventricular end-diastolic volume.
p < 0.05
p < 0.01, baseline versus 28 days after acute myocardial infarction and apelin-13 or saline injection.
p < 0.05
p < 0.01, compared apelin-13 treatment group with saline injection group at 28 days after treatment.
Discussion
We previously demonstrated that the apelin–APJ pathway is involved in the differentiation of cardiomyogenic cells derived from BM-MSCs after AMI in rats 20 . However little is known about whether apelin may mobilize endogenous myocardial progenitors and CSCs. The results of the current study demonstrated that small amounts of apelin-13 administered to the infarcted rat myocardium leads to a significant increase in the number of c-kit+, Sca-1+, and Flk-1+ eCSCs or progenitors in the damaged myocardium. This is accompanied by significant increases in the expression of c-kit, Sca-1, and Flk-1, as specific CSC markers, on levels of transcription and translation measured by RT-PCR and Western blot. We propose that apelin-13 treatment resulted in a significant reduction of infarct size and that improvement of cardiac function post-MI might occur through cardiac regeneration due to activation of eCSCs.
Recent studies have shown that the adult heart contains small populations of committed, multipotent CSCs, which are adapted to the cardiac microenvironment and participate in postnatal physiological and pathological cardiac renewal or repair 22 . These cells were c-kit+, Sca-1+, and Flk-1+ cells derived from autologous myocardium, that can be isolated, expanded in culture, and administered therapeutically to improve cardiac function in preclinical studies 22 . Intracoronary infusion of c-kit+ CSCs in patients with chronic ischemic cardiomyopathy and heart failure produces a striking improvement in both global and regional LV function, a reduction in infarct size, and an increase in viable tissue that persists for at least 1 year posttreatment 23 . Undoubtedly, the best replacement for the lost myocardium after MI is functional autologous myocardial tissue6,7. Unfortunately, that therapeutic approach is not always practical clinically13,22. Some studies indicated that these progenitor cells are highly active in the neonatal period because they are main participants in the hyperplastic growth and maturation of the myocardium through adulthood. However, most eCSCs in the adult myocardium are quiescent12,13. Recently, molecules modulating eCSC fate have been identified and described14,24. eCSCs possess insulin-like growth factor 1 (IGF-1) and hepatocyte growth factor (HGF) signaling pathways that regulate their growth, survival, and migration 11 . Ellison et al. demonstrated that intracoronary administration of IGF-1/HGF induces expression of cardiomyogenic progenitor markers and myocardial regeneration, and improves ventricular function 25 . The beneficial effects seen in these studies may be linked to stimulation of eCSCs26,27. There is a critical need to develop strategies to specifically activate the intrinsic cardiac regenerative potential represented by the resident eCSCs in situ using endogenous bioproteins11,14,25–27.
Apelin-13 has previously been reported to induce potent positive inotropic benefits and to reduce ventricular preload and afterload through triggering nitric oxide-dependent vasodilatation in normal and failing hearts16,17. Li et al. further demonstrated that apelin-13 promotes the homing of vascular progenitor cells to the infarcted myocardium, thereby mediating repair and functional recovery post-MI in mice 28 . We previously reported that expression of the apelin–APJ pathway during the differentiation of BM-MSCs into cardiomyogenic cells contributed to myocardial regeneration and functional recovery after BM-MSC transplantation in rat AMI models! 20 21 . In the current study, we provide direct evidence that intramyocardial injection of apelin-13 indeed reduced infarcted size, limited ventricular remodeling, and improved heart function in rats. Furthermore, these changes are associated with a significant upregulation of expression of c-kit, Sca-1, and Flk-1 as specific markers of CSCs or progenitors. Our experimental data further strengthen the understanding of the mechanism by which apelin promotes cardiac repair and functional recovery post-MI by activating CSCs or progenitors in animal models 27 .
Conclusion
Our study demonstrated that apelin-13 is indeed able to activate eCSCs or progenitors, which provides further mechanistic insights for how apelin-13 might repair the heart and improve cardiac function in rat AMI models. Importantly, our data suggest that apelin-13, or pharmacological agonists of the APJ receptor, could act as novel therapies for heart regeneration. Therefore, further experimental developments and refinements of this approach are needed for clinical application.
Footnotes
Acknowledgment
This work was supported by the National Natural Science Foundation Grant of China (No. 811 70094). We thank JianQing Zhang, Ph.D., and Jacques Riby, Ph.D., of the Department of Epidemiology University of Birmingham for their valuable comments and suggestions in writing and revising the manuscript. The authors declare no conflicts of interest.
