Abstract
Acute myocardial infarction (AMI) results in ischemic damage and death of cardiomyocytes and loss of vasculature. Stem cell therapy has emerged as a potentially promising strategy for maximizing cardiac function following ischemic injury. Issues of cell source, delivery, and quantification of response have challenged development of clinically viable strategies. In this study we investigate the effects of a well-defined bone marrow-derived allogeneic cell product delivered by catheter directly to the myocardium via the infarctrelated vessel on global and regional measures of left ventricular (LV) function in a porcine model of anterior wall myocardial infarction. Multipotent adult progenitor cells (MAPCs) were derived and expanded from the bone marrow of a donor Yorkshire pig. Anterior wall myocardial infarction (AMI) was induced by 90 min of mid-LAD occlusion using a balloon catheter. Two days after AMI was induced, either vehicle (Plasma Lyte-A, n = 7), low-dose (20 million, n = 6), or high-dose (200 million, n = 6) MAPCs were delivered directly to the myocardium via the infarct-related vessel using a transarterial microsyringe catheter-based delivery system. Echocardiography was used to measure LV function as a function of time after AMI. Animals that received low-dose cell treatment showed significant improvement in regional and global LV function and remodeling compared to the high-dose or control animals. Direct myocardial delivery of allogeneic MAPCs 2 days following AMI through the vessel wall of the infarct-related vessel is safe and results in delivery of cells throughout the infarct zone and improved cardiac function despite lack of long-term cell survival. These data further support the hypothesis of cell-based myocardial tissue repair by a paracrine mechanism and suggest a clinically translatable strategy for delivering cells at any time after AMI to modulate cardiac remodeling and function.
Keywords
Introduction
Acute myocardial infarction (AMI) results in loss of myocardial tissue and ventricular remodeling leading to decreased cardiac function and increased risk of chronic heart failure. Stem cell therapy is a promising approach for minimizing cardiac myocyte loss and optimizing ventricular remodeling in an attempt to improve regional and global cardiac function (13,18,30). While cell therapy has shown some promise in improving cardiac function after AMI (4,23,31) there are multiple issues surrounding not only the mechanism of benefit but also the details of clinical delivery and assessment that remain to be resolved (5,10,11,17,20).
One cell type that has garnered significant interest is the multipotent adult progenitor cell (MAPC) (12,22). MAPCs are an adherent adult bone marrow-derived culture that have been demonstrated to possess differentiation capability encompassing the epithelial, endothelial, neural, myogenic, hematopoeitic, osteogenic, hepatogenic, chondrogenic, and adipogenic lineages. MAPCs have shown engraftment and differentiation ability along multiple developmental lineages in nonobese diabetic severe combined immunodeficient (NOD-SCID) mice without evidence of teratoma formation. Several laboratories have demonstrated functional benefits associated with autologous and allogeneic MAPC delivery following AMI, making it a potentially useful source of allogeneic stem cells for maximizing cardiac function after AMI (2,19,27,32).
The goal of this study was to investigate a potential clinical strategy for the delivery of an allogeneic “off the shelf” stem cell to the myocardium using a strategy that could allow for stem cell delivery to the myocardium via the infarct-related vessel, obviating the need for additional vascular access, endoventricular injections in newly injured myocardium, and perhaps better retention than that seen with antegrade infusion (24).
Materials and Methods
Cell Culture
Pig MultiStem® (MAPC product) from Lots AP4 bgal WB 15, 17, 26, and 27 were used for all of the in vivo experiments. Human MultiStem®, a clinical-grade adult human bone marrow-derived stem cell product from Lonza (Walkerville, MD), from Lot 7CC 165 were used for the in vitro biocompatibility experiments.
Pig MAPCs were expanded in 60% Dulbecco's modified Eagle's medium low glucose (DMEM-LG)/40% MCDB-201 supplemented with 1× insulin-transferrin-selenium (ITS), 0.5× linoleic acid bovine serum albumin (LA-BSA), 0.1 mM l-ascorbic acid 2-phosphate, 100 U penicillin and 1,000 U streptomycin, 50 nM dexamethasone, and 2% prescreened fetal bovine serum (FBS) with 10 ng/ml each human platelet-derived growth factor BB (PDGF-BB) and human epidermal growth factor (EGF). Culture flasks were coated with 10 ng/ml fibronectin and the cells were seeded at 500 cells/cm2. Cells were passaged at a preconfluent stage and 0.25% trypsin was used to lift the cells off the culture flask. Trypsin was then neutralized by using FBS and cells were centrifuged at 400 × g for 5 min, and after removal of the supernatant the cells were resuspended in complete medium. While freezing, the cells were harvested, centrifuged, and resuspended at 10 million cells/1 ml cryopreservation medium that consists of 70% growth media, 20% FBS, and 10% dimethyl sulfoxide (DMSO) in 1-ml cryovials or bags. Cells were frozen at ~1°C/min to −80°C and transferred into liquid nitrogen for long-term storage. All cell counts and viability analyses were done according to the instructions for usage of the Guava PCA system (Guava Technologies, Hayward, CA).
Transarterial Catheter
The Cricket™ Micro-Infusion Catheter (transarterial catheter) from Mercator MedSystems (San Leandro, CA) has 510(k) clearance for the delivery of therapeutic agents into the vessel wall and perivascular tissues around coronary arteries and veins. The catheter has a length of 145 cm and an outer diameter of 2.0 mm. The balloon at the tip of the catheter sheaths a perpendicular-oriented, 35-gauge microneedle. The catheter is advanced into the desired treatment site, and upon inflation, the balloon expands and extrudes the microneedle through the vessel wall to deliver the therapy. Delivery is monitored with dilute contrast medium mixed into the therapeutic agent, and agents diffuse cylindrically around the vessel from the single needle penetration site. Following injection of the therapeutic agent, the delivery port is retracted into the sheathing structure with negative pressure on the balloon actuator. In this experiment, we used the catheter in vitro to assess the postinfusion cell recovery and viability.
Biocompatibility Testing with Catheter
Human MultiStem® (clinical-grade product from Lonza) were reformulated (200 million at 50 million cells/ml in Plasma Lyte A) to test their biocompatibility with the transarterial catheter. The reformulated cells were stored in shipping container for 6 h as per Athersys' standard operating protocol. The cells were then mixed with the contrast agent (20%) and filtered through a sterile 25-mm filter holder with 41-μm filter. The balloon of the catheter was inflated to a maximum of 2 atm (the safety valve on the balloon portal of the catheter started leaking if more than 2 atm pressure was applied) by using the inflation device. The cells (200 million in a total volume of 4 ml at 50 million cells/ml in Plasma Lyte A) were infused into the catheter at a rate of 100 μl/5 s (1.2 ml/min). A sample (0.1 ml) of the cell solution was collected before and after infusion of the total volume of the cells to test the pre- and postinfusion cell number and viability. After completion of the cell infusion, the balloon of the catheter was deflated.
VEGF Production
The mechanism of myocardial improvement following MAPC delivery has been suggested to be paracrine mediated, leading to angiogenesis and decreased cardiac myocyte death in peri-infarct area. One potential paracrine mediator of this effect is vascular endothelial growth factor (VEGF). Among other factors, VEGF has been demonstrated to play a major role in angiogenesis or new blood vessel growth in injured myocardium (3) and the improvements in cardiac function following MAPC delivery have previously been correlated with an angiogenic response (19,27). Therefore, as a part of biocompatibility testing of the cells and catheter, we determined the amount of VEGF secreted by the cells pre- and postcatheter infusion. As VEGF production is a function of cell number; the concentration of VEGF is calculated on a per cell basis to adjust for the difference in the number of cells in each well of the culture plate.
Induction of AMI and Cell Injection
Female and male domestic Yorkshire crossbred swine (n = 38) 5–6 months old were induced to have an acute myocardial infarction by obstruction of blood flow through the left anterior descending (LAD) artery by balloon catheter occlusion just distal to the first diagonal branch followed by reperfusion and percutaneous myocardial delivery of either pig stem cells or vehicle (Plasma Lyte A). An initial set of animals (n = 7) received 50 or 200 million cells within 30 min after AMI to evaluate cell persistence and distribution. A subset (n = 12) of the remaining animals that received cell injections at 48 h after AMI was used for toxicological assessment at 1-week post-cell injection. The remaining animals (n = 19) were enrolled in the efficacy study that has been discussed in detail in this article. Vessels were angiographically confirmed to be patent immediately following transarterial delivery.
Cell Injection
An incision was made over the femoral artery. An arterial sheath was introduced, and a guide catheter was advanced into the artery. Heparin was administered as needed to achieve and maintain an activated clotting time (ACT) of approximately ≥ 250 s.
The injection device was inserted along the guide catheter and advanced into the LAD. The cells/Plasma Lyte A were injected through the catheter using a 1-ml leur-lock syringe. In the preliminary cell persistence and distribution study, either 50 or 200 million β-galactosidase-labeled cells (at a concentration of 50 million/ml) were infused into the myocardium through the transarterial approach within 30 min post-AMI. For those animals that received 50 million cells (50 million/ml in a total volume of 1 ml), one injection was performed, whereas those animals that received 200 million cells (50 million/ml in a total volume of 4 ml) received cells through two injections of 100 million cells (~2 ml) each (Fig. 1A).

(A) Graphically represents injection sites (light ovals) in left anterior descending and method of sectioning the left ventricle into cross-sectional rings (B) for cell distribution analysis. Tissue blocks from cross sections were divided into segments (B) and circumferential distribution (C) was measured as number of blocks in one ring with >100 β-galactosidase (β-gal) + cells (red areas in B) divided by total number of blocks in that ring × 100%. Whereas radial distribution (D) was measured as number of β-gal + cells in each of the three layers divided by total number of β-gal + cells in full thickness × 100%.
The remaining animals receiving the low dose, 20 million cells, were dosed at one site (distal to the occlusion) with at volume of ~1 ml of cells (20 million/ml). To ensure delivery of complete cell dose, the catheter lumen was flushed with ~0.4 ml of Plasma Lyte A immediately following cell injection to wash out the cells in the dead space of the catheter. Animals in the high-dose group, 200 million cells, were dosed at two sites and received ~2 ml of cells (50 million/ml) each at site 1 and site 2. The first dose (site 1) was in the distal LAD, and the second dose was at least 2 cm proximal to site 1. Plasma Lyte A (~0.4 ml) was infused through the catheter lumen to ensure complete delivery of cell dose at both sites. Animals assigned to the control group were dosed with Plasma Lyte A at a volume of ~2 ml each at site 1 and site 2. All injections were made at a rate of approximately 1.2 ml/min. After administration of the vehicle and/or test article, the injection catheter was withdrawn, the skin and subcutaneous layers were closed with absorbable suture, and the skin was closed with skin glue.
Evaluation of Cell Persistence and Distribution
The left ventricle was sectioned into five rings of approximately 1 cm thickness, and one ring was cut into 10–14 blocks (see Fig. 1A, B). The tissue blocks were stained overnight with β-galactosidase (X-gal) solution and embedded in OCT. Sections 10 μm thick were then cut from each of the blocks. The slides were evaluated for β-galactosidase-positive cells with bright field and fluorescence microscopy. Cell persistence and distribution (circumferential and radial distribution) were evaluated by trained observers blinded to treatment.
Evaluation of Echocardiographic Endpoints
Echocardiographic examinations were conducted in a blinded fashion on all surviving animals in the safety and efficacy arm of this study, where the animals received either 20 or 200 million cells or vehicle. Echocardiography was performed prior to surgery, on day 2 prior to injection, at 1 month ± 3 days, and 3 months ± 3 days posttreatment. Images were obtained for each animal in short axis view (at apical, papillary, and mitral valve level), long axis view, and a two- and four-chamber view. All measurements of digital data were performed in triplicate and averaged to ensure quality.
The primary outcome variable was defined as left ventricular (LV) ejection fraction (EF), and as secondary outcome end-systolic volume (ESV), circumferential strain, torsion, and wall motion score index (WMSI). WMSI was calculated by first scoring the segments as being normal (1), mildly hypokinetic (2), severely hypokinetic (2.5), akinetic (3), or dyskinetic (4), and then by dividing the sum of wall motion score with the number of visualized segments. Focused wall motion analysis was performed using only the segments (i.e., apical cap, four apical segments, and septal, anteroseptal, anterior, and anterolateral segments at the midventricular level) that are primarily affected by LAD ligation as performed in this experiment.
Strain was measured utilizing speckle tracking technology of Echopac software (Version 3.x, GE Medical systems). Affected segments were defined as those in the LAD territory, specifically septal, anteroseptal, anterior, and lateral in short axis apical views and septal, anteroseptal, and anterior in short axis papillary muscle views. Torsion measurements were made offline using the same Echopac software. Basal and apical rotation was measured as degrees rotation between end diastole and end systole, and left ventricular torsion was determined by subtracting basal rotation from apical rotation. Care was taken to verify that basal planes included the mitral valve and that apical planes were below the level of the papillary muscles. All measurements were performed in triplicate and by separate observers to ensure accuracy.
Statistical Evaluation of Echocardiography Data
Data are presented as mean ± standard error of measurement. Initial statistical analysis was performed with statistician being blinded to treatment and control groups. The identity of the groups was disclosed to compile the final conclusions. Simple comparisons between two samples were performed by paired and unpaired t-testing, as appropriate. Comparisons between three samples were compared by one-way analysis of variance. The three samples used in this analysis are group 1 (control/Plasma Lyte A), group 2 (low dose/20 million cells), and group 3 (high dose/200 million cells).
The impact of treatment was assessed by calculating the difference between the value of outcome variable at 1 month (or 3 months) and their value at day 2. We used this approach because the infarct size, even in well-controlled experiments, always shows individual variability. By using the outcome variable difference, adjustments were made for possible differences in the infarct size.
To quantify the impact of treatment on outcome variable difference, a General Linear Model approach to univariate analysis of variance was used with groups and time as fixed factors and animal ID as a random factor. The model tests for the difference between groups, the difference between times, and the interaction between group and times. A significant interaction means that over the course of time the tested variable evolves in the opposite direction in two groups, with the difference between groups either converging or diverging. In treatment trials, detection of convergence or divergence of data over time means that the treatment effect is either increasing or decreasing. Absence of interaction means that the treatment effect over a time period tested is stable. Finally, the difference between the three groups was assessed by post hoc Tukey HSD test (25). Additionally, Games Howell post hoc test (25) was performed to ascertain the stability of post hoc analysis if assumptions of homogeneous variances was violated.
Statistical analysis was performed by SPSS 10.0. All of the authors had full access to the data and take responsibility for its integrity. All authors have read and agreed to the manuscript as written.
Results
Biocompatibility of Cells with the Catheter
Cell Recovery and Viability
Cryopreserved human stem cells were thawed, reformulated, and infused through transarterial catheters in vitro. A total of four transarterial catheters from two different lots (two from each lot) were used to infuse the cells (200 million at 50 million cells/ml) and tested for pre- and postinfusion cell recovery and viability. An average of 209 ± 16 million viable cells was recovered postinfusion with no change in the viability of the total infused cell product. The total number of viable cells and the viability of the cells postinfusion were similar to that of preinfusion viable cell number and viability.
VEGF Production
Based on the results from four in vitro catheter runs, the average concentration of VEGF produced by the preinfusion and postinfusion cell product was 0.014 ± 0.010 and 0.012 ± 0.009 pg/cell, respectively. Thus, catheter infusion did not alter the ability of the cells to produce VEGF.
Cell Persistence and Distribution
An initial set of animals (n = 7) was studied to verify MAPC delivery to the myocardium and distribution of MAPCs delivered immediately after AMI by transarterial delivery. Animals were sacrificed at 2 and 8 weeks after cell delivery and the myocardium was analyzed to assess the distribution of MAPCs by β-galactosidase (β-gal) staining. The data were analyzed on a per treatment group basis for the number of persistent cells in one ring per animal. The data are reflected as the percentage of cells counted. The β-galactosidase-stained cells were identified in several segments around the circumference of the LV (Fig. 1B, C), suggesting active MAPC migration within the myocardium following cell injection.
Further analysis of the MAPC staining in the tissue blocks indicated that the animals that received 200 million cells showed a trend towards higher circumferential cell distribution compared to animals that received 50 million cells at 8 weeks posttransplantation (Fig. 1B, C). The radial distribution analysis across doses shows that the cells are equally distributed through all three layers of the myocardium (Fig. 1D).
Induction of AMI in Pigs
AMI was induced in adult pigs by transient ligation of the LAD followed by reperfusion. Two days after infarct creation, when the creatine kinase muscle brain isoforms (CK-MB) had decreased from the peak (Table 1), the animals received vehicle, 20 (low-dose) or 200 million (high-dose) MAPCs by transarterial catheter delivery into the adventitial layer of the LAD. A subset of animals [n = 12; low dose (4), high dose (4), and Plasma Lyte A (4)] was sacrificed at 1 week post-cell injection for toxicological assessment. Administration of the test article was not associated with any adverse clinical observations, body weight changes, hematology or coagulation parameters, or macroscopic or microscopic findings at 1 week post-cell injection (data not shown). In the remaining animals [n = 19; low dose (6), high dose (6), and Plasma Lyte A (7)] echocardiography was performed at baseline (prior to AMI), 2 days (just prior to MAPC injection), 1 month, and 3 months post-AMI in order to characterize LV function through the measurement of several parameters, including measurement of EF and ESV using the Single-plane Simpson's method (Table 2). Regional function was quantified by wall motion score index (WMSI) and radial and circumferential strain. Global mechanics were quantified by measuring left ventricular torsion.
Cardiac Enzyme (CK-MB and Troponin I) Levels

CK-MB, creatine kinase muscle brain isoforms; AMI, acute myocardial infarction.
End Systolic Volume (ESV) and Ejection Fraction (EF) Measurements as a Function of Time After Acute Myocardial Infarction and Treatment

Two days after AMI was induced, the animals were examined by echocardiography to determine heart function. The WMSI and focused WMSI were significantly increased in all groups compared to the initial baseline measurements that were performed prior to AMI (p < 0.001) (Table 3), demonstrating a significant decline in contractility of the affected territories and that the animals in different groups received comparable infarcts. Consistent with these observations, there was a significant decline in circumferential strain in the affected areas compared to noninfarcted territories. There was no significant change at day 2, prior to cell injection, compared to baseline in EF (p = 0.20) or ESV (p = 0.09) in any of the groups. The lack of change in EF is most likely due to hyperdynamic contractility of the noninfarcted territories of myocardium. Analysis of serial cardiac enzyme (CK-MB and troponin I) measurements from induction of AMI through 48 h post-AMI was consistent across groups. These data are consistent with the wall motion results indicating that a large infarct was created in all the animals and that the degree of myocardial necrosis was similar among all three groups prior to receiving MAPCs (Table 1).
Wall Motion Measurements as a Function of Time After Acute Myocardial Infarction and Treatment

fWMSI, focused wall motion score index.
Functional Effects of MAPCs After Delivery by Transarterial Catheter
MAPCs were delivered by a transarterial catheter microsyringe through the artery wall of the infarct-related vessel into the adventitial layers of the LAD at 2 days following AMI. We chose to deliver MAPCs after 2 days following AMI because this allowed the confirmation that the cardiac enzymes, CK-MB and troponin, had peaked and were returning back to pre-AMI levels (Table 1). The effects of MAPCs on cardiac enzymes were then monitored after cell delivery to determine if cell delivery by the transarterial catheter caused further myocardial injury. Importantly, no significant changes in the cardiac enzymes were observed at 48 h following MAPC delivery (Table 1).
Analysis of the echocardiography data (Fig. 2A, B) indicated that there were significant differences in cardiac function between the three groups 1 month after AMI. EF was significantly improved in animals that received MAPCs (p = 0.042) compared to the vehicle-treated (control) animals. Compared to the baseline measurements, there was no significant change in EF or ESV in the low-dose MAPC group at 1 month, whereas cardiac function was worse in the control group as evidenced by a decrease in EF and an increase in ESV. As a result, left ventricular function (i.e., EF) was better preserved (p = 0.026) and adverse remodeling (i.e., ESV increase) was less prominent (p = 0.013) in the low-dose group compared to control animals. Interestingly, in the high-dose MAPC group there were trends similar to the low-dose group; however, these changes were not statistically different from control animals (p = 0.4 and p = 0.7 for ESV and EF, respectively).

Graphs representing EF (A) and end systolic volume (ESV) (B) at baseline and 1 month postinfarction. At 1 month, ESV for the low-dose group was significantly reduced compared to control (p = 0.013) (B). Low-dose-treated animals had a significantly smaller decrease in EF between day 2 and 1 and 3 months (p = 0.0003 and p = 0.001) compared with control and high-dose groups (C). Post hoc analysis (D) showed an increase in ESV in all groups with the low-dose group exhibiting a significantly smaller increase between day 2 and 1 and 3 months (p = 0.014 and p = 0.014) than control and high-dose groups, respectively.
EF significantly worsened between month 1 and month 3 (p = 0.003) in all treatment groups (Table 1), possibly due to the weight gain (35.6 ± 4.9%) that occurred in the animals during this period, resulting in a concomitant increase in the growth of their heart chambers and increased cardiovascular load. The absence of interaction between EF and cell treatment (p = 0.385) signifies that the EF decrease between month 1 and month 3 was similar in all three groups. Post hoc testing (Fig. 2C, D) showed that the decrease in EF between day 2, month 1, and month 3 was less pronounced in the low-dose group than in the control or high-dose groups (p = 0.0003 and p = 0.001, respectively).
There was a trend approaching significant difference between the three groups in ESV change (p = 0.054) over all times. ESV significantly increased between month 1 and month 3 (p < 0.0001) in all treatment groups. Again, absence of interaction between ESV and cell treatment (p = 0.285) signifies that the increase in ESV was similar in all three groups between 1 and 3 months. Consistent with our observations between 2 days and 1 month, post hoc testing showed that the low-dose group had consistently less dilation of the LV chamber as evidenced by a lower increase of ESV after MI than the control or high-dose groups at all time points (p = 0.014 and p = 0.012, respectively).
In addition to EF and ESV, echocardiography was also used to evaluate wall motion (Fig. 3A) and focused wall motion limited to territories perfused by the LAD (Fig. 3B). Similar to what was observed for EF and ESV, the low-dose group had a significant improvement in WMSI at 1 month compared to the control (p = 0.004). At 3 months, there was a trend approaching significant difference between all three groups in WMSI change (p = 0.064). Post hoc testing (Fig. 3C) showed that the low-dose group had a consistently smaller increase in WMSI after MI than the control or high-dose groups at all time points (p = 0.005 and p = 0.003, respectively).

Global wall motion score index (WMSI) (A) and focused WMSI (B) over baseline, 2 days, and 1 month postinfarction). Only the low-dose animals were observed to have a decrease in wall motion observed for both the WMSI and focused WMSI from day 2 to 1 month, with the remaining groups showing an increase in WMSI and focused WMSI. There was a consistently smaller increase in the low-dose groups compared to control and high-dose groups in both WMSI (C) (p = 0.005 and p = 0.003, respectively) and focused WMSI (D) (p = 0.002 and p < 0.001, respectively) between day 2 and 1 and 3 months.
Focused WMSI (fWMSI) analysis of LAD-related territories showed significant differences between the three groups in WMSI change (p = 0.016) over all time points (Fig. 3B). As observed for WMSI, the low-dose group also had a significant improvement at 1 month compared to the control (p = 0.001). Post hoc testing (Fig. 3D) showed that the low-dose group had a consistently smaller increase in fWMSI after MI than the control or high-dose groups (p = 0.002 and p < 0.001, respectively).
Consistent with the preservation of global and regional function observed in the MAPC-treated animals, we also observed improvements in circumferential strain with MAPC treatment (Fig. 4A). At 1 month following infarction, there were differences in circumferential strain of at-risk segments between groups that approached significance (p = 0.052). However, on post hoc analysis, circumferential strains in the area at risk were better in both low-dose and high-dose groups when compared to control (p < 0.001 for both). There were no differences between two treatment groups (p = 0.95). Importantly, there was no change in circumferential strain in the noninfarct area in any of the treatment groups. At 3 months the difference in circumferential strain between groups continued to increase and reached statistical significance (p = 0.016). Post hoc analysis of circumferential strains in the area at risk showed that strain was increased in the low-dose group (p = 0.001) and showed a trend towards significance in the high-dose group (p = 0.18) when compared to control. There were no differences between two treatment groups (p = 0.3).
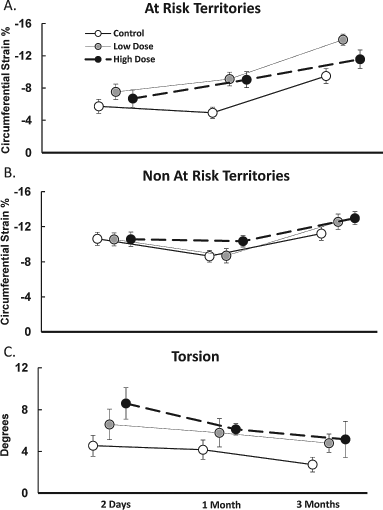
Circumferential strain of at-risk (A) and non-at-risk (B) territories showing a significant difference (p = 0.016) in circumferential strain of at-risk segments between animals receiving multipotent adult progenitor cells (MAPCs) compared to control with the low-dose group, having consistently higher strain at all three time points. There were no significant differences between the groups over time in circumferential strain of non-at-risk segments (B) or in torsion (C).
There was no significant difference in baseline torsion between treatment groups. Torsion steadily declined over the 3-month study period in all three groups. We did not observe any statistically significant difference in torsion between groups at any time during the study.
Discussion
The increasing use of reperfusion therapy including primary percutaneous coronary intervention has significantly increased survival in patients presenting with acute myocardial infarction. This increase in the survival has heightened the need to develop strategies to further minimize myocardial damage or optimize cardiac remodeling in order to prevent the development of chronic heart failure. One of the strategies currently being advanced to maximize cardiac function after acute myocardial infarction is cell therapy. While several clinical trials have suggested potential benefit of cell delivery in patients with acute myocardial infarction, several issues remain to be determined, including cell type, mode of delivery, and method of cardiac assessment. Here we demonstrate that MAPCs can be safely delivered by transarterial catheter into the adventitial layer of the target vessel at 2 days following an AMI. Furthermore, echocardiography can provide a number of parameters to assess LV function, including EF, ESV, wall motion, circumferential strain, and torsion.
We have previously demonstrated that autologous and allogeneic MAPCs result in similar improvements in left ventricular function and remodeling. In these studies we used allogeneic MAPCs obtained from a donor Yorkshire pig. The animal used as donor for these studies was obtained several months prior the experimental animal group as well as from a distinct supplier; thus, the donor animal and the experimental animals were not closely related.
The delivery of MAPCs led to improved cardiac function and muted negative remodeling following AMI. Multiple groups have demonstrated that the delivery of MAPCs to ischemic myocardium leads to an increase in vascular density (27). Consistent with increased vascular density as a potential mechanism, previously we have demonstrated that the MAPCs secreted significant levels of VEGF (27). Importantly, we also showed that passage of the cells through the transarterial needle did not result in loss of cell viability or altered VEGF secretion.
Like allogeneic mesenchymal stem cells that similarly have been shown to improve cardiac function in porcine models of AMI (1,16), MAPCs have both been shown to be anti-inflammatory and immunomodulatory, including inhibiting mixed lymphocyte reactions (MLRs) (7,14,15). Thus, in addition to increasing vascular density, MAPC delivery could lead to a decrease in neutrophil activity in the infarct zone. While we were not able to quantify neutrophil numbers in this study, preliminary results in our laboratory demonstrate that MAPC delivery at the time of AMI leads to a significant decrease in neutrophil accumulation 3 days after AMI.
To date the majority of clinical studies have used autologous bone marrow mononuclear cell preparations. Because of the complexities associated with harvesting and processing autologous bone marrow it is not possible to deliver bone marrow mononuclear cells at the time of primary percutaneous coronary intervention. An off-the-shelf allogeneic cell product, like MAPCs, offers the best opportunity for delivering cells at the time of mechanical reperfusion. The system that we implemented in these studies offers the potential to deliver an efficacious cell therapy at any time after reperfusion.
The delivery of MAPCs 2 days after AMI in the efficacy study allowed for cardiac biomarkers to decline following AMI. Cell delivery following decline in cardiac biomarkers allowed us to determine if cell delivery led to additional myocardial damage. Based on the levels of CK-MB and troponin I at 6 and 48 h post-cell delivery, compared to pre-cell delivery, there was no increase in cardiac biomarkers following cell or vehicle delivery, demonstrating that there was no evidence of reinfarction following cell injection (Table 1).
The transarterial catheter we used in our studies offers the possibility of delivering a cell product directly to the infarct zone without any evidence of inducing additional myonecrosis as has been seen with the intracoronary delivery with some cell products (6,28). The delivery of the cells by a transarterial catheter into the adventitial layer of the infarct-related coronary artery did not cause pericardial effusions following the procedure, although the animals had activated clotting times greater than 250 s at the time of cell or vehicle injection. Adventitial delivery of the cells obviates that need for cell homing to the myocardium, binding to the endothelium, and migration into the infarct zone, perhaps making the process of cell delivery more efficient. That said, it is noteworthy that while both the low- and high-dose arms of our study demonstrated improvement compared to vehicle injection, the low-dose arm demonstrated greater benefit. These findings are consistent with that seen with other cell types, such as the mesenchymal progenitor cell with endoventricular delivery (9), and suggest the possibility of a threshold effect, at least for a single dose, beyond which increased cell therapy provides no further benefit or even reduced benefit.
We used echocardiography to assess both global and regional effects of cell therapy on cardiac function and remodeling following cell delivery. The majority of clinical studies in AMI have implemented either left ventriculography and/or cardiac MRI to assess global and regional changes in cardiac function (11,23). Echocardiography, which is widely available, is capable of quantifying regional wall motion as well as cardiac strain (8,21,26). In this study we demonstrated early improvement in global function 1 month following the delivery of 20 million MAPCs, similar to other reports (27,29). However, over the ensuing 2 months, the global benefits of cell therapy were less robust, possibly due to the large increase in weight gain that occurred during this time period resulting in an increased cardiac demand as well as heart size. That said, as has been observed in clinical trials (11), strain imaging demonstrated sustained improvement in regional wall function in cell-treated animals compared to control animals at 3 months. Consistent with cell therapy having a significant effect on injured tissue, but not resulting in restoration of global cardiac mechanics, we did not observe a significant improvement in cardiac torsion in MAPC-treated animals.
In conclusion, MAPC delivery via a transarterial catheter 2 days after AMI in a porcine model of anterior wall myocardial infarction led to a significant positive modulation of left ventricular function and remodeling. The combined system of the catheter and cell product we implemented in these studies allows for the direct and safe delivery of an efficacious cell product to the injured myocardium at any time following reperfusion in clinical populations.
Footnotes
Acknowledgments
We would like to thank the State of Ohio for the “Third Frontier Project,” and support from the Biotechnology Research and Technology Transfer program. We would like to thank Wouter Vant Hof, Robert Mays, Mark Frey, Emily Rebro, Abner Mhashilkar, Piradeep Suntharalingam, Sherry Boozer, and Pina Patel for their efforts in generating the pig cell banks and helping in the cell persistence arm of the study. We would like to thank Mark Johnson and other staff at MPI Research for their skills and support in performing high-quality animal research. We would like to thank the staff of Echocardiography Core Lab at the Cleveland Clinic who performed the echocardiographic analysis. We would like to thank Kirk Seward from Mercator MedSystems for assistance in implementation of the catheter delivery platform. Coauthors R.D., A.T., S.M., N.L., and A.R. are scientists employed by Athersys, Inc., who own the MultiStem® technology. All other authors declare no conflicts of interest.
