Abstract
To determine whether newer combination cytokine treatment with granulocyte colony-stimulating factor (G-CSF) and darbepoetin can improve efficacy of stem cell therapy, we evaluated safety and peripheral blood stem/progenitor cell (PBSC) mobilizing effects of combination cytokine in comparison with G-CSF alone in patients with acute myocardial infarction (AMI). We randomized 60 patients with AMI into two groups under 2:1 ratio; combination treatment with darbepoetin and G-CSF (n = 41: Combicytokine group) and the G-CSF alone (n = 19: G-CSF group). After coronary angioplasty, G-CSF was treated for 3 days with dose of 10 μg/kg/day in both groups. Only in the combicytokine group, additional single intravenous injection of 4.5 μg/kg of darbepoetin was administrated immediate after coronary angioplasty. Combination cytokine treatment was well tolerated as was G-CSF alone. PBSCs were obtained by apheresis for intracoronary infusion after completion of cytokine treatment and were analyzed by flow cytometry. The purity of proangiogenic cells was higher in combination cytokine group than the G-CSF group. Specifically, proportion of CD34+/KDR+ endothelial progenitor cells, CD3+/CD31+ angiogenic T cells and Tie2+/CXCR4+ cells in apheresis products were higher in the combicytokine group. These meant that the combicytokine treatment recruited PBSCs in higher purity and fewer unwanted inflammatory cells than G-CSF alone in apheresis products. Combination treatment with darbepoetin and G-CSF is safe and more efficient to mobilize and recruit proangiogenic cells than G-CSF alone in patients with AMI. (Trial registration: www.ClinicalTrials.gov identifier: NCT00501917)
Keywords
Introduction
We and another group reported that peripheral blood stem cell (PBSC) therapy is effective for improvement of left ventricular systolic function and perfusion in the patients with ischemic heart disease (4,9,10). Granulocyte colony-stimulating factor (G-CSF) has been popularly used for stem cell therapy for cardiovascular disease. We also demonstrated the feasibility and usefulness of G-CSF to mobilize PBSCs for stem cell therapy in patients with ischemic heart disease (3,20). Intracoronary infusion of mobilized PBSCs by G-CSF is effective for improvement of left ventricular systolic function (4,9,10). However, the extent of improvement is only modest. Additionally, there are concerns that G-CSF may attenuate homing effects of mobilized PBSCs by cleaving surface CXC chemokine receptor type 4 (CXCR4) expression, which is essential for homing to the ischemic region (6).
To enhance efficacy of current PBSC therapy with G-CSF, we need to improve homing and angiogenic efficiency and survival of transplanted PBSCs in ischemic myocardium. Expecting additive effects of adjunctive erythropoietin administration, we developed a novel stem/progenitor cell treatment strategy for ischemic heart disease combining erythropoietin with stem/progenitor cell therapy using G-CSF. Erythropoietin is a cytokine secreted by the kidney in response to hypoxia and regulates plasma hemoglobin concentrations. Its cytoprotective effect is repeatedly reported in preclinical studies (15). Erythropoietin can induce angiogenesis by mobilization of endothelial progenitor cells (5). Moreover, combination of erythropoietin with bone marrow transplantation improved efficacy of stem cell therapy in an animal study (25). We evaluated novel strategy of combination cytokine treatment with darbepoetin, a long-acting analogue of erythropoietin, and G-CSF to determine whether this newer strategy has a potential to improve efficacy of current G-CSF-based PBSC therapy.
Materials and Methods
Subjects
Patients evaluated in this study were enrolled in the “Myocardial Regeneration and Angiogenesis in Myocardial Infarction with G-CSF and Intra-Coronary Stem Cell Infusion (MAGIC Cell)-5-combicytokine” trial, which is a prospective randomized multicenter partly double-blind study. This study was performed as a substudy of MAGIC Cell-5-combicytokine trial in two participating centers. We prospectively collected peripheral blood and apheresis products from patients who were randomized to either treatment of G-CSF alone (the G-CSF group) or of combination cytokine with G-CSF and darbepoetin (the combicytokine group) with randomization ratio of 1:2 between June 2007 and April 2009. The study protocol was approved by the institutional review board of Seoul National University Hospital.
Patients with acute ST segment elevation myocardial infarction, who received successful revascularization to culprit vessels with percutaneous coronary intervention were enrolled. Patients with the following exclusion criteria were excluded from the study: elderly (>80 years), persistent severe heart failure, uncontrolled myocardial ischemia or ventricular tachycardia, malignancy, serious current infection or hematologic disease, and chronic renal failure with serum creatinine above 2.0 mg/dl. Informed consent was obtained from patients.
Cytokine Administration, PBSC Mobilization, and Collection
Patients randomized to the combicytokine group received 4.5 μg/kg (maximum 300 μg) of intravenous darbepoetin (Amegen, USA) infusion just after revascularization. Patients in the G-CSF group received placebo infusion. For both the G-CSF and combicytokine group, G-CSF was administrated subcutaneously twice a day at a dose of 5 mg/kg for 3 days thereafter. The blood tests for complete blood cell count, cardiac enzymes including creatine kinase MB isoenzyme (CK-MB) and electrocardiogram were checked daily, and 6 and 12 h after the apheresis procedure. A questionnaire for patients was acquired for monitoring the side effects of G-CSF and darbepoetin.
One day after completion of the scheduled G-CSF injection, PBSCs were collected from the mononuclear cell component (Cobe spectra® apheresis system, Cobe BCT, Inc, Lakewood, CO, USA). Peripheral blood samples were also obtained just before apheresis. Cells harvested from apheresis product and peripheral blood were analyzed with the following antibodies of cell surface markers: anti-human kinase insert domain-containing receptor (KDR), anti-CXCR4, anti-endothelial TEK tyrosine kinase (Tie2; R&D Systems, Wiesbaden, Germany), anti-CD133 (Miltenyi Biotec GmbH, Gladbach, Germany), anti-CD34, anti-CD3 (Dako cytomation, Copenhagen, Denmark), anti-CD31, anti-CD73, anti-vascular endothelial (VE)-cadherin (BD Biosciences, San Jose, USA), and anti-CD105 (Serotec, Oxford, UK) by fluorescence activated cell sorter (FACS) analysis (see Fig. 1).
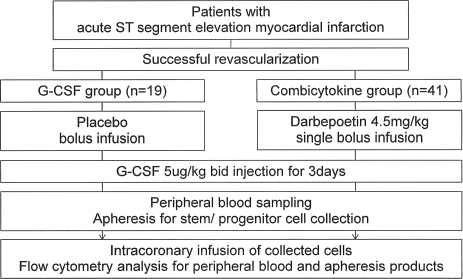
Study protocol. G-CSF, granulocyte colony-stimulating factor.
Statistics
Continuous variables are expressed as mean + SD and categorical variable as an actual number of patients and percentages. To compare the variables between the G-CSF group and the combicytokine group, we used Student's t-test for continuous variables and Pearson's chi-square test for categorical variables. Multiple linear regression analysis was performed to identify determinants of CD34+/KDR+ cell proportion in apheresis products and variables showed p < 0.1 in univariate analysis and clinical significance were included. All statistical analyses were performed with SPSS17.0 and values of p < 0.05 were taken to indicate statistical significance.
Results
Patient Characteristics
Nineteen patients from the G-CSF group and 41 patients from the combicytokine groups were included in this study. The baseline characteristics of the study participants are summarized in Table 1. There were no significant differences between two groups.
Baseline Characteristics of the Patients
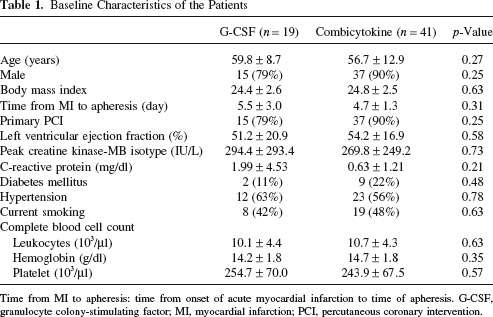
Time from MI to apheresis: time from onset of acute myocardial infarction to time of apheresis. G-CSF, granulocyte colony-stimulating factor; MI, myocardial infarction; PCI, percutaneous coronary intervention.
Mobilization by Cytokines and Collection by Apheresis
Increment of leukocyte counts from baseline after completion of cytokine administration was similar between the G-CSF and the combicytokine group. Hemoglobin concentration, mononuclear cell proportion, and platelet counts were also not different between two groups. Administration of G-CSF with or without darbepoetin bolus infusion was safe. There were mild adverse reactions in patients from the G-CSF group and the combination group, such as myalgia or bone pain in three and six patients, headache in one and two, fever in one and two, and dizziness in one and two, respectively. Most adverse reactions improved and disappeared within several days after completion of cytokine administration. There were mild but significant elevations of c-reactive protein (CRP) level after cytokine treatments both in G-CSF and combicytokine group. And the degrees of elevation were similar between the two groups (G-CSF: 2.29 ± 1.97 vs. combicytokine: 2.49 ± 1.77 mg/dl, p = 0.73). And PBSC collection with apheresis was completed without any major cardiac complication in all patients. During apheresis, transient hypotension occurred in one patient from the G-CSF group and one patient from the combicytokine group, and transient paresthesia in two patients from the G-CSF group. Adverse events during cytokine treatment and apheresis were similar between groups. The time spent on the apheresis machine and blood volume for processing was similar between the two groups. During a 1-month clinical follow-up after cytokine treatment, there were no additional major adverse cardiac events in these patients.
Apheresis Product Profiles
Hemogram of peripheral blood after cytokine administration was similar between the two groups. However, the composition of apheresis products was significantly different between the two groups. Apheresis products obtained from the combicytokine group contained fewer leukocytes with higher mononuclear cell proportion than those from the G-CSF group. Although leukocyte concentration in apheresis products was higher in the G-CSF group, the CD34+ cell concentration of apheresis products was not different between the two groups (Table 2). These meant that apheresis products obtained from the combicytokine group had higher CD34+ cell purity and fewer unwanted inflammatory cells than those from the G-CSF group. We could obtain sufficient numbers of CD34+ cells for intracoronary infusion in all patients from both groups. Additionally, apheresis products from the combicytokine group showed slightly higher hemoglobin concentration and lower platelet counts than those of the G-CSF group.
Apheresis Process and Apheresis Product Yield

CBC, complete blood cell count; FACS, florescence-activated cell sorting; KDR, kinase insert domain-containing receptor; VE-cadherin, vascular endothelial cadherin.
Apheresis products of the combicytokine groups had higher portions of proangiogenic cell populations, such as CD34+/KDR+ endothelial progenitor cells (EPCs), CD3+/CD31+ proangiogenic T cells (12), and Tie2+ and/ or CXCR4+ cells, than those of the G-CSF group. In MAGIC Cell-5-combicytokine study, the infusion cell dose was 2 × 109 mononuclear cells per patient. As a results, patients in the combicytokine groups received higher numbers of CD34+/KDR+ cells, CD3+/CD31+ cells, and Tie2+/CXCR4+ cells than the patients from the G-CSF group. However, proportions of CD73+/CD105+ mesenchymal stem cell population were not different between the two groups. The combicytokine group had higher circulating CD3+/CD31+ proangiogenic T cells and CXCR4+ cells than the G-CSF group did in both apheresis product and peripheral blood.
Proportion of CD34+/KDR+ EPCs was independently determined by age and use of darbepoetin in multivariate analysis (Table 3). In this study, the size of infarction measured by cardiac enzyme elevation and timing of apheresis did not influence yields of stem/ progenitor cells.
Multiple Linear Regression Analysis for Determinants of CD34 /KDR Cell Proportion in Apheresis Products
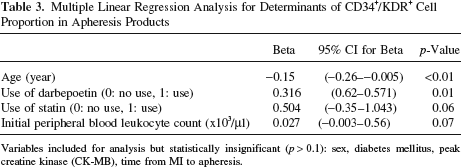
Variables included for analysis but statistically insignificant (p > 0.1): sex, diabetes mellitus, peak creatine kinase (CK-MB), time from MI to apheresis.
Discussion
Mobilization of PBSCs as an Essential Component for Cytokine-Based Cell Therapy
G-CSF is effective and safe as a stem cell mobilizer in patients with ischemic heart disease. The strategy to mobilize the stem cells with G-CSF and to collect them with apheresis is a well-established standard approach. But it still has limitations. Mobilization efficiency and cellular composition of mobilized PBSCs is variable and purity of stem/progenitor cell is quite low even with apheresis. Based on our previous experience, G-CSF mobilized and recruited higher numbers of CD34+/KDR+ cells in patients with AMI than patients with an old myocardial infarction (OMI) (4). In our MAGIC-Cell-3-DES trial, G-CSF-based cell therapy improved left ventricular ejection fraction only in patients with AMI but not with OMI (2). Thus, we thought that the mobilization efficiency would be one of several factors involved in therapeutic efficacy. Although we are not able to identify the ideal amount and types of stem/progenitor cells for cell therapy (1), it is suggested that the higher numbers of transplanted stem/progenitor cells would provide the bigger effects (12). Most researchers arbitrarily decided target cell numbers for cell therapy considering tolerability and efficacy. And maximum cell number feasible for transplantation is usually limited. Thus, increasing purity of stem/progenitor cells in infusates is a reasonable approach to improve the therapeutic efficacy. For this purpose, we can consider mechanical selection by surface markers and expansion by cultivation of specific cell population. But such approach has practical limitations due to cost, feasibility, and risk of contamination. In terms of feasibility, adjunctive darbepoetin to G-CSF can be a promising method. In our study, we observed that combicytokine treatment is more effective in mobilization and recruitment of proangiogenic stem/progenitor cell population in higher purity than G-CSF alone.
Superiority of Combicytokine to G-CSF Alone in Mobilization of PBSCs
Combicytokine treatment recruited higher numbers of proangiogenic populations including CD34+/KDR+ EPCs, CD3+/CD31+ proangiogenic T cells, and Tie2+ and/or CXCR4+ cells. Firstly, erythropoietin was a potent stimulus for the mobilization of EPCs into peripheral blood by previous studies (5,13,23) and our study also confirmed EPC mobilization efficacy of darbepoetin. Secondly, erythropoietin mobilized CD3+/CD31+ proangiogenic T cells, which is a novel cell population reported by our group (7). Angiogenic T cells initiate new vessel formation after transendothelial migration toward ischemic foci through CXCR4/stromal cell-derived factor (SDF-1) axis and secrete angiogenic cytokines such as vascular endothelial growth factor and interleukin-8, leading to activation of endothelial cells and EPCs. Thirdly, erythropoietin mobilized CXCR4+ or Tie2+ cells, which supports the recent studies that it stimulated CXCR4/SDF-1 and Tie-2/angiopoetin-1 axis in ischemic tissues (2,8,11,14,22,24). In myocardial infarction model, erythropoietin increased SDF-1 level in the infarct or circulating blood, and induced mobilization and homing of CXCR4+ bone marrow stem cell into ischemic myocardium (2). In rat cerebral ischemia or mouse hindlimb ischemia model, combination of erythropoietin with G-CSF synergistically increased expression of SDF-1, homing of bone marrow stem cell into ischemic lesion, and in vivo angiogenesis compared to G-CSF and erythropoietin alone (14). Erythropoietin and G-CSF can increase SDF-1 level by activating hypoxia-inducible factor-1 (HIF-1) and promoting its release from platelets (8,24). And as expected, darbepoetin did not mobilize CD73+/CD105+ mesenchymal stem cells.
In this study, darbepoetin showed potential to improve angiogenic efficacy of PBSC therapy in AMI patients by mobilization of proangiogenic stem/progenitor cells and potentiation of angiogenic stimuli. These effects of darbepoetin can overcome the hypothetical limitation of G-CSF that it may disturb the normal expression of CXCR4 on the surface of the mobilized cells resulting in the decreased homing of PBSCs into ischemic myocardium (5). At the 6-month follow-up, there were trends that patients in the combicytokine group showed similar recovery of coronary flow reserve and better protection against left ventricular remodeling in echocardiographic analysis compared with the patients with G-CSF group.
Potential Benefits of Adjunctive Darbepoetin to G-CSF
We expected several additional potential advantages by adjunctive use of darbepoetin to PBSC therapy in AMI patients. Erythropoietin has direct cytoprotective effects in ischemic conditions independent of PBSC mobilizing effects (14,16,19,21). When combined, erythropoietin and G-CSF exerted cytoprotective effects via their own receptor and interacted synergistically (14). Additionally, combination of erythropoietin with bone marrow stem cell therapy also showed additive benefits for improving cardiac function in AMI (25).
Safety
Safety of short-term use of high-dose erythropoietin was reported by previous studies (13,17,18). Use of erythropoietin did not further activate platelets in AMI patients (18). In our study, adjunctive use of high-dose single bolus darbepoetin infusion just after revascularization was well tolerated and did not increase risk of adverse events.
Conclusions
Combination treatment with darbepoetin and G-CSF is safe and more effective in mobilizing proangiogenic cell population compared to G-CSF alone in patients with AMI. These results suggest that combination cytokine treatment may have superior angiogenic therapeutic potential to PBSC therapy using G-CSF alone.
Footnotes
Acknowledgments
This study was supported by a grant from Stem Cell Research Center (SC4210), and the Korea Healthcare technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea (A062260). Dr. Hyo-Soo Kim is also a professor of Molecular Medicine & Biopharmaceutical Sciences, WCU program, Seoul National University, Seoul, Korea. The authors declare no conflicts of interest.
