Abstract
Cell therapy is a field of growing interest in the prevention of post acute myocardial infarction (AMI) heart failure. Stem cell retention upon local delivery to the heart, however, is still unsatisfactory. CellBeads were recently developed as a potential solution to this problem. CellBeads are 170-μm alginate microspheres that contain mesenchymal stem cells (MSCs) genetically modified to express glucagon-like peptide-1 (GLP-1) supplementary to inherent paracrine factors. GLP-1 is an incretin hormone that has both antiapoptotic and cardioprotective effects. Transplanting CellBeads in the post-AMI heart might induce cardiomyocyte salvage and ultimately abrogate adverse cardiac remodeling. We aimed to investigate the feasibility of intracoronary infusion of CellBeads in a large animal model of AMI. Four pigs were used in a pilot study to assess the maximal safe dose of CellBeads. In the remaining 21 animals, an AMI was induced by balloon occlusion of the left circumflex coronary artery for 90 min. During reperfusion, 60,000 CellBeads (n = 11), control beads (n = 4), or lactated Ringers' (n = 6) were infused. Animals were sacrificed after 2 or 7 days, and the hearts were excised for histological analyses. Intracoronary infusion did not permanently affect coronary flow in any of the groups. Histological analysis revealed CellBeads containing viable MSCs up to 7 days. Viability and activity of the MSCs was confirmed by qPCR analysis that showed expression of recombinant GLP-1 and human genes after 2 and 7 days. CellBeads reduced inflammatory infiltration by 29% (p = 0.001). In addition, they decreased the extent of apoptosis by 25% (p = 0.001) after 2 days. We show that intracoronary infusion of 5 million encapsulated MSCs is safe and feasible. Also, several parameters indicate that the cells have paracrine effects, suggesting a potential therapeutic benefit of this new approach.
Keywords
Introduction
Despite advances in pharmacotherapy and interventional cardiology, heart failure constitutes a growing patient population in the Western world (12). Acute myocardial infarction (AMI) is the major underlying etiology of congestive heart failure (14,28). Because an AMI leads to irreversible loss of cardiomyocytes and cardiomyocytes have limited regenerative or compensatory capacity, loss of cardiomyocytes irrevocably leads to fibrosis and scar tissue formation (5). Subsequent adverse left ventricular remodeling can cause the heart to fail and lead to the clinical symptoms of heart failure with poor prognosis (6,7).
The cornerstone of the treatment of AMI patients, which is reperfusion in combination with medical treatment, only partly prevents scar tissue formation and remodeling of the damaged heart (8). The prevention of cardiomyocyte loss and remodeling has therefore been the topic of extensive research. The field of regenerative cellular therapy is an area of growing interest, in particular in AMI patients. In fact, small-scale clinical cell therapy trials show promising results, using bone marrow-derived mononuclear cells (1,15).
The predominant working mechanism of post-AMI cell therapy is believed to be through the paracrine action of the grafted cells, resulting in myocardial salvage and neoangiogenesis (13,20,25). The mesenchymal stem cell (MSC) is currently hypothesized to be the most potent, nonembryonic cell with respect to secretion of relevant paracrine growth factors, antiapoptotic and prosurvival factors, as well as immunomodulatory cytokines and have been shown to exert cardioprotective effects in vivo (3,16,19,30).
Also, a number of recent studies has shown that glucagon-like peptide-1 (GLP-1), one of the most potent incretin hormones, has potential beneficial action on the ischemic and failing heart (2,17,22,24). GLP-1 is a naturally occurring incretin with both insulinotropic and insulinomimetic properties and has been shown to exert antiapoptic actions. Interestingly, when added to standard therapy, GLP-1 infusion improved regional and global left ventricular function in a clinical study with AMI patients with severe systolic dysfunction after successful primary coronary intervention (PCI) (18). However, due to its very short half-life, a prolonged infusion of 72 h with substantial side effects for the patients was needed to achieve this beneficial effect.
One of the biggest challenges in the cell therapy field today is the poor retention of therapeutic cells upon local delivery in the heart, with retention rates as low as 1% after intracoronary delivery (10,11,27). Even though permanent engraftment of stem cells is not required to elicit the cardioprotective effect, it seems logical that the greater the number of cells that are retained in the injured myocardium and the longer they reside there, the more pronounced the potential beneficial effect may be. Despite wide-ranging efforts to increase cell retention using various delivery techniques, results are still unsatisfactory. A new concept of stem cell delivery has recently become available owing to advances in the field of biotechnology, as it is currently possible to encapsulate MSCs in a biocompatible alginate shell (26,29). Alginate encapsulation of varying numbers of MSCs results in so-called CellBeads™, available in discrete sizes between 170 and 600 μm.
CellBeads are made from a highly purified alginate material, which is used to encapsulate clusters of adult human MSCs. These MSCs have been genetically modified to secrete a proprietary recombinant GLP-1 fusion protein, which consists of two GLP-1 molecules bound by an intervening peptide. This form of GLP-1 is more stable than endogenous GLP-1, rendering a longer half-life and thus prolonged therapeutic potential. The alginate coating of the CellBeads is permeable to the GLP-1 fusion protein, allowing for continuous delivery, while protecting the MSCs from the patient's immune system. Also, oxygen and nutrients can freely pass through the alginate shell, which renders the MSCs viable for a long period of time. Thus, CellBeads are potentially a unique, biological, long-term, local drug delivery platform that is capable of delivering GLP-1 or other therapeutic proteins, in addition to MSC-derived factors [vascular endothelial growth factor (VEGF), monocyte chemotactic protein-1 (MCP-1), interleukin (IL)-6, IL-8, glial-derived neurotrophic factor (GDNF), and neurotrophin-3 (NT-3)] to any target tissue.
We hypothesize that, when transplanted in the post-AMI heart, the synergystic effect of paracrine MSC-derived factors together with the cardioprotective GLP-1 peptide, might evoke myocardial salvage, reduce apoptosis, and influence the inflammatory response in the acute phase of the AMI. In the long term, CellBead therapy may induce angiogenesis, and decrease post-AMI adverse cardiac remodeling. The aim of the current study was to evaluate the feasibility and safety of intracoronary delivery of 170 μm CellBeads in the acute phase of an AMI in a relevant large animal model.
Materials and Methods
Experimental Animals
All procedures were approved by the local animal welfare committee. A total of 25 female landrace pigs (Van Beek SPF pigs, Putten, The Netherlands, 69.2 ± 1.0 kg) were used in this study.
Experimental Design
This study was subdivided in two phases. In the first phase (phase 1), the feasibility of intracoronary injection of CellBeads was assessed in naive, noninfarcted myocardium in four pigs. The goal of this phase was to determine the maximum dose of CellBeads to be delivered safely by continuously assessing coronary flow or the occurrence of fatal arrhythmias upon CellBead infusion.
In phase 2, 21 pigs underwent an AMI by balloon occlusion, followed by intracoronary infusion of CellBeads to assess the feasibility and safety of CellBead infusion in AMI. Animals received intracoronary infusion of lactated Ringers' (LR) solution (n = 6), alginate-only control beads (n = 4), or GLP-1-expressing CellBeads (n = 11). Coronary flow was assessed prior to, during, and after CellBead infusion using coronary angiography, Doppler-aided coronary flow (phase 1), and coronary flow reserve (CFR) (phase 2) measurements. Animals were sacrificed after 2 or 7 days, and the heart was excised for histological analysis (see Fig. 1 for study flow chart). In addition, the pigs received an event recorder to monitor for arrhythmic events.

Study flow chart.
Medication and Anesthesia
The animals were premedicated for 10 days with dual antiplatelet therapy (acetylsalicylic acid 80 mg qd and clopidogrel 75 mg qd) and antiarrhythmic therapy (only in phase 2; amiodarone 400 mg qd). General anesthesia was induced and maintained with intravenous infusion of midazolam, sufentanil, and pancuronium. Upon infarct induction and CellBead infusion, all animals were therapeutically heparinized and received intravenous infusion of eptifibatide (bolus of 180 μg/kg and 2 μg/kg/min). A fentanyl plaster was applied after the procedure for analgesia. Dual antiplatelet and antiarrhythmic therapy was continued until the sacrifice procedure.
Induction of the Acute Myocardial Infarction
A myocardial infarction was only induced in phase 2 of the current study. Although in phase 1, the left anterior descending coronary artery (LAD) was used to infuse CellBeads, we chose to switch to occlusion of the proximal left circumflex artery (LCX) for induction of the AMI due to an expected higher survival rate. Catheterization of the left coronary system was performed via the right carotid artery. A left coronary angiogram was made to determine the optimal position for balloon occlusion, followed by inflation of an angioplasty balloon (Voyager Rx 3.5–4.0 × 12 mm, Abbott, IL, USA) for 90 min in the proximal LCX to induce an acute posterolateral myocardial infarction.
CellBead Infusion and Coronary Flow Assessment
CellBeads were thawed rapidly and diluted in a large volume of LR (150,000 CellBeads in 250 ml of LR, rendering a concentration of 600 CellBeads/ml).
In naive animals (phase 1), a microcatheter (TwinPass, Vascular Solutions, IL, USA) was placed in the LAD coronary artery, distal to the first diagonal branch. The infusion rate in the first two animals was set at 4 ml/min or 2,400 CellBeads/min. In animals 3 and 4, the infusion rate was reduced to 2 ml/min or 1,200 CellBeads/min. During CellBead infusion, coronary flow was assessed regularly by coronary angiography and depicted using the conventional thrombolysis in myocardial infarction (TIMI) flow grade nomenclature (23). Also coronary flow was continuously measured during infusion using a Doppler flow wire (Combowire, Volcano, San Diego, CA, USA) and expressed as average peak velocity (APV).
In phase 1, the maximum amount of CellBeads to be safely delivered without compromising coronary flow was found to be 60,000. Thus, in phase 2 (AMI animals), either 60,000 CellBeads (n = 11), 60,000 control alginate beads (n = 4), or LR alone (n = 6) was infused intracoronarily. All animals received two 50-ml syringes, containing 30,000 beads or LR only each. These syringes were infused using a syringe pump (IVAC, Humberside, UK) at a constant flow rate of 2 ml/min (1,200 CellBeads/min). During infusion, settling of the beads was prevented by regular rocking of the syringe pump.
Beads were delivered after approximately 15 min of reperfusion, a TwinPass infusion catheter was placed in the proximal LCX. Coronary angiography was performed before infusion, at 25%, 50%, and 75% of infusion, and 10 min after infusion of the beads, to determine TIMI flow grade. Also, coronary flow reserve (CFR) was measured before and after bead or LR infusion. CFR was determined by dividing the APV at maximal vasodilatation during adenosine infusion by the baseline APV. Six animals were sacrificed two days after the AMI and bead infusion (four animals that received CellBeads, two control beads), and 15 animals were sacrificed at day seven (seven CellBeads, two control beads, six LR control) (see Fig. 1). At sacrifice, TIMI flow grade was measured, after which the animals were terminated and the hearts excised for histological analysis. TIMI flow grade was determined by an independent cathlab technician, who was blinded to the treatment of the individual animals.
Event Recorder
After the infusion procedure, all pigs were fitted with a REVEAL DX event recorder (Medtronic, Minneapolis, MN, USA). The recorders were placed subcutaneously in the left thorax. The occurrence of arrhythmias was assessed prior to termination using a CareLink™ programmer (Medtronic).
Macroscopic and Microscopic Analysis
In phase 2 of this study, animals were euthanized at day 2 or day 7 and the hearts were excised and sectioned in five to six bread-loafed slices from the apex to the base. The slices were stained in 2,3,5-triphenyltetrazolium chloride (TTC) to delineate the infarct area from noninfarcted myocardium. The noninfarcted myocardium was thoroughly screened for the presence of microinfarctions. The entire infarct area and biopsies of remote myocardial segments (anterior and anteroseptal wall) were taken and fixed in formalin, embedded in paraffin, and sliced into 5-μm sections. All sections were stained by hematoxylin and eosin staining (H&E staining) using a standard protocol. Sections were examined by a pathologist specialized in cardiac histopathology.
H&E-stained sections of remote myocardial segments were investigated for signs of microinfarctions or the presence of shedded CellBeads. Sections of infarct tissue of each study animal were investigated for the presence of CellBeads or control beads, inflammatory reactions, and/or foreign body response. More specifically, the composition and surface area of the inflammatory infiltrate surrounding each CellBead or control bead was assessed and measured using an Olympus BX45 microscope. We only investigated totally transverse cut beads and measured the distance from the bead to the periphery of the surrounding infiltrate.
Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling (TUNEL) Staining for Apoptosis
Apoptosis was assessed using an “In situ cell death detection kit” (Roche, Mannheim, Germany). Samples were counterstained with hematoxylin. At least 30 random pictures were taken from slides of animals that received CellBeads or control beads and were sacrificed at 2 or 7 days. TUNEL and hematoxylin double-positive nuclei were counted and expressed as positive cells/100 μm2.
Quantitative PCR Analysis of Infarct Specimens
For determination of recombinant GLP-1 expression in the target area, small samples of the infarct were taken of animals that survived 2 or 7 days. RNA was extracted using RNA-Bee (Tel-test, Inc., Friendswood, TX, USA) according to the manufacturer's protocol. Quality and quantity of the RNA was verified on an Agilent 2100 Bioanalyzer (Agilent Technologies, UK) and reverse transcribed. Quantitative PCR (qPCR) analysis was performed using an iCycler iQ Detection System (Bio-Rad, the Netherlands). Primers were designed to specifically amplify recombinant GLP-1 dimer (and not endogenous GLP-1), a porcine household gene (hypoxanthine-guanine phosphoribosyl transferase 1; HPRT), and a human household gene (HPRT). mRNA levels detected by qPCR were expressed relative to the porcine household genes. The primer sequences are provided in Table 1.
Primer Sequences Used for qPCR

qPCR, quantitative polymerase chain reaction; GLP-1, glucagon-like peptide-1; HPRT, hypoxanthine-guanine phosphoribosyl transferase 1.
Statistical Analysis
The current study was designed as a pilot feasibility study, so no formal power calculations were performed. All statistical analyses were performed post hoc using SPSS 16 statistical software (IBM, Chicago, IL, USA). An analysis of variance (ANOVA) was performed and a Bonferroni correction was used for pair-wise comparison between means. Data are presented as mean ± SEM. Values of p ≤ 0.05 were considered significant.
Results
Results of Phase 1 (Naive Animals)
All four experiments were a procedural success, and it proved to be possible to infuse CellBeads selectively via intracoronary infusion. In the first pig, 75,000 CellBeads were administered without any sign of ischemia or reduction of flow as depicted by APV (Fig. 2) (delta-APV 5/23) However, after infusion of approximately 80,000 CellBeads several premature ventricular complexes occurred that ultimately progressed into ventricular fibrillation and death of the animal.

Change of coronary flow upon CellBead infusion in naive, nonischemic myocardium. The individual points represent the APV that was measured every 15,000 CellBeads, whereas the corresponding TIMI flow is depicted on the right side of the graph. Intracoronary infusion of CellBeads reduces APV in a dose-dependent manner. An infusion rate of 2,400 CellBeads/min caused fatal arrhythmia in one animal and a steep decline of coronary flow (both APV and TIMI flow) briefly after the infusion of 60,000 CellBeads (arrow). Reducing the infusion rate to 1,200 CellBeads/min increased the amount of CellBeads that could be delivered before the APV dropped >50% from baseline. APV, average peak velocity; TIMI, thrombolysis in myocardial infarction.
In pig 2, APV gradually decreased until the infusion of 60,000 CellBeads, after which a steep decline in APV occurred accompanied by a decrease in TIMI flow from TIMI grade III to I. Hence, we decided to decrease infusion rate in the remainder of the animals from 2,400 to 1,200 CellBeads/min. As a result, we found that, in the remaining two animals, the amount of CellBeads that could be delivered before the decline in APV (defined as >50% of baseline value) and TIMI flow was significantly higher (90,000 CellBeads). Blood pressure remained stable in all four animals, showing no significant acute effect of CellBead infusion on cardiac performance. After reviewing the results of phase 1, we concluded that the maximal safe dose that can be infused in naive/non-AMI pigs is 60,000 CellBeads. Therefore, in phase 2, the feasibility of intracoronary delivery of 60,000 CellBeads was evaluated in a pig LCX AMI model.
Results in Porcine AMI Model (Phase 2)
Animal Experiments
A total of 21 pigs were included in this phase of the study. Three animals experienced ventricular fibrillation during infarct induction, but all animals were successfully resuscitated. Animals received 60,000 CellBeads (n = 11), 60,000 control beads (n = 4), or LR (n = 6). No lethal ventricular arrhythmias occurred during CellBead or control bead infusion, and all animals survived the dedicated follow-up time.
Coronary Flow Reserve
In control animals, average CFR before LR infusion was 1.67 ± 0.09 and 1.73 ± 0.12 after LR infusion (Fig. 3A). In the pigs that received intracoronary infusion of CellBeads or control beads, CFR remained unchanged with an average CFR of 1.63 ± 0.04 before infusion and 1.70 ± 0.09 afterwards (p = ns) (Fig. 3B).
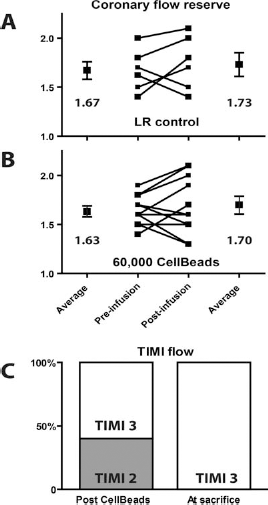
Coronary flow infusion in AMI pigs. (A) CFR remained unchanged after infusion of LR. (B) Intracoronary infusion of CellBeads did not alter CFR. (C) TIMI flow was slightly reduced in 40% of the animals that received intracoronary infusion of 170 μm beads but had recovered completely at sacrifice. AMI, acute myocardial infarction; CFR, coronary flow reverse; LR, lactated Ringer's solution.
TIMI Flow
All pigs had normal coronary flow after the induction of the AMI, defined as TIMI grade 3 flow. Infusion of 100 ml LR in control animals did not impede TIMI flow. Also, no effect on antegrade coronary flow was found in 60% (9/15) of the animals that received CellBeads or control beads. However, 40% (6/15) of the animals that received CellBeads or control beads experienced a slight reduction of coronary flow, resulting in sluggish flow (TIMI 2) in the culprit artery after the infusion of 60,000 beads (Fig. 3C). The occurrence of sluggish flow was predominant during the infusion of the final 15,000 beads, as coronary flow remained unchanged until 45,000 of all 60,000 beads were infused.
Coronary flow returned to normal TIMI 3 flow at the day of sacrifice (day 2 or day 7). There was no difference in coronary flow between animals that survived 2 or 7 days, or between animals that received CellBeads or control beads.
Analysis of Arrhythmias
None of the animals, either control or CellBead-treated, experienced ventricular arrhythmias during the 2- or 7-day follow-up period, as detected by the REVEAL arrhythmic detection device.
Histology
H&E Staining
H&E-stained sections of all myocardial segments were examined from all animals included in phase 2. No beads or microinfarctions could be detected in remote myocardial segments. On the contrary, in infarcted tissue specimens, multiple beads were detected per section, as the majority of 100–200 μm arterioles contained one or more beads. As far as we could determine with conventional histological analysis of sections of the whole infarct area, the CellBeads were distributed equally throughout the infarct. CellBeads in pigs that survived 2 days as well as in pigs that survived 7 days all contained viable MSCs, as was determined by the presence of basophilic nuclei (Fig. 4B, C).

Photomicrograph of micro CellBeads. (A) Light photomicrograph of a CellBead prior to infusion. The alginate shell (∗) and MSC-containing core (†) can be appreciated. (B) H&E-stained section of a coronary arteriole completely comprised of one CellBead, 2 days after infusion. Only a thin rim of fibrin and granulocytes surrounds the CellBead (arrows). (C) H&E-stained section of a coronary arteriole containing one CellBead, 7 days after infusion. Only a thin rim of fibrin and granulocytes surrounds the CellBead (arrows). ∗Intact alginate; †mesenchymal stem cells (MSCs).
Two days after infarction, the cardiac tissue showed infiltration of predominantly neutrofilic granulocytes and loss of cardiomyocyte viability. After 7 days, a clear fibrotic response was present with fibroblasts infiltrating the infarcted area. Also, there was a marked increase of the inflammatory infiltrate as compared to the 2-day animals, which is normal 7 days post-AMI.
Inflammatory Infiltration Surrounding CellBeads
All the beads showed a thin rim of fibrin with neutrophilic granulocytes (Fig. 5A). In addition, the beads were variably surrounded by a rim of lymphocytes and plasma cells. We noted that the control alginate beads showed significantly more inflammatory infiltrate directly surrounding the bead when compared to CellBeads (0.038 ± 0.004 mm2 vs. 0.027 ± 0.004 mm2, p = 0.003) (Fig. 5B, C) 2 days following the AMI.

Peri-bead inflammatory infiltration after 2 days. (A) A thin rim of neutrophilic granulocytes is evident surrounding a single CellBead (arrow). (B) A more pronounced inflammatory reaction around a control alginate bead (arrow). (C) Comparison of inflammatory reaction by measuring the surface area of the inflammatory infiltrate reveals a significant decrease in inflammatory response to the CellBeads as compared to the control alginate beads (∗∗p = 0.003).
At the 7-day time point, the natural healing process of the AMI caused such a background inflammatory infiltrate that measurements of the infiltrate surrounding the beads were not reliable. We therefore performed the analysis at the 2-day follow-up time point after AMI only. Since this inflammatory infiltrate after 7 days was equally intense in the LR control animals, we considered this as a normal phase in the healing process.
Apoptosis
Apoptosis was significantly reduced in infarct areas of animals that received CellBeads as opposed to animals that received control beads. More specifically, in infarct tissue specimens of animals that received control beads, on average 7.5 ± 0.44 cells were apoptotic per 100 μm2 after 2 days. This number was significantly reduced in animals that received CellBeads to 5.6 ± 0.36 per 100 μm2 (p = 0.003) (Fig. 6). After 7 days, the number of apoptotic cells had decreased to 3.3 ± 0.41 per 100 μm2 in animals that received control beads as opposed to 2.3 ± 0.29 per 100 μm2 in animals that received CellBeads (p = 0.34).

TUNEL staining for apoptosis. (Top) Histological section of infarct tissue 2 days after the AMI, demonstrating apoptotic cardiomyocytes (∗) throughout the tissue. (Bottom) Quantification of TUNEL-positive cells revealed a significant difference in the amount of apoptosis in CellBead-treated tissue compared to controls after 2 days (∗∗p = 0.003), but not after 7 days (p = 0.34). TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling.
Quantitative PCR Analysis of Infarct Specimens
No expression of recombinant GLP-1 or human genes was found in the hearts of the control pigs. However, expression of recombinant GLP-1, together with the human household gene HPRT was found in infarct segments of animals that received GLP-1 expressing CellBeads, confirming the presence of CellBeads in the infarcted heart that contain viable and actively transcribing human MSCs. Expression levels of recombinant human GLP-1 was comparable between specimens from animals that survived 2 and 7 days (Fig. 7).
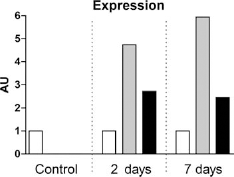
Quantitative PCR analysis of infarct samples. qPCR revealed expression of both the human household gene (grey bar) and recombinant GLP-1 (black bar) as compared to porcine household gene (white bar) in animals that received CellBeads after 2 and 7 days compared to no expression in control animals. The values of the porcine household gene were arbitrarily set to one. AU, arbitrary units; GLP-1, glucagon-like peptide-1.
Discussion
The current study demonstrates for the first time that it is feasible to intracoronarily infuse alginate, MSC-containing microspheres in infarcted myocardium. Infusion did not permanently affect coronary flow, resulted in precise targeting of the infarct area, and MSCs remained viable for at least 7 days. Interestingly, the inflammatory response to CellBeads was negligible in this xenogeneic transplantation model.
We found that intracoronary infusion of significant numbers of 170-μm particles did not severely hamper coronary flow. CFR remained unchanged in all animals that received 60,000 CellBeads or control beads, suggesting little or no microvascular obstruction. Although coronary flow as assessed by angiography was slightly decreased to sluggish flow in 40% of the animals, it returned to normal flow within 2 or 7 days follow-up. This suggests that, even though the coronary vasculature might have been partly obstructed by the beads, this obstruction was temporary and that alternative collateral routes were recruited to perfuse the inflicted myocardium. Also, we hypothesize that infusion of limited amounts of obstructive beads into already infarcted myocardium will not increase ischemic damage. Importantly, we did not see any proarrhythmic effects of the infused beads. Although longer-term follow-up is warranted to draw firm conclusions, these data add to the safety profile.
The current approach is the first that guarantees high stem cell retention. In fact, as 170-μm CellBeads are trapped in the arteriolar tree of the myocardium and cannot pass the capillary bed, retention will approximate 100%. Because CellBeads contain ~80 MSCs per bead, infusion of 60,000 beads translates to almost five million delivered MSCs that remain in the targeted area for a substantial period of time. For reference, in cell therapy studies performed thus far, maximally 200 million bone marrow-derived mononuclear cells were transplanted (1,21). Of these cells, 0.001–0.01% comprises MSCs (4), rendering only 20,000 MSCs infused at the best. Retention rates of 1–10% results in negligible cell numbers as opposed to the approach investigated in the current study.
In addition, we show that a substantial amount of the MSCs in the CellBeads remain viable for at least 7 days.
In this period of time, the MSCs secrete MSC-derived soluble factors that have been shown to exert cardioprotective and proangiogenic effects (3,13,16,20,25). Also, our cells produce therapeutic amounts of GLP-1 that has beneficial, antiapoptotic effects on the post-AMI heart (18,24). CellBeads are thus small delivery devices of GLP-1 and MSC-derived factors that might have synergistic, favorable effects on cardiac scar tissue formation and adverse remodeling.
Indeed, in our experiments, infusion of CellBeads significantly reduced apoptosis in the infarct area after 2 and 7 days when compared to control alginate beads. This is in line with one of the presumed working mechanisms of GLP-1 and suggests that GLP-1 is factually secreted into the area at risk of cardiomyocyte death following ischemia/ reperfusion. The additive effect of GLP-1 remains hypothetical though, as in this pilot study the appropriate control group (MSCs that do not express GLP-1) is lacking. However, qPCR analysis confirmed expression of recombinant GLP-1 and human genes in porcine tissue even after 7 days.
The fact that there is a significant difference in inflammatory infiltration between CellBeads and control alginate beads is another direct validation of our paracrine hypothesis. Immunomodulatory cytokines, like IL-10 and TGF-β, are secreted by the MSC and prevent the local acute immune reaction and rejection against these human-derived cells. In fact, we observed significantly less immune reaction against the CellBeads than against the biocompatible alginate control beads (9) that evoked some foreign body response in the highly inflammatory post-AMI environment.
There are two major limitations to this study. First, the short follow-up time of 7 days does not rule out long-term safety issues. Second, the current study did not thoroughly investigate the biodistribution of the CellBeads. Although we hypothesize that retention of the CellBeads in the target area will approximate 100%, additional studies in the near future using nuclear imaging techniques will render more definite answers to this question. Nonetheless, the results of this pilot study are promising, and future studies will focus on long-term safety effects and signs of efficacy. We are currently enrolling over 70 pigs in a large dose-finding and efficacy trial, in which we investigate three different doses of CellBeads versus three control groups. This study will answer questions concerning long-term safety, but also concerning the effect of CellBeads containing MSCs that do not express GLP-1.
Conclusion
We show that intracoronary infusion of CellBeads is feasible and appears to be safe in a large animal model of AMI. CellBeads were successfully targeted to the infarct area, and MSCs remained viable and active for at least 7 days. Also, several parameters indicate that the cells sort a paracrine effect, highlighting the potential for cardiovascular repair of this new therapy.
Footnotes
Acknowledgments
This study was funded by an unrestricted educational grant from Biocompatibles UK Ltd. Andrew Lewis and Peter Stratford are employees of Biocompatibles UK Ltd., and Christine Wallrapp is an employee of CellMed AG, owners of the intellectual property pertaining to the technology described in this article. The authors declare no other conflict of interest.
