Abstract
We describe a 40-year-old South Asian female with relapsing polychondritis-associated myocardial infarction with non-obstructive coronary arteries during an acute flare for which she was successfully managed with guideline-directed medical therapy. The clinician should be cognizant of this rare association, especially in patients who present with acute coronary syndromes and chronic, pre-existing autoimmune syndromes.
Keywords
Introduction
Myocardial infarction with non-obstructive coronary arteries (MINOCA) is a clinical syndrome characterized by the presence of acute myocardial infarction (AMI) criteria, the absence of obstructive coronary artery disease (CAD; ≥50% stenosis) on angiography, and the exclusion of an identifiable non-cardiac cause for the acute presentation.1-3 MINOCA typically comprises circa 15% of all acute coronary syndromes (ACS) and disproportionately affects the female gender.4,5 Sex-based disparities in coronary physiology include increased myocardial perfusion in women, which may exacerbate endothelial shear stress with resultant CAD progression. Additionally, hormonal factors may contribute to impaired vascular stiffness and accentuate cardiovascular risk. 6
Women have a predisposition to autoimmune disorders and face a marked risk of major adverse cardiovascular and cerebrovascular events compared to men. 7 Relapsing polychondritis (RP) is a rare autoimmune disorder characterized by episodes of inflammation in cartilaginous structures, primarily affecting the ears, nose, and respiratory tract. The condition presents with a heterogeneous clinical spectrum, often involving systemic manifestations. 8 Inflammation and autoantibodies may represent a common, central pathway linking autoimmune disease and cardiovascular disease (CVD). 9
We present a case of a 40-year-old South Asian female who developed MINOCA secondary to RP during an acute inflammatory episode. The patient was successfully managed with guideline-directed medical therapy (GDMT). This case highlights the importance of recognizing RP as a rare but potential etiology of MINOCA, particularly in patients who present with atypical ACS. Clinicians should maintain a high index of suspicion for vasculitic or inflammatory mechanisms in such scenarios to ensure timely diagnosis and appropriate management for optimal patient outcomes.
Case Presentation
A 40-year-old South Asian female with a documented history of RP for the prior 6 years, managed with oral low-dose methotrexate, presented to the emergency department with a 1-day episode of unstable angina (USA Canadian Cardiovascular Society (CCS) Class III-IV) and progressive, exertional dyspnea. Of note, her past medical history was only positive for her PM diagnosis, for which a rheumatologist was managing her. She denied illicit drug use, tobacco use, or significant alcohol consumption. Her family history was remarkable for a maternal aunt with rheumatoid arthritis. She did not report any antecedent infection or ill contacts. She denied the use of any complementary and alternative medications or pertinent travel and pet history. She maintained her usual routine self-care with respect to diet and physical activity prior to the onset of this symptomatology.
Upon presentation, her vital signs included a blood pressure of 164/95 mm Hg, a pulse of 92 beats/minute, a respiratory rate of 18 breaths/minute (rr−min), an oxygen saturation of 98%, and a temperature of 98.6 ° (afebrile). The patient was alert and oriented, with no overt focal neurological deficits. The cardiovascular examination revealed normal heart sounds (S1 and S2) without additional sounds or murmurs. Her jugular venous pulse was not elevated, and there was no sacral or pedal edema. Her apical pulse was not displaced with a normal character. The respiratory examination revealed bilateral vesicular breath sounds without wheezing or crackles. Her otorhinolaryngology examination revealed bilateral erythematous auricles, consistent with auricular chondritis, a characteristic sign of RP (Figure 1). 10 An initial 12-lead electrocardiogram showed a normal sinus rhythm at a rate of 84 beats/minute, with 2 to 3 mm ST-segment-elevation in leads II, III, and aVF (Figure 2).

The patient’s right and left auricles. (A) The patient’s right ear with the encircled erythematous auricle is consistent with auricular chondritis. (B) The patient’s left ear demonstrating the characteristic sign of RP within the red circle. RP, relapsing polychondritis.
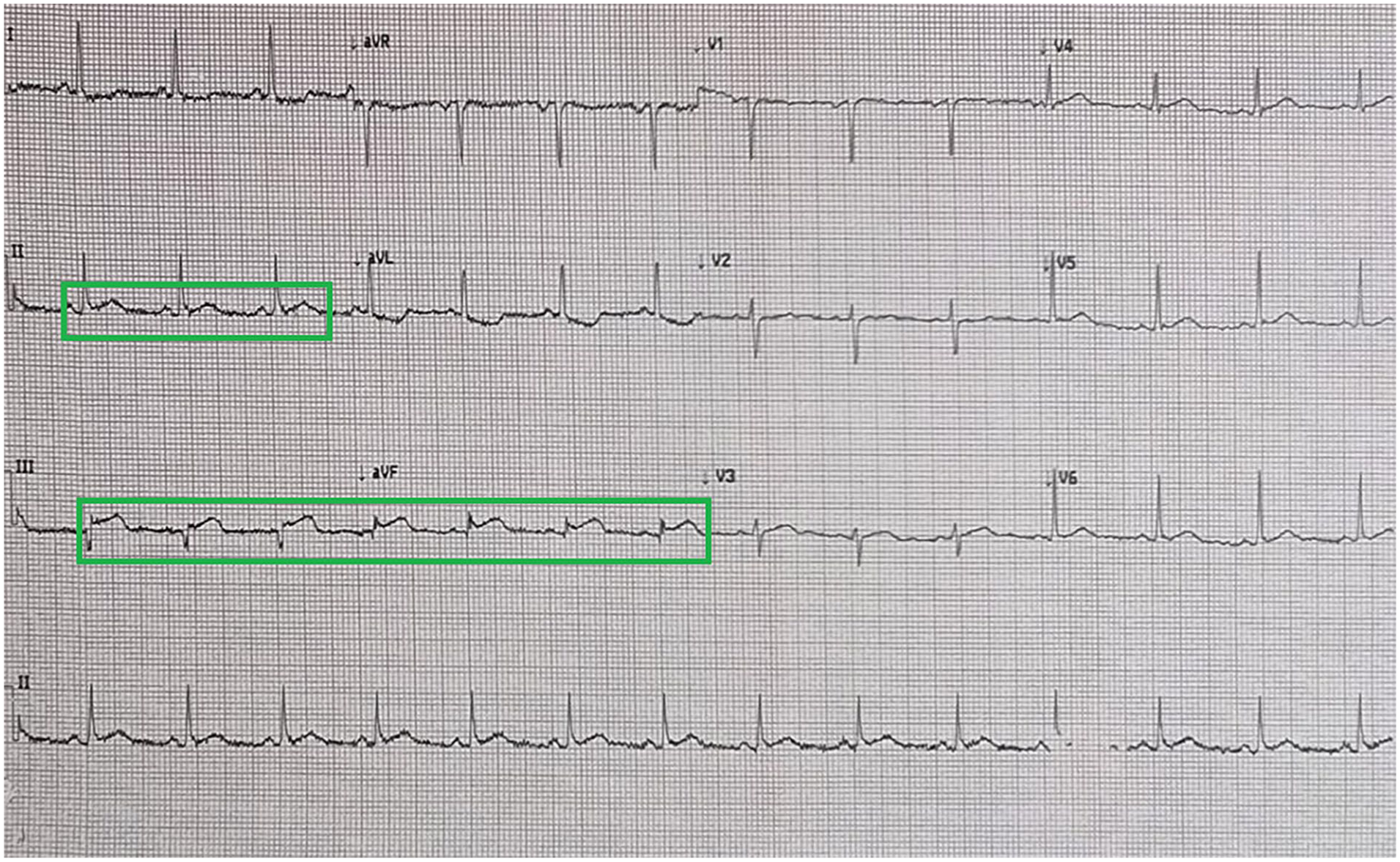
The patient’s 12-lead ECG demonstrating normal sinus rhythm at a rate of 84 beats/minute, with 2 to 3 mm STE in leads II, III, and aVF. ECG, electrocardiogram; STE, ST-segment-elevation.
A swift, bedside two-dimensional transthoracic echocardiogram demonstrated mild left ventricular inferolateral hypokinesis with an estimated left ventricular ejection fraction (LVEF) of 40% to 45%, normal tricuspid annular plane systolic excursion (TAPSE), and no severe valvular dysfunction or pulmonary hypertension. A high-sensitivity cardiac troponin I (hs-cTnI) biomarker was markedly elevated, at 82.7 ng/mL (normal range: <0.03 ng/mL), as well as NT-pro-brain natriuretic peptide, 373 pg/dL (normal range: <125 pg/mL). The patient proceeded directly to primary percutaneous coronary intervention, which revealed angiographically normal coronary arteries with no evidence of CAD but thrombolysis in myocardial infarction 2 (TIMI 2) flow (Figure 3).

The patient’s coronary cineangiography series. (A) A straight-cranial view demonstrating an angiographically normal LAD artery with 2 major diagonal branches. (B). A RAO-cranial image indicating normal LAD and LCx arteries with their respective subbranches (diagonals and obtuse marginals). (C) A LAO revealing a normal RCA along with the PDA and the PLV. RAO, right anterior oblique; LAD, left anterior descending; LCx, left circumflex; LAO, left-anterior oblique; RCA, right coronary artery; PDA, posterior descending artery; PLV, posterolateral branch.
During her ensuing hospitalization, she clinically stabilized within 24 hours in the cardiology ward. She was initiated on oral comprehensive, GDMT comprising aspirin 81 mg once daily, ticagrelor 90 mg twice daily, rosuvastatin 40 mg, valsartan/sacubitril 50 mg once daily, bisoprolol 2.5 mg once daily, finerenone 10 mg, and empagliflozin 10 mg once daily with intravenous hydrocortisone as per Rheumatology recommendations (the latter of which was eventually transitioned to oral prednisone). 11 The erythrocyte sedimentation rate was elevated at 63 units/hour (normal range <20 units/hour). Her glycated hemoglobin, lipid panel, thyroid function, D-dimer, and other routine tests were normal. Her repeat hs-cTnI levels gradually trended downward. She was further monitored for an additional 48 hours without any adverse clinical event and was subsequently discharged. She maintained her routine follow-up appointments with Cardiology and Rheumatology, where she was successfully weaned off her oral steroid therapy with interval normalization of her LVEF.
Discussion
RP is a sporadic autoimmune disorder characterized by episodic inflammation of cartilaginous tissues, primarily affecting the ears, nose, and respiratory tract.8,12 Mounting evidence suggests that immune-mediated mechanisms target proteoglycan, the primary structural component of cartilage. Both circulating and tissue-specific antibodies against collagen types II, IX, and XI have been detected in patients, implicating humoral immunity in its pathophysiology. 13 Additionally, cellular immune responses contribute to cartilage inflammation, with CD4+ T cells playing a pivotal role. These cells secrete proinflammatory cytokines, including interleukin-8, macrophage inflammatory protein-1β, and monocyte chemoattractant protein-1, which promotes the recruitment of monocytes and macrophages to sites of cartilage damage, thereby sustaining a complex milieu of chronic inflammation. 14 These maladaptive processes can potentially impair vascular endothelial function, exacerbating myocardial dysfunction. 3
In order to establish the diagnosis of MINOCA, the Fourth Universal Definition of Myocardial Infarction criteria, alongside angiographic confirmation of <50% stenosis in all major epicardial coronary arteries and no alternative explanation for the clinical presentation, must be met.3,15 Myocardial infarction with obstructive CAD and MINOCA display contrasting risk factor profiles. Conventional cardiovascular risk factors are less prevalent in MINOCA patients, whereas comorbidities such as anxiety, depression, and autoimmune pathologies are inherently more common.16,17 The pathophysiology of MINOCA is heterogeneous, encompassing both cardiac and extra-cardiac mechanisms, which can be further subclassified into atherosclerotic and non-atherosclerotic origins. 3 Atherosclerotic mechanisms chiefly involve plaque disruption or erosion, whereby, in contrast, non-atherosclerotic etiologies comprise coronary thromboembolism, coronary artery vasospasm, spontaneous coronary artery dissection, coronary microvascular dysfunction, and supply–demand mismatch.18,19 Additionally, non-ischemic myocardial injury, such as myocarditis and Takotsubo cardiomyopathy, may mimic MINOCA and result in troponin elevation without CAD. 20 A growing body of evidence alludes that vasospasm and microvascular dysfunction account for the substantial majority of the cases of MINOCA. 20 Advanced imaging modalities, such as cardiac magnetic resonance imaging, intravascular imaging with optical coherence tomography, intravascular ultrasonography, and provocative testing, are essential for ascertaining the pathophysiological mechanisms. 19 The management of MINOCA is overshadowed by limited evidence, with no established standardized treatment protocols. Pharmacotherapy, including antithrombotic agents, high-intensity statins, and neurohormonal heart failure therapies, should be individually tailored according to the identified pathology.21,22 Surveillance echocardiography and electrocardiography are essential for patients with RP. 23
This case presented several notable clinical features, with the patient’s underlying RP emerging as the incipient risk factor for precipitating MINOCA. With respect to the TIMI 2 flow, the patient responded favorably to the standard ACS protocol with intravenous glucocorticoids; thus, we postulate that this patient’s presentation was likely mediated by immune-mediated vascular dysfunction.24,25 She did not display any refractory features that required escalating immunosuppressive therapies, such as biologics for her RP.26-28 Additionally, our patient displayed parameters associated with cardiac involvement, such as age (40 years) and protracted disease duration (6 years), without airway-related involvement. 23 There are conflicting studies that implicate cardiac involvement as a negative prognosticator.29-31
Our patient also did not display traditional cardiovascular risk factors such as diabetes, hypertension, dyslipidemia, or tobacco use. In fact, the Predicting Risk of Cardiovascular Disease (PREVENT) equation estimated her 10- and 30-year risk for total CVD at 0.4% and 4.9%, respectively, even factoring in her South Asian ethnicity but excluding her chronic, autoimmune RP, likely underestimating her risk.32,33 Our patient eventually underwent lipoprotein(a) (Lp[a]) testing post-discharge, which returned borderline (34 mg/dL, normal <50 mg/dL), with the major caveat of being on high-intensity statin therapy. It is crucial to recognize that South Asians have a relatively higher prevalence of elevated Lp(a), which may account for the earlier onset of aggressive CVD phenotypes that carry an inherent negative prognosis for major adverse cardiovascular events. 34 The clinician should be cognizant of the association of subtle or seemingly innocuous autoimmune syndromes, such as RP, in patients presenting with AMI.
Conclusion
We describe a 40-year-old South Asian female with RP-associated MINOCA during an acute episode, for which she was successfully managed with GDMT. This case highlights the importance of considering atypical etiologies such as autoimmune disease in patients with ACS. Early recognition and appropriate treatment are paramount for optimizing patient outcomes.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
