Abstract
The aim of this study is to examine whether the histone deacetylase inhibitor, suberoylanilide hydroxamic acid (SAHA), can generate dendritic cells (DCs) with a stable tolerogenic phenotype to counteract autoimmune responses in an animal model of multiple sclerosis. We investigated if the tolerogenic potency of DCs could be increased by continuous treatment during in vitro differentiation toward DCs compared to standard 24-h in vitro treatment of already terminally differentiated DCs. We show that in vitro treatment with SAHA reduces the generation of new CD11c+ DCs out of mouse bone marrow. SAHA-generated DCs show reduced antigen-presenting function as evidenced by a reduction in myelin endocytosis, a decreased MHC II expression, and a failure to upregulate costimulatory molecules upon LPS challenge. In addition, SAHA-generated DCs display a reduction in proinflammatory cytokines and molecules involved in apoptosis induction, inflammatory migration, and TLR signaling, and they are less immunostimulatory compared to untreated DCs. We demonstrated that the underlying mechanism involves a diminished STAT1 phosphorylation and was independent of STAT6 activation. Although in vitro results were promising, SAHA-generated DCs were not able to alleviate the development of experimental autoimmune encephalomyelitis in mice. In vitro washout experiments demonstrated that the tolerogenic phenotype of SAHA-treated DCs is reversible. Taken together, while SAHA potently boosts tolerogenic properties in DCs during the differentiation process in vitro, SAHA-generated DCs were unable to reduce autoimmunity in vivo. Our results imply that caution needs to be taken when developing DC-based therapies to induce tolerance in the context of autoimmune disease.
Keywords
Introduction
Histone deacetylases (HDACs) and their counterparts, histone acetyl transferases, determine the acetylation status of histone proteins (16,20). They also target non-histone proteins, such as transcription factors, chaperone proteins, signal transduction mediators, and DNA repair enzymes (31). Acetylation of these proteins influences protein function, signaling, stability, and protein/protein–DNA interactions (17). In this way, cellular biology is heavily affected by the use of HDAC inhibitors. By regulating the natural process of acetylation and deacetylation, HDAC inhibitors selectively alter gene expression, making them promising anticancer drugs. Recent evidence indicates that HDAC inhibitors could be of use in autoimmune and inflammatory diseases (7,12,18,22). In these studies, the observed therapeutic effect was ascribed to a reduction in circulating inflammatory cytokines and possible immunomodulatory functions of these drugs. However, the exact molecular and cellular mechanisms remain elusive. One of the immune cell types affected by the HDAC inhibitor suberoylanilide hydroxamic acid (SAHA) are dendritic cells (DCs). Reddy et al. demonstrated that the expression of costimulatory molecules CD40 and CD80 was reduced on DCs when treated for 16–18 h with the HDAC inhibitor SAHA (22). Other studies revealed that 24-h SAHA treatment affects DC functionality through inhibition of DC-directed Th1 and Th17 cell polarization and suppression of cytokines (4,12,18). We hypothesize that the potency of SAHA to generate tolerogenic DCs can be boosted when DCs are continuously treated during their differentiation from bone marrow cells (10-day treatment) and that these SAHA-generated DCs have therapeutic potential in autoimmune disease.
Since DCs have the ability to shape the immune response by polarizing T cells into different effector cell types, there is significant interest to use DCs to limit immunopathogenic responses during transplantation, allergy, and autoimmune disease. In regard to this, SAHA decreased in vivo alloproliferation in an acute graft-versus-host disease mouse model (22). Injection of antigen-pulsed tolerogenic DCs may thus deliver immunoregulatory peptides in vivo leading to the induction of antigen-specific regulatory T cells or inhibition of autoreactive T cells (9,30). To date, however, there is no consensus on which stimuli guarantee the induction of stable tolerogenic DCs. Unstable tolerogenic DCs could reverse back in vivo to an immune stimulatory phenotype when in contact with proinflammatory stimuli and may thus provoke undesirable responses. It is therefore critically important to thoroughly test the potency and durability of tolerance-inducing drugs.
The goal of this study was to augment the immunomodulatory and tolerogenic capability of the HDAC inhibitor SAHA to generate stable tolerogenic CD11c+ DCs that counteract autoimmune responses in an animal model of multiple sclerosis (MS). SAHA-mediated effects on DC phenotype and function and the mechanisms involved were assessed in vitro. Consequently, the disease-modifying potential of SAHA-generated CD11c+ DCs was tested in mice with experimental autoimmune encephalomyelitis (EAE).
Materials and Methods
Mice
Eight-week-old C57BL/6J female mice were purchased from Harlan (Horst, the Netherlands). Animals were housed under standard conditions with water and food ad libitum in the animal facility of the Biomedical Research Institute of Hasselt University. The animals were allowed to acclimatize 1 week before the start of the experiments. Experiments were conducted in accordance with institutional guidelines and approved by the local Ethical Committee for Animal Experiments of Hasselt University.
DC Generation, Culture, and Treatment
DCs were generated out of bone marrow isolated from tibiae and femurs of C57BL/6J female mice based on Inaba et al. (13) and Lutz et al. (19). Briefly, bone marrow was rinsed with Iscove's modified Dulbecco's medium (IMDM; Lonza, Braine-L'Alleud, Belgium) supplemented with 10% fetal calf serum (FCS; Gibco, Ghent, Belgium). After centrifugation (350 × g), the pellet was resuspended in red blood cell lysis buffer (0.155 M NH4Cl, 0.01 M KHCO3, and 0.1 mM Na2EDTA; VWR, Leuven, Belgium) and incubated for 10 min at room temperature. Next, cells were placed in IMDM supplemented with 10% FCS, 20 ng/ml granulocyte macrophage colony-stimulating factor (GM-CSF; R&D Systems, Abingdon, UK), 10 ng/ml IL-4 (R&D Systems), and 1% penicillin–streptomycin (Gibco) and consequently plated at 3 × 106 cells/well and incubated at 37°C and 5% CO2. Culture medium was changed at days 3, 6, and 8. On day 6, cells were harvested with 5 mM ethylenediaminetetraacetic acid (EDTA; VWR), counted, and replated at 500,000 cells/well. DCs or bone marrow were treated for 24 h (standard treatment) or 10 days (from start of differentiation process) with 0.5 or 1 μM SAHA (Cayman Chemical Company, Huissen, the Netherlands). To generate mature DCs, cells were stimulated 24 h with 100 ng/ml lipopolysaccharide (LPS; Sigma-Aldrich, Bornem, Belgium). To assess the durability of the SAHA effect, DCs were treated for 10 days with SAHA, consequently placed on culture medium without SAHA, and finally restimulated with LPS on day 12 of culture. SAHA was dissolved according to manufacturer's instruction in dimethyl sulfoxide (DMSO; Sigma-Aldrich), aliquoted, and stored at −20°C. Prior to use, SAHA was diluted to the appropriate concentration in IMDM. An equivalent volume of sterile DMSO (vehicle) was used in the 0 μM SAHA condition. Morphology and purity of cell cultures are displayed in Supplementary Figure 1 (available at: https://drive.google.com/file/d/0B__9NJmGCuIAeFlidFJzcjl3N28/edit?usp=sharing).

Ten days of SAHA treatment limits the differentiation of CD11c+ DCs. Primary immature DCs were cultured out of mouse bone marrow in IMDM with GM-CSF and IL-4 for 10 days. Mature DCs were generated by adding LPS during the last 24 h of culture. Flow cytometry of CD11c expression on DCs treated with SAHA for (A) 24 h or (B) 10 days. Results are shown as mean percentage ± SEM; n = 13–22; with (+) or without (–) LPS. **p < 0.01, ***p < 0.001.
Cell Surface Phenotype Analysis
DCs were harvested and stained with CD80-FITC, CD86-PE, PD-L1-PE, FasL-PE, CCR5-PE, TLR4-PE, TLR-9, I-Ab-PE, F4/80-PE, CD14-PE, Ly6G/C-PE (0.20–0.30 μg/test; eBioscience, Vienna, Austria) combined with CD11c-APC (BD Biosciences, Erembodegem, Belgium). Data were acquired using the FACS Aria II (BD Biosciences) and analyzed with the FacsDiva software (BD Biosciences). As a gating strategy, a gate was set on the CD11c+ population. Expression levels of surface molecules were analyzed within this DC gate.
Myelin Phagocytosis
Myelin was purified from mouse brain tissue by means of density gradient centrifugation as described previously (21). Myelin protein concentration was determined by using the bicinchoninic acid (BCA) protein assay kit (Thermo Fisher Scientific, Erembodegem, Belgium). The Chromogenic Limulus Amebocyte assay kit (Genscript Inc., Aachen, Germany) was used to determine the endotoxin content. Isolated myelin contained a negligible amount of endotoxin (<1.8 × 10–3 pg/μg myelin). Myelin was fluorescently labeled with 1,1′-dioctadecyl-3,3,3′3′-tetramethylindocarbocyanine perchlorate (DiI; PE channel; Sigma-Aldrich) by incubating 12.5 μg DiI/ mg myelin for 30 min at 37°C. Endocytosis was determined by measuring the cellular uptake of DiI-labeled myelin. DCs were incubated for 90 min with 20 μg/ml DiI-myelin at 37°C and 5% CO2. Noningested myelin was removed by washing the plates with phosphate-buffered saline (PBS; Lonza). To determine endocytosis specifically within the DC population, a CD11c-APC staining was performed. Cells were analyzed by FACS Aria II (BD Biosciences) as described above. The mean fluorescence intensity (MFI) in the PE channel was used as a measure for the uptake of labeled myelin within the APC-CD11c+ DC gate.
T-Cell Proliferation Assay
SAHA-generated DCs (10 days, 1 μM) or untreated DCs were cocultured with concanavalin A (ConA)-stimulated (2.5 μg/ml; Sigma-Aldrich) carboxyfluorescein succinimidyl ester (CFSE)-labeled (2 μM; Life Technologies, Merelbeke, Belgium) splenocytes, isolated from naive C57BL/6J mice. CFSE diluted within the CD4+ population was measured after 5 days of culture using flow cytometry (FACSCalibur and CellQuest software; BD Biosciences).
Cytokine ELISA
Secretion of interleukin (IL-6), IL-10, and tumor necrosis factor-α (TNF-α) was measured in the supernatant of DC cultures using enzyme-linked immunosorbent assay (ELISA), following the manufacturer's protocol (eBioscience). Plates were read at a wavelength of 450 nm (reference filter 630 nm) using a Bio-Rad Benchmark spectrophotometer (Bio-Rad Laboratories, Eke, Belgium).
STAT Signaling
After 10 days of culture, DCs were allowed to rest in IMDM for 2 h at 37°C and 5% CO2. Cells were incubated for 15 min with IMDM supplemented with 10% FCS, 20 ng/ml GM-CSF, 10 ng/ml IL-4, and 1% penicillin–streptomycin. Additionally, mature DCs also received 100 ng/ml LPS. After 15 min, cells were immediately fixed by adding cytofix buffer (BD Biosciences) for 10 min at 37°C. Cells were stained with CD11c-APC and then permeabilized by adding Perm Buffer III (BD Biosciences) for 30 min on ice. Phosphorylation of signal transducer and activator of transcription 1 (STAT1) (pY701) and STAT6 (pY641) was determined by staining with, respectively, PE-pSTAT1 or PE-pSTAT6 antibodies (both obtained from BD Biosciences) for 60 min at room temperature. Data were acquired using the FACS Aria II and analyzed with the FacsDiva software. Phosphorylation of STAT1 and STAT6 was analyzed within the CD11c+ DC gate.
EAE Induction and Treatment with DCs
EAE was induced in 10-week-old C57BL/6J female mice from Harlan using the Hooke Kit™ (Hooke Laboratories, Lawrence, MA, USA) according to the manufacturer's instructions. Briefly, mice were injected subcutaneously with 100 μl of 1 mg/ml myelin oligodendrocyte protein peptide (MOG)35–55 emulsified in CFA containing 2 mg/ml mycobacterium tuberculosis H37Ra. Mice received an intraperitoneal injection of 100 μl of 2 μg/ml pertussis toxin (Hooke Laboratories) directly after immunization and 24 h later. DCs were treated with 1 μM SAHA for 10 days. Next, DCs were pulsed for 24 h with 5 μg/ml MOG35–55 to generate MOG-presenting DCs. Mice were then treated with intraperitoneal injections of 1 × 106 MOG-pulsed DCs, SAHA-treated MOG-pulsed DCs, or PBS (control) at the following time points: 3 days before EAE induction and 3, 6, and 9 days postinduction. Mice were evaluated on a daily basis for changes in body weight and disease severity, using a standard 5-point scale: 0, no clinical symptoms; 1, decreased tail tone; 2, paresis of hind limbs; 3, paralysis of hind limbs; 4, quadraparesis; 5, death.
MOG-Specific Immune Cell Proliferation Assay
At day 9 or 11 postinduction, spleens and inguinal lymph nodes were isolated from EAE mice. Single-cell suspensions were obtained by passage through a 70-μM mesh (BD Biosciences). Spleen cells underwent a Ficoll (Sigma-Aldrich) density gradient centrifugation after which the buffy layer was collected and washed with PBS. Lymph node and spleen cells were placed in RPMI-1640 (Lonza) medium containing 2% mouse serum (Harlan), 20 μM 2-mercaptoethanol (Sigma-Aldrich), 1% nonessential amino acids (Sigma-Aldrich), 1% sodium pyruvate (Sigma-Aldrich), and 1% penicillin–streptomycin and restimulated with 20 μg/ml MOG (Sigma-Aldrich). Unstimulated cells were used as a control. [3H]Thymidine (5 μCi) (PerkinElmer, Zaventem, Belgium) was added after 2 days of culture, and cells were harvested 24 h later using an automatic cell harvester (Pharmacia, Uppsala, Sweden). Radioactivity was quantified by means of a β-plate liquid scintillation counter (PerkinElmer). Stimulation indexes were calculated by dividing proliferation values in the MOG-stimulated cultures by those in the respective nonstimulated controls.
Statistics
Data were statistically analyzed using GraphPad (Software Prism, La Jolla, CA, USA) and are presented as mean ± SEM. Normality was checked with a D'Agostino–Pearson omnibus normality test. One-way ANOVA followed by Tukey's post hoc analysis for testing individual groups were performed. For EAE experiments, a two-way ANOVA was used, dependent on time and treatment, followed by a Bonferroni test to examine significance between treatment groups and the control PBS group. Significance was considered positive with a value of p < 0.05.
Results
SAHA Treatment Reduces the Generation of Bone Marrow-Derived CD11c+ DCs
We first investigated the effect of SAHA on the ex vivo generation of DCs from mouse bone marrow precursors by measuring CD11c expression in 10-day cultures. We tested the effect of SAHA either directly on already differentiated CD11c+ DCs (standard treatment) or on the differentiation toward DCs (treatment during the complete culture period, 10 days). A low dose (0.5 or 1 μM) of SAHA was chosen since higher concentrations (2.5–7.5 μM) of SAHA arrest cell growth (5). No increase in cell death was detected based on cell morphology and cell counts in tested conditions (data not shown). To generate mature CD11c+ DCs, an LPS stimulus was applied during the last 24 h of culture. When measuring the CD11c expression in standard SAHA-treated DCs, a modest but significant reduction of CD11c was observed in the 1 μM SAHA-treated group compared to the corresponding control group (Fig. 1A). In contrast, a strong reduction in the percentage of CD11c+ DCs was demonstrated when bone marrow was treated with 0.5 or 1 μM of SAHA during the whole differentiation process toward CD11c+ DCs (10 days) (Fig. 1B). These results indicate that standard SAHA treatment only slightly affects already differentiated DCs, whereas continuous treatment can significantly limit the generation of new CD11c+ DCs out of bone marrow precursors.
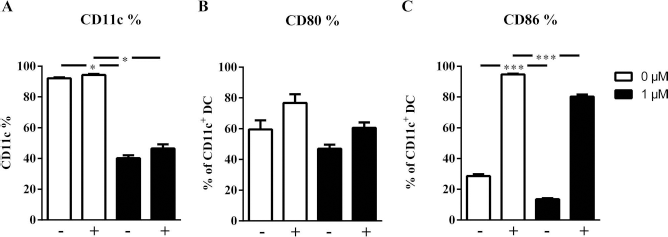
SAHA-Treated CD11c+ DCs Fail to Upregulate Costimulatory Molecules After LPS Stimulation
We next tested whether SAHA-treated CD11c+ DCs are able to provide costimulation when maturation is induced by LPS. Costimulation is one of the important signals a DC delivers to generate effective T-cell responses.
Untreated DCs significantly upregulated both CD80 and CD86 after 24 h of LPS stimulation (p < 0.001) (Fig. 2). This upregulation could not be blocked by the standard SAHA treatment (Fig. 2A,B). In contrast, upregulation of CD80 expression was blocked by SAHA treatment for 10 days (Fig. 2C). In addition, a significant inhibition of LPS-induced CD86 upregulation was detected when CD11c+ DCs were treated with 1 μM SAHA (Fig. 2D). The inhibition of the costimulatory molecules CD80 and CD86 in SAHA-generated DCs is a first indication that continuous SAHA treatment during differentiation of CD11c+ DCs is much more efficient in acquiring a tolerogenic phenotype, compared to the standard treatment of already differentiated DCs.

SAHA treatment for 10 days blocks the costimulatory upregulation of CD80 and CD86 after LPS stimulation. Primary immature DCs were cultured out of mouse bone marrow in IMDM with GM-CSF and IL-4 for 10 days. Mature DCs were generated by adding LPS for 24 h. (A) Flow cytometry of CD80 and CD86 on 24-h SAHA-treated DCs. (B) Flow cytometry of CD80 and CD86 on 10-day SAHA-generated DCs. Results are shown as mean percentage ± SEM; n = 13–18; with (+) or without (–) LPS. *p < 0.05, ***p < 0.001.
SAHA-Generated CD11c+ DCs Downregulate the Expression of Molecules Involved in Apoptosis Induction, Migration, and TLR Signaling
Next, we investigated whether SAHA-generated DCs undergo other phenotypical changes. Therefore, we analyzed molecules involved in apoptosis induction (PD-L1, FasL), migration (CCR5), or toll-like receptor (TLR) signaling (TLR4, TLR9). While SAHA (1 μM) significantly reduced the expression of FasL (Fig. 3A), programmed death-ligand 1 (PD-L1) was not affected by SAHA (Fig. 3B), indicating that the HDAC inhibitor influences specific cell death-inducing pathways. Moreover, migratory properties of SAHA-generated DCs may be influenced, as was detected by a significant diminished CCR5 expression after SAHA treatment (Fig. 3C). In addition, we detected a reduced cell surface expression of TLR4 and TLR9 (Fig. 3D,E) making SAHA-generated CD11c+ less prone to activation by danger signals. The reduction in TLR expression combined with the block in upregulation of costimulatory molecules (see above) suggests generation of stable tolerance-inducing DCs unresponsive to factors that induce an immunogenic shift in DC function when SAHA was added during the entire DC differentiation process.

SAHA treatment for 10 days reduces the expression of FasL, CCR5, TLR4, and TLR9 on CD11c+ DCs. Primary immature DCs were cultured out of mouse bone marrow in IMDM with GM-CSF and IL-4 for 10 days in the presence or absence of SAHA followed by flow cytometric analysis of the respective proteins. Mature DCs were generated by adding LPS for 24 h. Results are shown as mean percentage ± SEM; n = 4; with (+) or without (–) LPS. *p < 0.05, **p < 0.01, ***p < 0.001.
SAHA-Treated CD11c+ DCs Produce Less Proinflammatory Cytokines After LPS Stimulation
The cytokines produced by DCs play a pivotal role in the T-cell polarization. Therefore, supernatants of standard or 10-day SAHA-treated DC cultures were analyzed with ELISA for several cytokines, namely IL-6, TNF-α, and IL-10. Standard treatment of CD11c+ for 24 h with 1 μM SAHA significantly reduced the production of IL-6 and TNF-α after LPS stimulation (Fig. 4A,B). In contrast, no changes on the level of the anti-inflammatory cytokine IL-10 were detected (Fig. 4C). Similar results were obtained when DCs were generated in the presence of SAHA for 10 days. IL-6 secretion was significantly reduced with a dose of 1 μM, whereas TNF-α could be decreased with a minimum dose of 0.5 μM (Fig. 4D,E). As was the case with standard SAHA treatment, no change in IL-10 production was observed (Fig. 4F). IL-23 production was also measured, but was very low (<50 pg/ml) after 10 days of culture (data not shown).
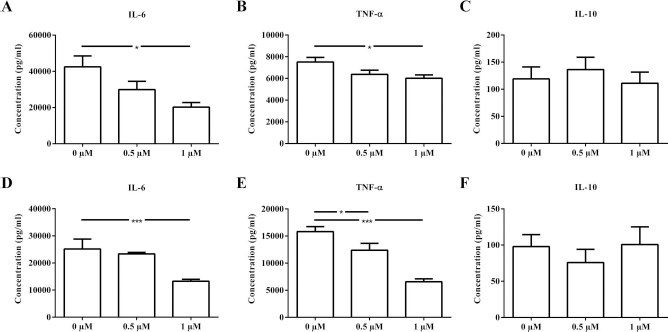
DC cultures treated with SAHA produce less proinflammatory cytokines after LPS stimulation. Production of IL-6 (A), TNF-α (B), and IL-10 (C) in response to LPS was measured with ELISA in the supernatant of cell cultures treated for 24 h with SAHA. Production of IL-6 (D), TNF-α (E), and IL-10 (F) in response to LPS were measured with ELISA in the supernatant of cell cultures treated for 10 days with SAHA. Results are shown as mean percentage ± SEM; n = 9–14. *p < 0.05, ***p < 0.001.
Altogether, SAHA treatment diminished the production of proinflammatory cytokines, whereas the anti-inflammatory cytokine IL-10 remained on the same level. Reduced secretion of proinflammatory cytokines upon TLR ligation is another hallmark of a tolerogenic DC phenotype, which in our study was previously suggested based on SAHA effect on costimulation and TLR expression.
SAHA-Generated DCs Show Diminished MHC II Expression and Myelin Endocytosis and Are Less Immunostimulatory
Providing costimulation and cytokines are important functions, but DCs are foremost specialized professional antigen-presenting cells. It is reported that immature DCs are exceptional in phagocytosing antigens from the environment, whereas the actual processing and presentation is done by mature DCs (2,8,27). Based on the observation that 10-day SAHA treatment potently inhibited upregulation of costimulatory molecules compared to the standard treatment, we investigated whether 10-day SAHA treatment also had an effect on myelin phagocytosis and the expression of major histocompatibility complex class II (MHC II) molecules. Myelin uptake of DCs is evident in postmortem brain of MS patients, suggesting DC recruitment and maturation in MS lesions. Therefore, when self-antigens are made available in vivo as a result of myelin damage, DCs can take up myelin antigens and contribute to the local activation and expansion of pathogenic T cells (25). To investigate myelin uptake, DiI-labeled myelin was added for 90 min to the cell cultures before flow cytometric analysis. In addition, the expression of mouse MHC II (I-Ab) was measured. LPS matured CD11c+ DC showed reduced myelin uptake compared to immature CD11c+ DCs (Fig. 5A). Interestingly, SAHA significantly reduced the uptake of DiI-labeled myelin regardless of maturation state. Furthermore, mature CD11c+ DCs show increased MHC-II levels in comparison with immature CD11c+ DCs (Fig. 5B). MHC II expression was almost completely absent after 1 μM of SAHA treatment.

CD11c+ DCs generated for 10 days with SAHA take up less myelin, show reduced MHC II levels, and are less immunostimulatory. (A) DiI-labeled myelin was added for 90 min to the cell cultures prior to flow cytometry. (B) MHC II or I-Ab expressions were simultaneously measured with flow cytometry. (C) ConA-stimulated CFSE-labeled splenocytes were cocultured for 10 days with SAHA-treated DCs, after which CFSE dilution of CD4+ splenocytes was measured using flow cytometry. Results are shown as mean MFI or percentage ± SEM within the CD11c+ gate; n = 4–6; with (+) or without (–) LPS. *p < 0.05, ***p < 0.001.
To investigate whether SAHA-generated DCs were able to suppress T-cell proliferation, these DCs were cocultured with ConA-stimulated splenocytes. SAHA-generated DCs (10 days) were significantly less immunostimulatory compared to untreated DCs (Fig. 5C).
Overall, 10-day SAHA treatment induced a reduced immunostimulatory phenotype in DCs as evidenced by a diminished antigen uptake and presentation together with the reduced induction of costimulatory molecules and proinflammatory cytokines.
SAHA Effects Are Paralleled with Reduced STAT1 Signaling and Are Independent of STAT6 Signaling
To reveal the downstream targets of SAHA, STAT phosphorylation is measured in 10-day SAHA-treated and untreated CD11c+ cultures. DC maturation requires activation of the STAT1 pathway, whereas immature DCs have constitutive activation of STAT6 signaling (15). SAHA reduced STAT1 phosporylation at Y701 compared to the corresponding control condition in agreement with the above-described block in DC maturation (Fig. 6A). In contrast, STAT6 phosphorylation (pY641) was reduced after LPS stimulation regardless of the presence of SAHA (Fig. 6B).

SAHA blocks maturation of CD11c+ DCs through diminished phosphorylation of STAT1 (pY701) and independent of STAT6 phosphorylation (pY641). Results are shown as mean percentage ± SEM; n = 4; with (+) or without (–) LPS. ***p < 0.01.
These results suggest that SAHA, when added at the start of differentiation, blocks CD11c+ DC maturation at least partially by interfering with STAT1 phosphorylation and consequently activation.
SAHA Treatment Did Not Enhance Clinical Potency of MOG-Pulsed CD11c+ DCs in EAE
To test whether SAHA-generated CD11c+ DCs have disease modulatory effects in vivo, 1 × 106 MOG-pulsed CD11c+ DCs pretreated in vitro with 1 μM SAHA were injected into EAE mice.
Injections of mice with MOG-pulsed DCs modestly, but significantly, reduced disease symptoms during 3 consecutive days compared to PBS-injected control mice. No differences in disease course were detected between SAHA-generated and untreated MOG pulsed DCs (Fig. 7A). Moreover, from day 16 onward, all groups followed the same clinical course, suggesting only a transient effect of the DCs, if any.
To test whether MOG-specific T-cell responses were affected by DC treatment of EAE mice, lymph nodes and spleens were collected. Mice injected with 10-day SAHA-generated or untreated DCs equally reduced MOG responses in spleen compared with PBS-injected controls (Fig. 7C). Untreated MOG-pulsed CD11c+ DCs also showed significantly less MOG reactivity in lymph nodes (Fig. 7B). Together these analyses explain the slight delay in onset of clinical EAE scores.

MOG-pulsed DCs reduced disease symptoms independent of SAHA treatment during 3 consecutive days. (A) Mice were injected intraperitoneally at four time points: –3 days, 3 days, 6 days, and 9 days postinduction with PBS, 1 × 106 MOG-pulsed CD11c+ DCs treated with 1 μM of SAHA for 10 days (n = 10 animals/group). (B) Stimulation index (SI) of MOG responses of lymph node cells ([3H]thymidine labeled) isolated on day 11 out of mice injected with PBS, 1 × 106 MOG-pulsed or MOG-pulsed 1 μM of SAHA-treated CD11c+ DCs (10-day treatment). (C) Stimulation index of MOG responses of spleen cells ([3H]thymidine labeled) isolated on day 11 out of mice injected with PBS, 1 × 106 MOG-pulsed or MOG-pulsed 1 μM of SAHA-treated CD11c+ DCs (10-day treatment). Results are shown as mean EAE score or SI ± SEM; n = 7–10. SI, stimulation index; LN, lymph nodes. **p < 0.01, ***p < 0.001.
Ex Vivo SAHA-Generated Tolerogenic DC Phenotype Is Not Stable
Given the transient effect on the EAE course, the question was raised whether in vitro CD11c+ DCs treated with 1 μM of SAHA for 10 days retain their tolerogenic phenotype once injected in vivo. To address this hypothesis, cell cultures were set up with 10-day treatment of SAHA, but before analysis cells were placed in culture medium for 2 days without SAHA. Next, maturation was induced by adding LPS for 24 h, and induction of costimulatory molecules was analyzed as described above.
This washout experiment indicated that the DCs regained the ability to upregulate the costimulatory molecules CD80 and CD86 in response to LPS (Fig. 8). CD86 was still significantly lower than untreated controls. However, the previously observed block (Fig. 2B) was abolished.
SAHA treatment for 10 days does not induce a permanent inhibition of CD80 or CD86 expression after SAHA removal. Primary immature DCs were cultured out of mouse bone marrow in IMDM with GM-CSF and IL-4 for 10 days followed by a resting phase of 2 days in culture medium without SAHA. Mature DCs were generated by adding LPS for 24 h before flow cytometric analysis of the respective molecules. (A) Flow cytometry of CD11c expression on 10-day SAHA-generated DCs. (B and C) Flow cytometry of CD80 and CD86 on 10-day SAHA-treated DCs. Results are shown as mean percentage ± SEM; n = 6; with (+) or without (–) LPS. *p < 0.05, ***p < 0.001.
From these experiments, it can be concluded that after removal of SAHA, CD11c+ DCs overcome the induced block during their differentiation process and regain their costimulatory potential.
Discussion
In this study, we examined the ability of the histone deacetylase inhibitor SAHA to ex vivo generate CD11c+ DCs with a tolerogenic phenotype out of mouse bone marrow to suppress autoimmune responses in an in vivo mouse model of MS. We looked at the increased potency of SAHA to generate these DCs when treatment was started during the differentiation toward DCs in comparison with the standard 24-h treatment of already terminally differentiated DCs. We showed that continuous SAHA treatment diminishes the number of newly generated CD11c+ DCs out of bone marrow. Furthermore, these SAHA-generated DCs acquired characteristics of tolerogenic DCs, namely a reduced ability to respond to the TLR ligand LPS as evidenced by a reduced induction of MHC II, costimulatory molecules, and proinflammatory cytokines, in contrast to standard treated DCs. All of these factors are essential features of immunostimulatory DCs to drive T-cell priming and polarization toward Th1 and Th17 cells in autoimmunity and downregulation, therefore suggesting reduced ability to prime and induce T cells. Furthermore, SAHA-generated DCs suppressed T-cell proliferation in vitro, which provides additional evidence for tolerance induction. Although the SAHA-generated DCs had a tolerogenic phenotype in vitro, they were not able to fully counteract detrimental autoimmune responses when injected into an experimental model of MS. In vitro analysis of SAHA-generated DCs showed that, after removal of SAHA, costimulatory molecules were again upregulated in response to LPS. Our results demonstrate that testing the stability of tolerogenic phenotypes should be a priority when developing DC-based therapies to restore tolerance in the context of autoimmunity.
The mechanism behind the induction of the tolerogenic DC features by SAHA was related to SAHA's interference with STAT1 phosphorylation, whereas STAT6 phosporylation was unchanged. STAT1 signaling is essential in the maturation process (15), in line with our observation of a complete block in upregulation of the costimulatory molecules CD80 and CD86 on immature DCs upon LPS challenge. Moreover, in our cell cultures, IL-4 and GM-CSF were used to generate CD11c+ or conventional DCs. A study of Jackson et al. proved that IL-4 and GM-CSF activate distinct and overlapping STAT pathways vital in DC differentiation and maturation (15). STAT1 phosphorylation and subsequent activation is detected in all stages of DC development. However, a more robust level is needed for complete maturation. In contrast to STAT1, STAT6 is only constitutively activated in immature DCs (15). CCR5's ligand CCL5 is proven to be important in mediating leukocyte adhesion toward the central nervous system (CNS) in EAE (10,28). Since SAHA-generated DCs had a reduction in the chemokine receptor CCR5, one can expect that these DCs are less capable of migrating to the CNS. FasL is involved in controlling cells by inducing apoptosis in autoreactive T cells. The observed reduction in FasL levels on SAHA-generated DCs could thus interfere with the control of autoreactive T cells. However, in contrast to FasL, PD-L1 was not changed after 10-day SAHA treatment. Expression by DCs of coinhibitory molecules such as PD-L1 is crucial for the downregulation of T-cell responses and the maintenance of immune homeostasis. PD-L1 expression is described as one of the markers for tolerogenic DCs (26,29). Wölfle et al. showed that PD-L1 expression is regulated via STAT3 activation (29). Since our data showed no changes in PD-L1 levels, we can speculate that there was no interference on the STAT3 pathway as was also the case with STAT6. Other studies in DCs revealed that STAT1 is involved in TLR4 signaling. This signaling leads to the induction of a range of genes involved in proinflammatory cytokines, chemokines, and cell surface molecules (1,23,24). Our experiments showed that besides surface molecules, proinflammatory cytokine production (IL-6 and TNF-α) was also strongly reduced when SAHA was present in the cultures. In contrast, no changes in basal levels of IL-10 secretion were detected with or without SAHA. However, we have to keep in mind that DCs were stimulated with LPS, which induces increased proinflammatory cytokine production and is therefore not the appropriate stimulus for IL-10 induction; it may be concluded that treatment with SAHA on its own did not increase IL-10 production.
Taken together, SAHA at the start of the DC differentiation process induces immature CD11c+ DCs with tolerogenic features such as reduced antigen uptake and presentation, low levels of costimulation, and diminished cytokine production that can be explained by the observed reduction in TLR4 and STAT1 signaling. Still, other pathways, like nuclear factor κ-light-chain enhancer of activated B cells (NF-κB), are also regulated by acetylation and hence may be blocked by SAHA (6).
When SAHA-generated DCs were injected into EAE mice, we observed only a minor and temporal effect on the disease course, which is in contrast to the findings of Iruretagoyena et al. (14). As illustrated by our in vitro experiments, we suggest that immature DCs mature in vivo before or soon after the appearance of clinical symptoms in the EAE model. Furthermore, there was no additional clinical effect of SAHA pretreatment as untreated immature DCs reduced the onset of clinical symptoms to the same extent. Moreover, a similar decrease in SAHA-generated and untreated immature MOG-DCs was found at the level of anti-MOG T-cell reactivity in secondary lymph nodes. This decrease in MOG reactivity can be explained by the immature DC phenotype, which already suggests intrinsic tolerogenic properties (2,3). Mechanistically, the transplanted immature DCs could have reduced the priming of MOG-specific effector T cells or alternatively induced MOG-specific Foxp3+ regulatory T cells that limit MOG-specific T-cell responses in the lymph nodes, as previously reported (11,14,32,33). Tolerogenic characteristics induced by SAHA in vitro were shown to be only temporal since the removal of SAHA overcame the block in maturation and restored the immunostimulatory characteristics in vitro. This is an experiment that is lacking in most studies, which describes promising results when testing new tolerance-inducing drugs in vitro. The instability of the SAHA-induced block in maturation explains why SAHA-generated DCs are not able to limit autoimmune responses in EAE mice, since they likely revert back to stimulating MOG-specific immune responses.
In summary, our work reveals that SAHA can have profound effects on DC differentiation leading to enhanced tolerogenic properties in vitro compared to the standard SAHA treatment on terminally differentiated DCs. DCs that differentiated out of bone marrow in the presence of SAHA are hampered in their specific functions, such as antigen uptake and presentation, providing costimulation and cytokines that are necessary for an optimal T-cell proliferation and polarization. Although in vitro results were promising, a stable induction of tolerogenic DCs to treat an autoimmune disease model could not be achieved with SAHA. Therefore, our results imply that caution needs to be taken when working on DC-mediated tolerance-inducing therapies since in vivo stimuli can undermine the stability of tolerogenic DCs leading to a lack of efficacy.
Footnotes
Acknowledgments
This work was supported by grant No. G.0168.09 of the Fund for Scientific Research–Flanders, Belgium (FWO-Vlaanderen). Further support was provided through the Flemish Institute for Science and Technology (IWT). The authors declare no conflicts of interest.
