Abstract
Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) is a rare primary cutaneous lymphoma composed of CD8+ cytotoxic T-cell that is primarily localized in the subcutaneous tissue. No standard treatments are currently available for SPTCL due to its rarity. Chemotherapy, radiotherapy, immunosuppressive agents, and hematopoietic stem cell transplantation (HSCT) have been used frequently, however, the effects of these treatment approaches remain controversial. In this report, we present an unusual case of SPTCL in a 47-year-old woman whose initial symptoms were atypical. The patient was started on etoposide, vincristine, cyclophosphamide, doxorubicin, and prednisone (EPOCH) chemotherapy once diagnosed. After two cycles of chemotherapy, her clinical symptoms were not significantly improved. Subsequently, histone deacetylase (HDAC) inhibitor chidamide was added to the chemotherapy from the third cycle. She recovered gradually and achieved complete remission (CR) after four cycles of chemotherapy combined with chidamide, followed by chidamide monotherapy for maintenance. More than 1 year after the therapy, she remained in CR. Our case illustrates, for the first time, chidamide can be an effective agent to induce long-term remission for rare SPTCL.
Keywords
Introduction
Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) is a distinct subset of primary cutaneous T-cell lymphoma that preferentially infiltrates subcutaneous adipose tissue, mimicking panniculitis, accounting for approximately 1%–2.3% of cutaneous lymphoma. 1 The typical manifestations of SPTCL are subcutaneous nodules and/or plaques of the extremities and/or trunk,2,3 but it can also involve extracutaneous viscerals, such as bone marrow, 4 breast, 5 liver, 5 spleen, 5 mesenteric fat, 6 lymph node, 7 lung,8,9 and muscle. 10 We report a 47-year-old woman with SPTCL who presented with serious pulmonary diseases as the first symptom, gradually she appeared swelling of right thigh as the disease progressed, thus the diagnosis is a challenge. In terms of treatment, worldwide, there is no standardized therapy currently available for SPTCL, although various treatments such as chemotherapy, radiotherapy, immunosuppressive agents, and hematopoietic stem cell transplantation (HSCT) have been reported.2,11 With the advancement of studies on abnormal epigenetic regulation of tumors, histone deacetylase (HDAC) inhibitors have become a novel anti-tumor drugs, and significant achievements have been achieved in the treatment of recurrent or refractory peripheral T-cell lymphoma (PTCL). 12 With reference to the PTCL regimen, we tried to treat this patient with HDAC inhibitor chidamide, which is the first approved oral epigenetic regulator in China, and the clinical response was better than we expected. She was followed up for more than 1 year with no signs of relapse. Chidamide not only has good clinical efficacy, but also has less adverse reactions and good compliance.
To our knowledge, there are no reports of chidamide in the treatment of SPTCL. We report a case of chidamide combined with chemotherapy for SPTCL and review the literature, which provides a new choice for the treatment of SPTCL patients in the future.
Case report
A 47-year-old woman presented with intermittent cough, shortness of breath, fever, and body temperature up to 38.5°C, accompanied by right eyelid swelling around June 17, 2017, which worsened day by day. Weight loss was about 9 kg. On July 5, 2017, she visited a local hospital and received both eyes and head computed tomography (CT) scan, no obvious abnormalities were found. Chest CT showed pulmonary infection with a high possibility of fungal infection, and a small amount of pleural effusion in the left lung. She received antibiotics about 10 days, but the symptoms appreciated no improvement. Later, the patient had aggravated the above symptoms and developed high fever with the highest body temperature of 39.4°C. As a result, the patient was transferred to our hospital with “pulmonary infection” on July 17, 2017.
Physical examination included a high body temperature of 39°C, right eyelid swelling and a painless enlarged lymph node about 2 cm × 2 cm in size was in the left axilla. The auscultation of the lungs had coarse respiratory sounds with the moist rale. No hepatosplenomegaly and edema in lower limbs. Laboratory data revealed decreased leucocyte (1.50 × 10–9/L) and hemoglobin (88 g/L), raised aspartate transaminase (AST) (61 U/L), and lactate dehydrogenase (LDH) (724 U/L). Galactomannan (GM) test was positive and plasma (1,3)-β-D-glucan (G) test was negative. Coagulation function, kidney function, ferritin, and erythrocyte sedimentation rate were all normal. An infectious origin was ruled out by sputum culture and acid-fast staining, and blood culture. HIV, hepatitis, and syphilis test panels were negative. Her autoimmune workup was negative. Reexamination of chest CT showed interstitial exudation in both lungs, mass lesions, and left pleural effusion, still considering fungal infection (Figure 1(a)). Bronchoscopy showed acute inflammatory changes in the bronchial mucosa. The above clinical findings indicated suspected aspergillosis pneumonia.

Chest CT image. (a) Interstitial exudation and mass lesions of both lungs can be seen before treatment. (b) Lung lesions were significantly diminished after four cycles of EPOCH chemotherapy combined with chidamide treatment.
After symptomatic treatment with antibacterial, fungal, and leucocyte-elevating, her systemic symptoms such as fever, cough, and eyelid swelling improved, and leucocyte increased. Reexamination of orbital and chest CT showed no abnormalities in the orbit, but interstitial exudation of both lungs and left pleural effusion were more than the previous CT scan. On August 4, 2017, positron emission tomography (PET)-CT images showed multiple spherical lesions and ground glass-like increased images in the lungs, with significant lower lobe, and mostly hypermetabolic activity; left pleural and right interlobular effusion. Biopsy of hypermetabolic nodules in the chest wall revealed difficulty in differentiating inflammation and tumor, and PCR was negative for tuberculosis in paraffin-embedded tissue. Anti-infective treatment was continued and the patient was discharged 1 week later due to improvement in symptoms. However, fever returned after a brief respite and did not improve. She revisited our hospital on August 28, 2017. Reexamination of chest CT exhibited a reduced volume of multiple lesions of both lungs, and the interstitial exudation was slightly absorbed compared to before. A blood reexamination revealed re-decrease of leucocyte, higher levels of AST and LDH than in the first admission, and raised blood β2 microglobulin (2549 ng/Ml). A of biopsy of the lung tissue was performed and revealed a high number of inflammatory cells infiltration in the pleural. Anti-infective treatment was ineffective, and her fever was unremitting, along with right thigh redness, swolling, heat, and painful (Figure 2(a) and (b)). On September 1, 2017, vascular ultrasound of the right lower limb showed intermuscular venous thrombosis, enlarged inguinal lymph nodes (multiple), and subcutaneous soft tissue edema with interstitial effusion on the lateral thigh. The patient underwent surgical decompression of fascial compartment and pathological biopsy of neoplasm (subcutaneous neoplasm, muscle interstitial neoplasm) of the right limb on September 4, 2017. Histopathological examination demonstrated focal rimming of individual adipocytes by infiltrating small to medium-sized atypical T lymphocytes with irregular and hyperchromatic nuclei. Immunohistochemistry exhibited positivity for CD3, CD8, granzyme B (GB), CD43, and T-cell intracellular antigen-1 (TIA-1), and negativity for CD4, CD56, CK, and CD79, Ki67 staining positive was 50% (Figure 3(a)–(d)), which led to a pathological diagnosis of SPTCL. A monoclonal rearrangement of TCR-β gene was confirmed by polymerase chain reaction (PCR). Bone marrow aspiration suggested proliferative anemia and infection, and bone marrow biopsy had no lymphoma cells infiltration. A final diagnosis of SPTCL (IIB phase, IPI scores of 2) was made.
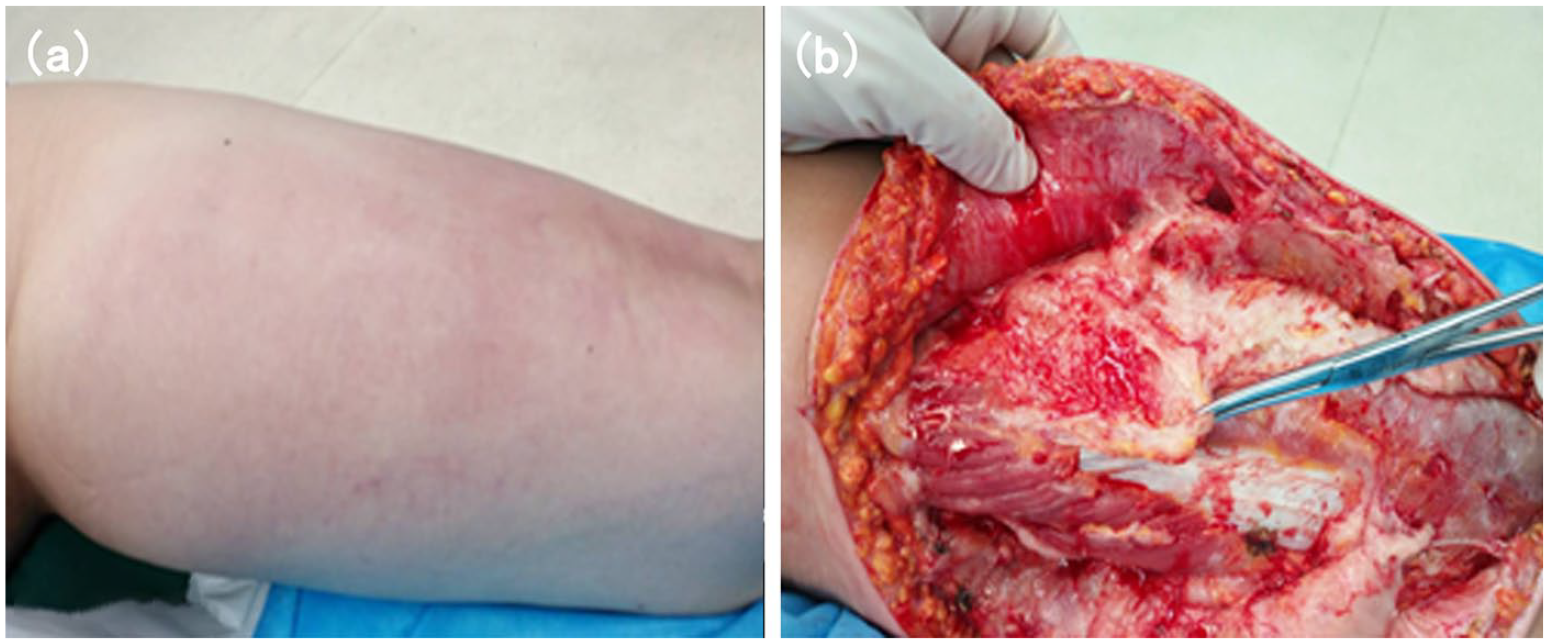
Showing images of the patient’s right thigh. (a) The patient exhibited prominent right thigh swelling. (b) Pathological biopsy showed gray–white and gray–yellow neoplasm in intermuscular space.

Histopathological findings. (a) Focal rimming of individual adipocytes by infiltrating small to medium-sized atypical lymphocytes. (b–d) Immunostaining showed infiltrating lymphocytes positive for CD8 (b), granzyme B (c), and T-cell intracellular antigen-1(TIA)-1 (d).
She was treated on September 15, 2017 with etoposide, vincristine, cyclophosphamide, doxorubicin, and prednisone (EPOCH) chemotherapy. After two cycles of chemotherapy, the patient’s clinical symptoms were not significantly improved, she still had intermittent fever and shortness of breath, the enlarged lymph node in the left axillary did not noticeably shrunk, and the swelling of right thigh was not significantly reduced. Considering obvious systemic symptoms of the patients and chidamide is effective in PTCL, an attempt was made to add chidamide (30 mg BIW oral) to the chemotherapy from the third cycle. Fortunately, the patient responded well with resolution of all systemic manifestations of disease within four cycles of combined chemotherapy with chidamide, the patient had normal body temperature, significant improvement in cough and shortness of breath, and complete resolution of enlarged lymph node of left axillary and swelling of right thigh. Additionally, the chest CT showed that the lung lesions were significantly diminished (Figure 1(b)), and the right thigh magnetic resonance imaging (MRI) showed no obvious abnormal signal in subcutaneous and layers of muscle group (Figure 4). The efficacy was evaluated as CR, and patient was recommended to undergo HSCT as consolidation for long-term disease control, but the patient declined HSCT treatment, so she continued to underwent two cycles of EPOCH chemotherapy combined with chidamide. CR was evaluated again after six cycles of combined treatment, followed by chidamide monotherapy for maintenance. The last follow-up date was on January 16, 2020, and remission has been maintained. Written informed consent was obtained from the patient’s legally authorized representative for publication of this case report and accompanying images.
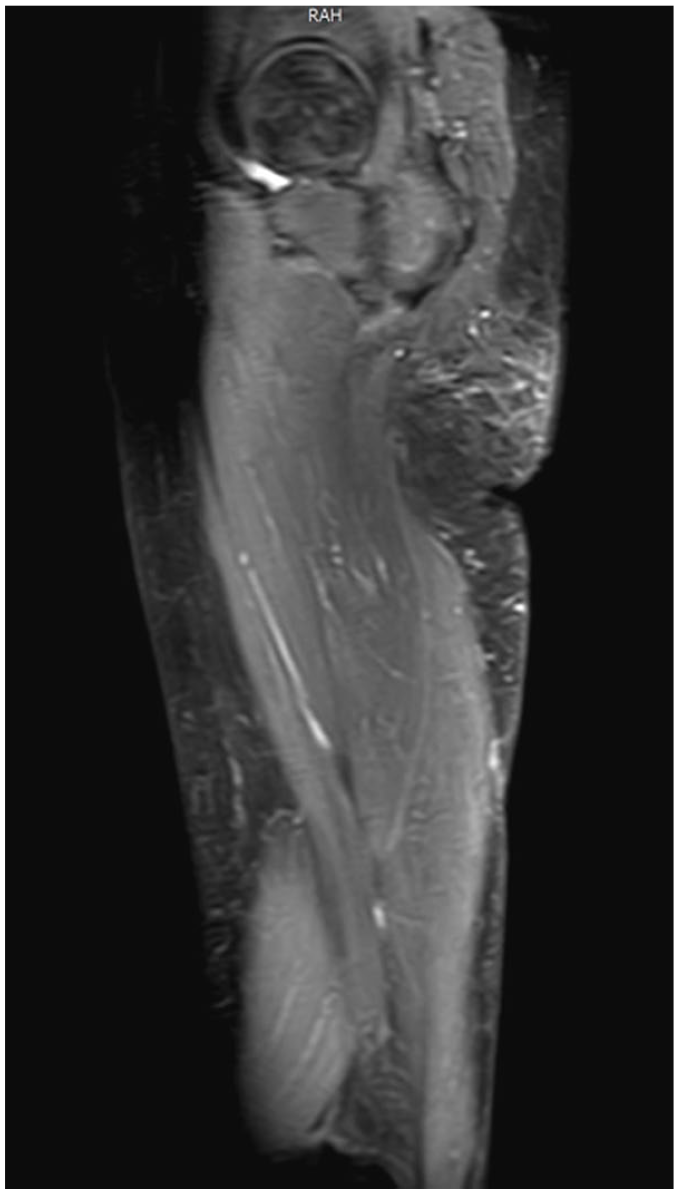
Right thigh MRI showed no obvious abnormal signal in subcutaneous and layers of muscle group.
Discussion
SPTCL is an uncommon primary cutaneous CD8+ cytotoxic T cell lymphoma.1,13 SPTCL was previously categorized into α/β-type and the γ/δ-type based on T-cell receptors (TCR) and immunophenotype. The immunophenotype of the former is CD4−, CD8+, CD56−, which is consistent with the immunophenotype of our patient reported herein, the latter is CD4−, CD8−, CD56+.7,14 The clinical manifestations, histopathology, and prognosis also differ between these two types, with the α/β-type typically limited to the subcutaneous tissue, having an indolent clinical course and a relatively good prognosis, while the γ/δ-type often involves the epidermis or dermis, has a rapid course, is more likely to be complicated by haemophagocytic syndrome (HPS), and has a very poor prognosis.7,13 In 2005, the World Health Organization-European Organization for Research and Treatment of Cancer (WHO-EORTC) classification for cutaneous lymphoma defined SPTCL as α/β T-cell lymphoma, while the γ/δ-type was classified as cutaneous γ/δ T cell lymphoma. 15
Clinically, subcutaneous nodules mainly on the extremities and trunk are commonly the initial symptoms of SPTCL patients. 2 However, occasionally, the earliest symptoms of some patients are characterized by facial or eyelid swelling and long-term fever, but no skin manifestations.7,16,17 In addition, SPTCL can also involve multiple extracutaneous viscerals such as bone marrow, 4 breast, 5 mesenteric fat, 6 lymph node, 7 lung,8,9 and muscle. 10 Common systemic manifestations, including B symptoms, hepatosplenomegaly, lymphadenopathy, hepatopathy, and increased LDH. More serious conditions include serosal effusions, HPS, and cytopenia.7,18 The manifestations of our patient in this paper is different from the common clinical manifestations of SPTCL reported previously. Her original symptoms were respiratory symptoms, accompanied by fever and eyelid swelling. Chest CT showed lung infection, biopsies of chest wall nodules and lung tissue were both conducted, but the results were just inflammatory changes. At this stage, the patient was easily misdiagnosed as a pulmonary infection. As the disease progresses, this case subsequently exhibited swelling and pain of right thigh, and the final diagnosis is confirmed by pathological biopsy of the thigh lesion. Although SPTCL involvement of lung and muscle is very rare, it has been reported previously. The case of SPTCL we report here presents with lung and thigh muscle involvement. Thus, the clinical manifestations of SPTCL are diverse, involving not only the skin but also a variety of extracutaneous viscerals, making the diagnosis of SPTCL difficult. In addition, SPTCL is a rare lymphoma, sometimes the diagnosis is a challenge. Clinicians need to be concerned about the patient’s clinical presentation, and patients with recurrent fever, eyelid or limb swelling should be highly suspected of cutaneous lymphoma including SPTCL, even if excluded early by pathologic biopsy, and consider re-biopsy when symptoms recur or worsen. The prognosis of SPTCL is generally good, the 5-year survival rate is reported to be approximately 80%. However, HSP had a negative effect on the prognosis of SPTCL, the 5-year overall survival rates of SPTCL patients with or without HPS were 46% and 91%, respectively. 13 Our patient did not develop HSP.
At present, the treatment of SPTCL is the most important and controversial issue, and there is a lack of standard and effective therapy for SPTCL. Currently reported treatments include chemotherapy, radiotherapy, immunosuppressive agents, and HSCT. ESMO clinical practice guideline proposed that for SPTCL patients without HPS, systemic steroids or other immunosuppressive agents are recommended as the preferred treatment option. Radiotherapy is recommended for patients with only single or limited lesions. Patients with progressive disease that does not respond to immunosuppressive therapy or those with HPS require multidrug chemotherapy. 19 Chemotherapy is the most common first-line treatment for patients with SPTCL, one study has reported that approximately 80% of Japanese SPTCL cases receive chemotherapy as first- or second-line treatment, and as well as two-thirds of Chinese SPTCL cases. 3 However, for patients with HPS, the effects of chemotherapy are not ideal, and because the overall prognosis of this disease is good, chemotherapy has been controversial. Immunosuppressive agents have been favored by scholars in recent years, and some studies have emphasized the effectiveness of immunosuppressive therapy. However, the immunosuppressive therapy has a short-term response, and once the drug is reduced or discontinued, the disease will progress, and the recurrence rate is higher than that of chemotherapy.11,20 HSCT has a certain efficacy in SPTCL patients with HPS. A recent study on HSCT for SPTCL in Asian population showed that of eight patients treated with HSCT, including one autologous HSCT, and seven allogeneic HSCT, seven of the eight patients achieved durable remission and maintained remission for more than 30 months. 21 However, the majority of patients were unable to receive HSCT due to illness or physical condition. Therefore, there is an urgent need to find new treatment options to improve the survival of SPTCL.
HDAC inhibitors are an important class of antineoplastic agents. In recent years, a variety of HDACs have been used in the treatment of cutaneous T-cell lymphoma or PTCL. 12 There are currently three HDAC inhibitors on the market for this indication, including romidepsin, belinostat, and chidamide, the first two of which are administered intravenously, while chidamide is the first oral epigenetic regulator approved in China. 22 Chidamide is a novel benzamide HDAC inhibitor with subtype selective activity against class I HDAC subtype of 1, 2, 3, and class II subtype of 10. 22 Chidamide has spectral anti-tumor mechanisms, including inhibiting the tumor cell cycle, inducing apoptosis and differentiation of tumor cells.23,24 Chidamide also has the potential to induce tumor stem cell differentiation and reverse the epithelial-mesenchymal phenotypic transformation (EMT) of tumor cells, thereby sensitizing drug-resistant tumor cells to drugs and inhibiting tumor metastasis and recurrence.25–27 In addition, chidamide can also enhance natural killer cells (NK) and antigen-specific cytotoxic T cells (CTL) mediated tumor killing, leading to tumor cell death. 28
The study of chidamide on PTCL was based on two clinical trials, the exploratory, and critical phase II clinical trial, respectively, with significant activity and controllable toxicity. 26 PTCL with a low International prognostic index (IPI) score (IPI score of 0–1) is recommended to be treated with chidamide monotherapy with better efficacy. Patients with intermediate and high-risk IPI scores (IPI scores of 2–5) who have a large tumor burden and aggressive tumor growth are recommended to use chidamide in combination with commonly used second-line chemotherapy. 26 In a recent multicenter clinical study conducted in China, 383 patients with relapsed or refractory PTCL were treated with chidamide, and the results showed that overall response rate (ORR) and disease control rate (DCR) of 256 patients treated with chidamide monotherapy were 39.06% and 64.45%, respectively, and the ORR and DCR of 127 patients received chidamide combined with chemotherapy were 51.18% and 74.02%, respectively. Progression-free survival (PFS) was 129 days in the monotherapy group and 152 days in the combination therapy group. 29 Chidamide can be combined with ICE, Gemox, GDP, DICE, DA-EPOCH, PET, thalidomide, lenalidomide, and many other regimens, and the ORR of these combined regimens exceeds 60%.29,30 To our knowledge, no case of SPTCL have been treated with chidamide. For our patient, the systemic symptoms were obvious in the early stages of the disease, and IPI score was 2, therefore, the combination therapy of chidamide and EPOCH chemotherapy was given.
Adverse events (AEs) of chidamide are controllable, most often grade 1–2, with the most common grade 3–4 AEs being mainly hematological adverse reactions, manifested by the decrease in platelets, leukocytes/neutrophils, and hemoglobin. Non-hematologic AEs are fatigue and gastrointestinal symptoms, there are also reports of abnormal liver and kidney function. The incidence of common AEs were increased in combination therapy, but no change in AE profile. 26 Compared with other HDAC inhibitors, chidamide has a lower incidence of gastrointestinal disorders and infections and is less severe.22,26 For this patient, hematologic AEs during the treatment of chidamide were mainly leukopenia (grade 3), which returned to normal with leucocyte-elevating therapy. Non-hematologic AE were mainly fatigue (grade 1) without special treatment.
Conclusion
The original symptoms of the patient reported in this paper are atypical, multiple systems are involved, and the pathological biopsy in the early stages of the disease is negative, which made the diagnosis difficult, thus the importance of multiple pathological biopsy is shown in this process. In terms of treatment, we applied chidamide for the first time to treat SPTCL. This patient was treated with chidamide in combination with EPOCH regimen for a total of six courses. At the end of chemotherapy, oral chidamide monotherapy was administered as maintenance therapy and the patient remained in CR. Chidamide is well tolerated orally, toxicity is controlled, and during the maintenance treatment of chidamide, the patient’s quality of life is improved, avoiding frequent hospitalizations and saving costs. At present, the patient is followed up regularly and all indicators are within the normal range. In the treatment of chidamide for this SPTCL patient, we have gained experience with the following clinical applications: (1) chidamide can be used as an effective agent to induce long-term remission for rare SPTCL; (2) chidamide not only has a good synergistic effect with chemotherapy drugs, but also has a good maintenance effect with monotherapy; (3) AE can be tolerated; (4) patient compliance is good, and oral chidamide may be considered for some patients who are not suitable for intravenous therapy or hospitalization. This report presents new options and opportunities for the treatment of SPTCL patients, but a more extensive clinical trial would be needed to fully validate it.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Ethics Committee of Lanzhou university second hospital (2020A-118).
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
