Abstract
Islet transplantation is limited by the difficulties in isolating the pancreatic islets from the cadaveric donor and maintaining them in culture. To increase islet viability and function after isolation, here we present a novel culture technique involving an histone deacetylase inhibitor (HDACi) to rejuvenate the isolated islets. Pancreatic islets were isolated from Sprague-Dawley (SD) rats and one group (FIs; freshly isolated islets) was used after overnight culture and the other group (RIs; rejuvenated islet) was subjected to rejuvenation culture procedure, which is composed of three discrete steps including degranulation, chromatin remodeling, and regranulation. FIs and RIs were compared with regard to intracellular insulin content, glucose-stimulated insulin secretion (GSIS) capacity, gene expression profile, viability and apoptosis rate under oxidative stresses, and the engraftment efficacy in the xenogeneic islet transplantation models. RIs have been shown to have 1.9 ± 0.28- and 1.7 ± 0.31-fold greater intracellular insulin content and GSIS capacity, respectively, than FIs. HDACi increased overall histone acetylation levels, with inducing increased expression of many genes including insulin 1, insulin 2, GLUT2, and Ogg1. This enhanced islet capacity resulted in more resistance against oxidative stresses and increase of the engraftment efficacy shown by reduction of twofold marginal mass of islets in xenogeneic transplantation model. In conclusion, a novel rejuvenating culture technique using HDACi as chromatin remodeling agents improved the function and viability of the freshly isolated islets, contributing to the reduction of islet mass for the control of hyperglycemia in islet transplantation.
Keywords
Introduction
Diabetes is one of the most serious health problems worldwide, affecting nearly 200 million people and predicted to increase to 400 million by 2030 (51). Type 1 diabetes consists of about 5% of all diabetic patients and is characterized by β-cell destruction from autoimmune attacks (24). Type 2 diabetes is common form of diabetes, and peripheral insulin resistance and gradual dysfunction of β-cells are the main features of this disease (7,47). Although the two forms of diabetes are different regarding the etiology of the disease, β-cell loss, whether it is absolute or relative, is a common pathophysiological outcome, and thus, the replacement or regeneration of β-cells has been extensively studied over the past several decades (13,31).
Islet transplantation is a preferred option for type 1 diabetes treatment, particularly when patients easily succumb to severe hypoglycemic episodes (45,46). However, donor shortage remains a serious limitation of this therapeutic regimen to broad usage. Moreover, islet isolation techniques are so imperfect that they fail in as many as half of the isolation attempts from cadaveric donor pancreata, thereby worsening the shortage of the transplantable islet mass (32). Therefore, various methods have been studied over the years to circumvent this problem. First, reagents required for islet isolation process such as collagenase have continuously improved with respect to purity and potency (23). Various parameters affecting islet quality and quantity such as cold ischemia time, organ storage solution, and collagenase incubation time have been tested to optimize islet yield (19,26,37). Second, several surrogate β-cells have been developed (6,15,28). Third, pancreatic progenitor cells have been directed toward islet-like clusters (3,42). Fourth, procedures of the differentiation of adult and embryonic stem cells into insulin-producing cells have been extensively studied (4,5,14,25). Finally, many researchers have tried to increase the viability of isolated islets through beneficial gene delivery (43,48,54) or with the treatment of antiapoptotic agents (10,11) or prosurvival agents (53).
Given the hot debate on the β-cell source of adult pancreatic islets on whether they are from the proliferation of preexisting β-cells or from the neogenesis of the precursor or progenitor cells (8,52), the clinically relevant β-cells from either proliferation of adult islets or differentiation of progenitor cells await further research. Likewise, differentiation of islet-like cells from stem cells has put forth serious ethical considerations in the case of embryonic stem cells and very low efficiency even when they are transplanted into diabetic animals in the case of islet-like cells derived from adult stem cells. Therefore, it is not surprising that, among various strategies, an improved collagenase blend is the only technique that has been applied to clinic, while others remain at animal experimental stages.
In the present study, we have developed a new culture technique, which is comprised of the following three steps: degranulation, chromatin remodeling, and regranulation. Using this novel culture technique, we were able to increase islet function with regard to intracellular insulin content, glucose-stimulated insulin secretion (GSIS) capacity, and resistance to oxidative stresses such as H2O2. Moreover, in the islet transplantation model, we were able to show that rejuvenated islets are at least twofold superior to freshly isolated islets regarding the correction of high blood glucose levels induced by streptotozocin (STZ).
Materials and Methods
Animals and Reagents
Nude mice (Balb/c slc nu-nu) and outbred Sprague-Dawley (SD) rats were purchased from Central Laboratory Animal Inc. (Seoul, Korea). STZ, trichostatin A (TSA), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), valproic acid (VA), and all other chemicals were obtained from Sigma-Aldrich Korea Co. (Seoul, Korea) unless otherwise indicated. Hank's balanced salt solution (HBSS), CMRL 1066 media, fetal bovine serum (FBS), antibiotics (penicillin and streptomycin), and other cell culture wares were purchased from Invitrogen (Carlsbad, CA, USA). Fibrin gel kit (Greenplast®) was purchased from Green Cross Corp. (Kyunggi-do, Korea). All animal experiments were performed in accordance with the protocols approved by the Korea University Institutional Animal Care and Experimentation Committee.
Islet Isolation
Pancreatic islets were isolated from SD rats weighing 250–280 g in exactly the same way as previously described (30).
Islet Culture
The isolated islets were incubated in 10% FBS-supplemented CMRL 1640 medium (complete media) overnight in a bacterial dish to prevent attachment. To minimize islet batch variation, these islets were divided into two portions and one was used as freshly isolated islets (FIs), and the other was used as rejuvenated islets (RIs) after rejuvenation procedure as described below. Briefly, during the degranulation step, islets were incubated in complete media supplemented with 15 mM glucose and 30 mM KCl to promote intracellular insulin release for 48 h. Then, during the chromatin remodeling step, media was switched to 1% FBS-supplemented CMRL 1640 media and islets were treated with 100 nM TSA for 24 h. In some cases, TSA was replaced by 1 mM of VA. During the regranulation step, islets were washed with complete media twice and then incubated in complete media for an additional 48 h.
Glucose-Stimulated Insulin Secretion (GSIS) and Insulin Content
Isolated islets were either incubated overnight in regular media or cultured as above. Batches of islets with the similar size distribution ranging from 150 to 200 μm in diameter (10 islets) were dispensed in Eppendorf tubes. GSIS was measured by 1-h static incubation in Kreb's Ringer bicarbonate (KRB) buffer containing 0, 5.6, and 16.7 mM glucose sequentially. Finally, islets were sonicated in acidic alcohol, and intracellular insulin was extracted overnight at 4°C. Insulin was measured in duplicate by an enzyme-linked immunosorbent assay (ELISA) according to the manufacturer's protocol (Mercodia AB, Uppsala, Sweden).
Western Blot and Histone Deacetylase (HDAC) Assay
Nuclear extracts from 1,000 islets sampled at each step during the rejuvenation culture period were prepared using NE-PER Nuclear and Cytoplasmic Extraction Reagents (Pierce Biotechnology, Inc., Rockford, IL, USA). HDAC activity was measured using colorimetric HDAC assay kit (Biovision, Inc., Mountain View, CA, USA). For Western blot analyses, 50 μg of nuclear extracts were resolved by sodium dodecyl sulfate (SDS)-polyacrylamide electrophoresis and transferred to a polyvinylidene fluoride membrane. The membrane was probed with anti-acetyl histone 3 and acetyl-histone 4 antibodies (Cell Signaling Technology, Inc., Danvers, MA, USA) and was followed by a peroxidase-conjugated secondary antibody. The same membrane was re-probed with anti-H3 and H4 to normalize protein amounts. Bands were visualized by enhanced chemiluminescence plus (ECL plus, GE healthcare Life Sciences, Inc., Piscataway, NJ, USA) and the LAS-3000 image analyzer (Fujifilm Corp., Tokyo, Japan).
Semiquantitative Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Total RNAs were isolated from FIs or RIs using Trizol reagent (Invitrogen) according to the manufacturer's protocols. Two micrograms of total RNAs was converted to cDNAs using Superscript II reverse transcriptase system, and resulting cDNAs were diluted twofold by adding the same volume of H2O. PCR was performed using 0.5 μl of cDNAs, 1.25 U of Ex-Taq polymerase (Takara, Japan), and 0.3 μM of particular gene-specific primer pairs. PCR cycles were chosen to ensure that PCR product was in linear amplification range with cycle number. PCR products were separated in 1–2% agarose gel and photodocumented using GelDoc system (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Bands from RT-PCR were digitized using Scion image analysis software (Scion Corp., Frederick, MD, USA) and the mean fold increase in values from at least three independent experiments was shown. Primer sequences are available upon request.
Oxidative Stresses
One thousand islets were made as single cell suspension by incubation in Accutase (Thermo Electron, Louisville, CO, USA) for 10 min at 37°C, gently triturated, and then followed by filtration using 70-μm cell strainer (SPL Life Sciences, Gyeonggi-do, Korea). Islet cells (2 × 105 per tube) were dispensed in a fluorescence activated cell sorting (FACS) tube and treated with different concentrations of H2O2 for 30 min at 37°C. Cells were then washed with complete media and the level of apoptosis was determined by staining with annexin-fluorescein isothiocyanate (FITC) and propidium iodide (PI) using BD Pharmigen FITC Annexin V Apoptosis Detection Kit according to the manufacturer's protocol. The stained cells were acquired with a FACSCanto and analyzed by FACSDiva software. The proportions of Annexin V alone (early apoptotic cells) and both Annexin V+ PI + cells (late apoptotic cells) were determined.
Islet Size Calculation
Batches of FIs and RIs were photomicrographed under 40× magnification using an inverted microscope equipped with a digital camera (Olympus CK-2, Japan), and then the images were printed out. The islet equivalent (IEQ) number was then calculated by classifying islets according to their diameter, and IEQ was using the Ricordi algorithm (44).
Diabetes Induction
Nude mice weighing 20–25 g were rendered diabetic by injecting STZ (200 mg/kg, IP) after a 12-h fasting period. STZ was freshly made in 0.04 M citric acid buffer (pH 4.0). Diabetic induction was confirmed by monitoring the tail blood glucose level to be >300 mg/dl for at least 2 consecutive days using an automatic glucometer (CareSens II, I Sense Co., Seoul, Korea).
Islet Transplantation
Varying amounts of islets (50–150 IEQ) were embedded in fibrin gels and transplanted into kidney subcapsule of diabetic nude mice as previously described (30). In our hands, survival rates of nude mice transplantation was >95%.
Terminal Deoxyuridine Nick-end Labeling (TUNEL) Assay
To determine apoptotic cells within the islet grafts, the left kidneys harboring the islet graft (75 IEQ FIs or RIs per animal, n = 2 at each time point) were harvested at day 1, 3, 7, 14, and 30 post-islet transplantation. A tissue section (every 10th section throughout the sample, n = 3) was prepared and immunostained with ApopTag® plus peroxidase in situ apoptosis detection kit according to the manufacturer's instruction (Roche Diagnostics, Inc., Minneapolis, MN). At minimum, eight independent areas (100 × 100 μm) were randomly chosen per kidney, and the apoptotic cells were measured in blind fashion. Total cell counts exceeded the minimal 365 cells. To determine viable β-cells within the graft, the adjacent section (every 10th section) was immunostained with an anti-insulin antibody as above. Cell counting was performed exactly the same way as above.
Statistics
All data are expressed as means ± SD. A difference was considered significant with a value of p < 0.05, using the Student's t-test or one-way analysis of the variance (ANOVA) followed by Bonferroni's post hoc test when multiple comparisons were made between groups.
Results
Development of Islet Rejuvenation Method
Islet β-cells have been shown to have an insulin receptor, insulin receptor substrate, and various downstream signaling components such as phosphoinositide 3-OH kinase, AKT/PKB, and so on, and insulin itself is deemed as an autocrine and/or paracrine survival factor for β-cells (29). Additionally, previous studies demonstrated that short-term exposure of the isolated islets to high glucose led to decreased autophagy and less sensitivity to subsequent oxidative stresses (39,40). Therefore, in the degranulation step (step 1), we wanted to increase islet viability by releasing intracellular insulin using well-known insulin secretagogues, glucose, and high concentrations of K+ ion rather than adding exogenous insulin to the culture media. Then, TSA, a histone deacetylase inhibitor (HDACi), was treated to induce chromatin remodeling (step 2) during which the relative lysine acetylation levels at the histone 3 and 4 are expected to increase, thereby inducing expression of the repressed subset of genes. Among these gene sets, several important genes for β-cell survival and function might be expected to increase because the isolated islets should recover from stresses during the harsh isolation process. Finally, after removing residual HDACi, islets were incubated in regular serum-containing media in which insulin biosynthesis and storage in the secretory vesicles are expected to occur (step 3). Because islets were cultured in suspension during the entire period, they became more and more rounded with time and very often fused with each other, giving rise to large multilobed giant islets (Fig. 1). As seen by the scanning electron microscope, their surfaces were very smooth and numerous microvilli were observed (supplementary Fig. S1A; available at http://blog.naver.com/ohbosle/100115366645). Interestingly, even cells in the innermost layer appeared to be healthy without any sign of necrosis as shown in representative semithin section and transmission electron micrograph images (supplementary Fig. S1B and S1C; available at http://blog.naver.com/ohbosle/100115366645). By contrast, central necrosis was frequently observed in islets cultured for the same period without the rejuvenation procedure (supplementary Fig. S1D; available at http://blog.naver.com/ohbosle/100115366645).
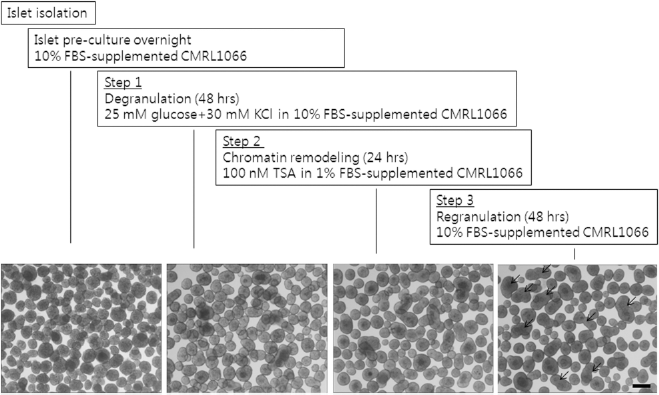
Scheme of the rejuvenating process and the gross morphology of islets during culture period. Isolated islets from Sprague-Dawley (SD) rats were preincubated in regular media overnight, and suspension was cultured according to rejuvenating method. The islets were incubated in regular media supplemented with additional 15 mM of glucose and 30 mM of KCl for 48 h (step 1), treated with histone deacetylase inhibitor [HDACi; 100 nM trichostatin A (TSA)] for 24 h (step 2), and then incubated with regular media for 48 h (step 3). Islets were gathered into the center of the dish by gentle swirling and photomicrographs were taken at 40× magnification under an inverted microscope. Arrows indicate multilobed islets, which were formed by a fusion of a few islets during the culture period. Scale bar: 250 μm.
Rejuvenated Islets Had Enhanced GSIS Capacity and Intracellular Insulin Content
As we measured intracellular insulin content and GSIS from the batch of size-matched islets, FIs had 70.8 ± 10.4 ng/IEQ of intracellular insulin and showed a 2.0-fold insulin secretion at 16.7 mM glucose over 5.6 mM glucose concentration (1.76 ± 0.41 ng/IEQ at 16.7 mM glucose vs. 0.87 ± 0.08 ng/IEQ at 5.6 mM glucose). However, RIs had 136.3 ± 8.81 ng/IEQ of intracellular insulin and 3.4-fold insulin response to glucose stimuli (3.72 ± 0.92 ng/IEQ at 16.7 mM glucose vs. 1.09 ± 0.30 ng/IEQ at 5.6 mM glucose). This represents 1.9 ± 0.28-fold more insulin in intracellular spaces and a 1.7 ± 0.31-fold better insulin response to glucose stimuli of RIs compared with FIs. In control experiments, when islets were cultured for the same period as the rejuvenation procedure with regular media changes, intracellular insulin increased to 104.8 ± 22.3 ng/IEQ, but their insulin response to glucose stimuli was completely blunted (0.5 ± 0.13 ng/IEQ at 16.7 mM glucose vs. 0.43 ± 0.13 ng/IEQ at 5.6 mM glucose), consistent with previous several reports (21,35,41). When the intracellular insulin content was measured during the course of the rejuvenation procedure, it decreased to 60% of the level of FIs after the step 1, but then gradually increased, reaching to 1.9 ± 0.28-fold after step 3. Importantly, valproic acid (1 mM), another HDACi, had similar effects on intracellular insulin content and GSIS (Fig. 2). These results indicate that our culture method would rejuvenate isolated islets as evidenced by increased intracellular insulin content and enhanced GSIS capacity. Moreover, through detailed protocols such as omitting the first step or second step, we could dissect out the effect of each step of the rejuvenation procedure on intracellular insulin and GSIS. The degranulation step contributed to the increase of intracellular insulin content and HDACi treatment contributed to the enhanced insulin response to high glucose stimulation (supplementary Fig. S2, available at http://blog.naver.com/ohbosle/100115366645).
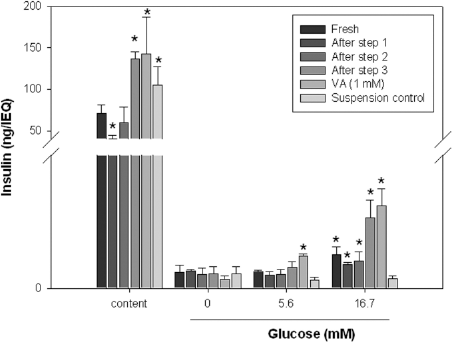
Intracellular insulin content and glucose stimulated insulin secretion (GSIS) of freshly isolated islets (FIs) and rejuvenated islets (RIs). The isolated islets were divided into multiple portions, and intracellular insulin content and GSIS were measured at indicated time points during the rejuvenation procedure. In all experiments, TSA was used as HDACi except for valproic acid (VA, 1 mM). A suspension control, which was cultured for the same period with regular media changes, was used as a control. Data are the mean ± SD of at least three independent experiments, and the statistical significance (p < 0.05) between control and test groups of intracellular insulin content or insulin secretion between 0 and 16.7 mM glucose was tested by Student's t-test with Bonferroni's correction.
Rejuvenated Islets Exhibited Increased Expression of Important Genes in β-Cell Survival and Function
Treatment of HDACi and observation of the increased biochemical capacity of RIs prompted us to hypothesize that the rejuvenation procedure might affect gene expression profiles of isolated islets leading to the enhancement of islet function. Prior to analyzing gene expression profiles, we measured histone acetylation status during rejuvenation using Western blot analyses. Indeed, during the chromatin remodeling step, the levels of histone acetylation revealed as acetylation both at Lys9 in histone 3 and Lys8 in histone 4 increased, while during step 3, their levels were completely normalized (supplementary Fig. S3; available at http://blog.naver.com/ohbosle/100115366645). Furthermore, as we directly measured HDAC activity in nuclear extracts obtained from islets at step 2, its activity is 50.5% of those in FIs (12.33 ± 3.71 vs. 24.42 ± 3.41 μmol equivalent of Boc-Lys-pNA as HDAC substrate, p < 0.05).
When RIs and FIs were subjected to semiquantitative RT-PCR analyses, important genes for β-cell function such as v-maf musculoaponeurotic fibrosarcoma oncogene homolog A (MafA), neurogenic differentiation factor 1 (NeuroD1), proprotein convertase subtilisin/kexin type 1 (Pcsk1), and pancreatic and duodenal homeobox 1 (PDX-1) were not increased (data not shown), but insulin 1 and 2, glucose transporter type 2 (GLUT2), and several genes for DNA repair were significantly increased by 1.18- to 2.79-fold in RIs compared with FIs. By contrast, β-actin was unaffected (Fig. 3A). Interestingly, all of the HDACs including class I (HDAC 1, 2, 3, and 4) and class II (HDAC 6, 7, 8, and 10) were expressed in isolated islets, and the expression of HDAC 4 and 7 was selectively decreased by TSA (Fig. 3B). These results indicated that HDACi treatment during the rejuvenation process led to chromatin remodeling at the various loci throughout the whole genome with some, but not all essential genes for β-cell survival and function was increased. Furthermore, wide expression of HDACs and selective regulation of HDAC 4 and 7 by TSA strongly suggested that HDACs would be important for maintenance and/or regulation of islet function.
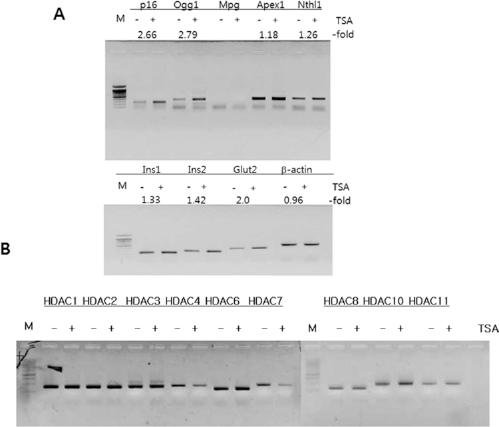
Gene expression pattern between FIs and RIs. The isolated islets were divided into two groups (FIs and RIs) and the gene expression pattern was examined by semiquantitative RT-PCR analyses. Essential genes for beta cell function (Insulin-1 and -2 and GLUT2) or DNA repair (Ogg1, APEX, and Nthl1) were significantly increased in the RI group compared with the FI group, whereas β-actin was similar between the two groups (A). Gene expression pattern of class I and class II HDACs was also measured (B). GLUT2, glucose transporter 2; Ogg1, 8-oxoguanine glycosylase; APEX, AP DNA endonuclease; Nthl1, endonuclease III-like protein 1; MPG, N-methylpurine-DNA glycosylase.
Rejuvenated Islets Were More Resistant Against Oxidative Stresses Than Freshly Isolated Islets
Islet β-cells are more vulnerable to oxidative stress than other cell types because levels of antioxidative enzymes handling this insult are relatively low (33). Therefore, prior to confirming the enhanced islet function of RIs in vivo by transplantation, we set out to examine islet viability and the apoptosis rate under oxidative stresses in vitro. To examine that in a quantitative way, we employed single cell suspension and FACS analysis as reported in the study (38). When FIs were treated with different concentration of H2O2, apoptosis rate of FIs significantly increased from 8.4 ± 0.4% to 30.9 ± 2.1% and 39.6 ± 1.9% upon 0, 250, and 500 μM of H2O2 treatment. By contrast, the corresponding values of RIs were 6.1 ± 0.2%, 21.3+1.2%, and 31.1 ± 1.9% at 0, 250, and 500 μM of H2O2, respectively. More importantly, only 14.8 ± 0.9% of cells from FIs were viable (Annexin V- PI-) and over half of other cells (53.3 ± 2.8%) were already experiencing apoptotic process at 250 μM H2O2 concentration as evidenced by Annexin V positivity (Fig. 4). Collectively, these results clearly demonstrated that FIs are more vulnerable to similar strength of oxidative stress, thus undergoing earlier apoptosis compared with RIs.

Apoptosis of FIs and RIs under oxidative stress. The isolated islets were divided into two groups and were processed as RIs and FIs according to the methods described in Materials and Methods. After being treated with 0, 250, and 500 μM H2O2 for 30 min, apoptosis of each group of islet cells was measured by propidium and Annexin V staining. Data are representative of three independent experiments.
Rejuvenated Islets Were at Least Twofold Superior to Freshly Isolated Islets with Respect to the Correction of Hyperglycemia Induced by STZ in Transplantation Model
To evaluate the glycemic control function of RIs in vivo, we used an islet xenotransplantation model in which islets from SD rats were transplanted into STZ-induced diabetic nude mice. Consistent with our recent data (30), we found that marginal islet mass (defined as the minimal mass of islets expressed as the IEQ needed to correct hyperglycemia in 50% of the STZ-induced diabetic recipients) in rat to diabetic nude mouse model was 100 IEQ of FIs (Fig. 5A). By contrast, in the cases of RIs, when 100 IEQ of RIs were transplanted into diabetic nude mice, all mice (5 out of 5 mice) restored normoglycemia within 1 week of the transplantation. When even half doses of RIs (50 IEQ) were transplanted, half of the mice (5 out of 10 mice) became normoglycemic (Fig. 5B), indicating that the marginal islet mass of SD RIs is 50 IEQ, and thus RIs are at least twofold superior to FIs with respect to glycemic control. We did another independent set of experiments and obtained consistent results even with low quality of isolated islets, of which case marginal mass of FIs was greater than 100 IEQ (supplementary Fig. S4; available at http://blog.naver.com/ohbosle/100115366645).

Blood glucose levels of the diabetic nude mice transplanted with FIs or RIs from SD rats. Diabetic nude mice were transplanted with 150 and 100 IEQ FIs or 100 and 50 IEQ RIs of SD rats at day 0, and their blood glucose levels were monitored for ~3 months after islet transplantation. Every symbol in all figures represents an individual animal that underwent islet transplantation. Asterisks denote individual animals that experienced persistent hyperglycemia, and eventually died during the follow-up periods.
Rejuvenatation of Islets Increased β-Cell Number with a Minimal Sign of Apoptosis
Previous animal and human studies demonstrated that about 50% of the transplanted islets were lost via apoptosis shortly after transplantation due to hypoxia, oxidative stress, or inflammation (9,12). Therefore, to compare the apoptotic rate during an early time period after the islet transplantation, we measured the apoptotic rate at day 1, 3, 7, 14, and 30 after transplantation of RIs or FIs using the TUNEL assay on a tissue section. As shown in Figure 6, the apoptotic rates of FIs at day 1, 3, 7, 14, and 30 posttransplantation were 1.78 ± 0.03, 0, 0.76 ± 0.03, 1.00 ± 0.04, and 4.05 ± 0.06 × 10−4 cells/μm2. Those of RIs were significantly lower at all time points, with a maximal difference of 8.1-fold (4.05 ± 0.06 × 10−4 cells/μm2 in FIs vs. 0.33 ± 0.09 × 10−4 cells/μm2 in RIs at day 30). By contrast, viable β-cell counts among the transplanted mass were significantly higher at all time points with a maximal difference of 1.4-fold (59.6 ± 0.89 × 10−4 cells/μm2 in FIs vs. 83.0 ± 2.74 × 10−4 cells/μm2 in RIs) at day 30. These results directly demonstrated that RIs show increased engraftment efficacy with significantly lower apoptotic rates compared with FIs.

Apoptotic cells and beta cells within the islet graft retrieved at indicated times after islet transplantation. Diabetic nude mice were transplanted with 75 IEQ FIs or RIs at day 0. Kidney harboring islets were retrieved at day 1, 3, 7, 14, and 30 and apoptotic cells (A) and β-cells (B) were counted on tissue sections using the Terminal deoxyuridine nick-end labeling (TUNEL) assay and insulin immunostaining, respectively. (C) A representative image showing more insulin* β-cells and less apoptotic cells in the RIs group (c, d) compared with the FIs group (a, b) on the tissue section retrieved at day 30 after islet transplantation. Data are mean ± SD of results from at least eight tissue sections. Scale bar: 50 μm.
Discussion
Islet transplantation is becoming more affordable as an efficacious therapeutic method for type 1 diabetes due to rapid progresses in developing innovative immunosuppressive regimens, making collagenase purer and more potent, and standardizing all procedures for islet transplantation. These achievements allowed the insulin independence rate to reach almost 60% at 36 months after transplantation during the last 5 years (2005–2009) compared to 35% during the last 15 years (1999–2004) (2). However, in successful islet transplantation, multiple donor pancreata are still frequently needed to meet adequate beta cell mass for achieving insulin independence. In this respect, our technique has significant implications because the rejuvenated islets according to the rejuvenation procedure have increased insulin content, GSIS capacity, and oxidative stress resistance and at least twofold superior engraftment efficiency to freshly isolated islets for correcting hyperglycemia of STZ-induced diabetes in xenogeneic islet transplantation model. When our technique was applied to human islets, insulin content and insulin secretion in response to high glucose increased similarly to rat islets and increased expression of GLUT2 was confirmed (supplementary Fig. S5; available at http://blog.naver.com/ohbosle/100115366645). These results strongly suggest that our culture technique is a viable option for human islets and should lead towards achieving the goal of a “one donor–one recipient” human islet transplantation.
HDAC inhibitors have originally been developed as anticancer agents and have been shown to be effective for diverse classes of cancer types (34). Recently, however, these interesting molecules have been reported to have other potent effects such as neuron-reviving activity (16), anti-inflammatory effects (1), and immunomodulatory activity (50). To our knowledge, four recent reports have examined the effect of HDAC inhibitors on islet function (49), survival (27), cyclin D promoter regulation (22), or development of the pancreas (20). In this study, we found an entirely novel aspect of HDAC inhibitors' effect on the isolated islets. During isolation, the islets experienced severe hypoxia, anoikis, and mechanical sheering due to severing of blood vessels, detachment from the extracellular matrix, and frequent shaking, respectively. This harsh condition inevitably leads to loss of islet viability, islet function, and even global gene expression changes. Thus, we hypothesized that many genes important for islet survival and function should increase during the recovery period and HDACi treatments may promote these processes. Indeed, we found that insulin-1, -2, GLUT2, and DNA repair enzymes (Ogg1, APEX, and Nthl1) were significantly increased compared with untreated control islets (Fig. 3). For more systematic analyses, total RNAs from FIs versus RIs were hybridized to Agilent rat whole genome 44K gene chip (41,012 transcript sequence genes and 1,841 controls), and about 1,000 genes whose expressions were differentially regulated by HDACi were found. Consistent with previous many data (18,36), differential expressions of most of the genes are marginal (<1.5-fold difference), with only 337 genes being increased or decreased greater than twofold compared with untreated controls (supplementary Table 1; available at http://blog.naver.com/ohbosle/100115366645). Among these differential genes, the several selected genes including glucagon receptor were indeed significantly increased by HDACi in real-time PCR analyses (supplementary Fig. S6; available at http://blog.naver.com/ohbosle/100115366645). Surprisingly, a very recent report showed that overexpression of glucagon receptors on β-cells enhanced insulin content and insulin secretion in response to glucose by 1.5- and 3.9-fold, respectively, compared with islets from nontransgenic littermates (17). This result is fully consistent with our result (Fig. 2; also supplementary Fig. S2; available at http://blog.naver.com/ohbosle/100115366645) and strongly suggests that HDACi treatment has selective effects on the beneficial genes for islet function and survival. Further mechanistic studies warrant future work.
Although the mechanism underlying rejuvenation of the isolated islets is only speculative at this point, we were able to show that RIs are more resistant to oxidative stress and are at least twofold superior to FIs with respect to engraftment efficacy in xenogeneic islet transplantation model. Therefore, we anticipate that our culture protocol should be modified to fulfill the best rejuvenation of the isolated human islets (HDACi concentration and treatment time) and be easily incorporated into clinical settings.
Footnotes
Acknowledgments
The authors thank Dr. Kwang-Won Kim at the Samsung Medical Center for his continuous support and helpful discussion on human islets. This work was supported by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea (Project No. A040004) and a grant of the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare & Family Affairs, Republic of Korea. (Project No. A092258).
