Abstract
As angiogenesis and vasculogenesis involve the complex network structures of various types of cells, extracellular matrix components, and cytokines, it is still difficult to exactly mimic the microenvironment of vascularization in vivo. In our study, we constructed a complex containing highly proliferative fibroblasts that can secrete extracellular matrix components and growth factors to chemotaxize endothelial progenitor cells (EPCs) in an attempt to create an ideal microenvironment for quick vascularization. Amniotic membrane microparticles (mAM) rich in type IV collagen (COL IV) and laminin (LN) were prepared, and human dermal fibroblasts (HDF) were infected with lentivirus (LV) of overexpression of SDF-1α to construct SDF-1αovHDF. Using the rotary cell culture system (RCCS), mAM was loaded with HDF or SDF-1αovHDF to construct HDF-mAM and SDF-1αovHDF-mAM complexes. The complexes were able to secrete various types of active peptides (IL-6, IL-8, TGF-β, and bFGF) during in vitro culture. In addition, SDF-1αovHDF-mAM complex highly expressed SDF-1α. Transwell assay showed SDF-1αovHDF-mAM complex had an apparent chemotactic effect on EPCs. Transplantation of complexes onto full-thickness skin defects of C57BL mice further demonstrated that SDF-1α expression and the number of peripheral EPCs at days 3, 5, and 7 in the SDF-1αovHDF-mAM group were significantly higher than that in other groups (p < 0.01). The local microvascular density at day 10 of transplantation showed that the microvascular density in the SDF-1αovHDF-mAM group was significantly higher than that in HDF-mAM group (p < 0.01). In conclusion, HDF-mAM had a strong proliferative activity and could be used to create a sound microenvironment for quick vascularization by secreting multiple cytokines and extracellular matrix components. Overexpression of SDF-1α could chemotaxize EPCs to reach local wounds, thus further accelerating angiogenesis in the transplant site. The technique described may prove to be a new model for accelerating vascularization of tissue and organ transplants and chronic ischemic wounds.
Keywords
Introduction
How to promote vascularization has become a hotspot of research in the evolving field of tissue engineering and wound healing (5). Researchers have attempted to transplant various growth factors such as vascular endothelial growth factor (VEGF), acidic fibroblast growth factor (aFGF), and platelet-derived growth factor (PDGF) to local wounds to promote vascularization through the gene transfection technique or bridging and slow-release technique (10,18,24). Some researchers tried using extracellular matrix component decoration or mixed culture of fibroblasts and endothelial cells in vitro to construct a vascularized skin substitute to promote vascularization of the graft (4,22). However, this method is complex, requiring high technical conditions. Recent studies have demonstrated that endothelial progenitor cells (EPCs) existing in circulating peripheral blood are a key participant in angiogenesis, not only directly participating in stem cell angiogenesis of the ischemic tissue but participating in local angiogenesis by differentiating into endothelial cells (2). However, the use of EPCs is largely limited due to unavailability of a mature method of isolating and culturing autogenous EPCs. The latest research has discovered that stromal cell-derived factor-1α (SDF-1α), as a powerful chemotactic agent of EPCs, can mobilize EPCs in bone marrow to enter peripheral blood within a short time by binding with the specific receptor CXCR4 on the surface of EPCs, and mediate the migration and homing of EPCs in peripheral blood to the locality of the ischemic tissue, thus participating in angiogenesis and reendothelialization of the injured vessel (23).
Although the above method can to some extent promote vascularization of the graft or tissue, there are still some limitations. As angiogenesis and vasculogenesis involve complex network structures including various cell types such as endothelial cells, EPCs, and fibroblasts, extracellular matrixes such as laminin (LN) and type IV collagen (COL IV), and growth factors such as aFGF and VEGF, the effect of a particular single intervention factor is limited (9). Therefore, simulation of the complex microenvironment of local wound vascularization would be of great significance in accelerating angiogenesis and vasculogenesis.
In the present study, we developed amniotic membrane microparticles (mAM) in a 3D culture model to load human dermal fibroblasts (HDF) or SDF-1αovHDF to construct a highly proliferative HDF-mAM or SDF-1αovHDF-mAM complex to reach local wounds through posttransplant secretion of SFD-1α to chemotaxize EPCs. At the same time, fibroblasts secreted several angiogenetic growth factors and extracellular matrix components to create a sound microenvironment for angiogenesis, thus markedly accelerating local vascularization of the graft.
Materials and Methods
Preparation of mAM
The study protocol was approved by the Ethics Committee of Changhai Hospital. mAM and the protocol were prepared as described in our previous study (7). Briefly, placenta tissues (nine fetuses: six male and three female) were obtained from parturients who underwent Cesarean section after obtainment of informed consent, and all donors were serologically negative for HIV, HBV, HCV, and syphilis. The amniotic membrane was separated from the underlying chorion by blunt dissection. Epithelial cells and fibroblasts on the amniotic membrane were removed by repeated freezing and thawing in combination with DNase (1 mg/ml; Gibco, Gaithersburg, MD, USA) digestion. Then amniotic membrane (AM) patches were homogenized into microparticles with a macrohomogenizer (Tuohejidian, Shanghai, China), freeze-dried under a closed vacuum condition, and finally filtrated through a metal mesh filter (Yibaiju, Shanghai, China) to obtain 300-600-μm microparticles.
Fibroblast Culture and Transfection with SDF-1α Gene
SDF-1αovLV was constructed by Shanghai Genechem Co., Ltd. (Shanghai, China). Briefly, using the cDNA library and PCR method, human target gene SDF-1α was obtained, and the obtained target gene was fused with the vector pGC-FU (Genechem) to obtain recombinant plasmids. The target plasmids were transfected to 293T (Genchen) cells. The multiplicity of infection (MOI) was 20. After real-time PCR analysis of the Ct value and the amount of expression in the samples infected with different concentrations of the virus, the titer of the virus was finally set at 2.00E + 8 TU/ml.
The normal human foreskin was isolated after obtainment of informed consent and cultured for fibroblasts as previously described (1). The fibroblasts (four to six passages) were transfected with green fluorescent protein (GFP)-labeled SDF-1αovLV (lentivirus) or GFP alone to obtain GFP-labeled SDF-1αovHDF (human dermal fibroblasts) or GFP-labeled HDF (GFP-HDF). The amount of relative expression of SDF-1α mRNA was detected by realtime RT-PCR and analyzed by the 2-ΔΔCT method using HDF without LV transfection as the reference. The transfection rate was defined as the ratio of the number of GFP cells to the number of total cells and analyzed by flow cytometry.
Detection of the SDF-1α Expression in Transgenic Fibroblasts
SDF-1αovHDF, GFP-HDF, and HDF were seeded onto a 12-well plate (Corning, Corning, NY, USA) and cultured in serum-free DMEM (Gibco) for 24 h until 90% confluence. The supernatant was collected, and the SDF-1α protein content was measured using the SDF-1α ELISA reagent kit (R&D Systems, Minneapolis, MN, USA).
Total RNA was extracted using TRIzol reagent (Invitrogen, Grand Island, NY, USA). All operations were RNase free and reverse-transcribed for real-time PCR (ABI, Waltham, MA, USA) using the following SDF-1α promoter-specific primers: I-F: GAGGAT CCCCGGGTACCGGTCGCCACCATGAACGCCA AGGTCGTG; I-R: TCACCATGGTGGCGACCGGCTT GTTTAAAGCTTTCTCCAGG, under the condition of 95°C 3 min; 40 cycles, 95°C 10 s, 60°C 20 s, 72°C 20 s; 84°C 15 s. SYBR® Green master mix was from TAKARA (Otsu, Shiga, Japan).
Chemotactic Ability of SDF-1αovHDF Upon EPCs
EPCs were isolated according to the procedure as described previously (19). Briefly, mononuclear cells were isolated from the bone marrow of C57BL mouse (n = 5; three male, two female) by density gradient centrifugation and plated on fibronectin-coated plates in endothelial growth medium-2 microvascular (EGM-2MV; Cambrex, East Rutherford, NJ, USA). After being cultured for 48 h, nonadherent cells were removed and the culture maintained for 7-10 days, and the markers of EPCs (CD34/KDR/CD133) were analyzed by immunohistochemistry. The CD34, KDR, CD133 (1:200; all from Santa Cruz, Santa Cruz, CA, USA) were used as the primary antibodies and incubated with the FITC-conjugated secondary antibody (1:300; Santa Cruz).
Three cell groups (HDF, GFP-HDF, and SDF-1αovHDF) were added to the lower chamber of a 24-well Transwell (8 μm pore size; Corning) and cultured to 80% confluence. After changing the serum-free medium, EPCs (1 × 105) were cultured in the upper chamber by keeping the fluid surfaces of the lower and upper chambers at the same level. After 24 h of culture, the upper layer cells of the upper chamber were scraped off and stained with crystal violet (0.1%; Sigma-Aldrich, St. Louis, MO, USA), photographed, and counted using Leica Q-Win image analysis software (Tokyo, Japan). Each experiment was repeated at least three times.
Preparation of HDF-mAM and SDF-1αovHDF-mAM Complexes
Using the 3D culture model, 10 ml 5 × 104 cells/ml HDF or SDF-1αovHDF and 100 mg mAM were added to the Rotary Cell Culture System (RCCS) (Synthecon, Houston, TX, USA) and cultured in a 37°C 5% CO2 incubator with the revolution controlled at 15~20 r/min, and 50% of the medium was changed every other day. The sample was harvested regularly and stained with Hoechst 33342 (0.5 μg/ml; Beyotime, Haimen, Jiangsu, China), and observed under a fluorescence microscope (Leica 3000; Leica, Tokyo, Japan) for adhesion and proliferation of the fibroblasts. Part of the samples were routinely fixed, dehydrated, paraffin waxed, sliced, and hematoxylin and eosin (H&E) (Yuanye, Shanghai, China) stained for microscopic observation, and the remaining part was fixed, dehydrated, and observed under a field emission scanning electron microscope (SEM) (Quanta 450 FEG; FEI, Hillsboro, OR, USA).
Detection of Active Peptides
The medium was sucked out 3 and 5 days after in vitro culture of the HDF-mAM and SDF-1αovHDF-mAM complexes. Cells were washed with phosphate-buffered saline (PBS; Beyotime) and continued to culture for 24 h by addition of the serum-free medium. The supernatant was centrifuged at a high speed and stored at −20°C. Interleukin-6 (IL-6), IL-8, transforming growth factor-β1 (TGF-β1), basic fibroblast growth factor (bFGF), and SDF-1α were determined by ELISA reagent kit (R&D Systems) according to the manufacturer's instructions. Optical density (OD) was measured at 450 nm using the Model 550 Bio-Rad microplate reader (Hercules, CA, USA), and converted into a corresponding concentration according to the standard curve. Laminin (LN) and hyaluronic acid (HA) were detected using a RIA reagent kit (Biotechnology Center of Shanghai Institute of Navy Medical Sciences, Shanghai, China). Radioactivity of the precipitate was detected using a mini-monitor (Morgan, Oxford, UK). Each experiment was repeated at least three times.
Transplantation of HDF-mAM and SDF-1αovHDF-mAM Complexes to Full-Thickness Skin Defects
Institutional approval of the protocol was obtained, and all animal experiments were performed according to the NIH Animal Care and Use Guidelines. C57BL mice of either sex aged 6~8 weeks were anesthetized via intraperitoneal (IP) injection of 1% pentobarbital sodium (Sinopharm, Beijing, China). A full-thickness circular skin defect about 1.4 cm was made on the mouse back. The 96 mice (48 male, 48 female) were equally randomized into four groups (12 male and 12 female in each group): SDF-1αovHDF-mAM complex group, HDF-mAM complex group, mAM group, and blank control group. SDF-1αovHDF-mAM complex, mAM-HD complex, and mAM were smeared on the wound and covered with Vaseline gauze, sewed up intermittently with a 4-0 suture (Jinhuan, Shanghai, China). Photographs were taken regularly from a certain distance after treatment. The wound healing rate was calculated using Image-Pro Plus Software (Media Cybernetics, Rockville, MD, USA).
Wound specimens were taken periodically and stained with H&E. Angiogenesis was observed by immunohistochemistry using CD31 (1:200; Santa Cruz) as the primary antibodies and developed by 3,3′-diaminobenzidine (DAB) (Thermo Fisher Scientific, Rockford, IL, USA). Wound SDF-1α expression was detected by Western blot using anti-SDF-1α (1:1,000; Abcam, Oxford, UK) as the primary antibody and GADPH as the internal reference (1:5,000; Kangchen, Shanghai, China) at days 3, 5, 7, and 10 after treatment. Five mice were used for material analysis each time.
Peripheral blood was drawn at days 3, 5, and 7 after treatment. Both stem cell-associated antigen CD133 (1:100; eBiosciences, San Diego, CA, USA) and endothelial cell-specific antigen VEGFR-2 (1:100; eBiosciences) were used to detect EPCs by flow cytometry (2,3,14). Five mice were used for material analysis each time.
Statistical Analysis
Data statistical analysis was performed by SPSS16.0 (IBM, Armonk, NY, USA), and the results of normal distribution and homogeneity of variance were expressed as mean ± SD. Intergroup comparison was performed by Student's t-test (between two groups), and comparison among multiple groups was performed by analysis of variance (post hoc multiple comparisons were tested using Bonferroni method, among more than two groups). Values of p < 0.05 were considered statistically significant.
Results
Verification of LV-Transfected Fibroblasts
The SDF-1α plasmid sequencing was correct, and the SDF-1α mRNA expression was 1.1-fold in GFP-HDF and 5.3-fold in SDF-1αovHDF compared with that in HDF. There was a significant difference in SDF-1α mRNA expression between SDF-1αovHDF and GFP-HDF groups (p < 0.01). After 24 h of transfection, the amount of SDF-1α expression was about 173 ± 2 pg/105 cells, which was significantly higher than that in GFP-HDF group (28 ± 2 pg/105 cells, p < 0.01) and nontransfection group (32 ± 3 pg/105 cells, p < 0.01).
As shown in Figure 1, the transfected cells grew well and shaped like spindles with no vacuole seen in the nucleus. Fluorescence microscopic observation showed that green fluorescence was concentrated in the cytoplasm in the SDF-1αovHDF group and in the whole cell in the GFP-HDF group (Fig. 1A, B). The possible reason was that GFP is expressed in both the cytoplasm and nucleus, and therefore the fluorescence is distributed throughout the cell, while SDF-1α is a secretory protein that is mainly distributed in the cytoplasm, and therefore the fluorescence is mainly located in the cytoplasm and hardly seen in the nucleus. As the time of photography was different in the two groups (5 days after transfection in the SDFovHDF group and 1 day after transfection in GFP-HDF group), the density of proliferation and the morphology of growth were different between the two groups. Flow cytometry showed that the transfection rate was 94.2% in the GFP-HDF group and 92.7% in the SDF-1αovHDF group.
Fibroblast transfection with LV, MOI = 20. (A, B) and (C, D) Transfected cells grew well and shaped like spindles with no vacuole seen in the nucleus under a fluorescence microscope and a phase contrast microscope. (A) Green fluorescence of the SDF-1αovHDF group is most concentrated in the cytoplasm. The GFP-HDF group (B) shows that the whole cell emits green fluorescence. (E) Detection of the viral transfection rate by flow cytometry. GFP-HDF group, 94.2%; SDF-1αovHDF group, 92.7%.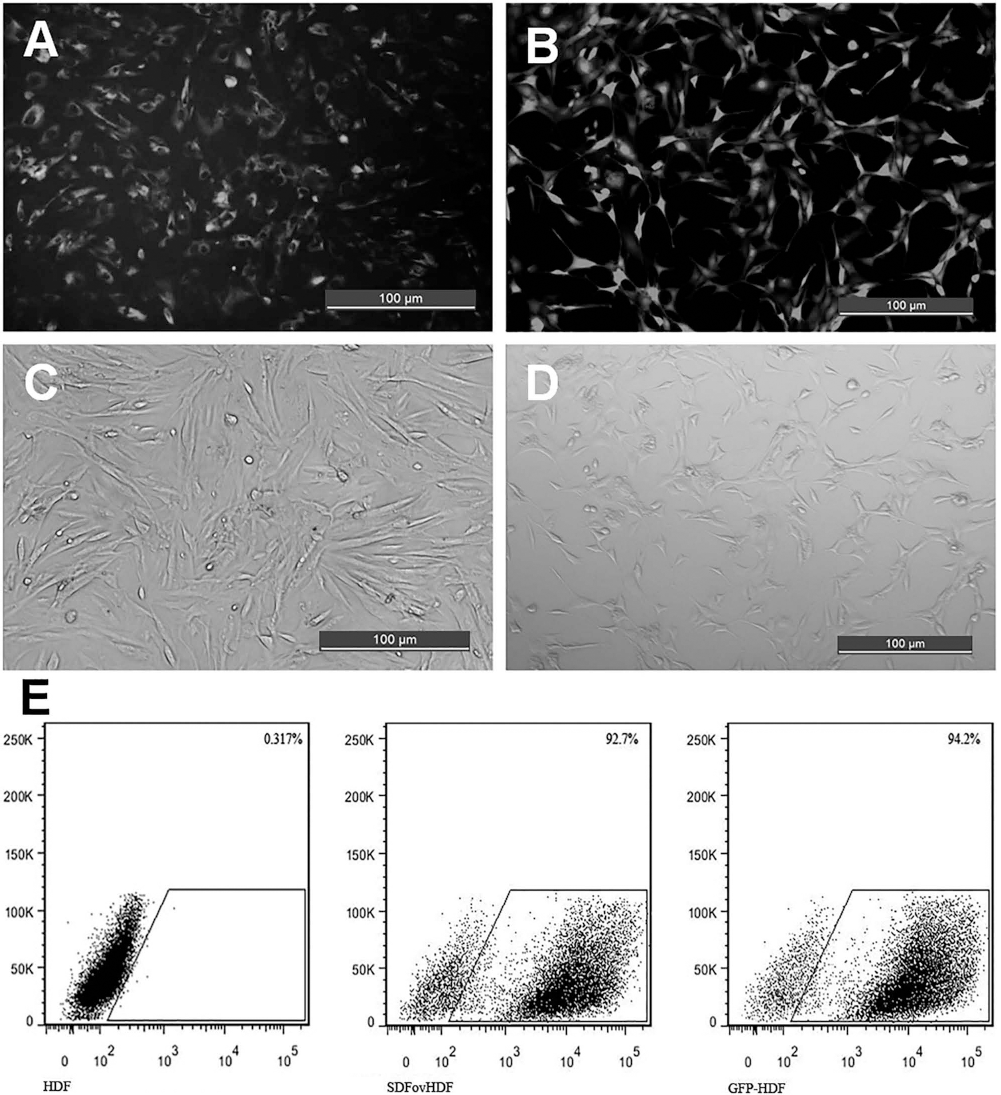
The Chemotactic Effect of SDF-1a ovHDF Upon EPCs
After a 24-h culture, SDF-1αovHDF showed a significant chemotactic effect upon EPCs (Fig. 2). The number of EPCs in the SDF-1αovHDF group was 392 ± 27/HP, which was significantly higher than 204 ± 17 in the GFP-HDF group (p < 0.01) and 181 ± 17 in the HDF group (p < 0.01).
EPC migration in Transwell chambers. Twenty-four hour culture of SDF-1αovHDF\GFP-HDF\HDF in the lower chamber and EPCs in the upper chamber shows that the ability of chemotaxizing EPCs (crystal violet staining) in the SDF-1αovHDF group is significantly stronger than that in the other two groups (*p < 0.01).
HDF Amplification on mAM Surface and Morphological Observation
Cell adhesion was observed on mAM surface 1 h after seeding. From day 1 of culture, cells adhering on the mAM surface were seen spreading gradually, and 3~5 days after culture, cells were seen spreading all over the mAM surface; the microflake-like mAM edges were seen coiling gradually to one side like “microsphere” (Fig. 3). H&E staining and SEM showed that the fibroblasts were evenly distributed on the surface of the microparticles, and most of the cells on the surface were confluent in a single-layer arrangement (Fig. 4).
Cell proliferation on mAM surface. Observation under a phase contrast microscope and a fluorescence microscope after Hoechst 33342 staining. It shows that fibroblasts adhere to the mAM surface and grow in a 3D manner at days 1 and 3 after culture. Cells were seen amplifying gradually and spreading all over the mAM surface at day 5, and the microflake-like mAM edges were seen coiling gradually to one side. H&E staining (A) and SEM (B) observation of SDF-1αovHDF-mAM. H&E staining (A) and SEM (B) show the morphology of fibroblasts on mAM. The fibroblasts were evenly distributed on the surface of the microparticles, and most of the cells were confluent in a single-layer arrangement, and some were seen between the microparticles.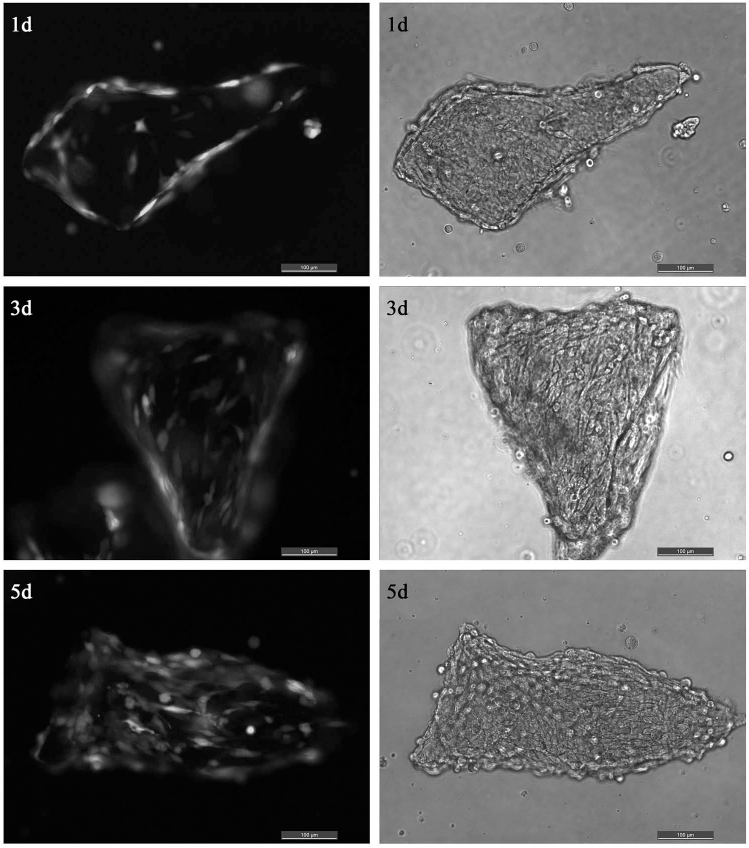

Determination of Active Peptides in the Supernatants of HDF-mAM and SDF-1αovHDF-mAM Complexes
Detection of Active Peptides in the Supernatants of HDF-mAM (IL-6, IL-8, TGF-β1, and bFGF pg/ml w/v, n = 5; LN and HA ng/ml w/v, n = 5)
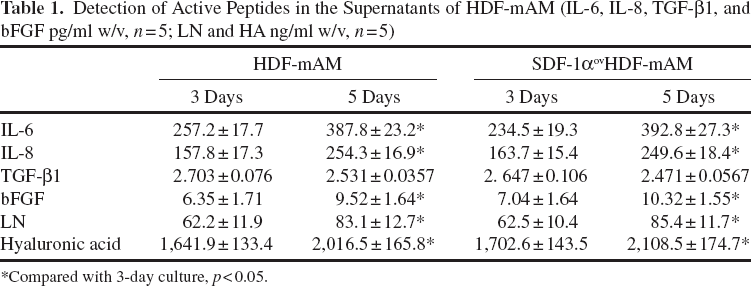
Compared with 3-day culture, p < 0.05.
Complex Transplantation Promotes Vascularization and Wound Healing
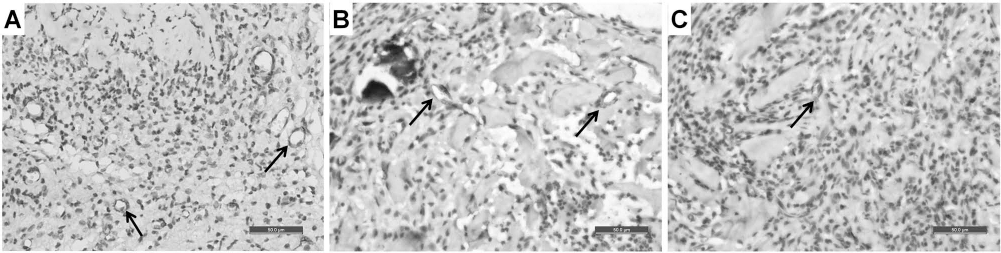
EPCs in peripheral blood detected by flow cytometry showed that the percentage of EPCs in the SDF-1αov HDF-mAM group (0.86 ± 0.09%) was significantly higher than that in the HDF-mAM group (0.34 ± 0.06%, p < 0.01), mAM group (0.18 ± 0.02%, p < 0.01), and blank control group (0.15 ± 0.05%, p < 0.01) after transplantation for 3 days. Then the percent of EPCs gradually decreased in all groups, but it was still higher in the SDF-1αovHDF-mAM group than that of the other groups (p < 0.01) (Fig. 5A). Correspondingly, Western blot of wound specimens showed that SDF-1α expression at days 3, 5, 7, and 10 was obviously higher in the SDF-1αovHDF-mAM group than that in the HDF-mAM and mAM groups, and the amount in the HDF-mAM group was higher than that in the mAM group (Fig. 5B, C). In addition, immunohistochemistry of CD31 showed that vascularization in the SDF-1αovHDF-mAM group was more evident than that in the other groups at day 10, and the local microvascular density in the SDF-1αovHDF-mAM group (33.6 ± 1.2/HP) was significantly higher than that in the HDF-mAM group (21.9 ± 1.1/HP, p < 0.01) and mAM group (13.9 ± 0.7/HP, p < 0.01) (Fig. 6).
The number of peripheral EPCs and the level of SDF-1α expression after transplantation. (A) Flow cytometry shows that the percentage of peripheral EPCs (CD133/VEGFR-2) in SDF-1αovHDF-mAM group was higher than that in the other groups (HDF-mAM, mAM, and blank control group; **p < 0.01) at days 3, 5, and 7 after transplantation. The percentage in the HDF-mAM group was higher than that in the mAM and blank control groups (*p < 0.01). (B, C) Western blot shows that the level of expression in the SDF-1αovHDF-mAM group was significantly higher than that in the HDF-mAM and mAM groups at days 3, 5, 7, and 10 (**p < 0.01). The amount in the HDF-mAM group was higher than that in the mAM group (*p < 0.05). Wound angiogenesis after transplantation. Immunohistochemistry of CD31 developed by DAB showed that the number of the newly formed vessels in the SDF-1αovHDF-mAM group (A) increased significantly. The microvascular density in the SDF-1αovHDF-mAM group was 33.6 ± 1.2/HP at day 10 after transplantation, which was significantly higher than that in the HDF-mAM group (B) (21.9 ± 1.1/HP) and mAM group (C) (13.9 ± 0.7/HP) (p < 0.01).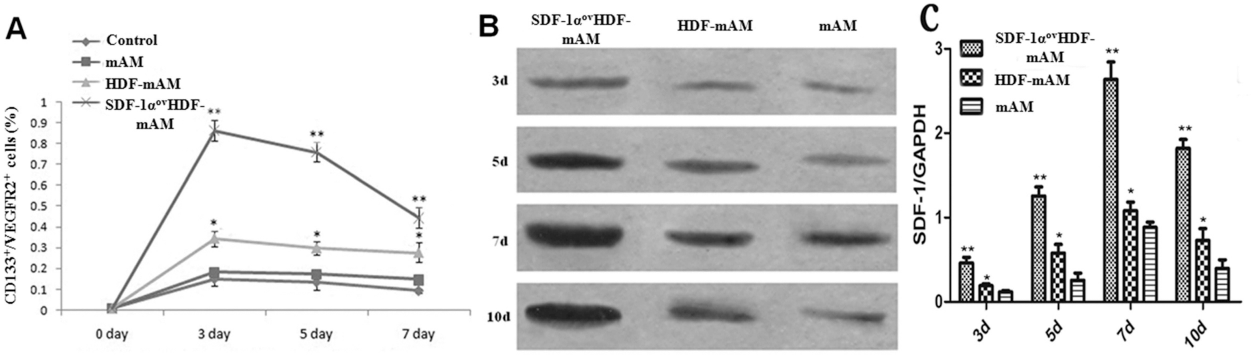
One week after transplantation, the SDF-1αovHDF-mAM and HDF-mAM groups survived well, and the wound was moist and ruddy, while dry scabs formed in the mAM group and the blank control groups. During the process of wound healing, the wound surfaces of the SDF-1αovHDF-mAM and HDF-mAM groups were flat and soft with minimal contraction; the mAM and blank control groups were seen shrunken obviously (Fig. 7). Two weeks after transplantation, the newly formed semitransparent epidermis was seen migrating in the SDF-1αovHDF-mAM group, where the healing time was significantly faster than that in the HDF-mAM and mAM groups (17.25 ± 0.25 days vs. 18.5 ± 0.42 days and 18.87 ± 0.40 days, p <0.05 and p < 0.01). Four weeks after transplantation, H&E staining of the sections showed that the dermal matrix was filled with microparticles in the SDF-1αovHDF-mAM, HDF-mAM, and mAM groups, and epithelial cells had formed six to eight layers versus three to four layers in the blank control group (Fig. 8A-D). In addition, the fibroblasts were seen between the microparticles in the SDF-1αovHDF-mAM and HDF-mAM groups (Fig. 8A, B). To further observe the epithelial morphology and dermal remodeling after wound healing, we obtained some material from SDF-1αovHDF-mAM 2 months after grafting, and H&E staining of the histological sections showed that mAM had grossly degraded, where collagen in the dermis layer was well arranged, and the epithelial layer was composed of three to four layers (Fig. 8E).
Wound healing of full-thickness skin defects. The wounds in the SDF-1αovHDF-mAM and HDF-mAM groups were seen flat with minimal contraction. Wound contraction was obvious in the blank control group, and the healing rate was faster than that in the other groups since day 7 (*p < 0.05). Two weeks after transplantation, the newly formed semitransparent epidermis was seen migrating in the SDF-1αovHDF-mAM group, and the healing rate was faster than that in the HDF-mAM and mAM groups (**p < 0.05). Histological sections after transplantation. Four weeks after transplantation, H&E staining showed that the dermal matrix was filled with microparticles in the SDF-1αovHDF-mAM (A), HDF-mAM (B), and mAM groups (C) with six to eight layers of epithelial cells versus three to four layers in the blank control group (D), and the fibroblasts were seen between the microparticles. Two months after grafting, H&E staining showed that mAM had grossly degraded, where collagen in the dermis layer was well arranged, and the epithelial layer was composed of three to four layers in the SDF-1αovHDF-mAM group (E).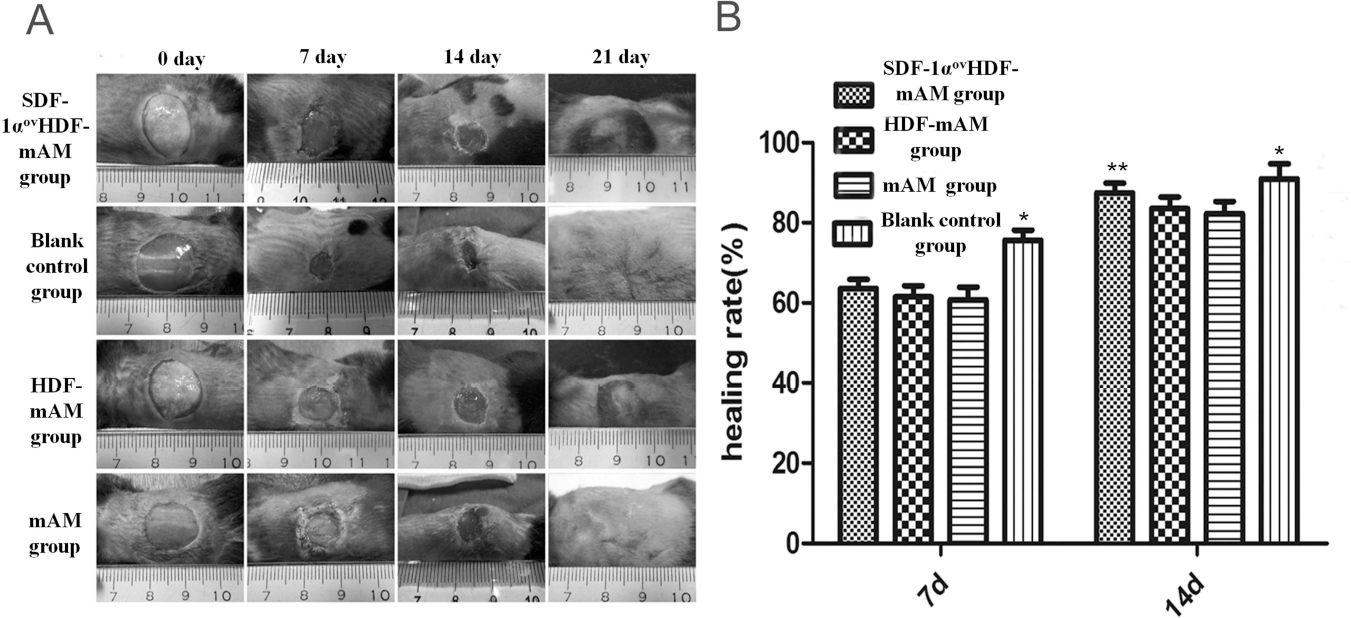

Discussion
Fibroblasts are the main cell component in the connective tissue of the dermis and play an important role in collagen formation during the process of wound healing and a key role in wound closure and vascularization. They participate in various important physiological processes of wound healing by regulating capillary formation via secretion of multiple types of extracellular matrix components, cytokines, and proteinases (11-13,15,21). In addition, mutual contacts and interactions between fibroblasts and endothelial cells are important factors affecting angiogenesis (4). Researchers mixed human umbilical vein endothelial cells with fibroblasts and seeded them onto the collagen gel and found the formation of microcapillary structures 1 week after in vitro culture (20). Based on the above experimental finding, we attempted to use fibroblasts to construct a microcomplex as a microenvironment for vasculogenesis in vivo.
In the present experiment, we used mAM as the vector for culture and amplification of fibroblasts. It was found that fibroblasts and SDF-1α gene-transfected fibroblasts grew and proliferated vigorously on the mAM surface in a 3D manner. After a 5-day culture, continuous single-layer cells were observed on the mAM surface, and these cells secreted multiple cytokines and growth factors including IL-6, IL-8, and TGF-β1, and extracellular components LN and HA. Secretion of these cytokines and growth factors reached the peak 5 days after HDF-mAM culture, probably because cells were in the logarithmic growth phase at the time. In addition, the 3D structure of mAM was beneficial to the space distribution of HDF and effective utilization of nutrients (7).
As angiogenesis and vasculogenesis involve the complex network structures of various cell types such as endothelial cells, EPCs and fibroblasts, extracellular matrix components such as LN and COL IV, and growth factors such as aFGF and VEGF, the effect of using a particular factor alone is not satisfactory. Growth factors such as IL-8, TGF-β1, bFGF, and aFGF secreted from HDF-mAM are all factors that stimulate the proliferation of endothelial cells. LN and HA are important components of the dermal structure, also playing important roles in adhesion and growth of epithelial cells, fibroblasts, and vascular endothelial cells (13,17). In addition, interactions between fibroblasts and endothelial cells can also promote angiogenesis. Animal studies have further demonstrated that HDF-mAM complex can promote vascularization, and the micronized amniotic membrane facilitates the infiltration of interstitial fluid and formation of new vessels. It was found in our study that mAM could retain their volume effectively without diffusion and accelerate vascularization because aggregation and invasion of large amounts of fibroblast were observed even in the early stage. Other than the above-mentioned growth factors and extracellular matrix components, SDF-1αovHDF-mAM complex also highly expressed SDF-1α. Both in vitro and in vivo experiments showed that the expressed SDF-1α possessed normal physiological functions and had obvious chemotactic and migration effects on EPCs as represented by the increased amount of EPCs in peripheral blood, which is consistent with the finding of other studies (16). The number of the newly formed vessels in the SDF-1αovHDF-mAM group increased significantly compared with that in the HDF-mAM group. When EPC chemotaxis reached the local wound, it directly participated in angiogenesis and vasculogenesis. Previous studies (8) have shown that the vasculogenic effect of EPC not only involves matrix metalloproteinases, adhesive proteins, growth factors, adhesion molecules and their interactions but also a series of factors between endothelial cells and nonendothelial cells in the surrounding tissue such as fibroblasts and smooth muscle cells. Therefore, HDF and the growth factors and extracellular components secreted by HDF can provide a sound microenvironment for vasculogenesis of EPCs.
We did not follow up the survival of fibroblasts after transplantation in the present study, but previous studies have shown that fibroblasts do not express MHC-II-like antigens. As fibroblasts have weak immunogenicity, transplantation of allogeneic fibroblasts would not induce significant rejection. Hansbrough et al. (6) showed the clinical effect of seeding autologous or allogenous fibroblasts onto PGA or PGL mesh as the dermal scaffold was similar. Analysis of the karyotype after transplantation of the mixture of female mouse fibroblasts and collagen gel as the dermal substitute onto a culture to male mice of the same line showed that 9 days after transplantation, 82% cells were transplanted fibroblasts rather than the invaded host fibroblasts; 1 month after transplantation, there were still 60-64% transplanted fibroblasts, and a 13-month follow-up observation still showed large amounts of transplanted fibroblasts (6). The above findings have demonstrated that allogenous fibroblasts can survive, grow, and proliferate for a long time. Fibroblasts play a certain role in inducing invasion of autologous cells and angiogenesis during the early key period of graft survival after transplantation of HDF-mAM and SDF-1αovHDF-mAM complexes, while survival of allogenous (human) fibroblasts may not be very important during the late stage of transplantation because there has been sufficient vascularization.
Conclusion
In summary, HDF-mAM and SDF-1αovHDF-mAM are constructed simply and conveniently and have a strong proliferative activity; they can secrete various cytokines and extracellular matrix components to provide a sound microenvironment for quick vascularization. Moreover, SDF-1αovHDF-mAM may prove to be a new model to accelerate vascularization of tissue and organ grafts and chronic ischemic wounds by chemotaxizing EPCs to reach the wound surface, thus accelerating angiogenesis of the local wound.
Footnotes
Acknowledgments
This project was supported by the National Natural Science Foundation of China (81372058, 81071555, 81120108015, 81501665), National Basic Research Program of China (973 Program, 2012CB518100), “Twelfth Five-Year” Scientific Program of China (AWS11J008), Shanghai Youth Sailing Program (14YF1405500), “1255” Academic Discipline Project of Changhai Hospital (CH125510200), and “Priority” for Clinical Key Discipline Project of Shanghai. The authors declare no conflicts of interest.
