Abstract
Effective mobilization of hematopoietic stem cells (HSCs) in injured organs has not been established. Matrix metalloproteinase-9 (MMP-9) is known to release HSCs from bone marrow (BM) into the peripheral blood, but its role in the recruitment of HSCs to injured organs is unclear. In this study we tried to clarify the role of the host MMP-9 in trafficking of HSCs toward the injured liver, especially the relation of MMP-9 with the chemokine receptor 4 (CXCR4)–chemokine ligand 12 (CXCL12) axis, and to examine whether MMP-9 deficiency affects BM cell trafficking to the injured liver in mice. In vitro, we investigated the effect of MMP-9 on migration activity and CXCR4 expression on lineage-negative (Lin-) BM cells. In vivo, we induced acute and chronic liver injury in MMP-9 knockout (KO) and control mice by inoculation of carbon tetrachloride, followed by transplantation of Lin- BM cells obtained from enhanced green fluorescent protein (EGFP)-transgenic mice, and counted the BM cells mobilized in the injured liver. In a migration assay, active MMP-9, but not proMMP-9, increased the number of migrated Lin- BM cells, which was inhibited by tissue inhibitor of metalloproteinase-1 or a MMP inhibitor. This chemoattractant function by MMP-9 was synergistic when cotreated with CXCL12. CXCR4 expression on Lin- BM cells was dose- and time-dependently increased by active MMP-9. At the same time, treatment with MMP-9 enhanced CXCL12 expression, and CXCL12 reciprocally increased MMP-9 expression in BM cells. In in vivo studies, many EGFP+ cells were seen in control recipient mice. In contrast, few EGFP+ cells were observed in MMP-9 KO mice. BM cells tended to differentiate into desmin+ cells. In conclusion, MMP-9 contributes to the mobilization of BM cells in the injured liver by upregulating the expression of CXCR4 on Lin- BM cells and attracting BM cells along its gradient of CXCL12. Therefore, host MMP-9 plays an important role in BM cell migration in the injured liver.
Keywords
Introduction
Recent studies have shown that bone marrow (BM) cells mobilize in the injured area when a pathological disorder occurs, and BM cells are involved in organ repair (17,18,28,31,47,48). However, the mechanisms by which BM cells mobilize in the damaged liver are not yet fully understood. There are two steps involved in successful migration and engraftment of hematopoietic stem cells (HSCs) into the target organ (27,35). First is the release of HSCs from BM into peripheral blood, and second is the recruitment of HSCs to the target organ. It has been shown that chemokine ligand 12 (CXCL12), together with its cognate receptor, chemokine receptor 4 (CXCR4), a highly expressed membrane receptor in all undifferentiated or progenitor BM cells (4,22), generates signals that regulate both steps (3,5,29,46). CXCL12, constitutively produced by various BM stromal cell types, epithelial cells, and various tissues, is usually highly expressed in damaged organs, including the liver. It is a potent chemoattractant for hematopoietic stem cells (HSCs) and regulates cell adhesion, survival, and cell cycle status (8,24). Circulating CXCR4+ cells can be directed to organ-specific locations that actively secrete stromal derived factor-1α (SDF-1α), such as the bone, brain, lung, and liver (14,41,45).
On the other hand, matrix metalloproteinase (MMP)-2 (gelatinase A) and MMP-9 (gelatinase B), both of which are zinc-dependent endopeptidases, are of particular relevance to the liver because of their critical involvement in the degradation of basement membrane components, such as type IV collagen and fibronectin, two main components in the space of Disse (2,21,25). In addition, they have been found to be highly active in the recruitment of BM cells in the injured liver. Khandoga et al. suggested that MMP-9 is involved in the early recruitment cascade of neutrophils and in cluster of differentiation 4-positive (CD4+) T-cell migration in an autocrine manner (21). Kollet et al. showed that irradiation or inoculation with carbon tetrachloride (CCl4) in mice could increase MMP-9 as well as CXCL12 levels in the injured liver, leading to increases in CXCR4 expression and CXCL12-mediated recruitment (25). However, there is no direct evidence showing the correlation between the CXCL12-CXCR4 axis and MMP-9 in mobilizing BM cells in acute and chronic liver injury models.
The purpose of the present study was to elucidate the cooperation between the CXCL12–CXCR4 axis and MMP-9 with respect to HSC mobilization in vitro and in vivo. For the in vivo study, we examined whether MMP-9 knockout (KO) and wild-type mice differed in the mobilization of BM cells to the CCl4-induced injured liver. Moreover, we examined the differentiation of the mobilized cells in a chronic injury model.
Materials and Methods
Animals
MMP-9 KO mice (FVBMMP-9-/-) were purchased from Jackson Laboratory (Bar Harbor, ME) and bred in our animal facility. Female MMP-9 KO mice at 8 weeks of age were used in this study. Control FVB strain mice were purchased at 8 weeks of age from CLEA Japan Inc. (Shizuoka, Japan). Enhanced green fluorescent protein (EGFP)-transgenic mice [TgN (β-act-EGFP) Osb] of the C57BL/6 strain, which were kindly provided by Masaru Okabe (Genomic Research Center, Osaka University, Osaka, Japan) (32), were used as BM donors. All animal procedures were performed in the Animal Facility of University of Toyama following institutional guidelines, approved by the Ethical Commission of the Faculty of Medicine. All animals received humane care in accordance with US National Institutes of Health guidelines.
Isolation of Lineage-Negative Cells
Lineage-negative (Lin) cells were purified from BM cells of 8-week-old male EGFP-transgenic mice. In brief, whole BM cells were collected from both femurs by flushing the BM cavity with heparinized saline. The cell suspension was centrifuged over a Ficoll step gradient (density 1.077 g/ml) at 2000 rpm for 30 min. The interface fraction was collected and Lin- cells were selected using the Lineage Cell Depletion Kit (Miltenyi Biotec, Germany), which specifically depletes mononuclear cells by retaining them on a magnetic activated cell sorter (MACS) separator (Miltenyi Biotec, Bergisch Gladbach, Germany), utilizing an antibody cocktail (Miltenyi Biotec) consisting of antibodies against CD5, CD45R (B220), CD11b, granulocyte differentiation antigen-1 (Gr-1) [lymphocyte antigen 6G/C (Ly-6G/C)], T-cell receptor beta variable 7–4, and lymphocyte antigen 76 (Ly76; antibody Ter-119).
BM Transplantation in Acute Liver Injury Model
MMP-9 KO and two wild-type mouse groups, each consisting of six mice, were used. They were administered a single 4.5 Gy dose of whole-body irradiation on day 1. Twenty-four hours after radiation, 2 × 105 Lin- BM cells obtained from EGFP-transgenic mice were injected intravenously into the recipients. In one of the wild-type groups, 500 nM of MMP-2/9 inhibitor (MPI) (Calbiochem, Darmstadt, Germany) was intraperitoneally administered on day 0. The mice were killed on day 3 and the livers were analyzed.
BM Transplantation in Chronic Liver Injury Model
Groups of MMP-9 KO and wild-type mice, each consisting of 12 mice, were divided into four subgroups and administered a single 4.5 Gy dose of whole-body irradiation on day 1. From day 2, each mouse was intraperitoneally injected with 1 ml/kg CCl4 dissolved in olive oil (1:4) twice a week for 6 weeks. On day 3, 2 × 105 Lin- BM cells obtained from EGFP mice [BM transplant (BMT) group] or the same volume of saline injection as a control [non-BM transplant (non-BMT) group] were injected intravenously in both the MMP-9 KO mice and the wild-type recipients. The mice were killed on day 43 and the livers were analyzed.
Reverse-Transcription PCR and Real-Time RT-PCR
Total RNA was isolated from 30 mg of liver tissue using Isogen (Nippon Gene, Tokyo, Japan). RNA was quantified by spectrophotometry. cDNA was synthesized by reverse transcription. The PCR primers and the conditions to detect EGFP, CXCR4, CXCL12, sex-related Y chromosome (Sry), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) are listed in Table 1. The number of PCR cycles required to detect the expression of each gene was determined in order to semiquantify the amount of messenger RNA within a linear range. Quantitative PCR was performed with an ABI PRISM 7300 Sequence Detection System (PE Applied Biosystems) using three-stage program parameters provided by the manufacturer, as follows: 2 min at 51°C, 10 min at 95°C, followed by 32 cycles of 15 s at 94°C and 1 min at 60°C. The specificity of the resultant amplification products was confirmed by examination of the dissociation reaction plots. A distinct single peak indicated that a single DNA sequence was amplified during PCR.
Primers Used for Quantitative PCR and RT-PCR

Primers, probes, and accession numbers used to detect gene expression of enhanced green fluorescent protein (EGFP), chemokine receptor 4 (CXCR4), glyceraldehyde-3-phosphate dehydrogenase (GAPDH), matrix metalloproteinase (MMP)-9, sex-related Y chromosome (Sry), and chemokine ligand 12 (CXCL12).
Fluorescence-Activated Cell Sorting (FACS)
BM cells were isolated from cell suspension using 24% metrizamide gradient separation. Cells were adjusted to 2 × 107/ml in staining buffer (i.e., saline containing 1% bovine albumin). The cell suspension (50 ml) was incubated with antibodies on ice for 30 min, washed with staining buffer, and fixed with 2% paraformaldehyde. FACS data were acquired using a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA, USA) set to capture all events. A 530/30 nm bandpass filter was used to measure the EGFP fluorescence intensity with excitation at 488 nm (argon ion laser). To detect CXCR4+ cells, phycoerythrin-labeled anti-mouse CD184 (CXCR4) antibody (BD Pharmingen, San Jose, CA, USA; 1:500) was used. FACS data were analyzed using CellQuest software (BD Biosciences).
Transwell Migration Assay
We performed the migration assay using 24-well Transwells, pore size 5 μm (Corning, NY, USA). RPMI (700 μl) plus 10% fetal calf serum containing 100 ng/ml SDF-1α (RD Systems), 100 ng/ml proMMP-9, 0-100 ng/ml active MMP-9 (Calbiochem, Darmstadt, Germany), 0-100 μg/ml MPI, or tissue inhibitor of metalloproteinases-1 (TIMP-1) (Calbiochem, Darmstadt, Germany) were placed in the lower chamber. ProMMP-9 was activated by p-aminophenylmercuric acetate with 24-h coincubation. BM cells (1 × 105) in 1 ml medium were placed in the upper chamber and incubated at 37°C for 3 h. Moreover, the cells that migrated to the lower chamber were collected and counted using a Vector (2D) Hysteresis analyzer.
CXCL12 ELISA
The CXCL12 level in each liver specimen was measured by ELISA as follows. In brief, 96-well plates were coated overnight with anti-CXCL12 antibody (MAb 79018; R&D Systems, UK) at a concentration of 2 mg/ml in bicarbonate buffer (15 mM Na2CO3; 35 mM NaHCO3, pH 9.6). Wells were washed three times, blocked for 1 h with phosphate-buffered saline/1% bovine serum albumin/5% sucrose at room temperature, and rewashed three times. The samples, which were obtained from mice livers homogenizing with radioimmunoprecipitation assay (RIPA) buffer, were then added in duplicate wells and incubated for 2 h at room temperature. Recombinant murine CXCL12 (Peprotech EC) was used to generate a standard curve. Wells were washed three times and treated with anti-CXCL12-biotin capture antibody (MAb BAF310; R&D Systems) for 2 h at room temperature. After three washes, streptavidin-horseradish peroxidase was added for 20 min at room temperature, and the wells were rewashed three times and incubated for 20–30 min with 3,3′, 5,5″-tetramethylbenzidine substrate solution (Sigma-Aldrich, UK). Reactions were terminated with 2 M H2SO4 and the wells were read at 450 nm on an optical plate reader. All analyzed samples were quantified for protein concentration.
Immunofluorescence Studies
EGFP cells were observed using a confocal laser scanning microscope (TCS-SP5; Leica Microsystems, Japan). The emission fingerprinting method was used to distinguish specific fluorescent signals from background autofluorescence. To detect desmin expression, sections were incubated with anti-mouse desmin antibody (Dako, Japan) using the Mouse-on-Mouse Immunodetection Kit (Vector Laboratories, Burlingame, CA, USA). Sections were incubated with streptavidin-conjugated Alexa fluor 555 (Molecular Probes, Eugene, OR, USA). Other antibodies recognizing platelet endothelial cell adhesion molecule-1 (PECAM-1) (CD31) (BD Pharmingen) and hepatocyte nuclear factor-4 (HNF-4) (Santa Cruz Biotechnology) were also used with appropriate secondary antibodies to analyze the differentiation of BM cells.
Statistical Analysis
Results are presented as mean ± SD. Statistical significance was evaluated using paired t-test or one-way analysis of variance followed by Bonferroni's post hoc multiple comparison test. A value of p < 0.05 was considered statistically significant and the number of samples examined is indicated by n.
Results
Identification of CXCR4+ BM Cells
Theoretically, higher CXCR4 expression on the cell surface makes it easier for BM cells to migrate to the injured liver, where CXCL12 is produced. We first examined whether Lin- cells, which contain a lot of HSCs, express more CXCR4 than Lin+ cells. We divided BM cells into two groups, Lin- cells, which contained HSCs, and Lin+ cells, which contained cells that had differentiated to leukocytes. Thirty-five percent of Lin- cells expressed CXCR4 on their cell surface, whereas only 23% of Lin+ cells expressed CXCR4 (Fig. 1a-c). Real-time PCR revealed that CXCR4 mRNA expression in Lin- cells was higher than that in Lin+ cells (Fig. 1d).

Fluorescence-activated cell sorting (FACS) analyses of chemokine receptor 4 (CXCR4) expression on Lineage (Lin+/Lin-) bone marrow (BM) cells. (a, b) 35% of Lin- cells expressed CXCR4 on their surface, whereas only 23% on Lin+ cells expressed CXCR4. (c) CXCR4+ percent on Lin+/- cells was analyzed. n = 5. (d) Real-time PCR revealed that CXCR4 mRNA expression was higher in Lin- cells than in Lin+ cells.
MMP-9 and CXCL12 Increased Migration of Lin- BM Cells
We examined whether MMP-9 itself can act as a chemoattractant for HSCs, using Transwell migration chambers. First we examined active MMP-9 in different concentrations. The number of migrated Lin- cells increased almost twofold when more than 50 ng/ml of active MMP-9 was added to the lower chamber, which was inhibited by MMP inhibitor (Fig. 2a). Next we focused on MMP-9 activation, and found that the number of the migrated cells was increased by active MMP-9, but not by proMMP-9 (Fig. 2b). TIMP-1 inhibited the migration activity of active MMP-9 (Fig. 2b). These data showed enzyme activity of MMP-9 itself is important as a chemoattractant. To compare the chemoattractive activities of MMP-9 and CXCL12, Lin- cells were placed in the upper chamber with RPMI, and RPMI with/without CXCL12, active MMP-9, and MPI were placed in the lower chamber (Fig. 2c). CXCL12 treatment strongly increased Lin- cell migration as already reported (1,13). Interestingly, MMP-9 increased the significant number of migrated cells after cotreatment with CXCL12 and MMP-9. In addition, MPI significantly suppressed this migration. Thus, MMP-9 and CXCL12 synergistically participate in Lin- cell migration, although MMP-9 alone has a weaker effect than CXCL12.

Roles of matrix metalloproteinase (MMP)-9 and chemokine ligand 12 (CXCL12) in the migration of Lin- BM cells. (a) Active MMP-9 was added to the lower chamber and the number of migrated cells was counted. MMP-9 exhibited chemoattractive activity for Lin- cells at concentrations above 50 ng/ml, which was inhibited by MMP-2/9 inhibitor (MPI). (b). To compare the chemoattractive activity of proMMP-9 with active MMP-9, 50 ng/ml of proMMP, active MMP-9, tissue inhibitor of metalloproteinases (TIIMP)-1, or MPI was added to the lower chamber. Migration was promoted by active MMP-9, but not by proMMP-9. TIMP-1 and MPI inhibited the migration. (c) To compare the chemoattractive activities of MMP-9 and CXCL12, the migrated cells were counted. CXCL12 treatment strongly increased Lin- cell migration, and the number was significantly higher after cotreatment with CXCL12 and MMP-9. MPI significantly suppressed this migration.
MMP-9 Enhanced CXCR4 Expression on Lin- BM Cells
To clarify the mechanism of chemoattractive activity by MMP-9, we analyzed the effect of active MMP-9 on the expression of CXCR4 on the surface of Lin+/- cells in different conditions. FACS analysis showed that the BM cells coincubated with active MMP-9 showed higher CXCR4 expression in Lin- cells than Lin+ cells in a concentration-dependent manner (Fig. 3a). Moreover, we confirmed that coincubation with active MMP-9 increased the number of CXCR4+ cells in a time-dependent manner (Fig. 3b).
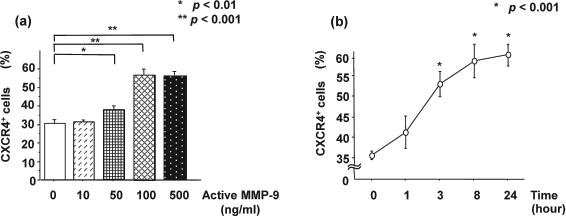
FACS analyses of CXCR4 expression on BM cells. (a) Incubation with MMP-9 caused significantly higher expression of CXCR4 in a concentration-dependent manner on Lin- cells than Lin+ cells. (b) FACS also showed that MMP-9 stimulated CXCR4 expression on Lin- BM cells in a time-dependent manner.
MMP-9 and CXCL12 Reciprocally Enhanced the Expression in Lin- BM Cells
We examined the expression of CXCL12 in Lin- cells coincubated with active MMP-9. RT-PCR study showed the increased expression of CXCL12 by the treatment of active MMP-9 (Fig. 4a). We also evaluated MMP-9 expression in Lin- cells coincubated with CXCL12. CXCL12 enhanced MMP-9 expression (Fig. 4b).
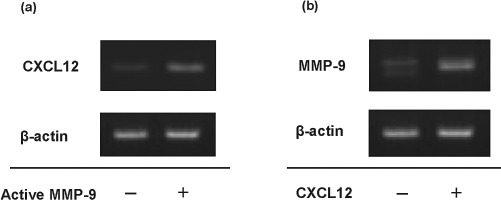
Gene expression of CXCL12 and MMP-9 in Lin- BM cells by RT-PCR. (a) CXCL12 expression was examined in Lin- cells treated with/without MMP-9. Active MMP-9 enhanced CXCL12 mRNA expression. (b) MMP-9 expression was examined in Lin- cells treated with/without CXCL12. CXCL12 enhanced MMP-9 mRNA expression.
CXCL12 Protein Levels and MMP-9 Expression in Acutely and Chronically Injured Liver
To determine the best condition for BM cell transplantation and to investigate the relationship between MMP-9 and the CXCL12-CXCR4 axis, we used ELISA to establish the best way to increase the concentration of CXCL12 protein. We tried various methods of irradiation and single doses of CCl4, and their combination, to induce acute liver injury, and confirmed that the concentration of CXCL12 protein increased the best with the combination after 24 h (Fig. 5a). Therefore, we induced acute liver injury using both irradiation and a single dose of CCl4. We then evaluated CXCL12 levels in MMP-9 KO and control mice. Significantly higher CXCL12 levels were detected by ELISA in the acute and chronic injured liver than each nontreatment liver (Fig. 5b). However, there was no significant difference between MMP-9 KO and control mice in either the acutely or the chronically injured liver model. MMP-9 mRNA expression was also quantified by real-time PCR (data not shown).
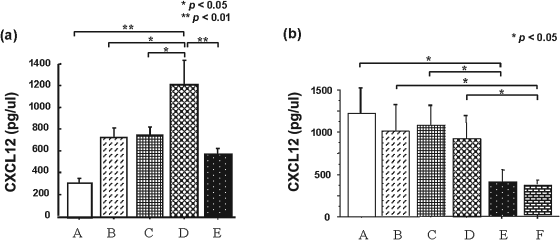
CXCL12 levels in mouse liver. (a) ELISA detected CXCL12 protein in liver injury model. A: control; B: 24 h after radiation; C: 24 h after single injection of carbon tetrachloride (CCl4); D: 24 h after radiation together with CCl4; E: 48 h after radiation together with CCl4. The level of CXCL12 was highest in the group receiving radiation plus CCl4. (b) CXCL12 levels in MMP-9 knockout (KO) and wild-type (WT) mice. CXCL12 levels were increased in both acutely and chronically injured liver models than no injury models, and there was no difference between the four injured models. A: MMP-9 WT, acute injury; B: MMP-9 KO, acute injury; C: MMP-9 WT, chronic injury; D: MMP-9 KO, chronic injury; E: MMP-9 WT, no injury; F: MMP-9 KO, no injury.
Donor-Derived Migrated Cells in Acutely Injured Liver
In the acute liver injury model we examined EGFP+ BM cells mobilized in the liver. EGFP+ cells were detected by confocal laser scanning microscopy. A considerable number of EGFP+ cells were observed along the vessels and in the necrotic area in control mice (Fig. 6a). In contrast, few EGFP+ cells were observed in MPI-treated and MMP-9 KO mice (Fig. 6b, c). VH analysis showed significant fewer migrated EGFP+ cells in MMP-9 KO and MPI-treated mice (Fig. 6d). There were no histological differences between the groups (data not shown). Real-time PCR revealed significantly lower expression levels of EGFP and sex-related Y chromosome (Sry) mRNA in MMP-9 KO and MPI-treated mice (Fig. 6e, f), suggesting that MMP-9 is closely involved in BM cell migration.

Donor-derived migrated cells in injured livers 48 h after CCl4 inoculation. (a–c) Confocal laser scanning microscope images showed the presence of enhanced green fluorescent protein (EGFP)-positive cells. (a) Wild-type/bone marrow transplantation (BMT): control mice with BMT. (b) Wild-type/BMT + MPI: control mice treated with MMP inhibitor. (c) KO/BMT: MMP-9 KO mice with BMT. Arrows indicate EGFP+ cells. (d) EGFP+ cell counts in the liver. 1: Wild-type/BMT; 2: Wild-type/BMT + MPI; 3: KO/BMT. (e, f) Real-time PCR for EGFP (e) and sex-related Y chromosome (Sry) (f) revealed significantly lower expression in MMP-9 KO- and MPI-treated mice.
Donor-Derived Migrated Cells in Chronically Injured Liver
After repeated CCl4 injections over 40 days, bridging fibrosis connecting the neighboring portal areas and central veins was invariably formed in the MMP-9 KO and control groups. Sirius red staining was performed to assess the fibrosis grade, which was not different between the control and MMP-9 KO groups (data not shown). Confocal laser scanning microscopy showed the presence of EGFP+ cells in the fibrous area. Significantly more EGFP+ cells were observed in control mice with BM transplantation than in MMP-9 KO mice in the chronically injured model (Fig. 7a, b, d). No EGFP+ cells were detected in nontransplantation groups (Fig. 7c). Realtime PCR revealed that EGFP and Sry mRNA was suppressed in MMP-9 KO mice (Fig. 7e, f).

Donor-derived cells migrated to the liver after BMT in a chronic injury model. Confocal laser scanning microscope images showing the presence of EGFP+ cells. Arrows point to EGFP+ donor-derived cells. (a) Wild-type/BMT: control mice with BMT. (b) KO/BMT: KO mice with BMT. (c) Wild-type/non-BMT: control mice without BMT. Confocal laser scanning microscope images showing the presence of EGFP+ cells. EGFP+ cells were observed along the fibrotic area. Many positive cells were detected in wild-type/BMT. (d) EGFP+ cell count was significantly suppressed in MMP-9 KO mice. (e, f) Real-time PCR for EGFP (e) and Sry (f) mRNA revealed suppression in the MMP-9 KO group.
Differentiation of Migrated BM Cells in Chronically Injured Liver
We performed an immunofluorescence study to clarify the differentiation of migrated Lin- BM cells in the chronic liver injury model. Desmin immunostaining was seen in about half of the EGFP+ cells (Fig. 8a, d). Desmin/EGFP double-positive cells had a round shape. CD31 immunostaining was also seen in EGFP+ cells, but to a much lesser extent than desmin immunostaining (Fig. 8b, d). CD31/EGFP double-positive cells were elongated, suggesting the differentiation to sinusoidal endothelial cells. On the contrary, immunostaining for HNF-4, which is expressed in premature and mature hepatocytes, was seldom detected in only one cell from all fields (Fig. 8c). These results suggest that Lin- BM cells differentiated into stellate cells or myofibroblasts in this liver injury model.

Differentiation of Lin- cells 6 weeks after BMT in chronic injury model. (a) Desmin+ cells (200x, red, desmin; green, EGFP; blue, DAPI). (b). CD31+ cells [100x, red, CD31; green, EGFP; blue, 4′,6-diamidino-2-phenylindole (DAPI)]. (c) Hepatocyte nuclear factor-4 (HNF-4+) cells (100x, red, HNF-4; green, EGFP; blue, DAPI). (d) Counts of desmin, CD31, and HNF-4/EGFP double-positive cells.
Discussion
Bone marrow cell transplantation has recently become clinically available in chronic liver failure (15). However, the efficient recruitment of transplanted cells is still being explored. Human and animal studies have shown that MMP-9 plays an essential role in releasing progenitor cells from BM into circulation (6,11,12,19,36,40,42,44). Moreover, MMP-9 is important in the recruitment of HSCs from peripheral blood into the injured liver (10). Kollet et al. demonstrated for the first time that CXCL12–CXCR4 signaling determined the migration of progenitors to the murine liver, and that CXCR4 expression on human CD34+ cell surface was greatly increased by coincubation with MMP-9 (25). However, the correlation between the CXCL12–CXCR4 axis and MMP-9 on HSC mobilization in the injured liver has remained unclear. In the present study, we evaluated the roles of MMP-9 in the mobilization of BM cells from peripheral blood to the injured liver.
First we showed Lin- BM cells migrated according to the gradient of active MMP-9 using migration assay, suggesting that MMP-9 itself participates in the recruitment of HSCs directly from peripheral blood into the injured liver where MMP-9 is increased. At the same time, we showed not proMMP-9 but active MMP-9 increased the migration of Lin- BM cells, which were inhibited by TIMP-1 or MMP inhibitor, suggesting enzyme activity may be related to the chemoattractant activity (43). Migration assay also indicated that MMP-9 synergistically affected the migration of BM cells when coincubated with CXCL12, but with a much weaker effect than CXCL12. The present study showed for the first time that MMP-9 and CXCL12 act synergistically for recruitment of BM cells. One mechanism to explain the synergistic activity of MMP-9 is that MMP-9 upregulated the expression of CXCR4 on Lin- BM cells in a concentration- and time-dependent manner, which is consistent with previous results (20,25). Higher expression of CXCR4 may make it easier for Lin- BM cells in peripheral blood to migrate into the injured liver, where a large amount of CXCL12 is produced. Thus, recipient MMP-9 together with the CXCL12–CXCR4 axis acts as a chemoattractant for Lin- cells. This is the first report indicating that MMP-9 plays a direct role in the recruitment of Lin- BM cells. The present in vitro study also showed that MMP-9 itself enhanced the expression of CXCL12 in Lin- BM cells, which reciprocally enhanced the expression of MMP-9. These data suggested that BM cells easily migrate to injured organs where both expressions of CXCL-12 and MMP-9 are increased.
In an in vivo study, we confirmed that there were markedly fewer migrated cells in MMP-9 KO mice in the acute and chronic injury models, although CXCL12 levels were same in wild-type and KO mice in both models. This clearly indicates that MMP-9 as well as the CXCL12–CXCR4 axis plays an important role in mobilizing BM cells.
We found an augmentation of CXCR-4 expression in BM cells after treatment with MMP-9 that showed both concentration and time dependence, which indicates a relationship between MMP-9 and the CXCL12–CXCR4 axis. This increase was greater in Lin- cells. In the present in vivo study, we only used Lin- cells for transplantation because Lin- cells are rich in HSCs, and may aid in demonstrating the relationship between MMP-9 and the CXCL12–CXCR4 axis. MMP-9 might act as a ligand for CXCR4. We did not demonstrate a relationship between MMP-9 and adhesion molecules, which might regulate the attachment of BM cells in the liver. Therefore, further study is required to demonstrate how MMP-9 acts on the mobilization of BM cells in the injured liver.
One of the remarkable results in the chronically injured model was that there was no significant difference in fibrosis between MMP-9 KO and wild-type groups with/without BM transplantation, although we had expected that fibrosis would be more prominent in MMP-9 KO compared with wild-type mice. The lack of only MMP-9 should not affect collagen degradation, and other MMPs, such as MMP-2, should compensate, as has been reported previously (26). On the other hand, we supposed that at least some migrated cells differentiated to stellate cells in the wild-type mice, and might promote liver fibrosis because the number of migrated BM cells was fivefold higher than in the KO group.
There is increasing evidence suggesting that BM cells could provide a transplantable source of hepatic progenitors (15,30). However, it remains controversial as to which the populations of BM cells are clinically promising. Sakaida et al. (39) and Higasihiyama et al. (16) suggested that whole BM cells express MMPs and contribute to the regression of liver fibrosis. Kuo et al. reported the effectiveness of BM mesenchymal stem cells in rescuing experimental liver failure and contributing to liver regeneration (26). Nakamura et al. suggested that endothelial progenitor cell transplantation halts liver fibrosis (30). On the other hand, there is a controversial report suggesting that BM produces additional hepatic stellate cells and myofibroblasts, which contributes functionally to liver fibrosis (7,37,38). Some reports also imply that myofibroblasts as well as hepatic cells in the liver include subpopulations derived from BM cells in fibrotic human and rat livers (23,34). Kisseleva et al. suggested that BM-derived fibrocytes participate in the pathogenesis of liver fibrosis (23). We performed an immunofluorescence study to clarify the differentiation of migrated BM Lin- cells in injured liver. We showed that half of the transplanted cells were desmin+, implying that Lin- BM cells differentiate into myofibloblast or stellate cells. HNF-4+ cells were seldom found, which suggests that transplanted cells undergo slight differentiation into hepatocytes and this finding is consistent with other reports (9,17,28,33). These results suggest that Lin- BM cells that migrate into the injured liver might differentiate to myofibroblasts and stellate cells, which contribute to liver fibrosis in a chronic mouse model.
In conclusion, recipient MMP-9 had the ability to upregulate the expression of CXCR4 on Lin- BM cells and played an important role in the trafficking of these BM cells into the injured liver. There was a close correlation with the CXCL12–CXCR4 axis and MMP-9 in the mobilization of HSCs in the injured liver.
Footnotes
Acknowledgments
The authors thank Yoshihiro Kuwabara for providing animal care and Toyomi Kozawa for technical assistance. The authors declare no conflict of interest.
