Abstract
Skin substitutes with existing vascularization are in great demand for the repair of full-thickness skin defects. In the present study, we hypothesized that a pre-vascularized skin substitute can potentially promote wound healing. Novel three-dimensional (3D) skin substitutes were prepared by seeding a mixture of human endothelial progenitor cells (EPCs) and fibroblasts into a human plasma/calcium chloride formed gel scaffold, and seeding keratinocytes onto the surface of the plasma gel. The capacity of the EPCs to differentiate into a vascular-like tubular structure was evaluated using immunohistochemistry analysis and WST-8 assay. Experimental studies in mouse full-thickness skin wound models showed that the pre-vascularized gel scaffold significantly accelerated wound healing 7 days after surgery, and resembled normal skin structures after 14 days post-surgery. Histological analysis revealed that pre-vascularized gel scaffolds were well integrated in the host skin, resulting in the vascularization of both the epidermis and dermis in the wound area. Moreover, mechanical strength analysis demonstrated that the healed wound following the implantation of the pre-vascularized gel scaffolds exhibited good tensile strength. Taken together, this novel pre-vascularized human plasma gel scaffold has great potential in skin tissue engineering.
Keywords
Introduction
The repair of large extensive venous ulcers, burns and diabetic wounds requires hospitalization that results in a reduced quality of life and substantial socio-economic burden 1,2 . Autografts are the gold standard treatment for wound repair, but are impractical for extensive wound treatments due to the limited availability of donor sites and the risk of secondary morbidity resulting from graft harvesting 3 . Biomaterial or biopolymer-based skin substitutes have been well developed and investigated in the past decades, including naturally derived polymers (alginate, gelatin, collagen, chitosan, fibrin and hyaluronic acid), synthetic molecules (polyethylene glycol, PEG) or their cross-linking agents 4,5 . Biomaterial-based scaffolds have been proven to promote cell infiltration and cytokine production, which provides physiological conditions that are favorable for tissue repair post injury 6,7 . Skin substitutes are usually prepared by seeding with fibroblasts and/or keratinocytes into the respective layers of the scaffold 8 . However, the lack of vascularization inside the scaffolds causes cell death due to hypoxia, which results in a hollow skin substitute. This issue remains a major challenge and limits the potential of these scaffolds for clinical applications.
Previous studies have demonstrated that pre-vascularized scaffolds perform better
In the present study, we produced and examined a novel artificial 3D skin substitute using human plasma that was aimed to induce skin regeneration. This umbilical cord blood (UCB) plasma gel provides a favorable 3D microenvironment for fibroblasts and EPCs to contribute to epidermal migration, collagen deposition and wound repair. In addition, 3D plasma gels are sourced from patients individually, so the risk of pathogen transmission or foreign body rejection is eliminated.
Materials and Methods
Culture of Endothelial Progenitor Cells
The isolation and culture of EPCs were performed as previously described with modifications
12
. In brief, human UCB was layered on the top of Ficoll-PaqueTM PLUS (GE Healthcare, Chandler, Arizona, USA) followed by centrifugation at 900
The Preparation and Biocompatibility of Plasma Gel
The schematic experimental procedures of cell-contained plasma gel scaffold are presented in Fig. 1. Human blood samples were centrifuged at 800
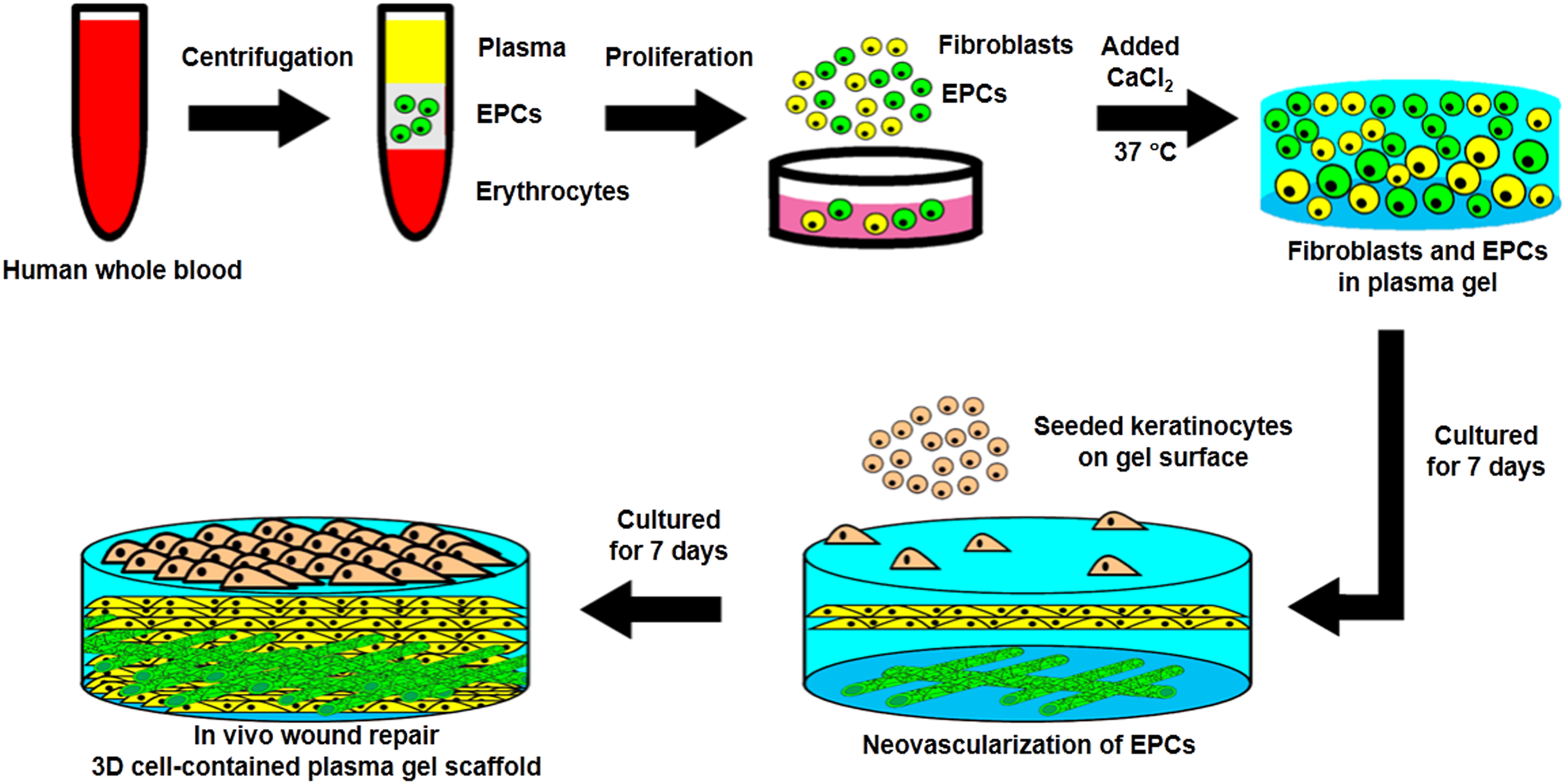
The schematic diagram for the preparation of cell-contained 3D plasma gel scaffold. After centrifugation, the endothelial progenitor cells were isolated from whole blood and mixed with fixed concentrations of CaCl2 for solidification. Endothelial progenitor cells proliferated and differentiated for 7 days to form microvascular-like structures, and keratinocytes were then seeded onto the surface of the plasma gel for 7 days to produce the epidermal layer. The cell-containing 3D plasma gel was prepared and then tested using a wound repair model on the dorsal skin of mice.
Preparation of Pre-Vascularized 3D-Gel Scaffold and Cell Proliferation Assay
EPCs and fibroblasts at various ratios were mixed with 250 μl of plasma gel in a 24-well culture plate and incubated in co-culture medium to form pre-vascularized 3D-skin substitutes. Typically, 2% FBS containing EBM-2 medium is used to induce the differentiation of EPCs to endothelial cells. In this experiment, EPCs and fibroblasts were mixed prior to seeding into the 3D-plasma gel scaffolds and co-cultured for 7 days to construct a pre-vascularized skin substitute. The co-culture medium was made up of equal amounts of DMEM-High glucose/F12 medium (3:1) and EBM-2 medium containing 1% PSA and 2% (or 20%) FBS. After 7 days, keratinocytes were seeded at the density of 1×106 cells/cm2 onto the external surface of plasma gel scaffolds and incubated for another 7 days. The co-culture medium was prepared with equal amounts of DMEM/F-12 medium (3:1, containing 5% FBS, 1% penicillin/streptomycin, 5 mg/ml insulin, 0.4 mg/ml hydrocortisone, and 1 mM isoproterenol) and EBM-2 medium (supplemented with 1% PSA and 20% FBS). In this study, the pre-vascularized skin substitute contained populations of EPCs, fibroblasts and keratinocytes that were seeded on the plasma gel. Non-vascularized skin substitutes, however, only contained fibroblasts and keratinocytes that were seeded on the plasma gel.
Cell proliferation of EPCs and fibroblasts in the plasma gel scaffolds were determined by WST-8 assay (Dojindo Laboratories, Kumamoto, Japan) at days 1, 3, 5 and 7. In brief, the medium was replaced with 300 µl of WST-8 reagent solution (90 µl WST-8 solution and 210 µl co-culture medium). After incubating with WST-8 reagent solution for 2 h, the absorbance was measured on a microplate reader (Sjeia Auto Reader II; Sanko Junyaku, Tokyo, Japan) with a test wavelength at 450 nm and a reference wavelength at 630 nm. Cell number was then derived using a standard curve. The differentiation of EPCs to endothelial cells was confirmed using an immunohistochemistry assay. As the ratio of EPCs to fibroblasts is a critical factor associated with the vascularization of EPCs, the cells were co-cultured at EPC:fibroblast ratios of 1:0.5, 1:1 and 1:2 in the plasma gel scaffolds. The morphology of EPC-derived endothelial cells was observed by an immunohistochemistry assay using CD31 antibody (ab9498, Abcam).
Immunohistochemistry Assay
Primary EPCs isolated from human UCB were incubated for 3 weeks before confirming the EPC phenotype using a CD31 marker for immunofluorescent staining. Cells were washed with phosphate buffered saline (PBS) twice and fixed in 4% paraformaldehyde (Sigma-Aldrich) for 24 h. The plasma gel was immersed in 100% alcohol for 1 min, washed with PBS three times, and then blocked with blocking buffer (1× PBS containing 5% BSA and 0.3% Triton X-100) at room temperature for 1 h. The plasma gel was then incubated with primary mouse monoclonal anti-human CD31 antibody (1:200 dilution) overnight at 4°C, and subsequently with a secondary alexa-conjugated 488 donkey anti-mouse IgG (ab150105, 1:100 dilution, Abcam) overnight at 4°C. The cell nuclei were stained with DAPI (Thermo Fisher Scientific, Waltham, Massachusetts, USA).
In Vivo Animal Studies
Tensile Tests
Mechanical properties were determined by using a universal testing machine (HT-8504 Hung Ta Instruments, Taichung, Taiwan) on the tensile mode setting. After 14 days post-surgery, the mice were sacrificed and the skin of the dorsal wound was excised. Each skin tissue was cut into a rectangular section of 1.5 cm × 4 cm. Tissue specimens were stretched at a crosshead speed of 50 mm/min until rupture. Young’s modulus and tensile strengths at breakage were calculated from the stress-strain data 14 . Six specimens per experimental group were measured to obtain the average values.
Angiogenesis Assay
To assess angiogenesis in the untreated control, plasma gel, non-vascularized and pre-vascularized groups 14 days after implantation, animals were intravenously injected with AngioSense 750 EX (Perkin Elmer, Waltham, Massachusetts, USA; 2 nmol/100 μl prepared in sterile PBS) via tail vein injection 15 . Fluorescence imaging (excitation: 745 nm; emission: 800 nm; high-pass filter cut off: 770 nm; illumination: 30%) in the anesthetized animal was performed 24 h after the injection of the fluorescence dye, using the IVIS Spectrum Imaging System (Perkin Elmer) 15 .
Statistical Analysis
Results were presented as the mean ± standard deviation of three replicates for each experiment. Statistical analysis was performed using SPSS version 22 for Windows (SPSS Inc., Chicago, Illinois, USA). The statistically significant differences between groups were assessed by one-way analysis of variance (ANOVA) with Tukey’s HSD Post Hoc Test.
Results
Preparation of the Plasma Gel Scaffolds
The gel scaffolds were prepared by using a series of CaCl2 concentrations at 2.5, 5, 10, 20 and 40 mM, into a mixture of equal amounts of plasma and culture medium (Fig. 2). Our results demonstrated that 10, 20, and 40 mM CaCl2 were able to induce agglutination of the plasma gel, but 2.5 and 5 mM CaCl2 were too low to successfully form the gel (Fig. 2A). Cell proliferation of fibroblasts inside the plasma gel scaffolds was analyzed using WST-8 assay (Fig. 2B). We found that 40 mM CaCl2 significantly inhibited cell growth from 12 h to 48 h, resulting in cell numbers decreasing from 1×105 cells at 0 h to 0.94×105 cells at 48 h. In contrast, an increase in cell number from 1×105 cells (0 h) to 2.07×105 cells (48 h) was observed in plasma gel scaffolds prepared using 10 mM CaCl2 (Fig. 2B). Therefore, plasma gel scaffolds prepared with 10 mM CaCl2 were used in the following experiments. This plasma gel scaffold is translucent and gelatinous, and can be easily handled and detached from a 6 cm dish plate after casting.
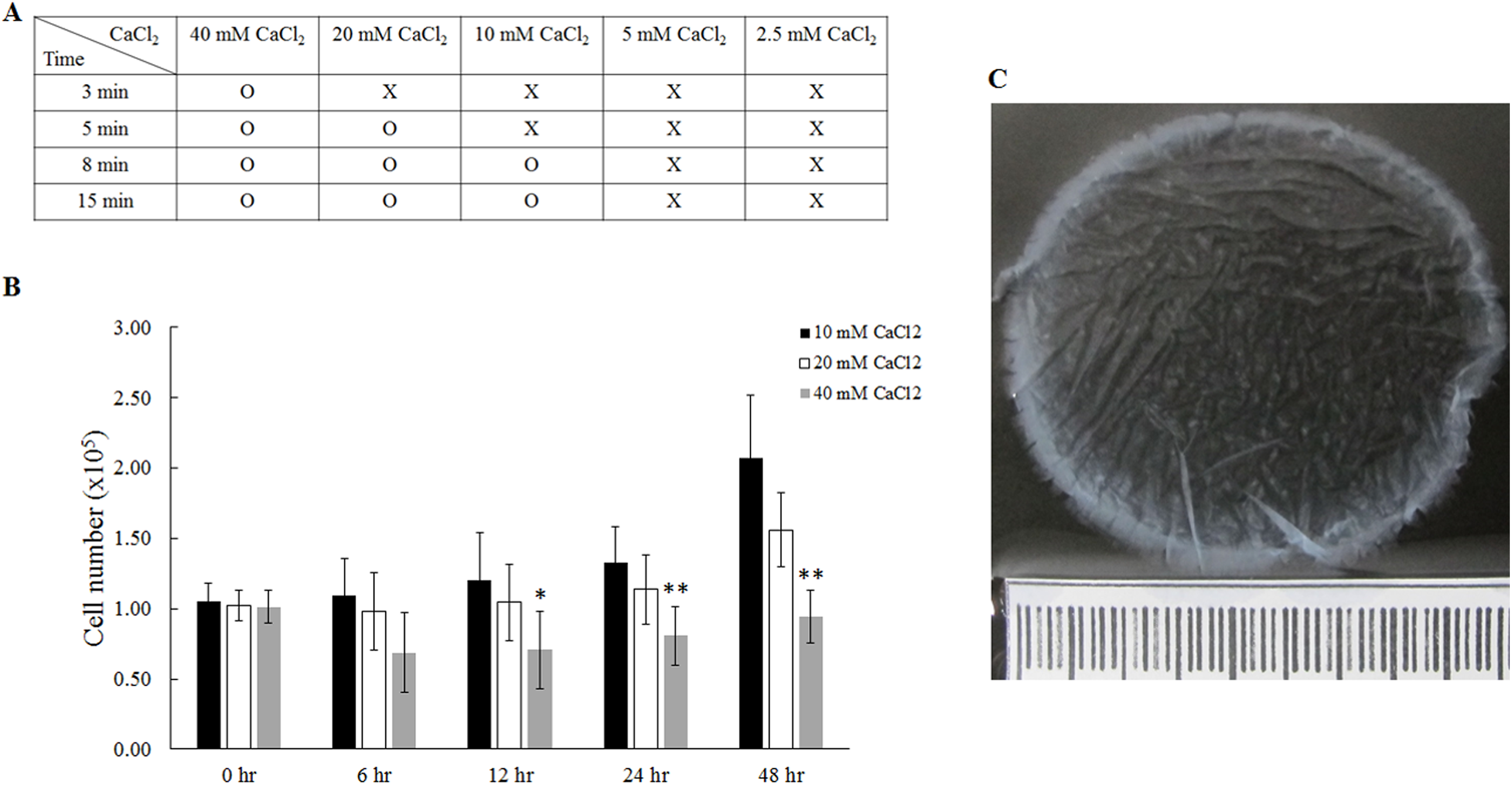
The preparation of plasma gel with addition of different concentrations of CaCl2. (A) The effect of plasma agglutination was tested by different concentrations of CaCl2. (B) The proliferation of fibroblasts for up to 48 h was investigated using plasma gel containing varying specific concentrations of CaCl2. (C) The morphology of plasma gel containing 10 mM CaCl2. *: indicates
Pre-Vascularization Using EPC Incorporated Plasma Gel Scaffolds
Primary EPCs were isolated and harvested from UCB samples. First, EPC phenotypes were identified using specific markers of endothelial progenitors such as CD31 (Platelet endothelial cell adhesion molecule), C-Kit (stem cell marker that is found in EPCs), Tie-2 (endothelial cell-specific tyrosine kinases 2) and VE-Cadherin (an endothelial-specific cell–cell adhesion molecule) in the present study (see Supplement S1). After 3 weeks of culture, the expression of CD31 surface bio-markers on EPCs was confirmed using immunofluorescence staining (Fig. 3). To determine if EPCs have the potential to differentiate into vascular-like endothelial cells in a 3D environment, EPCs were cultured in DMEM-High glucose: F12 (3:1 v/v) 2% or 20% FBS medium. As shown in Fig. 3A, a greater extent of vascularization was observed when EPCs were cultured in 20% FBS medium compared with 2% FBS culture medium over 7 days. Furthermore, cell proliferation assay results demonstrated that the cell growth of EPCs was significantly inhibited in the medium containing 2% FBS compared with 20% FBS medium (Fig. 3B). In contrast, the cell growth of fibroblasts was not affected by the concentration of FBS, resulting in very similar rates of cell growth from day 1 to day 7 (Fig 3C).
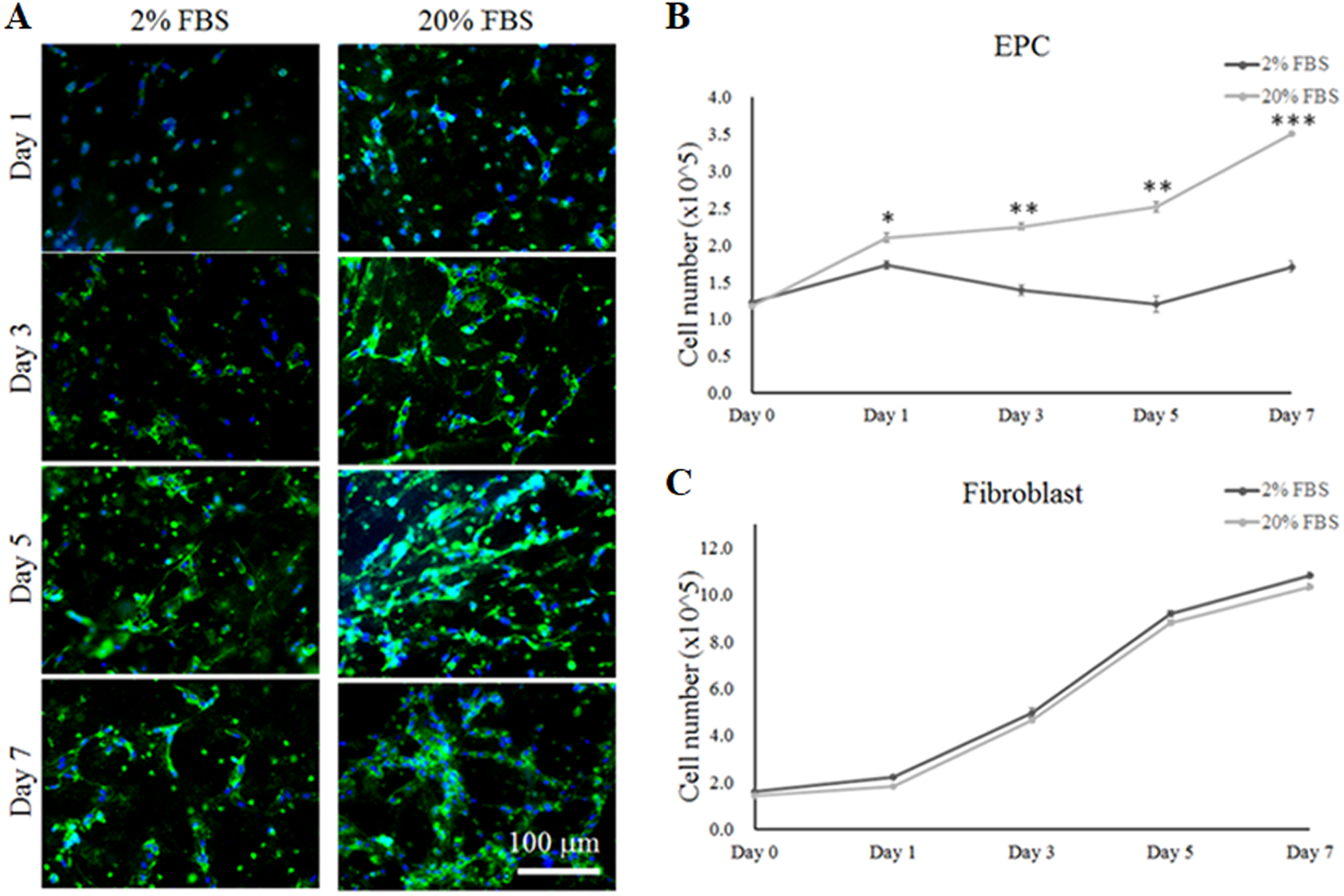
The differentiation and proliferation of EPCs and fibroblasts in plasma gel in co-culture medium containing 2% or 20% FBS. (A) Immunofluorescent staining for the pre-vascularization of EPCs using a labeled CD31 marker in plasma gel with the different co-culture media at 1, 3, 5 and 7 days. (B) The proliferation of EPCs in co-culture medium was observed at 1, 3, 5 and 7 days. (C) The proliferation of fibroblasts in co-culture medium was investigated at 1, 3, 5 and 7 days. *: indicates
Pre-Vascularization of EPC on the Plasma Gel Modulated by Fibroblast
The differentiation of EPCs for vascularization was examined in co-culturing conditions with fibroblasts at varying ratios of 1:0.5, 1:1 and 1:2. Positive staining of CD31 was used to assess vascularization of EPCs in the plasma gel (Fig. 4A). The density of vascularized area significantly increased when EPCs and fibroblasts were cultured at a ratio of 1:0.5 over 7 days. On day 3, the vascularization area was calculated at 17.83% and increased to 25.31% by day 7. In contrast, when ECPs:fibroblasts were cultured at either 1:1 or 1:2, the vascularized area was relatively low at approximately 9–12% over 7 days (Fig. 4B).

The differentiation of EPCs under different ratios of EPCs to fibroblasts in plasma gel. (A) Immunofluorescent staining presenting a vascular-like tubular structure from EPCs were labeled with CD31 marker and cell nuclei were labeled with DAPI at 3, 5, and 7 days. (B) The calculation of the vessel density by Image J Software. *: indicates
The Wound Repair by Neovascularized 3D Skin in Nude Mice
To assess the effect of this pre-vascularized 3D skin substitute on wound healing

The
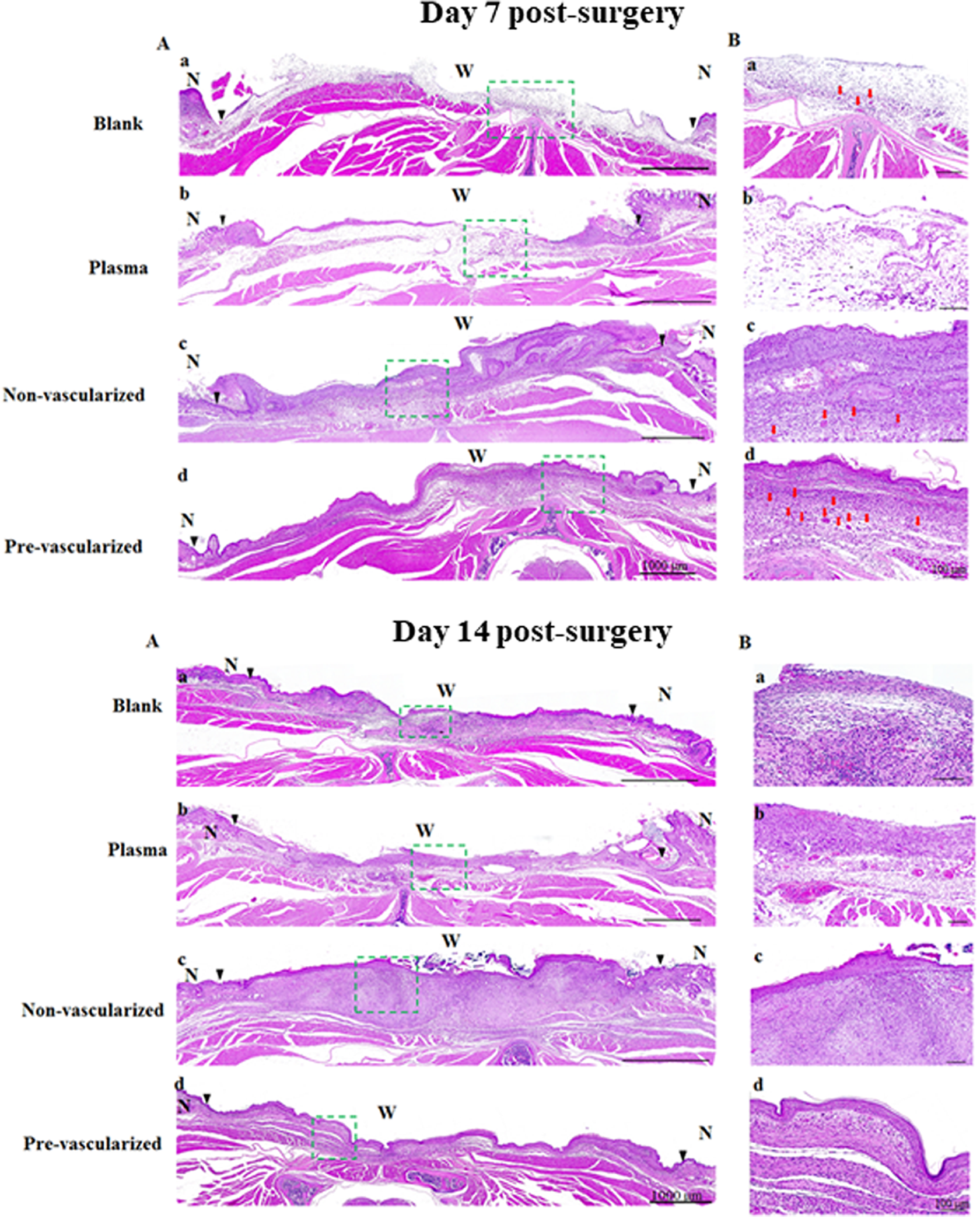
Histological examination of skin wound healing was observed by H&E staining of the whole skin wound (A) and the center of skin wound (B) wound engrafted by blank (a), plasma gel scaffold (b), non-vascularized (c), and pre-vascularized skin (d) under 20× magnification at 7 and 14 days post-surgery. Green dashed rectangle marks the wound center on the dorsal area of nude mice, and the magnification presented as (B) on the right side of images. The epidermal layer could not be found in the blank group. Scale bars = 100 μm. N indicates normal skin. W indicates wound bed. Black triangles in the images indicate the boundaries between the wound and the surrounding normal skin. Red arrows indicate micro-vessels.
Masson’s trichrome staining (Fig. 7A) was conducted to assess collagen formation post skin excision and grafting. Wounds grafted with pre-vascularized 3D skin substitutes displayed well-organized collagen fibers with corrugated structure. However, in the blank (Fig. 7A–b) and plasma gel scaffold treatment (Fig. 7A–c) groups, the density of collagen fibers found at the wound site was low. By day 14, more collagen fibers were obviously apparent in the non-vascularized (Fig. 7A–i) and pre-vascularized (Fig. 8A–j) gel groups compared with the other groups. The quantitative analysis of collagen density demonstrated that collagen production in non-vascularized and pre-vascularized groups was significantly higher compared with the blank and plasma gel groups 14 days post-surgery (Fig. 7B). The thickness of the epidermis layer in the central area of the wounds was measured using a ZEN slidescan tool 14 days post-surgery (Fig. 7C). The thickness of the regenerated epidermis was significantly greater in the non-vascularized 3D gel-treated wounds, but the thickness of the epidermis was found to be similar between the wounds grafted with pre-vascularized skin substitute and normal skin. Blank or plasma gel scaffold-treated wounds displayed limited re-epithelialization in 14 days. The thickness of the dermis layer was also examined 14 days post-surgery (Fig. 7D). Dermal thickness was significantly increased in the non-vascularized 3D gel-treated group, but not in the pre-vascularized groups. Thinner dermal thickness was observed in the blank and plasma gel groups.

(A) Masson’s trichrome staining for the histological analysis of repaired wound after treatment with blank (b, g), plasma gel scaffold (c, h), non-vascularized skin (d, i), pre-vascularized skin (e, j), and normal skin (a, f) at 7 and 14 days post-surgery. Scale bars = 100 μm. (B) quantification of collagen density after Masson’s trichrome staining (C) the calculation of the thickness of the epidermal layer and (D) thickness of the dermal layer as well as the evaluation of mechanical properties measured by (E) Young’s modulus and (F) tensile strength 14 days post-surgery in the five groups. *: indicated
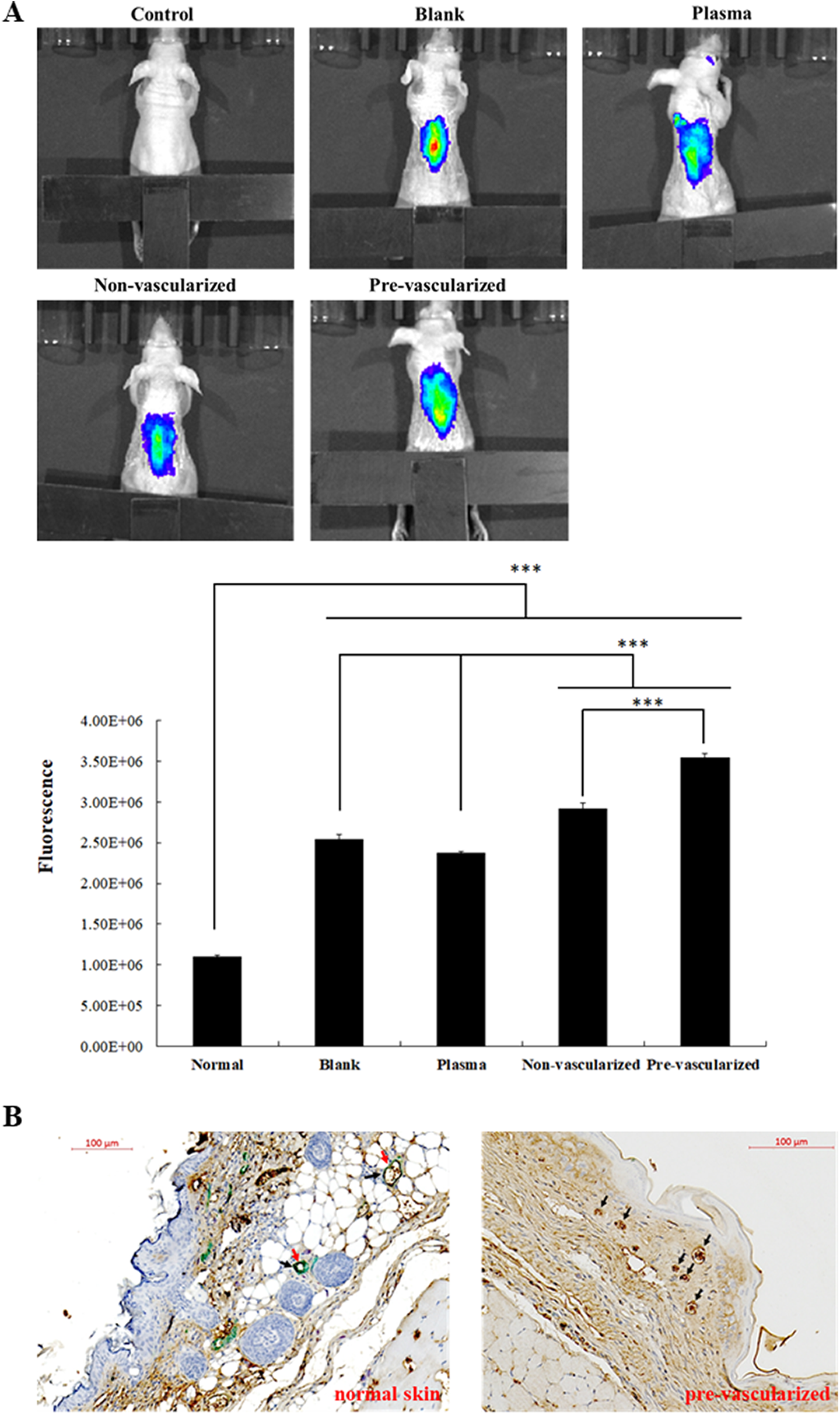
(A) IVIS imaging of neo-vascular blood perfusion of pre-vascularized 3D gel implants and quantification of fluorescent signals. 14 days after implantation, fluorescence imaging of nude mice were performed using IVIS Spectrum Imaging System as described in the Materials and Methods. *: indicates
Evaluation Skin Wound Restoration by Tensile Test
The mechanical strength of healed skin was determined using a universal testing machine (Fig. 7E and F). In the blank group, the wound area was not healed on day 14, therefore the strength assessment could not be performed for the blank group. The epidermal layer also could not be found in the blank group, as shown in the histological analysis (Fig. 6A–a). Non-vascularized and pre-vascularized healed skin maintained a similar Young’s modulus and tensile strength as normal skin. However, a significant reduction in Young’s modulus of 0.66 MPa and tensile strength of 0.22 MPa was observed in the skin healed following plasma scaffold treatment.
Angiogenesis
In order to assess the angiogenesis in the wound area, fluorescence probes (AngioSense 750 EX) were injected on day 14 and fluorescence imaging was recorded 24 h after the injection (Fig. 8A). The highest accumulation of Angiosense 750 fluorophores was found on the wound areas in the pre-vascularized skin group, indicating the formation of functional neovasculature for the regulation of blood flow (Fig. 8A). In addition, non-vascularized skin substitutes showed secondary high fluorescence signals compared with the blank or plasma groups, suggesting that the co-culture of fibroblast may benefit microvascular formation. Furthermore, wounds grafted with non-vascularized and pre-vascularized substitutes formed an epidermis on the surface of wound area. The differentiation of EPCs into smooth muscle cells was further examined using immunohistological double-staining analysis (Fig. 8B). In a normal control skin section, CD31 signals are shown in brown and αSMA expression was shown in green. However, only CD31 (shown as brown) expression was observed in pre-vascularized skin substitutes on day 14, indicating that EPC did not differentiate into smooth muscle cells in the 3D plasma scaffolds during the wound healing process.
Discussion
We successfully prepared a pre-vascularized 3D plasma gel scaffold to promote wound healing and skin regeneration. UCB plasma gels not only provide a favorable 3D microenvironment for skin dermal fibroblasts and EPCs to grow, but also contribute to epidermal migration, collagen deposition and wound repair. The aim of the present study was to form a novel skin substitute using EPCs, keratinocytes and fibroblasts within an optimized CaCl2 cross-linked plasma gel. We modified the CaCl2 concentrations, EPC-fibroblast co-culture conditions and the ratios of keratinocyte/fibroblasts, and found that the construction of a pre-vascularized 3D skin prior to
In previous studies, high concentrations of CaCl2 were reported to induce cell apoptosis and inhibit cell growth in the 3D environment 11 . In the present study, we found that using 10 mM CaCl2 to construct 3D plasma gel did not compromise cell expansion ability, resulting in a significantly higher cell proliferation rate of fibroblasts over 48 h. In addition, the 10 mM CaCl2 casted plasma gel provided a suitable niche for the vascularization of EPCs. Establishment of the co-culture system containing both fibroblasts and EPCs was achieved in this study by using mixed cell medium containing 20% FBS, which promoted cell proliferation of both EPCs and fibroblasts. We also found that the micro-vessel network was successfully developed from EPCs, demonstrating a favorable cell culture environment in the 3D plasma gel.
EPCs cultured in 3D systems undergo vascularization, elongation and coalescence for the development of the vascular-like structure containing lumen
16
. The formation of the vascular-like structure in 3D culture systems, composed of collagen type I or fibrin, is unique to endothelial cells
17
. Previous studies on synthetic hydrogel systems found that the micro-vessel formation of EPCs was influenced by the ratio of endothelial cells to smooth muscle cells
18
. From our
Pre-vascularization is a critical factor in tissue engineering and clinical applications
13,23,24
. Many biomaterial-based scaffolds fail due to poor graft take and integration with host tissue
25
, or lack neovascularization from the peripheral vessels of the wound edge
26
. A successful tissue-engineered scaffold is expected to accelerate dermal fibroblast proliferation, collagen deposition, neovascularization and, more importantly, the formation of the epidermal layer. Epidermal coverage of the wound is important as it acts as a barrier against moisture loss and infection. In this study, the pre-vascularized 3D gel with keratinocytes was engrafted on the wound area of mice. A functional micro-vessel network containing blood flow was apparent in the wound area of mice treated with the pre-vascularized 3D gel scaffold that contributed to accelerated skin regeneration by 14 days post-surgery. Furthermore, the pre-vascularized 3D gel showed mild inflammatory responses. Histological analysis confirmed that the established pre-vascularized network successfully integrated with the surrounding micro-vessels of the wound bed. The epidermal layer was also observed in engrafted pre-vascularized skin by day 7 and 14 post-surgery. On the other hand, dermal thickness was significantly increased in the wounds treated with non-vascularized 3D gel. However, collagen production between non-vascularized and pre-vascularized groups was not significantly different, indicating that the collagen density for pre-vascularized groups was higher than that for non-vascularized groups. Due to the lack of a neo-vascular network in plasma gel, and plasma gel scaffolds seeded with keratinocytes and fibroblasts, we can infer that the original neo-vascular network from the pre-vascularized 3D gel group was gradually integrated into the center of wound 14 days post-surgery. Moreover, our results indicate that the 3D plasma gel scaffold seeded with fibroblasts and keratinocytes can potentially contribute to repairing the wound bed, but the pre-vascularized skin substitute with the EPCs can accelerate the recovery of the skin wound. Based on previous literature, we postulate that the pre-vascularized skin substitute may play a role in shortening the period of vascular regeneration, and may contribute to decreasing hypoxia, inflammation and necrosis during the healing process of the wound bed
27
. Klar et al. (2017) reported improved outcomes in the early inflammatory response during the early phase of wound healing by the vascularized and non-vascularized bio-engineered dermo-epidermal skin transplants
Restoring the elasticity of healed skin remains a major challenge in the development of tissue-engineered skin substitutes 30 . Most manufactured skin substitutes are biocompatible and biodegradable, but do not address requirements for normal skin elasticity. In this study, we confirmed that the mechanical properties of skin engrafted with a plasma gel scaffold were lacking 14 days post-surgery. However, the elasticity of the wounds treated with the non-vascularized and vascularized scaffolds resembled normal skin elasticity.
Conclusions
A novel pre-vascularized plasma gel scaffold was introduced in the present study and was determined a favorable skin substitute to provide multiple requirements, including 3D niche and biocompatibility. Our
Supplemental Material
Supplement_S1_The_immunofluorescent_staining_of_EPCs_of_CD31 - Development of a Novel Pre-Vascularized Three-Dimensional Skin Substitute Using Blood Plasma Gel
Supplement_S1_The_immunofluorescent_staining_of_EPCs_of_CD31 for Development of a Novel Pre-Vascularized Three-Dimensional Skin Substitute Using Blood Plasma Gel by Niann-Tzyy Dai, Wen-Shyan Huang, Fang-Wei Chang, Lin-Gwei Wei, Tai-Chun Huang, Jhen-Kai Li, Keng-Yen Fu, Lien-Guo Dai, Pai-Shan Hsieh, Nien-Chi Huang, Yi-Wen Wang, Hsin-I Chang, Roxanne Parungao, and Yiwei Wang in Cell Transplantation
Footnotes
Authors’ Note
Dai and Wang contributed equally to this manuscript.
Acknowledgments
Dr. Niann-Tzyy Dai acknowledges the financial support of this work provided by the Ministry of National Defense, ROC (MAB-105-043, MAB-106-034), National Defense Medical Center, Tri-Service General Hospital, ROC (TSGH-C106-112), Zouying Branch of Kaohsiung Armed Forces General Hospital, ROC (ZBH 105-07, ZBH 106-10), Taoyuan Armed Forces General Hospital, ROC (AFTYGH-104-27, AFTYGH-105-27) and Ministry of Science and Technology, ROC (MOST-102-2314-B-016-009, MOST-103-2314-B-016-013). Additional financial support was provided by Teh-Tzer Study Group for Human Medical Research Foundation, ROC. The authors declare no conflicts of interest.
Ethical Approval
The study protocol for the collection of human cells was reviewed and approved by the Institutional Review Board (IRB) in the Tri-Service General Hospital, R.O.C. (TSGHIRB No. 100-05-251).
Statement of Human and Animal Rights
The study protocol for the collection of human cells was reviewed and approved by the Institutional Review Board (IRB) in the Tri-Service General Hospital, R.O.C. (TSGHIRB No. 100-05-251). A written informed consent according to the guidelines of Institutional Review Board (IRB) was obtained from each donor. In addition, the protocol for animal experiment was approved by the Institutional Animal Care and Use Committee (IACUC) in National Defense Medical Center, Taiwan, R.O.C. (IACUC-14-286).
Statement of Informed Consent
The study protocol for the collection of human cells was reviewed and approved by the Institutional Review Board (IRB) in the Tri-Service General Hospital, R.O.C. (TSGHIRB No. 100-05-251). A written informed consent according to the guidelines of Institutional Review Board (IRB) was obtained from each donor.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ministry of National Defense, ROC (MAB-105-043, MAB-106-034), National Defense Medical Center, Tri-Service General Hospital, ROC (TSGH-C106-112), Zouying Branch of Kaohsiung Armed Forces General Hospital, ROC (ZBH 105-07, ZBH 106-10), Taoyuan Armed Forces General Hospital, ROC (AFTYGH-104-27, AFTYGH-105-27) and Ministry of Science and Technology, ROC (MOST-102-2314-B-016-009, MOST-103-2314-B-016-013). Additional financial support was provided by Teh-Tzer Study Group for Human Medical Research Foundation, ROC.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
