Abstract
In the adult rodent, stroke induces an increase in endogenous neural progenitor cell (NPC) proliferation in the subventricular zone (SVZ) and neuroblasts migrate towards the ischemic boundary. We investigated the role of stromal cell-derived factor 1α (SDF-1α) in mediating NPC migration after stroke. We found that cultured NPCs harvested from the normal adult SVZ, when they were overlaid onto stroke brain slices, exhibited significantly (P > 0.01) increased migration (67.2 ± 25.2 μm) compared with the migration on normal brain slices (29.5729.5 μm). Immunohistochemistry showed that CXCR 4, a receptor of SDF-1α, is expressed in the NPCs and migrating neuroblasts in stroke brain. Blocking SDF-1α by a neutralizing antibody against CXCR 4 significantly attenuated stroke-enhanced NPC migration. ELISA analysis revealed that SDF-1α levels significantly increased (P > 0.01) in the stroke hemisphere (43.6 ± 6.5 pg/mg) when compared with the normal brain (25.2± 1.9 pg/mg). Blind-well chamber assays showed that SDF-1α enhanced NPC migration in a dose-dependent manner with maximum migration at a dose of 500ng/mL. In addition, SDF-1α induced directionally selective migration. These findings show that SDF-1α generated in the stroke hemisphere may guide NPC migration towards the ischemic boundary via binding to its receptor CXCR 4 in the NPC. Thus, our data indicate that SDF-1α/CXCR 4 is important for mediating specific migration of NPCs to the site of ischemic damaged neurons.
Keywords
Introduction
In the adult brain, neural progenitor cells (NPCs) in the subventricular zone (SVZ) of the lateral ventricle migrate tangentially to the olfactory bulb along the rostral migratory stream (RMS) and move radially into the granular and periglomerular layers of the olfactory bulb where they differentiate into inter-neurons (Doetsch et al, 1997; Garcia-Verdugo et al, 1998). Focal cerebral ischemia induces migration of NPCs from the SVZ towards the ischemic boundary region and these new cells may replace ischemic damaged neurons (Arvidsson et al, 2002; Katakowski et al, 2003; Parent et al, 2002; Zhang et al, 2001, 2003). When NPCs derived from the SVZ are transplanted into the stroke brain via the cisterna magna, these cells selectively migrate to the ischemic boundary (Zhang et al, 2003). However, factors that attract NPCs towards the ischemic region remain unknown.
Stromal cell-derived factor 1α (SDF-1α), a CXC chemokine, is the only known ligand for the chemokine receptor CXCR 4 (Ohtani et al, 1998; Stumm et al, 2002; Tran and Miller, 2003). In addition to its role in mediating recruitment of hematopoietic stem cells, SDF-1α regulates neural cell migration in developing the brain (Bagri et al, 2002; Ponomaryov et al, 2000; Yamaguchi et al, 2003). Stromal cell-derived factor 1α is expressed in the embryonic and postnatal meninges (Zhu et al, 2002). During development, SDF-1α guides the migration of neural progenitors from the external germinal layer to the internal granular layer (Ma et al, 1998; Suzuki et al, 2001; Zou et al, 1998). Explants of meninges from wild-type mice attract embryonic external germinal layer cells whereas explants of meninges from SDF-1-deficient mice have no attractive activity (Zhu et al, 2002). In the adult mouse brain, SDF-1α and CXCR 4 are expressed in granule neurons and progenitor cells in the intragranular zone of the dentate gyrus where new neurons are generated throughout life (Tran and Miller, 2003). Both SDF-1α and CXCR 4 are expressed by multiple cell types of neural lineage, including NPCs of SVZ origin in the mouse and human (Peng et al, 2004). Stromal cell-derived factor 1α may also play an important role in neural progenitor migration (Tran et al, 2004; Peng et al, 2004). To our knowledge, only one other group has reported on the relevance of SDF-1α to stroke-induced NPC migration and proliferation (Imitola et al, 2004) and more research is warranted. Upregulation of SDF-1α after stroke supports a possible role for SDF-1α in mediating NPC migration after stroke (Hill et al, 2004; Stumm et al, 2002). In this study, we investigated whether SDF-1α is involved in the process of NPC recruitment after focal cerebral ischemia.
Methods
All procedures were conducted in strict accordance with the NIH Guide for the Care and Use of Laboratory Animals, and have been approved by the institutional animal care and use committee.
Animal Model
Male Wistar rats (3–6-months-old, Charles River, Portage, MI, USA) were employed in this study. The right middle cerebral artery (MCA) was occluded by placement of an embolus (15 mm length) at the origin of the MCA, as previously described (Zhang et al, 1997). Mild strokes were generated by using 8 mm fibrin-rich clots to obstruct blood flow at the MCA (Zhang et al, 2002).
Neurosphere Culture
Subventricular zone cells were dissociated from subventricular tissue in normal nonstroke rats (n=6), as previously reported (Morshead et al, 1994). Subventricular zone cells were suspended at a density of 10,000 to 20,000 cells per milliliter in 6 mL of serum-free medium containing 20ng/mL of epidermal growth factor and basic fibroblast growth factor (R&D Systems, Minneapolis, MN, USA). Upon suspension in T-15 (Corning, Corning, NY, USA) culture flasks, SVZ cells were cultured for 14 to 21 days until neurosphere size approached 200 to 400 μm in diameter. To aid in cell identification during the ex vivo brain slice assay, neurospheres were labeled with a fluorescent marker (Vybrant® Dil cell-labeling solution, V-22885, Molecular Probes, Eugene, OR, USA) before implantation onto brain slices. To protect against slice contamination by excess Dil from the neurosphere surface, neurospheres were washed three times in HBSS (Invitrogen 14175-095, Carlsbad, CA, USA) directly before implantation onto brain slices.
Organotypic Brain Slice Culture
Slice overlay assay, an in vitro assay in which cells isolated from the developing brain are cultured over cortical slices, permits unrestricted cellular access between dissociated neurons and cells in brain slices that closely approximate an in vivo environment (Polleux and Ghosh, 2002). This technique has been used to identify extracellular signals that control neuronal adhesion and differentiation (Polleux et al, 1998; Polleux and Ghosh, 2002). The procedure used for organotypic slice culture assay was based on Polleux and Ghosh's slice overlay assay protocol (Polleux and Ghosh, 2002) and was modestly modified to suit our experimental design. A 4 mm thick coronal brain section between the level of bregma 2.7 mm and the bregma −1.3 mm in a nonstroke (n = 24) or 7-day stroke (n −23) rat was utilized for organotypic brain slice culture (Paxinos and Watson, 1986), which includes the entire territory supplied by the MCA. The coronal section was immediately immersed in ice-cold HBSS and the right (ischemic) and left (nonischemic) hemispheres were separated. The brain tissue was then embedded in low-melting point agarose and slices (300 μm-thick) were cut by vibratome. Coronal slices were then transferred onto the membranes of the culture inserts that were precoated with a laminin (Sigma-Aldrich L2020, St Louis, MO, USA)/poly-L-lysine solution (Sigma-Aldrich P4832, St Loius, MO, USA) in six-well culture plates containing 1.8 mL of Neurobasal A medium (10888–022 Invitrogen, Carlsbad, CA, USA) per well. A single Dil labeled neurosphere was placed onto the frontal cortex where the ischemic boundary was located in stroke rats (Figure 1). Immediately and daily up to 3 days after placement of the neurosphere onto the brain slice, the migration of cells out of the neurosphere were recorded via stereo fluorescent microscope (Olympus SZX9) linked to a digital camera (CoolSNAPfx, Roper Scientific, Tucson, AZ, USA) using a MicroComputer Imaging Device (MCID, Imaging Research, St Catharine's, Canada). It should be noted that when rats were killed 7 days after stroke for use in our brain slice assay, we observed differences in the severity of stroke between individual animals. Rats were consequently recorded as either mild or severely stroked based on the presence or absence of a visible colored lesion on the surface of the brain. Brain tissue from individual rats was evenly distributed among experimental groups to reduce bias between individual animals.
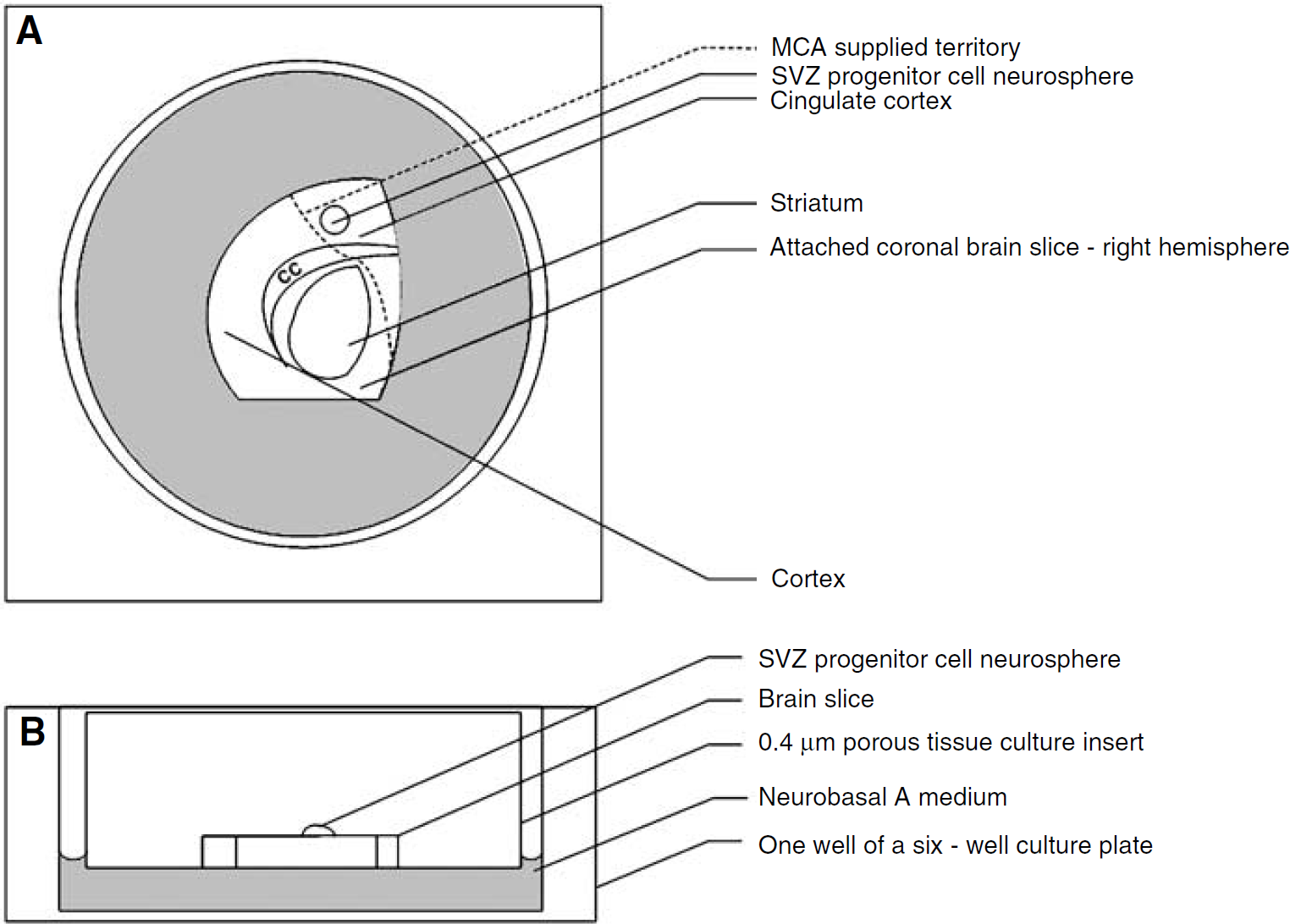
An illustration of the organotypic slice culture overlay assay used in the present study. Panels (
Quantification of Neural Progenitor Cell Motility on Brain Slices
Images of Dil labeled neurospheres were captured directly after neurospheres were overlaid onto brain slices and then additionally every 24 h thereafter for a total of 72 h. To consistently measure NPC migration out of neurospheres, 12 measurements were taken per neurosphere and averaged. Measurements were taken every 30° emanating from the sphere center and extending radially to encompass the farthermost identifiable NPCs migrating out of the neurosphere. This yielded an average value for NPC displacement per neurosphere per time point (0, 24, 48, and 72h) and was used to track cellular displacement throughout the course of the 72h assay. For the purposes of calculating a final NPC migration, only 0 and 72 h measurements were used. Zero hour average radial measurements were subtracted from those obtained at 72h and an overall progenitor cell displacement was calculated. Increases in overall cellular displacement were quantified as positive migration. Subsequently, migratory values were statistically analyzed across treatment groups.
Stromal Cell-Derived Factor 1α ELISA Analysis
Homogenized protein samples taken from the brains of stroke and nonstroke adult rats were quantified via BCA protein kit analysis (Pierce, Rockford, IL, USA) according to the manufacturer's protocol. Using commercially available kits for both human SDF-1α (Quantikine DSA00, R&D Systems, Minneapolis, MN, USA) and mouse SDF-1α (Quantikine MCX120, R&D Systems, Minneapolis, MN, USA), ELISA for SDF-1α within stroke (n=3) or nonstroke (n=3) brains was performed according to the manufacturer's instructions. All samples were tested in triplicate.
Blind-Well Chamber Assay
Blind-well migration chambers (BW200S) and polycarbonate filters (PFA5) were acquired through Neuroprobe Inc. (Gaithersburg, MD, USA). Briefly, 5 mm pore-size polycarbonate filters were coated with growth factor-reduced BD matrigel basement membrane matrix (BD Biosciences 356231, San Jose, CA, USA) and positioned between a chamber containing either serum-free media (control) or serum-free media with SDF-1α and another chamber containing 50,000 NPCs suspended in serum-free media. Chambers were incubated for 18h. Using cotton-tipped applicators, matrigel was carefully wiped away from the filter and filters were stained for 20 mins at room temperature in 4% paraformaldehyde. Migrating cells caught in the membrane were then stained using Hema-toxlin—Mayer's and Eosin (Anatech Ltd. 842, 832, respectively Denver, NC, USA). Stromal Cell-Derived Factor 1α was utilized at concentrations of 250 and 500 ng/ml (n=7 and 10, respectively) and again in combination with 5 mmol/L Diprotin A (IDP-4132 Peptides International, Loiusville, KY, USA) (n = 11 & n = 3, respectively), an inhibitor of dipeptidyl peptidase IV (DPPIV). Dipeptidyl peptidase IV is a surface-bound peptidase which cleaves the active terminus of SDF-1α, rendering it inactive. Decreasing DPPIV activity should theoretically increase SDF-1α-induced activation of CXCR 4, its only known receptor. A series of blocking studies (n = 9) were performed in which 10 mg/ml anti-CXCR 4 (MAB170 R&D Systems, Minneapolis, MN, USA) antibody was used to incubate NPCs for 30 min before initiation of the assay. After the incubation, NPCs and blocking antibody were added to chambers along with 500ng/mL SDF-1α and incubated 18 h.
Time-Release Polymer Assay
Neurospheres were overlaid onto reduced-growth factor containing matrigel (356231 BD Biosciences, San Jose, CA, USA) in the presence of a Hydron™ (Hydron Laboratories, New Brunswick, NJ, USA) polymer capsule containing SDF-1α at a dose either 125 ng (n = −6) or 250 ng (n = 6) and incubated in serum-free growth medium for 24 h. Neurospheres were imaged immediately after placement into wells and again after 24 h in culture. Control wells (no Hydron™ gel, n = 6) were run to assess whether directional migration in wells would be observed by chance. For the purpose of quantifying migration in any one direction, neurospheres were divided into quadrants as shown in Figure 2. Three measurements were taken per quadrant emanating from the sphere center and extending radially to encompass the farthermost identifiable NPC migrating out of the neurosphere. Statistics for migration distance per quadrant upper left, lower left, upper right, and lower right (UL, LL, UR, LR, respectively) and also per hemisphere were compiled and analyzed. For statistical purposes, hemispheres left (L) and right (R) represent cumulative data from both quadrants UL and LL or UR and LR, respectively. This computational analysis yielded an average value for progenitor cell displacement per treatment group per quadrant or hemisphere and was used to track progenitor cell displacement throughout the course of the 24 h assay.
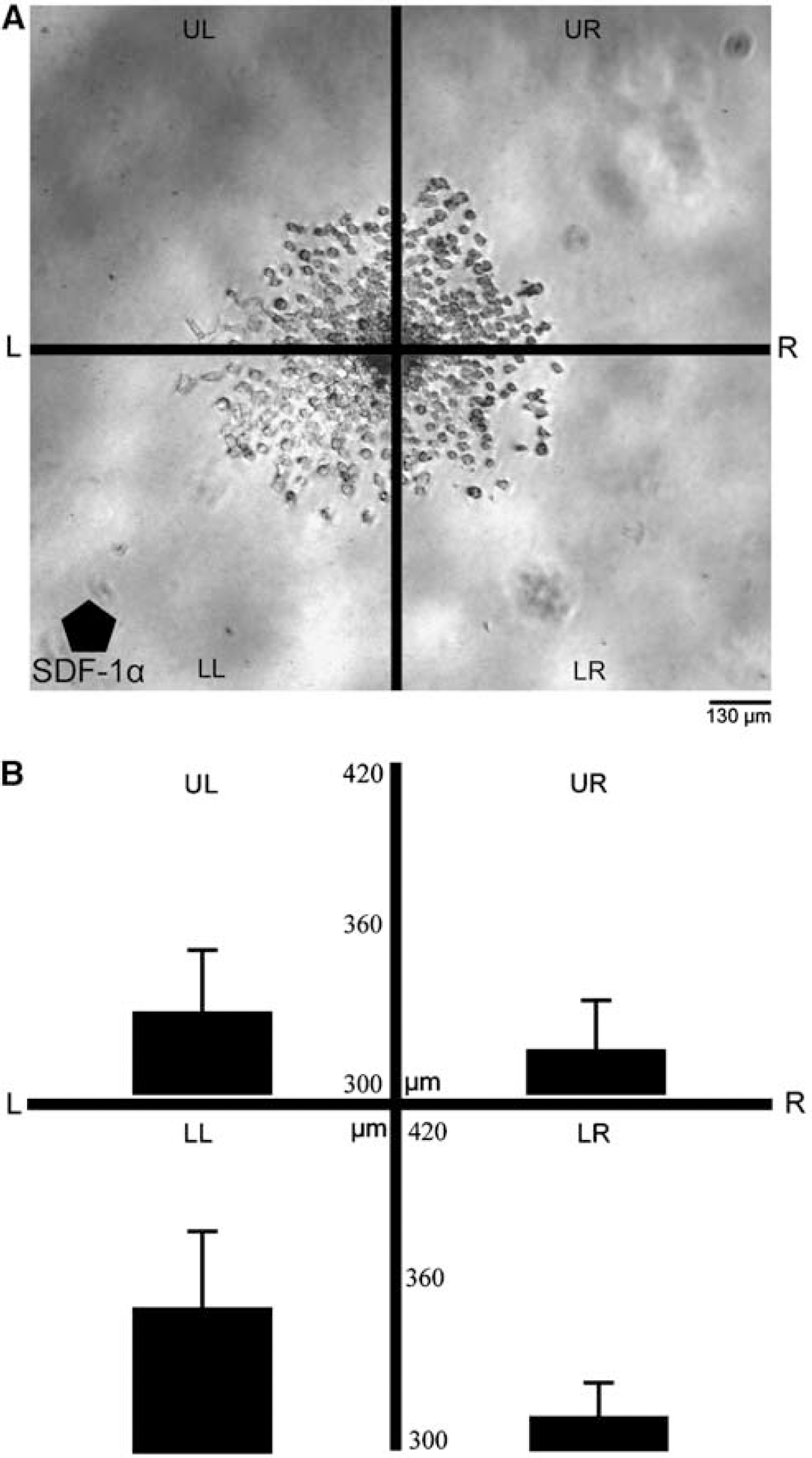
(
Immunohistochemistry
Immunohistochemistry was performed to characterize NPCs in a variety of conditions. The following primary antibodies were utilized in the present study: mouse monoclonal anti-β-tubulin III (TUJ1; 1:500; mms-435p, Covance, Princeton, NJ, USA), rabbit polyclonal anti-GFAP (1:10,000; z0334 DAKO Carpinteria, CA, USA), mouse monoclonal anti-Nestin (1:800; 556309 Pharmigen, San Jose, CA, USA), goat polyclonal anti-cd26 (1:300; sc-7704 Santa Cruz Biotech Inc., Santa Cruz, CA, USA), and mouse monoclonal anti-CXCR 4 (1:200; ab1846 Chemicon, Temecula, CA, USA). All secondary antibodies were acquired through Jackson Laboratories (Bar Harbor, ME, USA) and were used at a dilution of 1:200. For neurospheres differentiated 4 days on glass cover slips or neurospheres cultured and imaged in matrigel, cells were fixed in 4% paraformaldehyde and incubated in 1% BSA in PBS for 1 h at room temperature. After incubation with primary antibody, cells were reacted with FITC- or CY3-labeled secondary antibody for a period of 1 h at room temperature. For immunofluorescent staining of brain slices used for ex vivo assay, immunostaining was performed on frozen neurosphere sections (6 μm). The frozen sections were incubated with primary antibody overnight 4°C. FITC-conjugated secondary antibody was then applied also overnight at 4°C and then a fluorescent marker that stains the nuclei of viable cells was used to label SVZ neurosphere and brain slice tissue together. All immunofluorescent stains were viewed and imaged under a fluorescent microscope (Carl Zeiss Inc., Göttingen, Germany).
Experimental Protocol
(1) To examine whether stroke brain affects NPC migration, nonstroke (n = 24 brain slices, n = 3 rats) neurospheres were overlaid onto organotypic brain slices from rats killed 7 days after stroke (7-day stroke, n = 23 brain slices, n = 5 rats).
(2) The effects of SDF-1α on cell migration after stroke were investigated by blocking study using a neutralizing antibody against CXCR 4 (10 μg/well, MAB170, R&D Systems). Anti-CXCR 4 antibody was added to slice cultures in which NPC neurospheres were plated onto 7-day stroke brain slices (n = 15 brain slices, n = 3 rats). To verify cell viability, cultured brain slices and neurospheres were counterstained with 4‘,6‘-diamidino-2-phenylindole (DAPI) (Vector Laboratories, Burlingame, CA, USA) at the end of the experiment.
(3) The question of whether stroke increases secretion of SDF-1α was addressed by collecting brains of 7 day stroke (n = 3) and normal (n = 3) rats for homogenization and ELISA analysis.
(4) For further testing, the specificity of the response of NPC to SDF-1α/CXCR 4 complex activation, blind-well migration chamber assays were used in conjunction with SDF-1α, anti-CXCR 4 blocking antibody, and diprotin A, a drug designed to specifically increase SDF-1α-induced activation of CXCR 4.
(5) The capacity of SDF-1α to induce directionally selective migration in NPCs was explored using a time-release-dependent Hydron™ polymer capsule containing SDF-1α.
Statistical Analysis
All data are presented as mean±s.d. Data presented in figures or graphs are presented as mean±s.e. Significant differences among organotypic brain slice cultures and other migration assays were identified using a one-way analysis of variance (ANOVA). Statistical significance was set at P > 0.05 unless otherwise stated.
Results
Stroke Brain Slices Affect NPC Migration
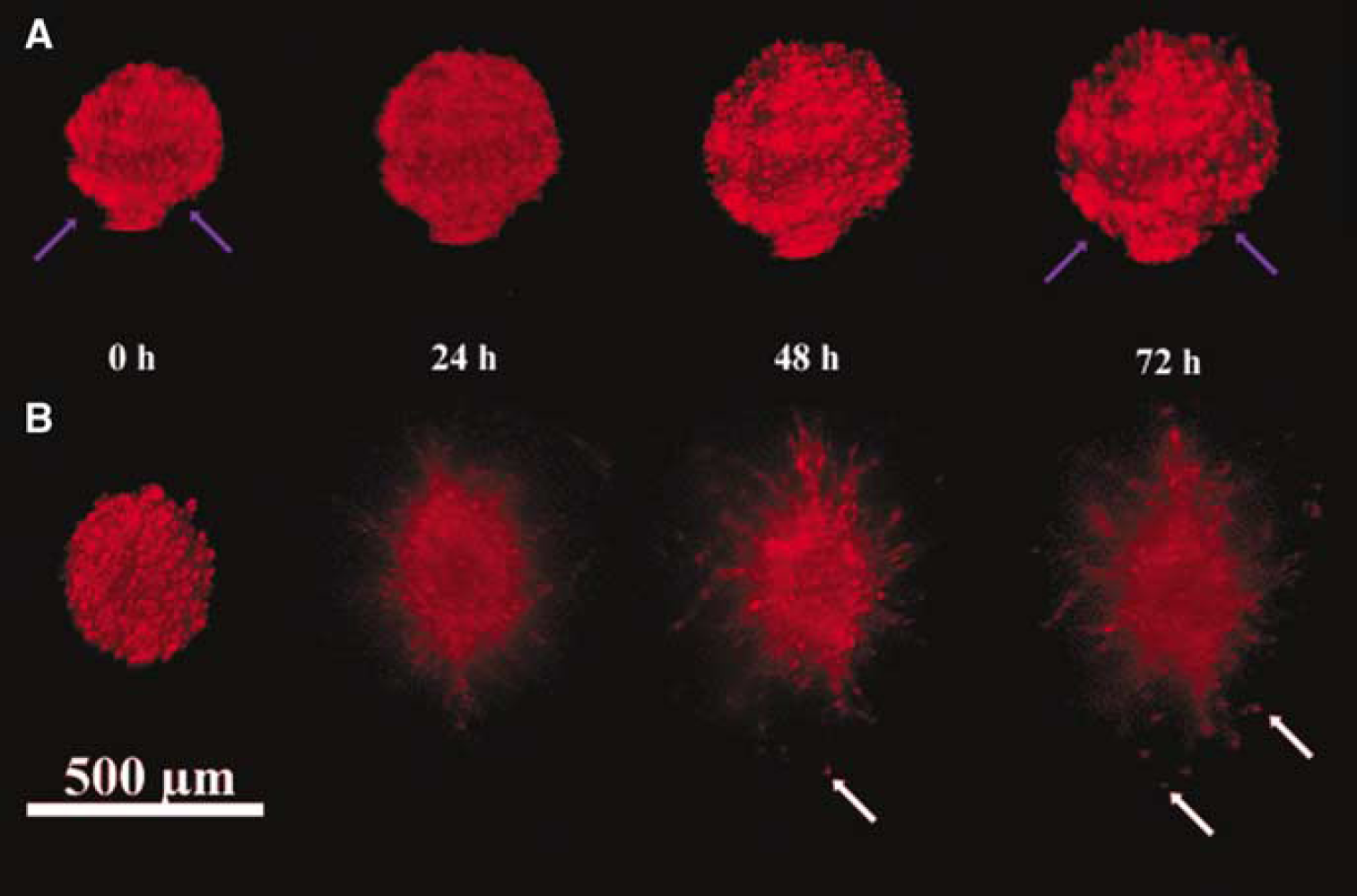
Neurospheres derived from non-stroke SVZ cells were overlaid onto nonstroke brain slices (naïve condition) and cell migration out of neurospheres was tracked for 72h in culture (n = 24). Quantitative data analysis revealed that the mean distance of cell migration after 72h in culture was 29.5± 29.5 μm. However, when nonstroke neurospheres were overlaid onto stroke brain slices (n = 23), cells in neurospheres showed more dispersal and a 2 to 3-fold increase (P > 0.01) in migratory distance (67.2 ±25.2 μm) than those cells in nonstroke neurospheres overlaid onto nonstroked brain slices, indicating that stroke brain increases progenitor cell migration. (Figure 3).
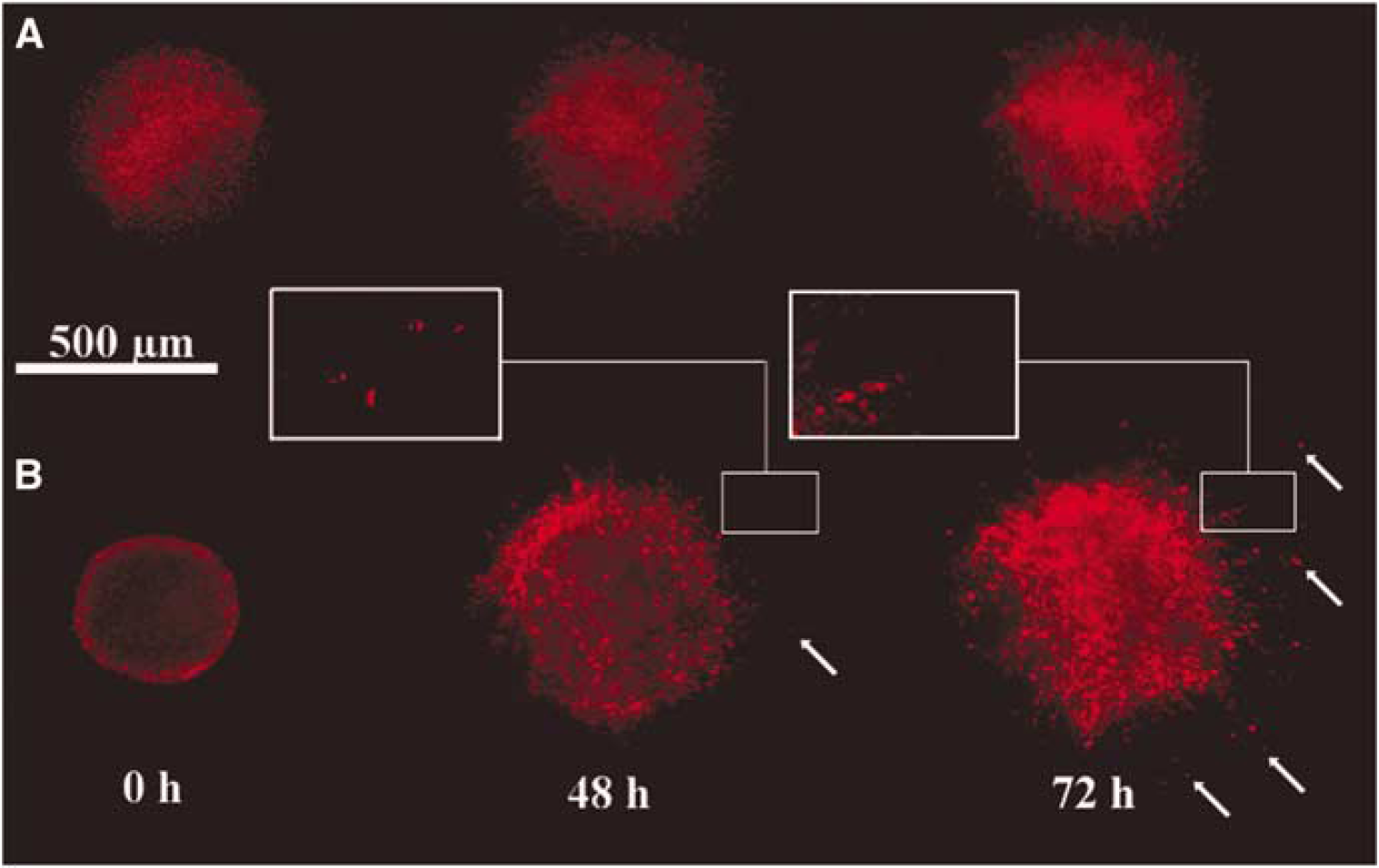
Temporal profile of progenitor cell migration. Dil fluorescently labeled neurospheres derived from nonstroke subventricular zone (SVZ) cells were overlaid onto nonstroke (
Moreover, cells in neurospheres overlaid onto mildly stroked brain slices (n = 10) traveled an average distance of 53.5±25.5 mm while cells in neurospheres overlaid onto severely stroked brain slices (n = 13) migrated 77.7±18.6μm (P > 0.01), suggesting that the severity of stroke affects progenitor cell migration. Both the mild and severe stroke group showed significantly (P > 0.05) increased migration when compared with the naïve condition (Figure 4A). Cell density, as determined by DAPI staining, shows that the number of cells within brain slices and neurospheres stayed largely unchanged over the course of the 72h culture, suggesting that cells and neurospheres are viable (Figures 4B–D).
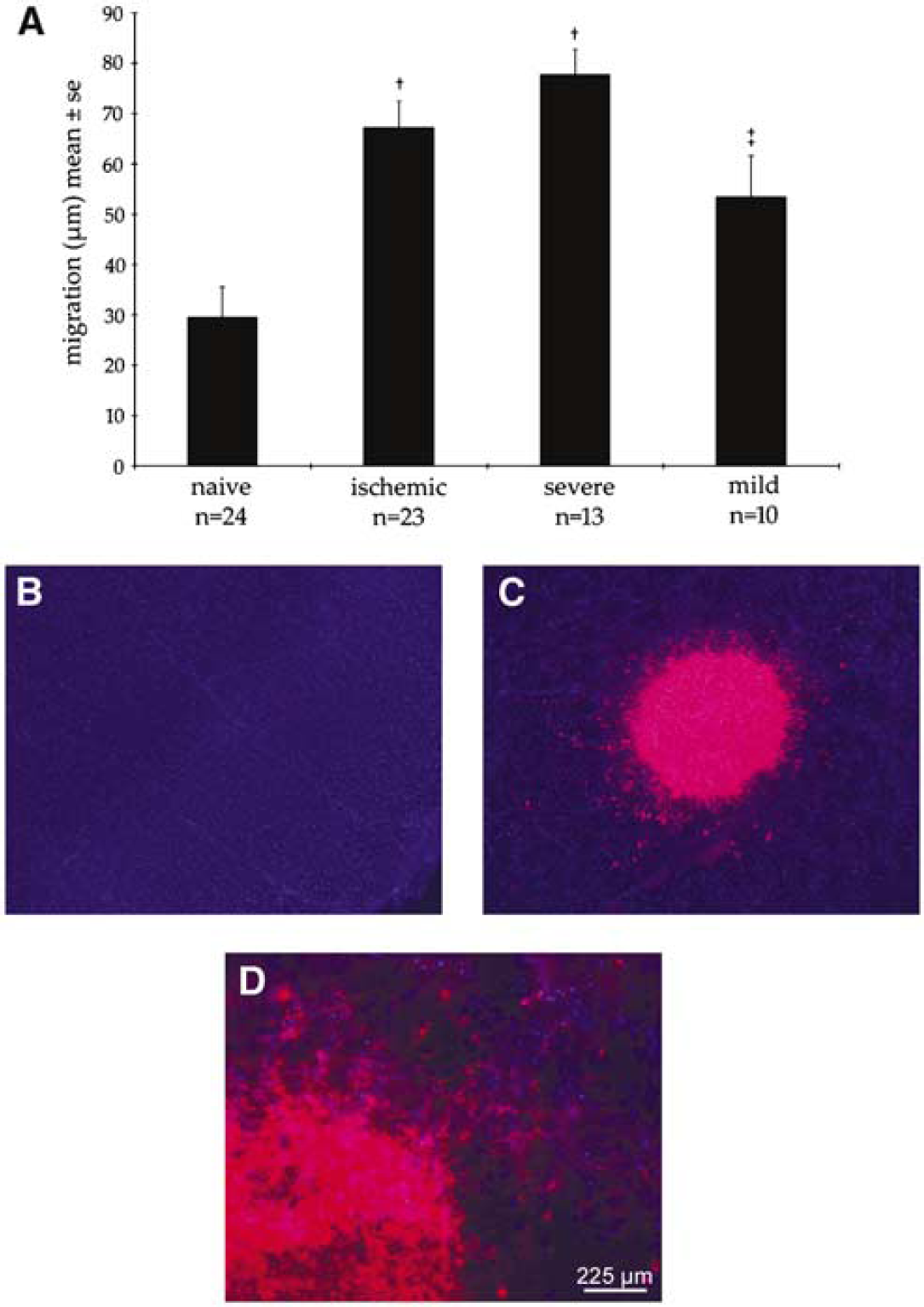
Neural progenitor cell (NPC) migration across adult organotypic brain slices is dependent on the severity of stroke (
Stroke Increases Secretion of SDF-1α and Blocking CXCR 4 Inhibits NPC Migration
To examine whether SDF-1α is involved in stroke-induced progenitor cell migration, we examined levels of SDF-1α in the brains of stroke and nonstroke rats. ELISA assay of homogenized brain revealed a 73% increase (P > 0.01) in SDF-1α expression in stroke brain (ipsilateral, 43.5±6.5 pg/ mg, n = 3) when compared with the normal brain (25.2±1.9pg/mg, n = 3). Biological activity of SDF-1α is mediated by its receptor CXCR 4 (Hill et al, 2004). We therefore considered whether NPCs in the SVZ express CXCR 4. Immunostaining showed that NPCs resembling migrating neuroblasts morphologically (arrows, Figures 5A–H) were CXCR 4 immunoreactive and double immunofluorescent staining showed that β-tubulin III positive cells were CXCR 4 immunoreactive (Figures 5I–L). In parallel to in vitro findings, double immunofluorescently stained brain sections showed that doublecortin (a marker for migrating neuroblasts) immunoreactive cells in the SVZ were CXCR4 positive (Figure 5M). These data suggest that SDF-1α/CXCR 4 may mediate NPC migration after stroke. Indeed, when nonstroke neurospheres were incubated with a neutralizing antibody against CXCR 4 before overlaying them onto stroke brain slices, cell migration out of neurospheres on stroke brain slices was significantly reduced (30.47 23.1 μm, n = 15, P > 0.05) when compared with neurospheres without treatment with the antibody (67.2 ± 25.2 μm, n = 23) (Figure 6). Blocking CXCR 4 did not affect neurosphere size or cell viability (Figure 6).
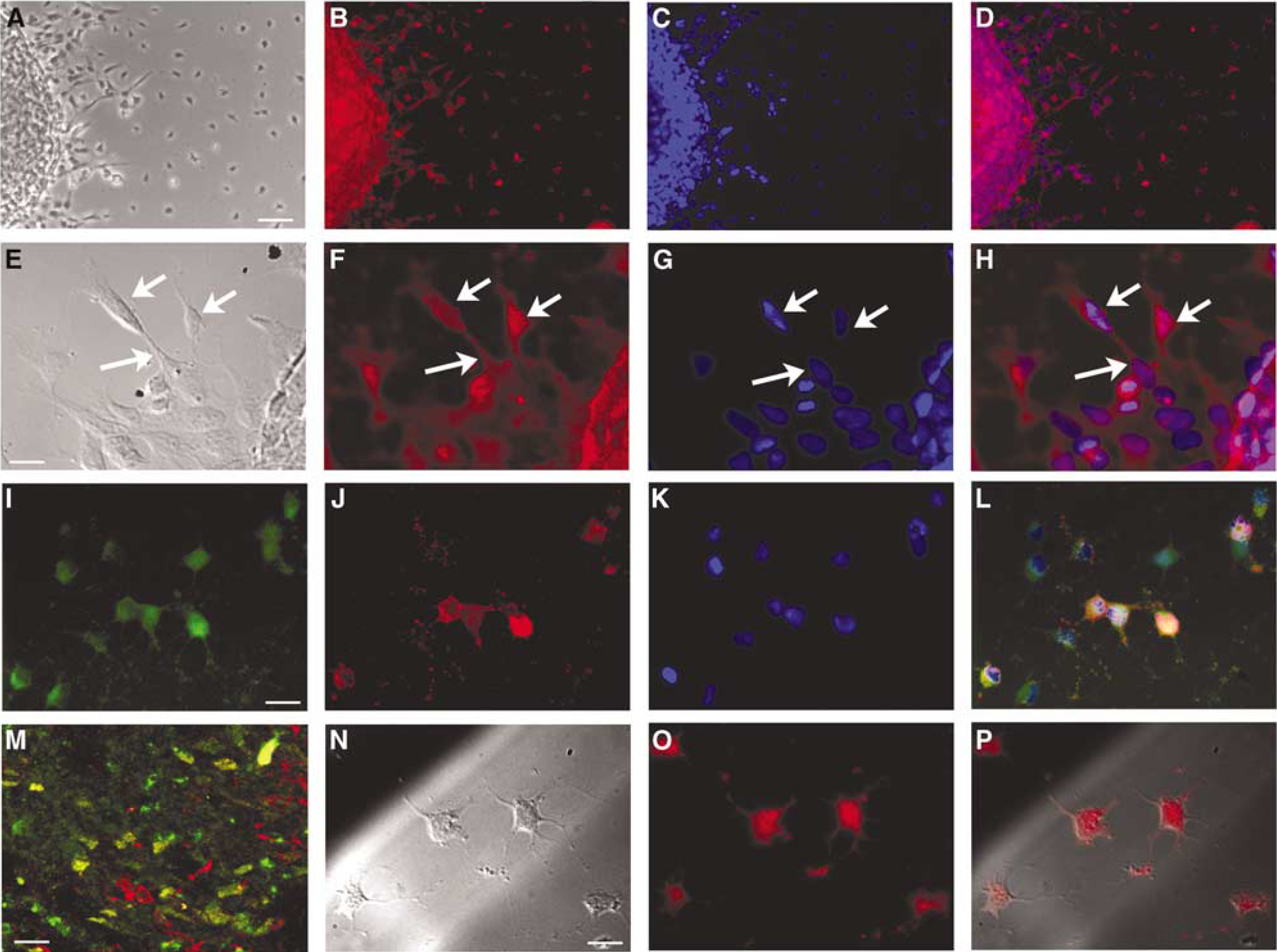
Photomicrographs (
Blocking CXCR 4 inhibits progenitor cell migration out of neurospheres. Dil fluorescently labeled neurospheres derived from nonstroke subventricular zone (SVZ) cells were placed onto 7-day stroke brain slices in the presence (
SDF-1α Specifically Induces NPC Migration
Further investigation of the role of SDF-1α in mediating NPC migration led to a series of in vitro migration assays. Briefly, NPCs were subjected to increasing levels of SDF-1α within a blind-well migration chamber system. Cells exhibited significantly increasing migration rates in a dose-dependent manner with a maximum increase at a dose of 500ng/mL (Figure 7). Treatment with the CXCR 4 neutralizing antibody significantly (P > 0.01) blocked the number of migrating cells (Figure 7). NPCs were immunoreactive for dipeptidyl peptidase IV (Figures 5N—P), an enzyme which cleaves SDF-1α near its active terminus, and disables the protein from binding and activating its CXCR 4 receptor. To further test the specificity for migration through the SDF-1α/CXCR 4 ligand—receptor complex, cells were incubated with 5 mmol/L diprotin A, an inhibitor of DPPIV. In the presence of diprotin A, incubation of NPC with SDF-1α at a dose of 250ng/mL significantly increased the number of migrating cells compared with the number with SDF-1α (250 ng/ mL) alone group (Figure 7). However, when cells were treated with both 5 mmol/L diprotin A and SDF-1α at 500ng/ml, the number of migrating cells significantly decreased (P > 0.05, Figure 7).
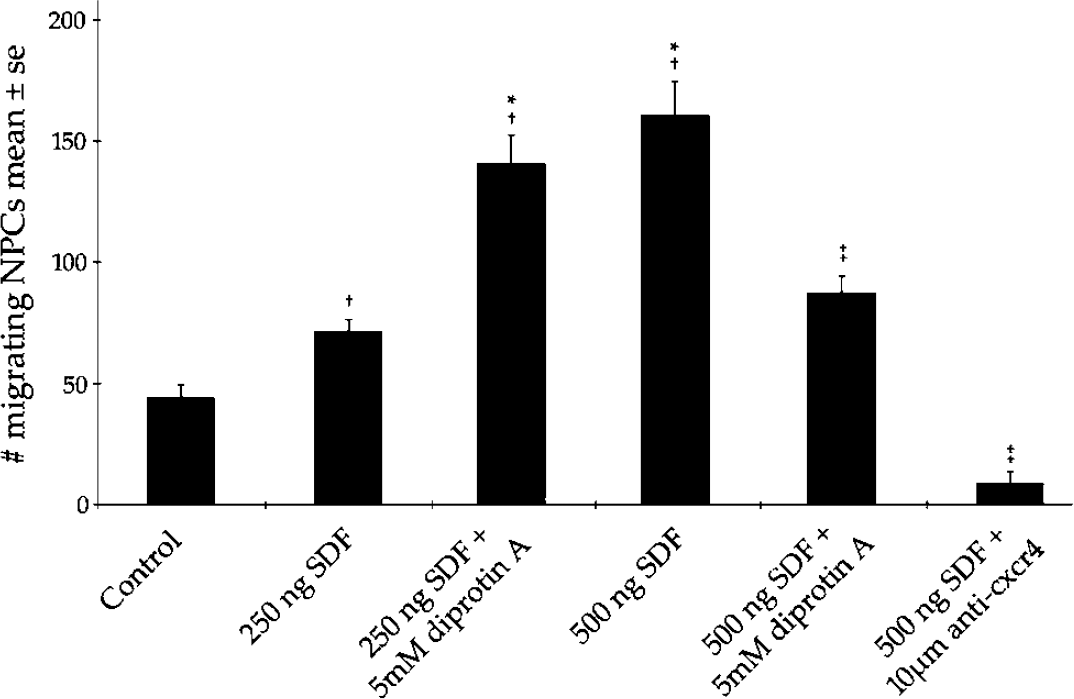
Neural progenitor cell (NPC) response to treatments that augment or abrogate activity through the stromal cell-derived factor 1 α (SDF-1α)/CXCR 4 ligand—receptor complex. Neural progenitor cell migration increases with SDF-1α treatment and decreases when blocking activity through CXCR 4. Neural progenitor cell motility is enhanced when NPCs are treated with low levels of SDF-1α and diprotin A in combination while high doses of SDF-1α and diprotin A can decrease NPC motility. † indicates a significant increase in migration with treatment (P > 0.05), †* indicates no statistical difference between marked treatment groups, ‡indicates a significant decrease in migration in comparison to SDF-1α treatment alone (P > 0.05).
SDF-1α Induces Directionally Selective Migration in NPC
To determine whether SDF-1α can induce directionally selective migration, neurospheres were overlaid onto reduced-growth factor containing matrigel in the presence of a Hydron™ polymer capsule containing SDF-1α at a dose either 125 ng (n = −6) or 250 ng (n = 6) for 24 h. After 24 h in culture, we observed increased migration in both the 125 ng SDF-1α and 250 ng SDF-1α groups when compared with controls (no Hydron™ gel, n = 6). Moreover, cells migrating out of neurospheres cocultured with 250 ng SDF-1α time-release capsules preferentially migrated (P > 0.01) in the direction of the capsule in comparison to migration distances in the direction opposite the SDF-1α containing polymer (Figure 2).
Discussion
Our data indicate that stroke increases NPC migration via upregulation of SDF-1α in ischemic brain slices (Figure 4). Blocking assays revealed that stroke-increased migration can be abrogated by an anti-CXCR 4 antibody (Figure 6). ELISA assay clearly shows that stroke brain expresses more SDF-1α than normal brain, and results from both blind-well migration chamber and time-release polymer assays indicate that SDF-1α causes NPCs to migrate preferentially down a chemotactic gradient (Figures 2 and 7). Further testing with an SDF-1α selective moderator indicated specific mediation of NPC motility through the SDF-1α/CXCR 4 ligand—receptor complex in vitro. Immunohistochemistry shows that CXCR 4 and β-tubulin III are coexpressed on NPCs and that CXCR 4 is expressed on NPCs migrating out of neurospheres (Figures 5I–L). Taken together, this data suggest that SDF-1α secreted by ischemic brain binds CXCR 4 expressed on the surface of NPCs and induces migration of NPCs out of the SVZ and possibly toward the ischemic boundary.
A consideration in choosing the brain slice assay for use in the study of NPC recruitment was the capacity of this assay to track NPCs as they migrate to the ischemic boundary across the brain slice. The modified version of Polleux and Ghosh's brain slice assay used in the current study allows for unfettered access to cells for observation as they migrate across brain, closely approximating an in vivo environment. Coronal slices taken from stroke and non-stroke adult brains support continued cell migration out of overlaid neurospheres during 72h in culture (Figures 3, 4, and 6). Coronal slices are suitable for manipulation of cell migration by blocking interactions between ligand and receptors, as exposure of NPCs to CXCR 4-neutralizing antibody abrogated NPC migratory response to SDF-1α (Figure 6). Although the treated neurosphere (Figure 6A) also increases in size, this may be because of a mechanical flattening of the three-dimensional spheroid onto the surface of the brain slice, an effect that is largely absent in the untreated neurosphere (Figure 6B) precisely because of the high level of cellular migration observed. The CXCR 4-neutralizing antibody-treated neurosphere (Figure 6A), in comparison to the untreated neurosphere (Figure 6B), undergoes little if any independent cell migration or dispersion away from the neurosphere and the untreated neurosphere (Figure 6A) maintains its original overall shape. According to ELISA analysis, ischemic tissue in our brain slice model provides a significant SDF-1α source, possibly arising from endothelial cells, perivascular activated astrocytes, and microglia, which have all been identified as expressing SDF-1α within tissue after stroke (Hill et al, 2004). It is apparent that the slice overlay assay can be used to analyze the overall migration distance of NPCs harvested from the SVZ across brain slices. Placement of neurospheres too close to the ischemic boundary for an adequate chemotactic gradient to be present within brain tissue at that location may account for our inability to quantify directionally selective migration of NPCs in the brain slice assay.
On treatment with SDF-1α in a blind-well chamber assay, NPCs exhibited a dose-dependent increase in the number of migrating cells and this response was significantly attenuated on addition of the same neutralizing antibody raised against CXCR 4 used in our brain slice experiments. It should also be noted that when cells were treated with diprotin A, an inhibitor of DPPIV, in combination with low or moderate levels of SDF-1α, the number of migrating NPCs rose significantly. Importantly, higher doses of SDF-1α may actually work to inhibit cell migration as is indicated by the drop in the NPC migration rate on treatment with 500ng/mL SDF-1α + 5 mmol/L Diprotin, a drug which specifically enhances biological activity via the SDF-1α/CXCR 4 complex. The decrease in NPC migration, which is correlated with higher doses of SDF-1α, may be because of receptor internalization after repeated activation (Signoret et al, 1997). The role of DPPIV in mediating SDF-1α-induced chemotaxis of CD34+ hematopoietic stem/progenitor cells has been thoroughly studied (Christopherson et al, 2002). We showed DPPIV expression and function on the NPC of the adult rat, suggesting a possible mechanism by which DPPIV could mediate SDF-1α-activation of CXCR 4, thus effectively arbitrating SDF-1α-induced recruitment of neuroblasts from the SVZ after stroke at the cell surface. This possibility warrants further investigation and our results may have important implications for enhancing recruitment of neuroblasts to replace stroke-damaged neurons via regulation of SDF-1/CXCR4 signaling.
Molecules that regulate the migration of NPCs in the adult brain after stroke remained largely unexplored. Stroke increases neurogenesis in the SVZ and enhances recruitment of NPCs in the SVZ to the stroke boundary in the adult animal brain (Zhang et al, 2001). SDF-1α is important for neural progenitor cell guidance and orientation in the developing mammalian brain (Bagri et al, 2002; Lu et al, 2002; Ma et al, 1998; Suzuki et al, 2001; Zou et al, 1998). Imitola et al (2004) recently report that CXCR 4-mediated activation of the p38, c-Jun, and ERK1 intracellular signaling pathways play a role in proliferation and migration of NPCs within a stroke-induced pathophysiologic state, which may mimic certain processes seen during development. Using selective PI3K inhibitors, we previously showed that the PI3K signal transduction pathway mediates neuroblast migration after stroke (Katakowski et al, 2003). In the present study, blocking interactions between SDF-1α and CXCR 4 on brain slices diminish NPC migration, suggesting that SDF-1α is providing migratory cues for NPCs in the brain slice assay. SDF-1α acts on its receptor CXCR 4 and the signal from CXCR 4 receptors can be transduced to the nucleus via the PI3K pathway (Curnock and Ward, 2003; Peng et al, 2004). Thus, it is likely that SDF-1α mediates NPC migration at least in part via the PI3K signal transduction pathway (Peng et al, 2004). Also, while we offer no data regarding the impact of SDF-1α on NPC proliferation, Akt, a downstream signaling component of the PI3K signaling cascade, has indeed been implicated in cell proliferation and survival and we propose PI3K plays at least a partial role in mediating the effects of upstream SDF-1α/CXCR4 ligand—receptor interactions. In addition to the role of SDF-1α in promoting NPC migration after stroke, other factors including slit, reelin, and epherinB2 have been implicated in the regulation of NPC migration (Adams et al, 2001). Studies that further elucidate roles for these factors in stroke-induced neuroblast migration are warranted. Identification of factors responsible for recruitment of neural progenitors to the ischemic boundary may lead to the development of therapies designed to augment brain plasticity and functional recovery after stroke.
