Abstract
In recent years, several investigators have successfully regenerated axons in animal spinal cords without locomotor recovery. One explanation is that the animals were not trained to use the regenerated connections. Intensive locomotor training improves walking recovery after spinal cord injury (SCI) in people, and >90% of people with incomplete SCI recover walking with training. Although the optimal timing, duration, intensity, and type of locomotor training are still controversial, many investigators have reported beneficial effects of training on locomotor function. The mechanisms by which training improves recovery are not clear, but an attractive theory is available. In 1949, Donald Hebb proposed a famous rule that has been paraphrased as “neurons that fire together, wire together.” This rule provided a theoretical basis for a widely accepted theory that homosynaptic and heterosynaptic activity facilitate synaptic formation and consolidation. In addition, the lumbar spinal cord has a locomotor center, called the central pattern generator (CPG), which can be activated nonspecifically with electrical stimulation or neurotransmitters to produce walking. The CPG is an obvious target to reconnect after SCI. Stimulating motor cortex, spinal cord, or peripheral nerves can modulate lumbar spinal cord excitability. Motor cortex stimulation causes long-term changes in spinal reflexes and synapses, increases sprouting of the corticospinal tract, and restores skilled forelimb function in rats. Long used to treat chronic pain, motor cortex stimuli modify lumbar spinal network excitability and improve lower extremity motor scores in humans. Similarly, epidural spinal cord stimulation has long been used to treat pain and spasticity. Subthreshold epidural stimulation reduces the threshold for locomotor activity. In 2011, Harkema et al. reported lumbosacral epidural stimulation restores motor control in chronic motor complete patients. Peripheral nerve or functional electrical stimulation (FES) has long been used to activate sacral nerves to treat bladder and pelvic dysfunction and to augment motor function. In theory, FES should facilitate synaptic formation and motor recovery after regenerative therapies. Upcoming clinical trials provide unique opportunities to test the theory.
Keywords
Introduction
In the past decade, several prominent laboratories have shown axonal regeneration in animal spinal cord injury (SCI) models, but locomotor recovery did not follow regeneration (277). For example Lu et al. (159,160) and He's group (156,282) regenerated many axons that synapsed with neurons in the lower spinal cord, but the animals did not recover walking. To explain the lack of locomotor recovery, the investigators assume that insufficient numbers of regenerated axons have formed synapses with appropriate neurons in the lower spinal cord to support locomotion. Another explanation for the lack of locomotor recovery after complete SCI and regeneration is that the animals did not learn to use their newly regrown connections. Regenerated fibers are very unlikely to be contacting the same neurons they used to. To walk again, animals must not only establish synaptic connections with appropriate lumbosacral neurons but also learn to turn on different neurons to control locomotion.
Locomotor recovery is common after incomplete SCI. Dobkins et al. (54) found that >90% of people with incomplete SCI recover independent locomotion within a year, whether they undertake treadmill or conventional overground walking training. Many groups have reported that locomotor training improves walking recovery in people with chronic incomplete SCI (92,97,99,100,115,126,143, 162,258,262–266). In 2008, Behrman et al. (19) described a 4-year-old child who recovered walking with intensive locomotor training and continued to improve 2 years after severe incomplete SCI (68).
Few investigators, however, have observed improved walking in people with “complete” SCI. Murillo et al. (181) recently described a 15 year old with complete SCI who recovered walking after locomotor training. Knikou et al. (136) trained 16 subjects with incomplete and complete SCI in 45 sessions, 1 h/day, 5 days a week, and found that the training promoted intralimb and interlimb coordination and reduced cocontraction of knee and ankle antagonists in both incomplete and complete subjects. However, the complete subjects were not walking.
In the last 3 years, several groups reported that epidural stimulation (57,58,180,244) of the spinal cord of people with chronic severe SCI restore weight-bearing (9,220) and voluntary motor function (5,98). Many clinical studies suggest that both central and peripheral electrical stimulation improve long-term motor (90) and sensory recovery (204), including standing (48), walking (96,105,130,204), and hand function (106,129).
The mechanisms underlying stimulus-induced recovery are not well understood. In this review, I will critically assess the role of training, the central pattern generator (CPG), Hebbian mechanisms of synaptic facilitation, and various stimulation paradigms used to facilitate recovery of motor function after SCI, including epidural cortical stimulation, epidural spinal stimulation, and functional electrical stimulation (FES) of peripheral nerves.
Locomotor training should activate diverse descending tracts entering the spinal cord, including regenerated motor axons. In theory, stimulation of peripheral nerve of arms and legs should activate ascending volleys of afferents that would facilitate synapses made by regenerating axons on appropriate interneurons and motoneurons. If patients were to try to move their arms or legs in synchrony with FES activating hand grasp and foot flexion, we should see improved motor control of these important muscles.
This theory should be straightforward to test in animals and in humans treated with regenerative therapies. In animals, descending volleys can be activated by either motor cortex stimulation or epidural stimulation of the spinal cord, timed to coincide with antidromic and afferent volleys elicited by peripheral nerve stimulation. In humans, FES can be used to activate hand grasp and foot flexion repeatedly, while the person attempts to move the hands or feet in synchrony. Upcoming clinical trials assessing regenerative therapies such as mononuclear cells and neural stem cell transplants provide opportunities to test this theory.
Importance of Locomotor Training
Treadmill training improves locomotor recovery in animals (124,164). For example, Ward et al. (257) studied rats that received a T10 spinal cord contusion and then trained for 1 h/day to walk. After 3 months of training, the rats improved limb kinetics, gait, and hindlimb flexor–extensor bursting patterns compared to nontrained controls. The training increased expression of nerve growth factor, but not brain-derived neurotrophic factor or neurotrophin-3. The rats not only improved locomotor function but also improved bladder function and neuropathic pain. Shin et al. (228) did gene expression profiling of the rat lumbar spinal cord at 1 and 3 weeks after contusive SCI at T9 and locomotor training. Treadmill training did not influence injury-induced upregulation of inflammation-related genes, but increased the expression of genes associated with neuroplasticity (activity-regulated cytoskeleton-associated protein, neuronal cell adhesion molecule) and angiogenesis [a disintegrin and metalloproteinase domain 8 (Adam8), tyrosine kinase with immunoglobulin-like and epidermal growth factor-like domains 1 (Tie1)]. Locomotor training increased the regeneration of the soleus muscle (124).
Wernig et al. (162,258–266) first reported that weight-supported treadmill walking improves locomotor function of people with incomplete SCI (92,97,99,100,115,126,143). Many investigators have reported beneficial effects of automated (222,270), electromechanical (22), robotic (185,234, 275), cable-driven (274), conventional treadmill training (61), daily passive cycling (45), exoskeletal robotic orthoses (110,175), and other activity-based training (157) on locomotor function and balance (67), spasticity (28,163), gait impairment (113), and respiratory function (247). Less intensive locomotor training (twice a week) also appears to be beneficial (187). Morrison et al. (178,179) estimated that locomotor training reduced lifetime care costs. Two reviews (169,176) suggested that while some of these approaches show potential for ambulatory improvement, randomized clinical trials are needed to show that these instrument-intensive programs are better than progressive overground training (55).
Despite many studies, however, the best approach, intensity, timing, and duration of locomotor training are still controversial (176). In Australia, a randomized controlled trial has started to compare regular and intense activity-based training (70). Laburuyere and van Hedel (148) studied nine subjects with chronic incomplete SCI, randomized to 16 sessions of robotic assisted gait training (RAGT) over 4 weeks followed by 16 sessions of strength training or the reverse order of strength training followed by RAGT. Strength training improved maximal walking speed (10MWT) more than RAGT, and both interventions reduced pain. Behrman et al. (19,68) reported a 4.5-year-old child who recovered walking with intensive locomotor training and continued to improve 2 years after severe incomplete SCI.
Locomotor training may help patients with complete SCI as well. Knikou et al. (136) studied 16 patients with complete and incomplete SCI after 45 locomotor training sessions, 5 days a week and 1 h/day. The training potentiated homosynaptic depression, promoted intralimb and interlimb coordination, and altered cocontraction of knee and ankle antagonistic muscles in both complete and incomplete patients. Presynaptic facilitation of soleus Ia afferents occurred only in patients with incomplete SCI. Murillo et al. (181) likewise described a 15 year old with complete SCI who recovered walking after locomotor training.
Perhaps the most impressive training effect on locomotor recovery was reported by Zhu et al. (281) in 2008. They recruited 30 subjects within several weeks of complete SCI, that is, American Spinal Cord Injury/International Spinal Cord Society Impairment Scale A (AIS/ISCOS A), decompressed the spinal cord by removing adhesions between the spinal cord and surrounding tissues and then trained the subjects to walk 6 h a day, 6 days a week, for 3 months. Before surgery, none of the subjects could stand without help. By 17 days after surgery, without rehabilitation, 10 subjects (33%) were walking using a rolling walker without assistance. Over 3 months, 18 subjects (60%) recovered walking in a rolling walker without assistance.
Dobkin et al. (54) showed that 90% of patients with motor incomplete (AIS/ISCOS C) will recover unassisted walking within a year after SCI, regardless of the type of locomotor training program. The patients were randomized to either treadmill walking or overground walking for 3 months after injury. The two groups showed no significant difference, but 92% of motor incomplete patients with AIS/ISCOS C recovered unassisted walking, while 100% of patients with AIS/ISCOS D recovered unassisted walking.
Activity Dependence of Synaptic Formation
In 1949, Donald Hebb (102) wrote on page 63 of his famous book The Organization of Behavior; a Neuropsychological Theory:
Let us assume that the persistence or repetition of a reverberatory activity (or “trace”) tends to induce lasting cellular changes that add to its stability… When an axon of cell A is near enough to excite a cell B and repeatedly or persistently takes part in firing it, some growth process or metabolic change takes place in one or both cells such that A's efficiency, as one of the cells firing B, is increased.
This rule is sometimes summarized as “cells that fire together, wire together,” but this summary is a gross oversimplification. As Caporale and Dan (37) pointed out, the synaptic modification has critical windows of tens of milliseconds and involves homosynaptic and heterosynaptic mechanisms, including long-term potentiation (LTP) due to trains of activity, release of neurotransmitters such as serotonin, and neuromodulatory heterosynaptic interactions between synapses that involve both facilitation and depression.
In 2000, Bailey et al. (10) wrote an article entitled, “Is heterosynaptic modulation essential for stabilizing Hebbian plasticity and memory?” Homosynaptic facilitation or LTP is primarily for learning and short-term memory, while heterosynaptic facilitation involves long-term memory that requires protein synthesis and synaptic formation. Bailey et al. (11) showed that synaptic connections between glutamatergic sensory neurons and motoneurons could be facilitated by heterosynaptic mechanisms or repetitive homosynaptic activation. Serotonin mediates long-term facilitation in Aplysia, converting short-term Hebbian plasticity into persistent, protein synthesis-dependent synapses. Both homosynaptic and heterosynaptic activity may be necessary to consolidate regenerated synapses, explaining why intensive locomotor activity or epidural stimulation enhances locomotor recovery, by ensuring descending activity to establish synapses on lumbosacral neurons. However, specifically timed heterosynaptic activation may be necessary to consolidate synapses for volitional activation of individual muscles.
Since the classic experiments of Hubel and Wiesel, who found that formation of ocular dominance columns requires visual experience, most scientists have accepted that neuronal activity maintains and refines synaptic connections through Hebbian and other competitive mechanisms. While synaptogenesis can proceed without neuronal activity (253) or neurotransmitter release (255), neuronal activity also regulates synaptic formation (4). Using live imaging and genetic control of tetanus toxin, Soto et al. (233) showed that increased spontaneous activity selectively increases synaptic formation in the developing retina.
In 2003, Rushton (215) proposed an interesting hypothesis. He pointed out that FES used to mimic or augment a weak or paralyzed muscle sometimes is followed by specific recovery of voluntary power. He proposed that FES may somehow promote adaptive changes in cortical connectivity, specifically that the corticospinal to neuronal synapses were a Hebb-type modifiable synapse (i.e., one that is strengthened by coincidence of presynaptic and post-synaptic activity, then FES, combined with coincident voluntary effort through a damaged pyramidal motor system should facilitate synaptic formation and modification).
In 2010, Everaert et al. (62) pointed out that long-term use of foot-drop stimulator applying FES to the common peroneal nerve improves walking performance even when the stimulator is off. They hypothesized that this therapeutic effect may be a result of neuroplastic changes in the spinal cord, related to Hebbian plasticity. They studied 10 patients with nonprogressive (such as stroke) and 26 people with progressive disorders (such as multiple sclerosis) who used a foot-drop stimulator for 3–12 months while walking in the community. They recorded surface electromyograms (EMGs) of the tibialis anterior muscle elicited by stimulating motor-evoked potentials (MEPs) using transcranial magnetic stimulation, by maximum voluntary contraction (MVC), and maximum motor wave from stimulating the common peroneal nerve. They found that FES significantly improved MEPs and MVCs by 50% and 48% in the nonprogressive and progressive groups, respectively. Walking speed also increased.
Lumbar Central Pattern Generator
A lumbar spinal cord motor center that controls locomotion was postulated over a century ago by Graham-Brown (80) who pointed out that hindlimb locomotion (then called “progression”) may be elicited in “late spinal animals” (p. 308) and that a mechanism confined to the lumbar part of the spinal cord is “sufficient to determine in the hind limbs an act of progression, which is probably very nearly a normal one … the act of progression is automatic and conditioned by the integration of reflex movements which follow each other successively, and each of which determines the stimulus which calls the following movement into being.”
Sherrington (225) had earlier noted in 1910 that cutting of superficial sensory nerves of the foot (carrying information from the skin) did not abolish the reflexive stepping and trotting. Graham-Brown (80) created a decerebrate low spinal animal preparation by cutting the spinal cord at T12 and demonstrated that regular walking motions continued and that only proprioceptive information from the muscles is necessary and sufficient for walking and trotting. Cats recover hindlimb locomotion 2–3 weeks after a hemisection and then rerecover such walking after the spinal cord is transected 3 weeks later.
In 1981, Dimitrijevic and Larsson (53) pointed out the importance of the CPG in human recovery of locomotion. In 1998, Dimitrijevic et al. (51,202) provided the first direct evidence for the presence of the CPG in the second lumbar segment of the spinal cord. Minassian et al. (173,174) showed that stepping-like movements could be activated in humans with complete SCI with extrinsic tonic input, showing that the CPG can convert continuous 5- to 15-Hz epidural stimulation to rhythmic walking activity.
The CPG can be activated by intrathecal administration of the neurotransmitter serotonin in lamprey (101), neonatal rats (43,235), and adult rats with SCI (6,231). Feraboli-Lohnherr et al. (63,64) showed the sublesional transplants of serotonergic neurons could restore locomotor function in rats with chronic SCI. In fact, Guertin et al. (89) showed that oral administration of buspirone, levodopa, tand carbidopa could activate locomotor activity in paraplegic mice. Koopmans et al. (137) studied functional recovery, lumbar serotonergic sprouting, and increased endogenous progenitors in rats exposed to delayed environment enrichment after SCI. They concluded that environmental enrichment improves interlimb coordination associated with serotonergic innervation of the L1-L2 segments, where the CPG is located.
Activation of the CPG can be nonspecific. As shown in Figure 1, descending brain and incoming sensory activity can initiate CPG activity. For example, walking starts with hip extension, knee extension, foot extension, then foot flexion, knee flexion, and hip flexion. Tail stimulation can activate cats to walk (280), and vibration applied to the foot alone can elicit step-like behavior in people with chronic motor incomplete SCI (66). Activation of CPG can initiate locomotor behavior even when subjects cannot activate individual muscles. The CPG is located at the top of the lumbar spinal cord, a shorter distance for regenerating axons to reach. Finally, CPG can initiate walking movements in both legs in the case of hemiplegia or unilateral regeneration.
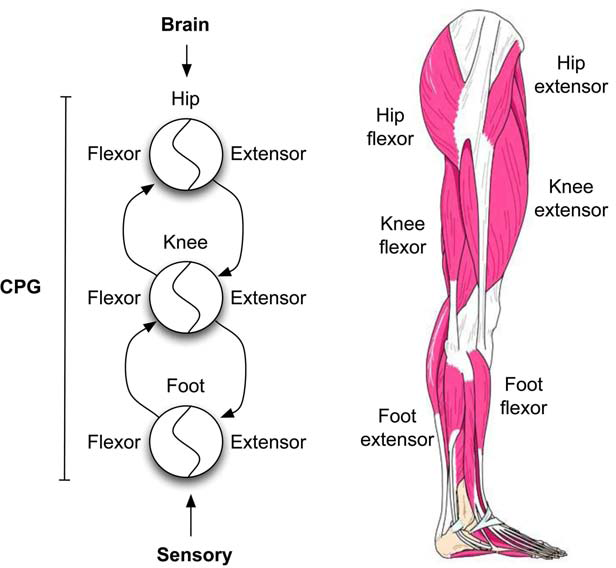
CPG for locomotor function. Located in the lumbar spinal cord (L2), the locomotor CPG contains programs that control movement of hip, knee, and foot muscles. The CPG activates walking by first causing hip extension, then knee extension, then foot extension, followed by foot flexion, knee flexion, and hip flexion. This sequence of activation can be initiated by activating the CPG and subsequent reflexive and sensory inputs resulting from the walking motions. Likewise, running, hopping, skipping, jumping, and dancing motions can be initiated, sustained, and modulated by the brain without direct activation of motoneurons. The leg muscle illustration was adapted from http://www.irishhealth.com/content/image/2262/Image1.jpg.
In 2006, Righetti et al. (209,210) pointed out that nonlinear oscillating circuits, such as the CPG, are particularly susceptible to Hebbian type of learning. They showed that the learning of rhythms is successful even when the teaching signal is noisy and that the encoded trajectory, once practiced, is stable against perturbations. Many rhythms of locomotion are intrinsic properties of oscillatory networks, including biped gaits of two-legged hop, run, and gallop (203) that can be reinforced with simple Hebbian rules (3). Therefore, it is reasonable that the CPG should be sensitive to training, that is, repeated walking motions to reinforce patterns.
Given the likelihood that walking training can reinforce locomotor rhythms and that nonspecific activation of the lumbar spinal cord can activate the CPG, it would seem to be a natural target for regenerating axons. Much evidence suggests that the CPG can be entrained even in people with complete SCI. If even a small number of axons were to reach the CPG and were able to release neurotransmitters to activate it, people should be able to recover walking. This may explain why most people with incomplete SCI recover walking (54).
Motor Cortex Stimulation
Spinal cord activity can be influenced in several ways. One way is of course to shower the spinal cord with activity in descending spinal tracts. Locomotor training should stimulate descending activity of many pathways. Another way is to stimulate the motor cortex, which in turn can activate midbrain, brainstem, and spinal cord. The motor cortex can be readily activated with epidural electrodes or transcranial electrical currents or magnetic fields. Subthreshold motor cortex stimuli are not particularly uncomfortable or painful but can activate the motor cortex without overt movements.
Motor cortex stimulation has long been used to treat pain (31,177,183,273). In 2004, Lefaucheur et al. (151) reported successful repetitive transcranial stimulation treatment of subjects with drug-resistant intractable pain due to stroke, SCI, brachial plexus injury, or trigeminal injury, compared to sham stimulation. Tani et al. (243) successfully used bilateral cortical stimulation to treat a woman with neuropathic pain after SCI. Fregni et al. (69) did a phase II clinical trial to assess transcranial direct current to alleviate central pain after SCI, showing that active anodal stimulation of the motor cortex reduced pain compared to sham stimulation.
In 2012, Wang et al. (256) showed that weak electrical stimulation of rat sensorimotor cortex causes long-term changes in spinal H-reflexes and motoneuron γ-aminobutyric acid (GABA) receptors. Within weeks of initiating stimulation, the numbers and sizes of GABAergic spinal interneurons increased, while GABAA and GABAB receptor labeling on soleus motoneurons decreased. Several months after terminating stimulation, the interneuronal and GABA terminal changes disappeared, but the H-reflex increase and receptor decreases remained. Ahmed and Wieraszko (2) found that trans-spinal direct current enhances corticospinal output and evoked release of glutamate analog dehydroascorbic acid in the spinal cord in rats. Hou et al. (113) found that combined treadmill and transcranial magnetic stimulation improved locomotor recovery in rats.
Carmel et al. (32,33,39,41,42) stimulated rat motor cortex after unilateral section of the corticospinal tract, finding that 10 days of electrical stimulation leads to increased sprouting of spinal axons that partially restores the sectioned corticospinal tract as well as cortical projection to the magnocellular red nucleus (40), which projects the rubrospinal tract to the spinal cord. The stimulation also restored skilled motor function in the forelimbs of the rats, bringing error rates of food retrieval to prelesion levels. Reversible inactivation of the stimulated motor cortex reinstated the impairment.
Roche et al. (212) have assessed the effects of transcranial direct current simulation in 15 healthy human subjects, showing that such stimulation can modify lumbar spinal network excitability and decrease cervical propriospinal system excitability. Stein et al. (236) reported similar results. Rebesco and Miller (208) showed that cortical stimulation can be used to produce Hebbian conditioning within the cortex in healthy subjects. Solopova et al. (232) used transcranial magnetic stimulation to facilitate voluntary control and triggering of stepping behavior. Benito et al. (21) used high-frequency repetitive transcranial magnetic stimulation in patients with motor incomplete SCI and found improved lower extremity motor scores. Thus, motor cortex stimulation is not only feasible but also safe and may improve motor function.
Motor cortical stimulation, however, has several drawbacks. First, having electrodes or magnetic paddles on the head requires a helmet or some kind of head covering. Second, electrical activation of the motor cortex may interfere with daily activity. Third, the effects of motor cortical stimulation are relatively nonspecific and therefore should be coupled with exercise or stimulation of the impaired limb. On the other hand, it is not difficult to imagine people placing a helmet on while exercising their impaired limbs for several hours a day. If this is effective in restoring motor control, this would be worth the trouble.
Epidural Spinal Cord Stimulation
Epidural spinal cord stimulation has long been used to treat intractable pain in patients (29,111,116,145,146,149, 177,183,188,227,230). Sayenko et al. (220) showed that lumbosacral epidural stimulation activated widespread rostral and caudal areas of the lumbar spinal cord, involving afferent and efferent pathways, in a patient with chronic SCI. Maruyama and Shimoji (165) showed that epidural spinal cord stimulation in 105 patients inhibited segmentally evoked spinal cord potentials and reduced cerebrospinal fluid norepinephrine concentrations.
Epidural stimulation activates widespread activity in the spinal cord. Sato et al. (218) compared 0.5-h and 6-h stimulation at 50%, 75%, and 90% motor threshold in rats, showing that stronger and longer stimulation showed the largest increase in mechanical withdrawal threshold. Ichiyama et al. (118) showed that epidural stimulation enhanced hindlimb stepping in rats with complete spinal cord transections. Gerasimenko et al. (75) and Ichiyama et al. (117) used a combination of epidural stimulation and quipazine administration to facilitate walking in rats.
In 1980, Sherwood et al. (226) used epidural stimulation to augment motor performance in 28 patients with upper motor neuron disorders. In 1985, Barolat-Romana et al. (18) reported that epidural spinal cord stimulation reduced intractable spasms in six patients after SCI and spasticity (15,17), while enhancing voluntary motor function (16). Campos and Dimitrijevic (36,52) have reported that epidural stimulation have a long-lasting effect on spasticity, while others (171) found that the treatment lacks long-term efficacy and is not cost-effective.
Subthreshold epidural stimulation improves locomotor function in patients with incomplete SCI. In 1998, Herman et al. (104) used epidural electrodes to activate the lumbar CPG in a patient with severe incomplete SCI. When the stimulation was on, the subject walked faster and expended less energy over 15 m. After a few months of training, the subject walked longer distances of 50–250 m and performed multiple functional tasks within the home and community with epidural stimulation (114). Other investigators have used epidural lumbar stimulation (9,144,180) to reduce the motor threshold for locomotor activation.
In 2011, Harkema et al. (57,98) reported that lumbosacral epidural stimulation restored lower extremity voluntary control in chronic motor complete patients (5,9,98,214,220). Four individuals are able to move their legs voluntarily even when the stimulation was not on. However, none of the subjects were able to walk. Although the mechanisms of the voluntary recovery are not well understood, one attractive possibility is that epidural stimulation enhances sprouting and causes both homosynaptic and heterosynaptic facilitation of synapses.
Epidural stimulation has several advantages over cortical stimulation. First, implanted epidural electrodes are unobtrusive, and the stimulus can be left on through the day and even at night. Second, especially for people with SCI with reduced motor and sensory function in the lower body, the stimulation should not interfere with daily activities. Third, although surgically placed epidural electrodes may be associated with some complications (77,158), many thousands of people have had epidural electrodes without problems. Finally, Hofstoetter et al. (107) showed that non-invasive transcutaneous spinal cord stimulation can ameliorate spasticity in people with incomplete SCI. Electrodes can also be introduced percutaneously (135). Such stimulation directly activates sensory and motor roots (213) and can also activate locomotor circuits (168,172).
The mechanisms by which epidural stimulation improve motor function are not well understood. However, one attractive hypothesis is that widespread activation of both ascending and descending activity in the spinal cord may strengthen synaptic activity in lumbosacral motor centers through Hebbian mechanisms, including walking, micturition, and defecation. Recent unconfirmed reports (46,123, 170,278) suggest epidural stimulation may improve sexual function, another programmed activity of the lower spinal cord. If confirmed, epidural stimulation is likely to become a popular approach for this purpose. At least one company has patented such a device (268).
Peripheral Nerve Stimulation
Sacral nerve stimulation (SNS) has been used since 1967 to activate bladder voiding (91,132,182,241), to treat bladder incontinence (20,34,44,59,78,108,119,120,122, 221,229), to alleviate pelvic pain (1,38,190,279), to prevent fecal incontinence (50,72–74,103,153), and to relieve constipation (56,128,133,134,161,166,167,186,189,248,252,272). In 1998, Hamdy et al. (93) discovered that transcranial motor cortex stimulation modifies anal sphincter electromyographic responses evoked by SNS in 11 healthy subjects, suggesting direct cortifugal pathways to sacral motor centers. In 2005, Sheldon et al. (224) found that SNS depressed corticoanal excitability for as long as 2 weeks after SNS cessation.
In 1980, Peckham and Mortimer (194) used implanted electrodes to stimulate forearm musculature of a quadriplegic patient. In 1981, Dimitrijevic and Larson (53) stimulated peripheral nerves to modify locomotion. In 1984, Kralj et al. (141) applied multichannel stimulation to strengthen muscles for standing and to assist gait. Petrofsky and Phillips (197) developed a feedback-based stimulator for an isokinetic leg trainer and exercise bicycle. Bajd et al. (12) used surface electrodes to restore reciprocal walking in patients with incomplete SCI. Krajl et al. (139) stimulated quadriceps to enhance locking of knee joints (140) during walking in people with incomplete SCI.
The advent of commercially available stimulators, feedback, and computers that control multichannel systems (87, 112,138,147,198,199,219) led to many devices (152,201) for improving walking (81,237), activating hand function (24) through voice commands (94), enhancing coughing (154), and even relieving seating pressures (65). Called neuroprostheses (25,193), these devices often are designed to correct specific deficits, such as foot drop (27,217,245) and hand grasp weakness (47,249,267). Other devices, such as the Parastep (71), use multichannel stimulators to activate leg muscles for standing and walking (184,238,239,242).
FES implies electrical stimuli applied to skin surface over muscles, usually activating local nerves innervating muscle. For this reason, FES cannot be used to stimulate denervated muscles (79). For many years, most physicians regarded FES as a means of activating muscle (192,195,200) to exercise paralyzed limbs (206,207) and to rebuild muscle (86,127), bone (95,150), and joints (23). Many FES devices have been approved for exercise (109,121,142,196,216).
The usefulness of FES devices varies. Taylor et al. (245) evaluated 126 patients who used a common peroneal nerve stimulator activated by a heel switch for 3.6 years and 33 who continued to use the device after 11.1 years. The stimulator allowed people with stroke to walk 45% faster and 52% of the subjects to improve their functional walking, for an average cost of £3,095. Hitzig et al. (76,105) randomized 34 subjects with incomplete SCI to thrice weekly stimulation-assisted and resistance training programs. At 12 months, the former group showed a significant increase in muscle bulk and had improved SCIM mobility subscores, but did not otherwise improve walking. Karimi et al. (130,131) reviewed the literature and found that user performance with mechanical orthoses was generally better than hybrid FES orthoses.
Several recent clinical trials, however, have reported that FES combined with activity-based therapies can contribute to walking recovery in people with chronic SCI. Jones et al. (125) randomized 48 subjects with chronic incomplete SCI to early or delayed activity-based therapy, including developmental sequencing, resistance training, repetitive pattern motor activity, and task-specific locomotor training for 9 h a week for 24 weeks. FES was part of the training. Trained subjects had significant increases in total motor scores and lower extremity motor scores, as well as walking speeds, and total distances walked. Kapadia et al. (129) studied eight participants with subacute cervical SCI. Five received 39 h of FES training of hand function over 13–16 weeks and three did not. The FES-treated group improved their hand scores and self-care scores. Hoffman et al. (106) compared FES and somatosensory stimulation and found that both were associated with improved hand use and corticomotor control in subjects with chronic tetraplegia.
Mechanisms of Fes-Mediated Motor Control
Peripheral stimulation has central effects (26,88,211,246). In 1992, Ragnarsson (205) pointed out that FES suppresses spasticity. FES also causes endocrine changes, including increases in β-endorphin-like immunoreactivity and cortisol levels (82,251). Likewise, FES induces autonomic dysreflexia (7). In 1994, Barbeau and Rossignol (14) proposed that FES should have long-term beneficial effects on locomotor recovery.
Many studies have reported that FES can improve long-term motor (90) and sensory recovery (204) in people, including standing (48), walking (105,130), and hand function (106,129). In 2007, Hardin et al. (96) reported a 22-year-old patient who was 18 months after a C5–6 incomplete (AIS/ISCOS C) SCI and had an implanted eight-channel FES system for walking. After 12 weeks of overground locomotor training, his maximal walking distance increased from 14 m to 309 m, maximal walking speed increased by 10-fold, and the physiological cost index fell fivefold. Before implantation of stimulating electrodes, he underwent 36 preparatory sessions using surface FES in a robotically assisted treadmill device that bore 60% of his body weight.
In 2014, Possover et al. (204) described four patients who had neuroprosthetic devices implanted to assist locomotor training combined with continuous low-frequency pelvic lumbosacral stimulation. Two subjects were sensory incomplete (AIS/ISCOS B), one was motor incomplete (AIS/ISCOS C), and one was complete with flaccid paralysis (AIS/ISCOS A). All four subjects recovered sensory and voluntary motor function: three recovered voluntary weight-bearing standing and walking a few meters using a walker without FES, one patient was able to walk 900 m with two crutches and electrical stimulation and 30 m without stimulation.
Edgerton et al. (68) have long claimed that animals with complete spinal cord recover weight-bearing stepping capability without reconnection of supraspinal tracts. Askari et al. (8) studied the timing effects of electrical stimulation during robotic treadmill training (RTT) of rats with severe SCI. The rats that had FES synchronized to RTT had much more (112%) EMG activity during the early swing phase of the gait cycle, exhibited burst-to-step latencies that were shorter (41%), and had greater ankle flexion than rats that received random stimulation. Tillakaratne et al. (250) reported that such stepping recovery in rats after complete neonatal spinal cord transection is not due to regrowth across the injury site.
FES not only stimulates peripheral motor axons to activate muscles but also sensory axons that return to the spinal cord and activate both motoneurons and interneurons, as well as the brain. If sensory activation coincides with descending supraspinal activity, heterosynaptic facilitation of synapses on spinal cord motoneurons and interneurons may occur. In addition, peripheral nerve stimulation also cause antidromic activation of motoneurons and Renshaw neurons (254), which may facilitate synapse formation (13,35,60,223,269) and release neurotransmitters that facilitate synaptic consolidation (240). Figure 2 illustrates how descending supraspinal activity and incoming afferent activation may facilitate synapses. Hebbian facilitation of synapses can occur at multiple levels in the cortex, brainstem, and spinal cord, as shown in Figure 3.
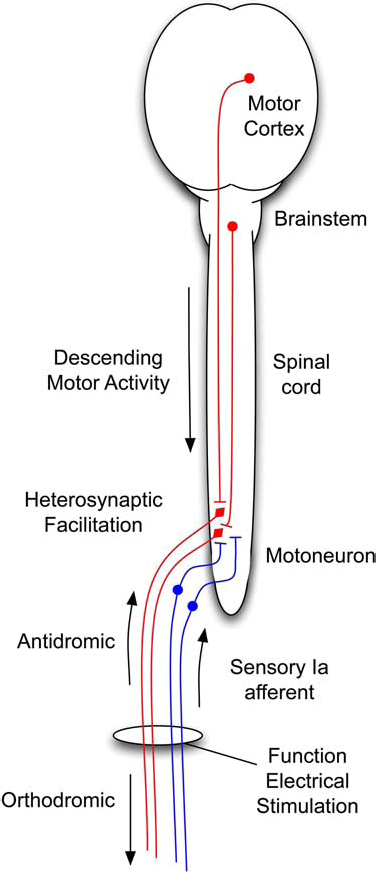
Diagram of spinal cord circuits that may be affected by functional electrical stimulation. Action potentials activated by peripheral nerve stimulation descend to the muscles, ascend antidromically to activate motoneurons, and ascend orthodromically in sensory Ia afferents to enter the spinal cord and activate interneurons and motoneurons. Descending supraspinal tracts from motor cortex and brainstem activate the motoneurons and interneurons (not shown).

Multiple levels of Hebbian learning in the brain and spinal cord. Hebbian learning can occur in the brain, brainstem, and spinal cord. Cortical stimulation (A) can reinforce incoming sensory activity coming from the dorsal column pathway through the nucleus gracilis and thalamus. Brainstem cell stimulation (B) can reinforce descending connections to the central pattern generator. Epidural stimulation or peripheral nerve stimulation can reinforce descending connections to motoneurons (C) or to lumbosacral spinal cord (D).
Not all FES-induced effects on the central nervous system are desirable. In 1986, Woolf and Wall (271) showed that stimulation of C-fibers can have long-lasting effects on flexor responses. Young et al. (276) described how neonatal hindpaw injury disrupts acquisition of instrumental responses in adult spinal cords. Grau et al. (49,83–85, 155,191) showed that noncontingent painful stimulation of the legs markedly impairs locomotor learning. The timing of exercise therapy and intervention is important (30).
When combined with appropriate exercises synchronized to the timing of stimulated activity, FES should induce long-lasting changes in motor function, especially in situations when new synapses are being formed by regenerating axons. The theory that FES stimulation can induce Hebbian-type facilitation of synapses formed by regrowing axons can be tested in upcoming clinical trials that are planning to test regenerative therapies. Not only will FES and exercise improve the results of such trials but also the trials should be able to ascertain whether Hebbian mechanisms are involved in FES-induced recovery of motor function.
Conclusions
Electrical stimulation of the motor cortex, the spinal cord, and peripheral nerves has long-lasting effects on motor function in people with SCI. Many investigators have reported beneficial effects of locomotor training after SCI. Despite many studies, the best approach, timing, duration, and intensity of locomotor training are still controversial. A few patients with complete SCI have been reported to recover walking.
In 1949, Donald Hebb (102) presciently proposed that close timing of activation of a target neuron by two neurons enhances synaptic connections of the neurons with the target neuron. This Hebbian rule, sometimes summarized as “cells that fire together, wire together,” has been extended to include heterosynaptic and homosynaptic facilitation and depression. Many studies have confirmed that neuronal activity regulates synaptic formation and consolidation.
The lumbar spinal cord contains the CPG. Postulated more than a century ago by Graham-Brown (80) and Sherrington (225), the CPG plays an important role in locomotor recovery in humans. The CPG can be nonspecifically activated by epidural and peripheral nerve stimulation, and even by intrathecal and oral neurotransmitter administration. It is an obvious target locomotor recovery mechanism.
Spinal cord activity can be influenced by stimulating the motor cortical, spinal cord, and peripheral nerves. Motor cortex stimulation has long been used to treat pain. Such stimulation affects spinal reflexes, changes spinal circuits, enhances sprouting and synapse formation, and restores skilled forelimb function in rats after corticospinal lesions. Transcranial magnetic stimulation improves lower extremity motor scores in people with SCI.
Epidural stimulation has long been used to treat intractable pain and spasticity, by evoking widespread spinal cord and peripheral nerve activation. Subthreshold epidural stimulation reduced the threshold for activation of the CPG. In 2011, Harkema et al. reported that lumbosacral epidural stimulation can restore lower extremity voluntary control (98). Others have reported that the spinal cord can be activated with electrodes placed on the skin surface.
Peripheral nerve stimulation has similar effects to epidural stimulation. SNS, for example, has been used since 1967 to activate bladder voiding, to treat bladder incontinence, to alleviate pelvic pain, to prevent fecal incontinence, and relieve constipation. FES has been used to stimulate arm and leg activity for exercise and to augment function. For many years, physicians considered FES as an electrical means of exercise.
FES can have long-term effects on motor recovery. In theory, FES should facilitate and consolidate synapses formed by regenerating axons. This hypothesis should be readily testable in animal studies and human clinical trials involving regenerative therapies. For example, it would be of interest to assess the effects of FES stimulation on hand grasp and foot flexors synchronized with voluntary efforts.
Footnotes
Acknowledgment
The author declares no conflict of interest.
