Abstract
Neuropathic pain following spinal cord injury (SCI) is a devastating disease characterized by spontaneous pain such as hyperalgesia and allodynia. In this study, we investigated the therapeutic potential of ESC-derived spinal GABAergic neurons to treat neuropathic pain in a SCI rat model. Mouse embryonic stem cell–derived neural precursor cells (mESC-NPCs) were cultured in media supplemented with sonic hedgehog (SHH) and retinoic acid (RA) and efficiently differentiated into GABAergic neurons. Interestingly, low doses of SHH and RA induced MGE-like progenitors, which expressed low levels of DARPP32 and Nkx2.1 and high levels of Irx3 and Pax6. These cells subsequently generated the majority of the DARPP32- GABAergic neurons after in vitro differentiation. The spinal mESC-NPCs were intrathecally transplanted into the lesion area of the spinal cord around T10–T11 at 21 days after SCI. The engrafted spinal GABAergic neurons remarkably increased both the paw withdrawal threshold (PWT) below the level of the lesion and the vocalization threshold (VT) to the level of the lesion (T12, T11, and T10 vertebrae), which indicates attenuation of chronic neuropathic pain by the spinal GABAergic neurons. The transplanted cells were positive for GABA antibody staining in the injured region, and cells migrated to the injured spinal site and survived for more than 7 weeks in L4–L5. The mESC-NPC-derived spinal GABAergic neurons dramatically attenuated the chronic neuropathic pain following SCI, suggesting that the spinal GABAergic mESC-NPCs cultured with low doses of SHH and RA could be alternative cell sources for treatment of SCI neuropathic pain by stem cell-based therapies.
Introduction
Spinal cord injury (SCI) results in numerous devastating events including loss of motor function below the level of lesion and central neuropathic pain (45). Neuropathic pain following SCI is characterized by spontaneous pain syndromes including hyperalgesia (exaggerated responses to painful stimuli) and allodynia (pain elicited by normally nonpainful stimuli) (45). Approximately half of patients with SCI suffer from chronic neuropathic pain, which predominantly occurs at or below the level of the lesion (16). These pain syndromes are long-lasting and may contribute to persistent problems in the rehabilitation of SCI patients, but the underlying mechanism has not fully been elucidated. Despite various therapeutic approaches for SCI, adequate control of central pain has not yet been achieved (47).
Damage to the spinal cord leads to plastic changes including anatomical, physiological, and molecular changes within the cord and at supraspinal levels. Many studies have indicated an increased excitability of spinal dorsal horn neurons (18) due to the loss of inhibitory influences involving γ-aminobutyric acid (GABA), which acts on the interneurons in the spinal dorsal horn (34) and contributes to the development and persistent neuropathic pain following SCI (12,15,34). For example, spinal ischemic injury results in behavioral and neuronal hypersensitivities that are associated with reduced GABA immunoreactivity in the spinal dorsal horn (24,50). Previous studies have shown that the activation of spinal GABA receptors can alleviate behavioral hypersensitivity to mechanical stimuli in both ischemic and hemisected SCI rats (19,24). In particular, Gwak et al. demonstrated an analgesic effect of intrathecally (IT) injected GABAA or GABAB receptor agonists in animals with SCI; this analgesic effect is blocked by GABA receptor antagonists (19). This result suggests that the recovery of GABAergic inhibitory tone and GABAergic neurons in the lesion area of the spinal cord may be therapeutic for neuropathic pain following SCI. However, the pharmacological approaches targeting the GABAergic system have had limited success due to their side effects, which include drowsiness and muscle weakness (38).
Many studies have used GABAergic cells as an alternative therapeutic source for treatments for various neurogenic diseases and have shown that neuropathic pain was alleviated (26–28,36). Neuropathic pain in the lesion area of the spinal cord was alleviated by transplantation of GABAergic neurons rather than spinal GABAergic neurons. In addition, stem cells have been directly injected into the spinal cord lesion sites, although this may cause further injuries due to injection into the lesion area or the expanding cavity of the lesion area becoming filled with cerebrospinal fluid (CSF).
In this study, we investigated the therapeutic effects of spinal GABAergic neurons from mouse embryonic stem cells (mESCs) on central neuropathic pain following contusive SCI. As an alternative method for SCI treatment, the spinal GABAergic cells were transplanted into the lesion area of the spinal cord in SCI rats by IT transplantation, using a catheter to increase the survival rate of the transplanted cells (2,3,31,35). After transplantation, the changes in mechanical hypersensitivity were examined both at and below the level of transplantation to observe the alleviation of neuropathic pain by transplanted spinal GABAergic neurons. The mESC-derived neural precursor cells (mESC-NPCs) were differentiated into spinal GABAergic neurons in vitro and subsequently transplanted into an SCI rat model using an IT method, which is generally considered noninvasive. The central neuropathic pain in the SCI rat model was efficiently reduced or prevented, but motor function was not improved.
Materials and Methods
Maintenance and Differentiation of mESCs
The mESC line J1 (purchased from the American Type Culture Collection, Manassas, VA, USA) was maintained as described previously (7,25). In brief, ESCs were cultured with ESC medium consisting of Dulbecco's modified minimal essential medium (DMEM) containing 10% horse serum (Sigma-Aldrich, St. Louis, MO, USA), 2 mM L-glutamine (Gibco, Carlsbad, CA, USA), 0.1 mM MEM NEAA (Gibco), 10 mM β-mercaptoethanol (Gibco), 500 U/ml LIF (Millipore, Billerica, MA, USA), and penicillin/streptomycin (Gibco). We used the five-stage in vitro differentiation procedure to generate GABAergic neuronal cells based on a modification of previously published methods (29). In brief, mESCs were differentiated as embryoid bodies (EBs) on nonadherant bacterial dishes for 4 days in EB culture medium consisting of DMEM supplemented with 10% fetal bovine serum (FBS; Gibco), 2 mM L-glutamine, 0.1 mM MEM NEAA, 10 mM β-mercaptoethanol, and penicillin/streptomycin. The EBs were transferred to gelatin-coated dishes. After 24 h in culture, differentiation of EBs was initiated by replacing the medium with serum-free insulin/transferrin/selenium (ITS; Gibco) medium for another 10 days, and the differentiated cells were characterized with markers and genes for three germ layers. For in vitro differentiation of GABAergic neurons, 200 ng/ml sonic hedgehog (SHH) (R&D Systems, Minneapolis, MN, USA) and 1 μm/ml retinoic acid (Sigma-Aldrich) were added to mESC-NPCs (stage 4) for 4 days (27,32,43), and these cells were allowed to fully differentiate into spinal GABAergic neurons for 14 days.
Lentivirus Construction and GFP+ Cell Sorting by FACS
The self-inactivating (SIN) lentiviral vectors (25,41) used in this study were based on the original pHR' CMV-lacZ vector previously described (37). These SIN lentiviral vectors contain the Woodchuck hepatitis virus posttranscriptional regulatory element (WPRE) as well as a ubiquitin C promoter as described before (41). The vectors were packaged in 293T cells (Life Technologies, Grand Island, NY, USA) by cotransfection with four expression vectors encoding the VSV-G envelope, Gag-Pol, Rev, and Tat. Three days after transfection, vector particles were harvested from the cultured DMEM (Hyclone, South Logan, UT, USA) medium containing 10% FBS (Hyclone), penicillin (100 IU/ml), and streptomycin (100 mg/ml; Gibco) by ultracentrifugation and titrated using 293T cells. To determine the titers, two independent methods were used. To estimate the concentration of functional transducing units (TU/ml), FT-293 cells were transduced using serial dilutions of an aliquot of each vector preparation. After 72 h, the cells were harvested and evaluated by flow cytometric analysis (25,41). Undifferentiated and differentiated ESCs were transduced for 3 h in a volume of 300 ml with lentiviral vectors (3.3 × 105 TU/ml; MOI = 1) in the presence of 2 mg/ml polybrene (Sigma-Aldrich). Media was changed on the following day.
Control (mock-transduced) and vector-transduced ESCs were dissociated by trypsinization with trypsin-EDTA (Gibco) and pelleted, washed, and resuspended in phosphate-buffered saline (PBS; Hyclone) containing 2% FBS (Hyclone), penicillin (100 IU/ml), and streptomycin (100 mg/ml; Gibco), and filtered using a 70-mM cell strainer (BD Falcon, San Jose, CA, USA). Green fluorescent protein (GFP)-expressing cells were sorted by FACSVantage SE (BD Biosciences, San Jose, CA, USA) on the basis of their GFP fluorescence signals. Flow cytometric (FACS) analysis of GFP-expressing cells was performed on a FACScan analyzer (BD Biosciences) according to their fluorescence intensity levels. Non-GFP-expressing cells were used as negative control for background fluorescence.
Immunocytochemistry
For the immunofluorescence assay, cells grown on coverslips were fixed in 4% paraformaldehyde (Sigma-Aldrich)/PBS (Hyclone) for 20 min at 25°C and then permeabilized with 0.01% Triton X-100 (Sigma-Aldrich) in PBS for 5 min. Cells were rinsed in PBS, blocked for 30 min with PBS containing 10% normal donkey serum (Millipore), and incubated overnight at 4°C with rabbit anti-GABA (1:10,000; Sigma-Aldrich), mouse anti-GABA (1:1,000; Sigma-Aldrich), rabbit or mouse anti-Tuj1 (1:500; Covance, Princeton, NJ, USA), mouse anti-MAP2 (1:500; Sigma-Aldrich), rabbit anti-GAD65/67 (1:500; Millipore), rabbit anti-DARPP32 (1:1,000; Chemicon, Billerica, MA, USA), rabbit anti-5-HT (1:500; Sigma-Aldrich), sheep anti-TH (1:500; Pel-Freez, Rogers, AR, USA), mouse anti-nestin (Rat-401) (1:250; Cell Signaling Technology, Danvers, MA, USA), and rabbit anti-SOX1 (1:300; Abcam, Cambridge, MA, USA) antibodies. Cells were rinsed three times with PBS and then incubated for 60 min at room temperature with Alexa Fluor 488- or 594-conjugated secondary antibodies (1:1,000 in PBS; Invitrogen, Eugene, OR, USA). After further rinsing, nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI, 1:10,000 in PBS; Sigma-Aldrich). Images were captured using a confocal microscopy FV1000 FluoView camera (Olympus, Tokyo, Japan), and quantitative analysis was performed using TissueQuest software (TissueGnostics, Tarzana, CA, USA).
RT-PCR
Total RNA was extracted with TRIzol (Molecular Research Center, Cincinnati, OH, USA) following the manufacturer's instructions and then reverse transcribed to cDNA using M-MLV reverse transcriptase (Fermentas, Waltham, MA, USA) and an oligo (dT) primer. PCR products were electrophoresed on 1.6% agarose gels. The primer sequences are listed in Table 1.
RT-PCR Primers

Experimental Animals and Contusive SCI
All animal protocols were approved by the Korea University Institutional Animal Care and Use Committee (KUIACUC-2013-102). Adult male Sprague–Dawley rats (200–220 g, n = 24; Orient Bio Inc., Seoul, Korea) were used in this study. Animals were housed under a 12 h/12 h light/dark cycle (08:00–20:00) with free access to water and food.
Under isoflurane (Hana Pharm, Seoul, Korea) anesthesia (2% isoflurane and 98% O2), contusive SCI was performed at T12 using the NYU Impactor (New York University, W. M. Keck Center, Piscataway, NJ, USA). A laminectomy was performed, and the spinal column was stabilized by clamping the T10 and T12 spinous processes with tissue forceps. A 10-g weight was released onto the exposed spinal cord (the T12 spinal segment) from a 12-mm height. After SCI, the overlying muscles and skin were closed in layers with 4–0 silk. Postoperative treatments included saline (2.0 ml, subcutaneous injection) for rehydration. Bladders were manually expressed twice a day until spontaneous urination returned. Unasyn (ampicillin/sulbactam, 100 mg/kg; Pfizer, Seoul, Korea) was injected intraperitoneally (IP) once a day to prevent urinary tract infection.
Intrathecal Catheterization and Cell Transplantation
Two weeks after SCI, to identify the effect of IT-injected cells in SCI rats, a sterile catheter was introduced through an incision in the atlanto-occipital membrane. Under isoflurane anesthesia (4% isoflurane and 96% O2), sterilized PE 10 tubing was inserted and threaded down to the lesion site of the spinal cord. The location of the IT catheters was confirmed with Evans blue (Sigma-Aldrich) injection after the experiment was finished.
Neuropathic pain in rats is developed around 2 weeks and is fully established 3 weeks after contusive SCI (10,48). In the present study, we transplanted mESC-NPCs into SCI rats 3 weeks postinjury to investigate effects of mESC-NPCs on neuropathic pain following SCI (1 week after IT catheterization). Prior to transplantation, prebehavioral assessments for pain and motor function in 24 rats with SCI and catheterization were performed. All behavioral tests in this study were assessed by an experimenter who was blinded to the treatment groups. Rats with SCI were excluded if they did not develop neuropathic pain; a previous study has shown that 67% of rats with contusive SCI develop allodynia (44) or had a deficit of motor function by IT catheterization (n = 10). Fourteen rats included were randomly allocated into saline or cell injection group using random allocation software. Either saline or cells (n = 7, respectively) were injected under light anesthesia (a mixture of 2% isoflurane + 98% O2). For cell injections, 1 × 106 mESC-NPCs in N2 medium (DMEM/F12 containing N2 supplement; Gibco) were injected 3 weeks after SCI into the subarachnoid space via PE 10 tubing inserted to the T10–T11 spinal segments. As a control, saline was injected at the same time points. To improve the survival of transplanted cells, the immune suppressor cyclosporine (IP, 10 mg/kg; Chong Kun Dang, Seoul, Republic of Korea) was injected daily from the day of transplantation until sacrifice.
Below-Level Mechanical Sensitivity Test
Below-level pain following SCI was assessed by measuring the threshold of brisk paw withdrawal response to graded mechanical stimulus with a series of von Frey filaments (0.41, 0.70, 1.20, 2.00, 3.63, 5.50, 8.50, and 15.10 g; Stoelting, Wood Dale, IL, USA). The rat was placed under a transparent plastic dome (28 × 28 × 10 cm) on a metal mesh floor, and a von Frey filament was applied to the plantar surface of the foot. The von Frey filament was applied for 3–4 s to each hind paw while the filament was bent. The 50% withdrawal threshold was determined using the up-down method (6), beginning with the 2.0 g (4.31 mN) strength of filament, in the middle of the series of eight von Frey filaments with logarithmically increasing stiffness (0.41–15.10 g). A withdrawal response led to presentation of the next weaker stimulus, and lack of withdrawal led to presentation of the next stronger stimulus. Stimuli were presented at intervals of several seconds. A brisk foot withdrawal to von Frey application was regarded as a positive response. Interpolation of the 50% threshold was performed according to the method of Dixon (11).
Vocalization Threshold Test of Lesion Area
To investigate the pain around the lesion area, the vocalization threshold to graded mechanical stimulus with a series of calibrated von Frey filaments (10, 15, 26, 60, 100, 180, and 300 g; Stoelting) was measured. The rats' torsos were shaved, and 12 dots were placed on typical allodynic skin areas following SCI (24); four dots were placed 0.5 and 1 cm to the left and right sides of the T10, T11, and T12 vertebrae. During the examination, rats assumed a prone posture, and the experimenter gently held the rat's head and masked its eyes. The filaments were applied to the dots until the filament became bent. Although the rats showed several reactions to the stimuli, the threshold to vocalization was the only response recorded. The pressure that induced vocalization responses to more than 60% (three or more of five trials) of stimuli was recorded. A lack of vocalization led to presentation of the next stronger stimulus, and stimuli were presented at intervals of several seconds. The values from four dots on each area of T10, T11, and T12 vertebrae were combined and averaged.
Motor Function
Motor function was assessed to test motor recovery after cell transplantation and to determine whether spinal GABAergic neurons from mESCs affected motor function. The Basso, Beattie, and Bresnahan (BBB) locomotor rating scale and a modified combined behavioral score (CBS) were used (4,17).
Immunohistochemistry
Five rats that had scores close to mean of behavior results at the end point of the experiment in each group were selected, and their spinal tissue was extracted. The remaining animals were sacrificed immediately after finishing behavioral tests. Tissue was dissected from the rostral, epicenter, caudal, and lumbar spinal segments (L4–L5) 7 weeks after SCI from the cell transplantation (n = 5) and saline (n = 5) groups. Rats were deeply anesthetized with sodium pentobarbital (50 mg/kg, IP; Sigma-Aldrich) and perfused with 4% paraformaldehyde (Sigma-Aldrich)/PBS (Hyclone). Tissue was postfixed for 6–8 h in the same fixative and then cryopreserved with 30% sucrose at 4°C overnight. Tissues were sectioned at 20 μm in a cryostat. After washing in PBS, the tissues were blocked for 30 min in 1% horse serum (Sigma-Aldrich) and incubated overnight at 4°C with rabbit anti-GABA (1:1,000; Sigma-Aldrich) and mouse anti-GAD65/67 (1:200; Millipore) containing 0.3% Triton X-100, and the sections were then rinsed and incubated for 60 min at room temperature with Alexa Fluor 594-conjugated secondary antibodies (1:1,000; Invitrogen). After further rinsing, nuclei were counterstained with DAPI (1:10,000 in PBS; Sigma-Aldrich). Images were captured using a confocal microscopy FV1000 FluoView camera (Olympus) and quantitative analysis was perfor med by TissueQuest software (TissueGnostics).
Cell Counts and Analysis
The total number of surviving engrafted cells was described previously and estimated by counting all GFP+ cells in each spinal cord section. The average number of GFP+ cells per section was then extrapolated to the total number of spinal cord sections that contained GFP+ cells, using the formula total GFP+ = A × B/2 (where A is the average number of GFP+ cells per section and B is the total number of spinal cord sections containing GFP+ cells) (5). In addition, because the same nuclei could be seen in two adjacent sections, we counted every other section to avoid counting cells twice. The proportions of surviving GABAergic and GAD65/67+ cells were examined using coronal sections in spinal cord. At least nine sections were analyzed for GABA and GAD65/67 markers in each animal (n = 5). Images were captured using a confocal microscopy FV1000 FluoView camera (Olympus), and cell counting was performed from coronal sections of the lesion area.
Statistical Analysis
All values were expressed as the mean ± SEM. All statistical tests were evaluated at an a level of 0.05. For cell counting, three coverslips were used and statistical analysis was performed by the two-tailed Student's paired t-test. Behavioral results were analyzed by repeated-measures ANOVA followed by unpaired Student's t-tests. Statistical calculations were performed with IBM SPSS Statistics 20.0 (IBM, Armonk, NY, USA).
Results
Efficient Differentiation of mESCs Into Neural Precursor (mESC-NPCs) Cells and GABAergic Neurons
It has been reported that SHH patterns cells in the lateral ganglionic eminence (LGE) to adopt a forebrain fate and then generates striatal projection neurons from medial ganglionic eminence (MGE)-like progenitors (39). The spinal GABAergic neurons were generated from mESC-NPCs using a five-stage protocol (32,42,43) (Fig. 1A, B). After production of EBs and selection for in vitro differentiation of mESCs into neural cells, the mESC-NPCs were fully differentiated into neuronal cells in the culture conditions without SHH and retinoic acid (RA) or with 200 ng/ml SHH or with 100 ng/ml SHH and 1 μM RA. The percentages of β-tubulinIII+ pan-neurons out of total cells were 44.5 ± 2.2%, 48.1 ± 5.8%, and 46.8 ± 4.5%, respectively, and the percentages of GABA+ neurons out of the Tuj1+ cells were 7.2 ± 2.2%, 73.9 ± 6.8%, and 68.9 ± 5.5%, respectively (Fig. 2A–D). MAP2+ neurons were efficiently generated from all culture conditions, and GAD65/67+ neurons were efficiently generated in SHH-treated conditions but very rare without SHH and RA (Fig. 2B). Our RT-PCR results show that the neuronal marker genes, β-tubulinIII (shown with Tuj1 stain) and MAP2, were expressed in all culture conditions, and the expression level of GAD65, an enzyme required for GABA synthesis, was low in the absence of SHH and RA (Fig. 2E). These results demonstrate that GABAergic neurons were efficiently generated from mESC-NPCs in our established culture conditions.
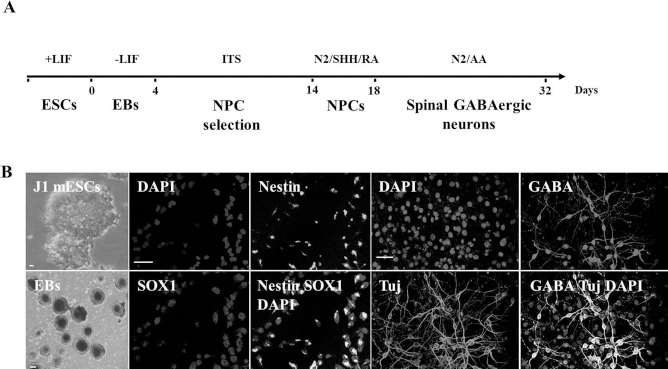
A scheme for in vitro differentiation of ESCs into GABAergic neurons. (A) Experimental procedures for spinal GABAergic neuron differentiation. (B) Representative immunostaining images. mESCs were maintained with LIF. EBs were made from mESCs, and the EBs were cultured in ITS media for 10 days for induction of NPCs (NPC selection). Nestin+/SOX1+ NPCs were obtained and cultured in N2 media supplemented with basic fibroblast growth factor (bFGF). For differentiation of GABAergic neurons, NPCs were cultured in N2 media supplemented with SHH and RA for 4 days, after which the precursor cells were fully differentiated into GABAergic neuronal populations for 14 days in N2 media with ascorbic acid (AA) but without bFGF, SHH, and RA. Scale bar: 200 μm in mESCs and EBs; scale bar: 40 μm in mESC-NPCs and GABAergic neurons.

In vitro differentiation of mESC-NPCs into GABAergic neurons. (A) At day 14 after beginning in vitro differentiation, neurons were analyzed by immunostaining with Tuj1 and GABA antibodies in 200 ng/ml SHH or 100 ng/ml SHH and 1 μM RA or without both SHH and RA. (B) Neurons were analyzed by immunostaining with MAP2 and GAD65/67 antibodies in 200 ng/ml SHH or 100 ng/ml SHH and 1 μM RA or without both SHH and RA. (C, D) Quantitative analysis of Tuj1+ and GABA+ neurons. (E) RT-PCR expression analysis of GABAergic genes from three different culture conditions.
Differentiation of mESC-NPCs Into Caudalized GABAergic Neurons Using Low Doses of SHH and RA
It has been reported that ESC-derived cells can be treated with RA to generate caudalized NPCs, which can subsequently differentiate into spinal GABAergic pheno types, but not striatal GABAergic phenotypes (33,39). Cells of striatal and spinal origin can be distinguished based on their expression of DARPP32, which is known to be expressed in striatal GABAergic neurons (1) but not spinal GABAergic neurons. We therefore examined whether the differentiated neurons in our study were DARPP32+. We found that 11.5 ± 2% of the β-tubulinIII+ neurons observed in the culture medium supplemented with only SHH (200 ng/ml) were also DARPP32+, and only 1 ± 0.5% of the β-tubulinIII+ neurons in cultures supplemented with SHH (100 ng/ml) and RA (Fig. 3A, B) were DARPP32+.

Restriction of mESCs to dorsal spinal progenitors by SHH and RA. (A) Immunostaining analysis for the striatal marker DARPP32 in the 200 ng/ml SHH or 100 ng/ml SHH and 1 μM RA conditions. (B, C) Quantitative analysis of DARPP32+ cells by immunostaining and RT-PCR. Most of the DARPP32+ cells were observed in the 200 ng/ml SHH condition, and most of the cells treated with 100 ng/ml SHH and 1 μM RA were DARPP32-. (D) RT-PCR analysis showing the expression of dorsally expressed transcription factors in cells cultured in without SHH and RA or 200 ng/ml SHH or 100 ng/ml SHH with 1 μM RA for 4 days.
Based on developmental principles, it has been shown that the progenitor cells in dorso-ventral domains are regulated by class I and class II proteins (30,32,42). To examine whether SHH and RA treatments define the progenitor domains and subsequently control the expression of class I and II genes, we performed RT-PCR in mESC-NPCs at 4 days after treatment with SHH and RA. High concentrations of SHH (200 ng/ml) induced relatively strong expression of Nkx2.1 genes (MGE progenitors) but weak expression of the class I genes Irx3, Pax6, and Pax7. These results indicate that high SHH induced the generation of cell types found in the ventral forebrain. However, low concentrations of SHH (100 ng/ml) with RA (1 μM) strongly induced the expression of the class I genes Irx3, Pax6, and Pax7, but weak expression of the Nkx2.1 gene, compared to both the conditions without SHH and RA and with high SHH only. These results suggest that low doses of SHH and RA can induce dorsal spinal GABAergic cell types, although some cells displayed ventral spinal GABAergic cell phenotypes. Next, when we differentiated the mESC-NPCs into neuronal cells, the ventral spinal cord markers Nkx2.2 and HB9 were expressed (Fig. 3C, D). Additionally, we found that 8.6 ± 2.1% of 5-HT+ serotonergic neurons and 6.7 ± 1.8% TH+ neurons were generated with culture conditions containing 100 ng/ml SHH and 1 μM RA (data not shown). These results demonstrate that our mESC-NPCs were mostly dorsal spinal cell types, but some cells were ventral spinal cell types. Moreover, the RA treatments not only dramatically inhibited the forebrain cell fate, but also robustly specified the dorsal spinal cord cell type.
Expansion of Mouse NPCs
Prior to transplantation, the mESC-NPCs were seeded on dishes coated with poly-L-ornithine/fibronectin (Sigma-Aldrich) and cultured for 4 days in N2 culture medium supplemented with RA and SHH as shown in Figure 1A.
Construction of SCI Rat Model and Motor Function After mESC-NPC Transplantation
Prior to cell transplantation, the mESC-NPCs were cultured for 4 days in N2 culture medium supplemented with 100 ng/ml SHH and 1 μM RA. Spinal contusive injury was made at T12 using the NYU impactor by releasing a 10-g weight onto the exposed spinal cord under isoflurane anesthesia (2% isoflurane and 98% O2). Postoperative treatments included saline (2.0 ml, subcutaneous injection) for rehydration. Prior to the contusion, all animals were evaluated for locomotor function in both hind limbs using the BBB scale with the open field test.
Prior to the injury, rats showed normal locomotion with a BBB score of 21. However, 1 day after SCI, all rats showed paralysis in both hind limbs, and the BBB scores in the stem cell and the saline groups decreased sharply from 21 to 0.64 ± 0.24 and 0.79 ± 0.21, respectively. These results demonstrate that the SCI induction protocol successfully produced SCI. To examine the improvements in locomotor function of both hind limbs of rats with contusive SCI after cell transplantation, mESC-NPCs and saline were IT injected into the lesion area of the spinal cord around T10–T11 at 21 days after SCI (Fig. 4A). When we observed the behavior in both groups during the entire experimental period, the BBB before transplantation and at 21 days after cell transplantation were 12.00 ± 0.82 and 12.18 ± 0.60, respectively (Fig. 4B). These results indicate that there were no significant differences in the BBB scores, suggesting no significant improvements in locomotor function of SCI model rats transplanted with the mESC-NPCs.

Motor function in SCI rats after mESC-NPC transplantation. (A) A scheme for IT transplantation. Contusive SCI was made at the T12 spinal segment (vertebra T11), as indicated by the arrow. Three weeks after SCI, GFP fluorescent cells indicated with dots were injected into the subarachnoid space around the T10–T11 spinal segments via a PE 10 tube. The behavioral changes in motor function after cell transplantation were investigated (n = 7, each group, B and C). Locomotor function (B) and combined motor function (C) were analyzed by repeated-measures ANOVA, which did not show an overall significant effect of cell transplantation. N, normal condition; PI, postinjury; Pre, 21 days after SCI.
We also examined the changes in combined motor function in rat models with contusive SCI after cell transplantation. Prior to the injury, all rats showed normal motor function with a CBS of 0. However, 1 day after SCI, the rats showed abnormal motor function, and their CBS values in the cell and saline groups increased to 69.64 ± 0.36 and 68.93 ± 2.37, respectively. During the experimental periods, the CBS values before transplantation and at 21 days after cell transplantation were 25.00 ± 0.94 and 24.29 ± 3.12, respectively. There were no significant differences in the CBS values before or after transplantation, indicating that the mESC-NPC transplantation produced no significant improvements in combined motor function in contusive SCI rats.
Reduced Neuropathic Pain in SCI Rat Model After mESC-NPC Transplantation
To examine the effects of ESC-derived spinal GABAergic neurons on below-level pain after transplantation in the SCI rat model, we applied the paw withdrawal threshold (PWT) test to the plantar surface. Prior to the injury, rats in the cell and saline groups had normal scores for PWT (14.17 ± 0.40 and 13.78 ± 0.48, respectively). After the injury, unlike the presurgical values, the withdrawal and vocalization threshold to mechanical stimuli were significantly decreased, accompanied by aversive behaviors such as vocalization, paw licking, and attacking the stimulus; this was considered an index reflecting nociceptive behaviors. After IT injection of mESC-NPCs into the contusive SCI rat models, the change of PWT was significant (p = 0.001), and the difference between groups was also significant (p = 0.007). The PWT significantly increased from 4 weeks to 7 weeks (4 weeks, p = 0.026; 5 weeks, p = 0.002; 6 weeks, p = 0.043; 7 weeks, p = 0.041) after mESC-NPC transplantation compared to that of the saline group (Fig. 5A). These results show that spinal GABAergic neurons reduced the neuropathic pain occurring below the level of the lesion area after SCI.

PWTs and VTs to mechanical stimulation after mESC-NPC transplantation on below-level and at-level areas following SCI. The time course of the responses to mechanical stimulation after cell transplantation on below-level areas and lesion areas after SCI was investigated. The change of PWT and the VT over time and the differences between groups over time were significant. (A) The PWT significantly increased from 4 to 7 weeks after transplantation. (B) The VT significantly increased at 7 weeks for the T12 area; (C) from 4 to 7 weeks for the T11 area; and (D) 4, 5, and 7 weeks for the T10 area after transplantation. The VT between groups also had marginal significance 5 weeks at T12 and 6 weeks at T10. Asterisks indicate values significantly different from the VT value of the saline group at each time point (n = 7, each group). N, normal condition; PI, postinjury; Pre, 21 days after SCI.
Next, we examined the vocalization threshold (VT) in the lesion area of the SCI rat models after cell transplantation. Mouse ESC-NPCs were IT injected into the lesion sites of the SCI modeled rats. There were no significant differences in the pretransplantation values for VT for T12, T11, and T10 between the saline (T12: 86.31 ± 35.87, T11: 77.81 ± 26.63, and T10: 75.44 ± 37.05) and transplantation (T12: 79.96 ± 21.31, T11: 75.11 ± 19.71, and T10: 78.39 ± 23.85) groups. As shown in Figure 5, cell transplantation significantly reduced pain in the lesion area. The changes of the VT at the lesion site were significant (T12, p < 0.001; T11, p < 0.001; T10, p < 0.001), and the difference between groups was also significant (T12, p = 0.034; T11, p < 0.001; T10, p = 0.014). The VT significantly increased 7 weeks (p = 0.035) at T12, from 4 weeks to 7 weeks (4 weeks, p = 0.036; 5 weeks, p = 0.009; 6 weeks, p = 0.028; 7 weeks, p = 0.020) at T11 and 4, 5, and 7 weeks (4 weeks, p = 0.049; 5 weeks, p = 0.004; 7 weeks, p = 0.040) at T10 after transplantation. The VT between groups also had marginal significance at 5 weeks (p = 0.067) at T12 (Fig. 5B) and 6 weeks (p = 0.062) at T10 (Fig. 5D). These results show that the spinal GABAergic neurons distinctly reduced the neuropathic pain occurring in the contusive SCI rat model after transplantation.
In Vivo Histological Analysis
To examine the survival and migration of engrafted cells at 7 weeks after transplantation, we counted cells in coronal sections of the spinal cord. GFP+ cells were observed as having accumulated around the transplant site, but they were not observed in the same areas in sham rats (Fig. 6A, B). Immunohistochemical analysis detected GFP+ cells in a length of spinal segment from T11 to T13, at a depth of 20 μm (Fig. 6C, D). Stereological measurement shows that the surviving GFP+ cells were 0.992 ± 0.067% of the total engrafted cells at 7 weeks after transplantation. Among GFP+ cells, the number of GABA+ and GAD65/67+ cells was 27 ± 9 and 25 ± 8 per section, respectively. The percentage of GABAergic neurons among surviving engrafted cells was 29.3 ± 4.0% for GABA+ and 27.2 ± 3.2% for GAD65/67+ cells, respectively (Fig. 6C, D). Interestingly, GFP+ engrafted cells were detected in the rostral and caudal segments from the lesion area and the L4–L5 segment of the spinal cord. The GFP+ cells expressed GABA and GAD65/67, which were dispersed at the lesion area (Fig. 7). These results indicate that the engrafted cells can survive at around the lesion site and some of the engrafted cells can migrate into the lesion area, suggesting neuropathic attenuation by GABAergic neurons.
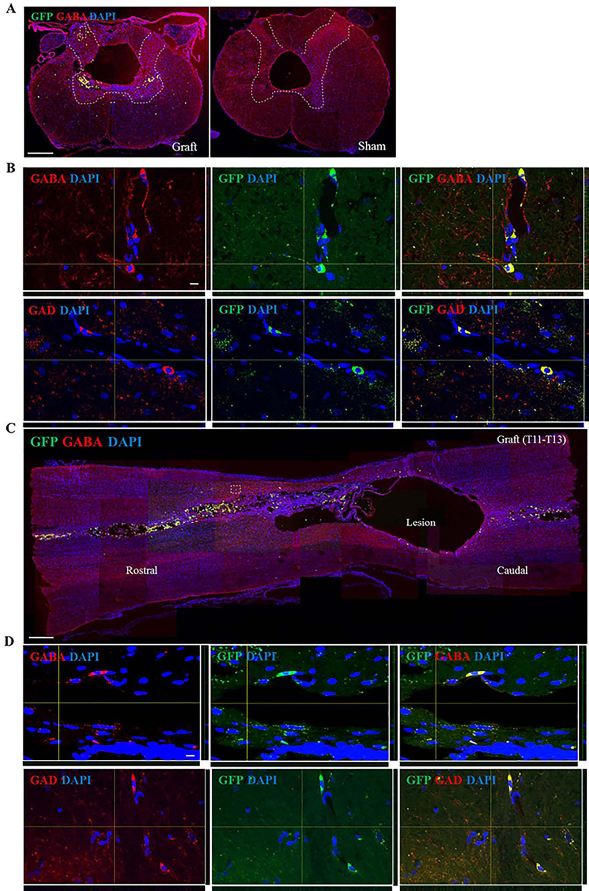
Colocalization of GFP+ cells and GABA or GAD at the lesion area of the spinal cord after transplantation. (A–D) Confocal images were obtained from transverse and coronal sections of the lesion epicenter in the spinal cord. (A, B) GFP+ engrafted cells were detected at the lesion area of the spinal cord after cell transplantation, but they were not detected in the saline group. Engrafted cells differentiated into GABAergic neurons in the spinal dorsal horn (C, D). GFP+, GABA+, and GAD65/67+ cells were detected at the lesion epicenter of spinal cord. Scale bar: 500 μm (A, C). Scale bar: 10 μm (B, D).

Colocalization of GFP+ cells and GABA or GAD at segments rostral and caudal from the lesion area and in the L4–L5 segment of the spinal cord after transplantation. (A–C) Confocal images were obtained from transverse sections of segments rostral and caudal from the lesion area and from the L4–L5 segment of the spinal cord. Engrafted cells migrated to segments rostral and caudal from the lesion area and from the L4–L5 segment and differentiated into GABAergic neurons (GABA+ and GAD65/67+ cells) in spinal dorsal horn. Scale bar: 500 μm (A). Scale bar: 10 μm (B, C).
Discussion
To determine whether IT transplanted spinal GABAergic neurons from mouse mESC-NPCs could be used in the treatment of neuropathic pain, we used a contusive rat model of SCI in which a lesion of the spinal cord at T12 induced paralysis and neuropathic pain in both hind limbs (Fig. 4). Previous studies have reported that cell-based therapies using transplantation into the spinal subarachnoid area for chronic pain syndromes did not result in sustained pain reduction (8,13,23,46).
In this study, we demonstrate that the spinal GABAergic neurons from mESC-NPCs that were injected into the lesion of spinal cord around T10–T11 by IT transplantation using a catheter significantly reduced neuropathic pain near T12 by 2 weeks posttransplantation and persistently alleviated pain at the lesion site up to 7 weeks after transplantation. In addition, spinal GABAergic neurons from mESC-NPCs transplanted into the lesion area significantly increased the PWT to mechanical stimuli; this was interpreted as evidence of mechanical allodynia in the hind paw. These results suggest that the IT transplantation of cells into the space around lesions of the spinal cord is an effective tool to treat neuropathic pain, particularly at-level or below-level neuropathic pain, following SCI. In addition, we demonstrate that mESC-NPCs specifically differentiated into a spinal GABAergic phenotype in vitro instead of striatal GABAergic neurons, suggesting that spinal GABAergic neurons are a useful source of cells for alleviating neuropathic pain after SCI.
Our data support the hypothesis that less GABAergic inhibition in the spinal cord can cause neuropathic pain. IT transplanted spinal GABAergic neurons may decrease pain, as indicated by observation of behavior, by restoring GABAergic inhibitory input after the inhibitory interneurons are lost at T12 following spinal cord injury. The transplanted spinal GABAergic neurons may be able to restore the inhibitory function to the spinal cord dorsal horn to decrease the hyperexcitability that develops in dorsal horn projection neurons. The increased GABA production is a potential mechanism of behavioral improvement in the allodynic rat after thoracic SCI. These results are consistent with prior studies investigating analgesic effects of neuronal cells bioengineered to secrete GABA that were transplanted into the subarachnoid area of allodynic rats (13,46).
In addition, the small percentage of cells surviving at 7 weeks after transplantation contributed to alleviation of neuropathic pain, which suggests that the transplanted cells were successfully integrated into or around the damaged tissues. Interestingly, some of the GFP+ cells migrated to the rostral, caudal, and L4–L5 portions of the injured spinal cord, suggesting that the transplanted cells could be delivered to the site of injury through a homing process (40).
In particular, differentiation of mESC-NPCs into the spinal GABAergic phenotype before transplantation may be critical to alleviate neuropathic pain after SCI. The mESC-NPCs treated with low doses of SHH and RA used in our study differentiated mostly into spinal GABAergic neuronal populations, which were DARPP32-/low cells; further, they showed high levels of Irx3, Pax6, and Pax7 expression and low levels of Nkx2.1 and DARPP32 expression. In addition, there were striatal GABAergic (DARPP32+), serotonergic, and dopaminergic neurons (Fig. 3 and data not shown) in our experiments, which may have contributed to the improvement in pain symptoms This conclusion is supported by previous studies using transplanted cells secreting serotonin (14,21,22) and catecholamines (9,20,49).
Although our results demonstrate significant improvement in neuropathic pain behavior after IT transplantation of spinal GABAergic neuronal populations, it may be necessary to combine this approach with other GABA agonists to maximize the therapeutic effects in the future.
Footnotes
Acknowledgments
This work is supported by grants from the Ministry of Science, ICT, and Future planning (20100023160, 2012M3A9C6050131, and 2014M3C1B2048632) and the Ministry of Health and Welfare (A120392) of the Government of the Republic of Korea. The authors declare no conflicts of interest.
