Abstract
Human fetal neural stem cells (hNSCs) are used to treat a variety of neurological disorders involving spinal cord injury (SCI). Although their mechanism of action has been attributed to cell substitution, we examined the possibility that NSCs may have neuroprotective activities. The present article studied the action of hNSCs on protecting neurons and promoting corticospinal tract (CST) axon regeneration after SCI. hNSCs were isolated from the cortical tissue of spontaneously aborted human fetuses. The cells were removed from the NSC culture medium to acquire NSCM, thus excluding the effect of cell substitution. Continuous administration of the NSCM after the SCI resulted in extensive growth of the CST in the cervical region and more than tripled the formation of synaptic contacts between CST collaterals and propriospinal interneurons that project from the cervical level of the spinal cord to the lumbar level. NSCM reduced the number of caspase 3-positive apoptotic profiles at 7 days and protected against loss of the neurons 6 weeks after injury. NSCM promoted locomotor recovery with a five-point improvement on the BBB scale in adult rats. Thus, hNSCs help to set up a contour neural circuit via secretory factors, which may be the mechanism for their action in SCI rats. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Introduction
Traumatic injury to the adult spinal cord leads to a massive loss of cells and permanent functional deficits, inflicting enormous suffering and economical loss. Various types of treatment, such as cell transplant (33,39) or tissue engineering (16), growth factor infusion or counteracting inhibitory molecules (13,14), altering gene expression, and/or increasing physiological activity have been investigated to enhance functional recovery after spinal cord injury (SCI) (27,28,37,38,43). However, none of these approaches have observed long-distance regeneration of the corticospinal tract (CST), which was thought to be the main reason for the locomotor function loss after SCI.
Although long-distance CST regeneration remains a major challenge, the other pathways can form compensatory circuits that help restore some function after incomplete lesions. Transection of the CST at the thoracic level causes severed axons to sprout collateral branches that synapse with long propriospinal interneurons (LPSNs) at the cervical level, forming “detour circuits” that restore some cortical control to the hindlimbs (5,9). Undamaged axons can also form collateral branches that contribute to recovery. Many agents such as brain-derived neurotrophic factor (BDNF) and inosine promote LPSN projection relays and could help improve the outcome after partial lesions of the spinal cord (21,44).
Recent studies shifted attention toward stem cell transplantation as a therapy (30). Simple substitution of transplanted cells for damaged or dead cells was thought to be the main mechanism of action for its multidifferentiation potentials and the ability of neural stem cells (NSCs) to be incorporated into the tissue (35,36). SCI represents a complex injury cascade (20). The main pathological damage in SCI is the failure of the axon regeneration of the CST (41). Until now, there has been no clear evidence that the stem cell-differentiated neurons present with the whole biological properties of the propriospinal neurons, for example, participating in the signal transduction from the brain to the distal spinal cord.
Whether the LPSNs would respond to the NSC treatment is not known. NSCs contain many secreting substances and factors. We hypothesized that these agents can protect propriospinal neurons, thus augmenting the formation of relay detour circuits, and thus improving the functional outcome after transection of the CST and other pathways in the dorsal spinal cord of mature rats. The present study investigated the role of the agents secreted from NSCs to reconnect the LPSN relays to the neural circuit after thoracic SCI in rats.
Materials and Methods
NSC-Conditioned Medium Preparation
All experiments on human subjects were carried out with the adequate understanding and written consent of the subjects. We also obtained the approval from the ethical board of Harbin Medical University (Harbin, China). NSCs were acquired from 20-week postconception human fetuses (four males, three females) that had been spontaneously aborted. The cells were pooled together. The stem cell isolation and identification methods have been reported previously (26). The cell pellet was cultured in Dulbecco's modified Eagle's medium/Ham F12 nutrient mix (DMEM/F12; 1:1) containing 9.6 μg/ml putrescine, 6.3 ng/ml progesterone, 5.2 ng/ml insulin, 0.1 mg/ml transferrin, 10 ng/ml epidermal growth factor (EGF), and 20 ng/ml basic fibroblast growth factor (bFGF). All culture reagents were from Gibco (Tokyo, Japan). NSCs were serially passaged every 7~10 days to retain a long-term culture at 37°C and 5% CO2 in vitro. All the cells were removed from the NSC culture by centrifugation (400 × g for 7 min) 3 days after passaging to acquire NSC-conditioned medium (NSCM) (4). To prepare for infusion, NSCM (0.2 ml) was loaded into the reservoir of an Alzet 2001 osmotic pump (0.2 ml, Durect Corp., Cupertino, CA, USA) and incubated overnight in normal saline at 37°C before tube insertion. As a control, the same volume of NSC culture medium in which NSCs had not been cultured was used to rule out the effect of the added growth factors (bFGF and EGF).
Animals
All animals received humane care in compliance with the guidelines for the care and use of laboratory animals of the Institute of Laboratory Animal Resources of Harbin Medical University and the National Institutes of Health. Female Wistar rats (n = 65, 8–10 weeks of age, weighing between 260 and 300 g) were obtained from the experimental animal center of Harbin Medical University. The rats were randomly divided into three groups, and each group was divided into two subgroups. Rats received a SCI plus treatment with either NSCM (n = 23: subgroup 1 = 12; subgroup 2 = 11) or a control solution (n = 22: subgroup 1 = 11; subgroup 2 = 11). NSCM control rats received culture medium in which NSCs had not been cultured. Sham-operated animals served as normal controls (n = 20: subgroup 1 = 10; subgroup 2 = 10) and underwent all procedures described except for the dorsal hemisection of the spinal cord.
Spinal Cord Injury and NSC-Conditioned Medium Intrathecal Infusion
All the rats were deeply anesthetized with ketamine/xylazine (75 mg/kg; 10 mg/kg, Fort Dodge Health, Fort Dodge, IA, USA). A laminectomy of half of one vertebral lamina at Th8 was followed by a dorsal hemisection using customized microscissors (Xiaoshan, Shanghai, China). The surgeon was blinded to the subsequent treatment of the animals. Following the lesion, the dorsal back musculature was closed in layers using 3-0 silk sutures (Johnson & Johnson, Shanghai, China). Immediately after surgery, NSCM and NSCM control rats were fitted with Alzet osmotic pumps attached to a silicon tube delivering either control medium or NSCM. Pumps were changed weekly. The tube was advanced slowly into the subarachnoid space. The tip of the silicon tube was placed just rostral to the level of injury. During surgery, the rats were kept on a heating pad to maintain the body temperature at 37.0 ± 0.5°C. All rats received postoperative treatment including the subcutaneous injection of Bicillin (20,000 U/day, Hayao, Harbin Pharmaceutical, Heilongjiang, China) for 7 days, and bladder massage for 7–10 days. The rats received infusions through loaded osmotic pumps four times. Pumps were pulled out at 4 weeks when the tracing surgery was performed.
Apoptosis Detection
At 7 days after injury, all the subgroup 1 animals were anesthetized and perfused. Frozen tissue blocks containing cervical levels C3–C5 were cryosectioned at 10 μm in the coronal plane (CM1860UV, Leica, Wetzlar, Germany). Every fifth slide was rinsed three times in phosphate-buffered saline (PBS; Hongzhou, Guangzhou, China) and blocked in 10% normal goat serum (Kangwei, Beijing, China) in PBS for 30 min at room temperature (RT). To detect active caspase 3-positive apoptotic cells, slides were incubated with a rabbit monoclonal antibody for active caspase 3 (1:100; Sigma-Aldrich, St. Louis, MO, USA). After three washes in PBS, slides were incubated for 1 h at RT with the fluorescein-conjugated goat anti-rabbit secondary antibody (fluorescein isothiocyanate; FITC) (1:200; Molecular Probes, Eugene, OR, USA). Slides were then washed three times in PBS and submerged in Hoechst 33258 (1 μg/ml; Sigma-Aldrich) to detect condensed nuclei indicative of apoptosis. To assess the cell identity of apoptotic profiles for neurons, neuron-specific nuclear protein was used (neuronal nuclei; NeuN; 1:1,000; Chemicon, Temecula, CA, USA). A Cy3-conjugated donkey anti-mouse secondary antibody (1:400; Jackson ImmunoResearch, West Grove, PA, USA) was used to visualize the mouse primary antibodies listed above. Active caspase 3-positive neurons with a condensed nucleus in the ventral horn in each section in the gray matter were counted and averaged across the 10 sections. The amount of apoptosis was expressed as the mean number of active caspase 3-positive profiles with condensed nuclei per section. All photographs were taken with a confocal microscope (LSM 510 Meta; Zeiss, Oberkochen, Germany). Counts were performed blind with respect to treatment.
Double Tracing of CST and Propriospinal Neurons
One month after the SCI, subgroup 2 animals were anesthetized, and a burr hole was drilled through the cranium to expose the right sensorimotor cortex. A 10% solution of biotinylated dextran amine (BDA; 10,000 mw; 10% w/v solution in sterile saline; Molecular Probes) was injected into eight sites at a depth of 0.7 mm from the cortical surface (0.5 μl /site) to cover the hindlimb region. Two weeks later, histology was performed. Retrograde tracing of LPSNs was carried out at the same time by injecting 1 μl of cholera toxin subunit B (CTB; 1%; List Biological, Campbell, CA, USA) into each side of the lumbar spinal cord (level L2–L3; 200–300 μm lateral to midline; 500 μm depth) using a glass micropipette and Nanoject II apparatus (Drummond Scientific, Broomall, PA, USA). The capillary remained in placed for 1 min following each injection. The skin and muscles were then sutured closed, and rats were allowed to recover on a warming pad before being returned to their cages.
Two weeks after tracer injections, rats were euthanized with an overdose of anesthesia by isoflurane (Shanghai Yuyan, Shanghai, China) and perfused transcardially with 0.9% saline followed by 4% paraformaldehyde (PFA, Sigma-Aldrich). The brain and spinal cord were dissected out, postfixed in 4% PFA overnight at 4°C, and cryoprotected in graded sucrose solutions (10% for 24 h, then 30% for 24 h; Hayao, Harbin Pharmaceutical). A 2-cm segment of the tissue block was embedded in gelatin–albumin (Type-A; Sigma-Aldrich) and cut at 40 μm using a Vibratome (Leica VT 1000s). For immunofluorescence detection, frozen tissue blocks were cryosectioned at 40 μm in the coronal plane.
To estimate the lesion depth of the injury, we used the fast blue and cresyl violet staining methods. The sections were deparaffinized and hydrated to 95% ethyl alcohol; then they were left in luxol fast blue solution (Sigma) in a 56°C oven overnight. After rinsing off excess stain with 95% ethyl alcohol, the slides were differentiated in the lithium carbonate solution (Sigma) for 30 s and 70% ethyl alcohol for 30 s. The differentiation steps were repeated if necessary. Then the slides were counterstained in the cresyl violet solution (Sigma) for 30–40 s.
Cell Number Detection
Six weeks after SCI, sections dissected from the cervical region were stained with rabbit anti-NeuN antibody (1:1,000), followed by an anti-rabbit biotinylated secondary antibody; then the sections were stained using the ABC and DAB kit (Vector Labs, Burlingame, CA, USA) to visualize BDA-positive axons. Total neuron number in the gray matter was assessed. NeuN-positive cells in 10 equally spaced 30-μm sections were counted in the gray matter. Results were reported in units of density (cells per cubic millimeter). NeuN-positive cells whose nucleus came into focus within the inclusive zone of each dissector frame of Stereo Investigator (MBF Bioscience, Willistion, VT, USA) were counted. Counts were performed blind with respect to treatment. Figures were prepared using Adobe Photoshop 10.0 (San Francisco, CA, USA). In Photoshop, a counting grid was imposed on each image. The number of NeuN immunoreactive cells in each area examined was counted and averaged.
CST Sprouting
Free-floating sections dissected from the cervical or thoracic region were stained using the ABC and DAB kit (Vector Labs) to visualize BDA-positive axons. Each section was visualized under low magnification. The number of collaterals emanating from the CST was counted. To correct the variation of the tracing method and interanimals, we divided the collateral number by the traced numbers in the main CST. Axons were quantified using Stereo Investigator and averaged across the 10 sections to determine the number of labeled axons in the CST per section for each case.
CST Contacts Onto LPSNs
Free-floating sections through the cervical enlargement (C3–C5) were blocked with a solution containing 3% bovine serum albumin (BSA; Kangwei) and 10% normal goat serum in PBS with 0.5% Triton X-100 (Kangwei), then incubated in a rabbit anti-CTB antibody (1:10,000, Genway, San Diego, CA, USA) in 3% BSA and 5% goat serum containing 0.5% Triton X-100 overnight at 4°C. Sections were rinsed thoroughly, then incubated with goat anti-rabbit IgG conjugated to Alexa fluor 594 (1:500; Invitrogen, Carlsbad, CA, USA) and streptavidin conjugated to Alexa fluor 647 (1:1,000; Invitrogen). Images containing CTB-positive cells or dendrites were taken using a Spinning Disk confocal microscope (Perkin Elmer, Waltham, MA, USA). The number of BDA-labeled boutons, defined as swellings at least twice the thickness of the axon (17), and falling within 10 μm of a CTB-labeled soma or dendrite, were quantified using the Stereo Investigator program. Results were averaged over 10–12 sections for each animal. The contacts between the sprouting collaterals and the LPSNs were counted and calculated in two ways. First, the absolute number of contacts was counted. The numbers were further divided by the traced BDA fibers. Second, the contact numbers, which had been normalized to the CST fibers, were divided by the labeled LPSNs.
Synaptophysin-Positive Contacts
A separate set of free-floating sections through the cervical enlargement were rinsed in PBS, transferred to a preheated solution of 10 mM sodium citrate (pH 8.5, 80°C, 30 min; Kangwei), then to 3% H2O2 in methanol (10 min; Kangwei), blocked with 5% normal goat serum in PBS, and incubated overnight with a monoclonal anti-synaptophysin antibody (1:200, Millipore, Billerica, MA, USA) in PBS containing 3% Triton X-100 at 4°C. Sections were then rinsed in PBS and incubated in the secondary antibody (Alexa Fluor 488-conjugated antibody to mouse IgG, made in goat, 2 mg/ml; Invitrogen) in 0.3% Triton X-100 in PBS and for 2 h at RT. We then carried out staining to visualize two other antigens that required different incubation times, one for presynaptic axons (BDA) and the other for LPSNs (CTB). Sections were washed and incubated in a rabbit antibody to CTB (1:10,000, in 3% BSA and 5% normal goat serum; Genway) overnight at 4°C, rinsed, and incubated 2 h at RT with an Alexa Fluor 594-conjugated antibody (2 mg/ml to rabbit IgG made in goat in 3% BSA and 5% normal goat serum; Invitrogen) to visualize CTB+ neurons and Alexa Fluor 647-strepavidin (2 mg/ml in 3% BSA and 5% normal goat serum; Invitrogen) to visualize BDA+ axons. Sections were then rinsed, mounted, and coverslipped. Stacked images were collected of CTB-positive cell bodies that had BDA-positive axons within approximately 20 μm of their cell bodies, as well as all objects at a prespecified position in the spinal gray matter using an Ultraview Vox Spinning Disk confocal microscope (Perkin Elmer).
Behavioral Analysis
The motor function was assessed using the Basso–Beattie–Bresnahan (BBB) Locomotor Rating Scale (6). For scoring, each animal was videotaped for 3 min. The BBB score was determined from sequences of four steps or more from digitized videos projected on a computer screen at one-fourth speed. Three different examiners blind to the treatment participated in all of the behavioral tests. They were segregated into three teams of two observers each. Functional recovery was assessed every week by a different team. All tests were videotaped, and two investigators blind to the treatment evaluated the functional outcome of each animal by reviewing the videotapes. Rats' responses were only scored when the conclusions obtained by in situ observers and from the videotapes were the same.
Statistical Analysis
All data were analyzed by SPSS 19 (IBM, Armonk, NY, USA). Results are presented as group means ± SEM. Group comparisons were evaluated by a one-way ANOVA. A Tukey–Kramer post hoc analysis was used for follow-up tests of between-group differences when the overall F was found to be significant. For all statistical analyses, significance was accepted at p ≤ 0.05 or less.
Results
NSCM Does Not Affect the Dystrophy of the Spinal Cord After SCI
With the microneurosurgical technique (all the injury operations were done by the neurosurgeons), we can accurately cut the spinal cord to a half depth with the purpose of completely cutting the CST and preserving the LPSNs. If the cut depth control was not well done between each rat, it will not only affect the results but also may change the recovery mechanism after surgery (discussed below). We measured the lesion depth on every rat, which extended from 51% to 56% (data not shown). After histological analysis with fast blue and cresyl violet, we found that the CSTs were completely cut, and the ventral horns were left intact. The NSCM treatment did not change the extent of the glial scar, the cavity of the lesion, or the dystrophy of the spinal cord (Fig. 1a–d). Although NSCM treatment would promote the recovery of the neural circuit (results shown below), it really cannot prevent the local secondary injury after SCI through our experiment.

The dorsal hemisection of the thoracic spinal cord. Six weeks after the injury, schematic diagram of the injured tissue showed there was almost the same amount of tissue scar between the control (nonconditioned media) rat (a) and NSCM-treated rat (b); the spinal cord did not exhibit significant dystrophy rostral and caudal to the lesion. Fast blue (showing the axons, blue) and cresyl violet (showing the neurons, purple) staining demonstrated that half of the spinal cord was cut accurately (c, d); the caudal white matter, which contained the propriospinal tract, was preserved intact. The lesion depth, glial scar formation, and the extent of the dystrophy in the thoracic spinal cord did not differ significantly between control group (c) and NSCM-treated group (d). Scale bar: 1 mm. Arrows show the scope of the glial scar and the cavity.
NSCM Promotes the Axon Regeneration
The transected CST axons at the thoracic level were found to sprout collateral branches; however, there were only sparse and short collaterals (Fig 2a) after hemisection of the spinal cord. The number of the CST collaterals did not increase after NSCM treatment (Fig 2b). Next, we observed the BDA-labeled CST axons at the cervical level and found there were numerous sprouting collaterals growing from the main CST, traversing the white–gray matter interface, then reaching the ventral lateral horn (Fig. 3). These collaterals contacted the LPSNs whose tract axons innervated the lumber motor neurons. This has been shown in other studies (5,10). We found that BDA-labeled boutons that contacted LPSNs expressed synaptophysin and were therefore likely to represent synapse formation (Fig. 4). These results indicated that CST collaterals form synapses with LPSNs at the cervical level, forming an LPSN relay neural circuit, which helped to restore supraspinal control to the lumbar spinal cord after SCI.
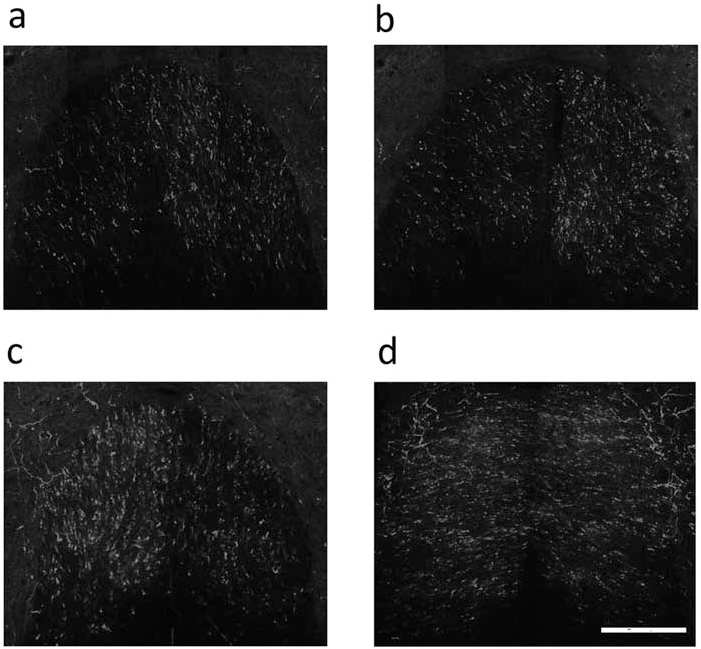
Spinal cord lesion significantly increased the collaterals of the CST axons in the cervical cord but not in the thoracic cord. Few sprouting fibers were seen in the transverse section located just rostral to the thoracic injury region in the medium control rat (a); the axons in the thoracic cord after NSCM treatment remained unchanged (b). The number of the sprouting fibers increased in the cervical region in the medium control rat (c); NSCM treatment increased many more collaterals in the cervical cord compared to the control rat (d). Scale bar: 100 mm.

NSCM increased the axon collaterals of the corticospinal tract, (a) Transverse section through the cervical enlargement of the sham rat showed very few collaterals, (b) There was an increased quantity of the CST axons in the gray matter after spinal cord injury, (c) NSCM further increases CST sprouting compared to the control rats, (d) Quantitation of CST axon collaterals entering into the gray matter. Scale bar (a–c): 100 urn. ##p<0.01 compared with sham-operated controls (sham); **p<0.01 compared with NSCM control rats (SCI). ANOVA with post hoc Tukey's test; F= 101.325. Axon collaterals were divided by the total number of axons counted in the main CST at the cervical spinal cord.
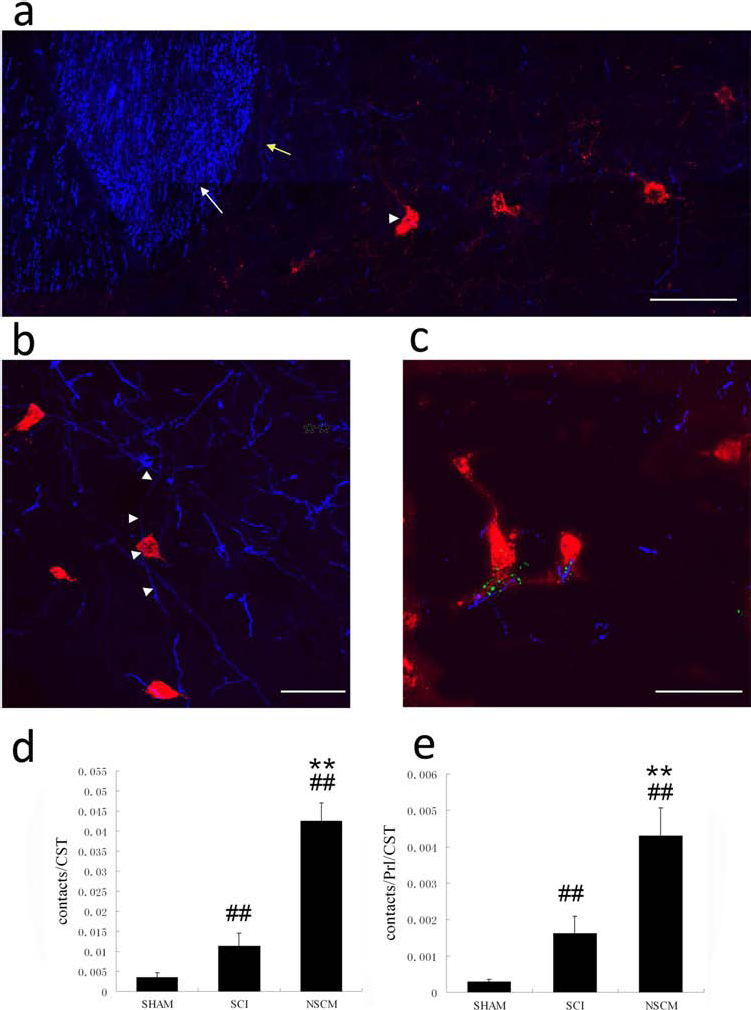
NSCM increases CST contacts onto LPSNs. (a) Trans verse section shows many collaterals grow from the main CST into the gray matter and make contact with the LPSNs. White arrows show the main CST. Yellow arrows show the regenerating fibers sprouting from the main CST. Arrowheads show the fibers contacting an LPSN. Scale bar: 100 mm. (b) Many LPSNs (red) contact BDA-labeled CST collaterals (blue). Arrowheads show BDA-labeled CST collaterals contacting an LPSN cell body. Scale bar: 50 mm. (c) Confocal image displaying triple staining for BDA-labeled boutons (red), presynaptic protein synaptophysin (green), and CTB-labeled LPSN (blue), which show the expression of synaptophysin gathered in or around the gap between the neurons and the axons, indicating synapse formation between the increased CST collaterals and LPSNs in the NSCM-treated rats. Scale bar: 50 mm. (d, e) Quantitation of close apposition formed by CST axons and LPSN somata or dendrites. After counting the number of contacts of bouton-like structures from CST collaterals and LPSNs, we normalized these counts to either the normalized number of collaterals (d) or to the number of normalized collaterals and LPSNs. (e) Normalized collaterals refers to the number of collaterals divided by the total CST number. Following normalization to the normalized collaterals, we found SCI alone increases axosomatic synapses compared to sham-operated controls, and NSCM increases these to a greater extent. Following normalization to the number of normalized collaterals and LPSNs, there was still a significant increase in the group with NSCM application compared to the NSCM control group. However, the amplitude of the increase is a little lower than the former. #p < 0.05, ##p < 0.01 compared to sham-operated controls (sham), **p < 0.01 compared to NSCM control (SCI) rats. ANOVA with post hoc Tukey's test: F = 117.253 (d); 91.787 (e). Error bars indicate standard error of the mean.
We investigated the sprouting of hindlimb CST axons, considering that the absorbance volume of the injected total BDA was different between each rat. The results were normalized by the total number of BDA-labeled axons in the dorsal CST to eliminate this effect. BDA labeling of the hindlimb motor area in normal rats resulted in minimal (fewer than 1%) total axons extending outside the dorsomedial CST at the cervical level of the spinal cord.
Just like earlier studies, dorsal hemisections of the spinal cord at the thoracic level increased sprouting of hindlimb CST fibers, more than tripling the number of hindlimb axons that extended into the gray matter at the cervical level (p < 0.01) (Fig. 3b, d). NSCM treatment approximately tripled the number of hindlimb CST axons that sprouted in the cervical spinal cord after SCI compared to control SCI rats (p < 0.01) (Fig. 3c, d). The number of the sprouting fibers was highest adjacent to the principal bundle of the CST and decreased progressively at greater distances from the CST. The long distance of the sprouting axon was observed projecting into the ventral horn of the spinal cord.
We next investigated whether hindlimb CST fiber collaterals contact LPSNs to form a detour circuit that can potentially restore cortical control to the lumbar region. Following the methods of Bareyre et al. (5), we injected CTB into the lumbar enlargement to retrogradely label cervical LPSNs that project from the cervical level, past the lesion site, to the lumbar cord (i.e., the level of hindlimb control). Surgery caused an appreciable change in the number of LPSNs that became labeled in the cervical enlargement following CTB injections into the lumbar area (normal, mean ± SEM = 50.7 ± 2.6; saline, 24.8 ± 1.2). NSCM-treated rats displayed increased labeled neurons (37.1 ± 1.8), almost 1.5-fold more than the controls. The setup of the potential novel circuits should be premised by the contacts of BDA-labeled CST collaterals with CTB-labeled interneurons in the cervical spinal cord. We therefore quantified the number of bouton-like swellings made by BDA-labeled CST axons that contacted or traversed CTB-labeled somata or dendrites (Fig. 4). SCI alone showed a threefold increase in the number of BDA-positive boutons contacting CTB-positive somata (p < 0.01) (Fig 4d). NSCM more than tripled the number of CST contacts with LPSN somata compared to medium-treated controls (p < 0.01) (Fig 4d). The neuronal loss decreased CTB-labeling neuron numbers, and so we normalized the number by propriospinal neurons. Then we found that NSCM also increased the contact number, although the increasing amplitude is a little lower than the absolute number after being normalized to the CST fibers (Fig 4e). These results indicated that the contact increases could be attributed to both increased sprouting fibers and attenuated neuron loss.
NSCM Attenuates Neuron Apoptosis
We sought to examine whether NSCM could have an antiapoptotic activity after SCI in a rat model.
To explore whether the induction of apoptosis involves the executioner caspase 3 in neurons, we combined immunostaining for cleaved (active) caspase 3 and the neuronal nuclear protein NeuN. Our findings revealed large numbers of active caspase 3-positive apoptotic profiles (Fig 5a). Systemic NSCM treatment significantly attenuated the number of apoptotic neurons (Fig 5b). Seven days after SCI, we found caspase 3 localized in the nuclei of neurons in the gray matter (NSCM, 8.3 ± 2.1 vs. control, 21.35 ± 2.7; p < 0.01) (Fig 5c). Continuous administration of NSCM after the injury reduced the number of apoptotic profiles, indicating that the injury-induced apoptosis of gray matter neurons depends on caspase activation.

NSCM inhibits apoptosis of the neurons after SCI. Spinal cord sections were processed for double labeling using neuronal nuclei (NeuN) antibody, a marker for neurons, and anti-cleaved capase 3 antibody. (a) Immunocytochemical analysis shows that a number of cleaved (activated) caspase 3-positive neurons was observed in the control group. (b) NSCM treatment decreased the number of cleaved caspase 3-positive neurons compared with that observed in the control group (transverse sections). Scale bar: 30 μm. (c) Quantitative analysis of caspase 3-positive neurons shows that NSCM significantly inhibited the apoptosis of neurons when compared with that in the control group rats after injury. Statistical tests were performed on total cell counts and are significantly different as indicated: ##p < 0.01 compared with sham-operated controls (sham); **p < 0.01 compared with NSCM control (SCI) rats. ANOVA with post hoc Tukey's test: F = 113.051.
Six weeks after SCI, the number of neurons decreased significantly in the gray matter, compared with the control group (control, 11,345 ± 1,523 vs. sham, 21,230 ± 2,053; p < 0.05). Continuous treatment with NSCM for 4 weeks protected against the loss of neurons (control, 11,345 ± 1,523 vs. NSCM, 16,270 ± 3,581; p < 0.01) (Fig. 6), indicating that the decrease in the number of neurons likely resulted from apoptosis.

NSCM attenuates loss of the neurons after SCI. NeuN-positive cells were counted in the gray matter. Many NeuN-stained neurons were observed in the NSCM treatment rat (b); however, few were observed in the NSCM control rat (a). Scale bar: 100 μm. Unbiased stereology was used to count cells in equally spaced transverse sections located in the cervical region. Neurons were counted 4 weeks after SCI and expressed as mean ± SD. (c) Statistical tests were performed on total cell counts and are significantly different as indicated: #p<0.05, ##p<0.01 compared to sham-operated controls (sham), **p<0.01 versus NSCM control (SCI). ANOVA with post hoc Tukey's test: F=33.8294.
NSCM Promoted the Functional Recovery of Paraplegic Rats
The result of the functional restoration after SCI is shown in Figure 7. After laminectomy and surgery, the rats with the dorsal hemisection of CST were paralyzed and moved by pulling themselves forward with the forelimbs. The SCI group and NSCM group were severely impaired in open-field behavior after surgery (BBB score = 0). Moreover, they presented with urinary and fecal incontinence. We found there were two rats that were paralyzed instantly after the injury; however, they could move a little after 1 day. The paralysis reflected spinal shock after extensive primary and secondary damage. Such animals were excluded from the group to make sure that all the rats underwent complete CST destruction. The sham group rats' locomotor function was affected at an early time after surgery (BBB score = 6.5), mainly because of the spinal cord shock following the surgery. However, they recovered quickly. Motor function of the rats was recovered continuously in the NSCM group. Gait analysis demonstrated that transplanted rats showed weight support and forelimb–hindlimb (FL–HL) coordination that was not found in controls. ANOVA showed that there was a significant difference between these groups. Over the next 4 weeks, the scores of medium-treated controls rose to an average of 10.1 ± 1.5. NSCM-treated rats began to outperform controls (media) after 1 week and maintained this superiority for the remainder of the testing period, averaging six points higher than controls on the BBB scale at 4 weeks (p < 0.05). The mean BBB scores reached 16.2 ± 0.5 in the NSCM group and got close to the sham group, 18.9 ± 1.0, after transplantation (p < 0.01).

BBB open-field walking scores for the functional recovery analysis of four groups. At 1 month, statistical analysis indicated an overall significant difference (ANOVA with post hoc Tukey's test: F = 101.757, p < 0.01). Further statistical analysis between the groups of animals at 4 weeks after injury indicated that the NSCM (medium) group versus sham-operated (sham) controls was significant (#p = 0.013), as was the NSCM (medium) group versus NSCM control (NSCM) group (**p < 0.01).
Discussion
Here we report that, in rats, after a thoracic SCI, NSCM could reduce the “delayed apoptosis” of gray matter neurons, attenuate the loss of neurons, promote axon sprouting, and help to form compensatory circuits via propriospinal neuron tracts. In addition, NSCM markedly improved limb coordination after SCI.
NSC transplantation has been widely used to treat animals with SCI (2,42,46). It has even been tried to treat patients in some countries (12). NSC transplantation has been shown to displace injured cells, promote CST regeneration and axonal myelination, and, most importantly, improve the functional outcome after SCI (19,42,48). Despite good results and outcome, the mechanism of NSC function in SCI has still not been clearly elucidated. Most of these studies reported that improved function was a result of differentiation to oligodendrocytes or, less frequently, neurons, weeks to months after transplantation (3). Different results were obtained in other studies; that is, there was no detection of transdifferentiation in the transplanted cells, even though functional improvement was noted (22). The majority of the engrafted NSCs in the spinal cord were found to differentiate into a glial lineage phenotype (8). Only a small fraction of the cells differentiated into neurons, in contrast with the high percentage of neuronal differentiation seen in other CNS disease models such as Parkinson's disease after stem cell transplantation. As a result, hNSCs pretreated in vitro to increase and optimize neuronal differentiation (42) or neuronal progenitors (11,24) were preferred for transplantation into SCI rats. Although oligodendrocytes differentiated from the stem cells promoted myelin formation (46), most of the stem cells migrated to the injury site under the chemotactic effect of inflammatory factors, rather than aligning with the long axis of the CST like the olfactory ensheathing cells (23,34). The question remains as to whether the mature cells derived from the stem cells are functional and incorporate into the host. Cell substitution is perhaps not the absolute or the predominant role of the stem cells in the injured spinal cord.
In this study, we ascertained NSCs' effect apart from cell replacement. To exclude the effect of cell replacement, we utilized NSCM from which the cells had been removed and then infused the NSCM into the injury site. We chose the animal model in which the CST was transected but the LPSN tract was preserved. This kind of injury mimics most of the actual injuries found in the clinic and can be used to better study the recovery mechanism after SCI. This model allowed us to study the role of compromising circuit formation and the restoration after SCI. We accurately cut the CST but did not injure the ventrolateral horn.
The disruption of the CST is the main reason for the movement failure after SCI, so we observed the CST axons of the rostral end of the lesional thoracic spinal cord at first. We found there were axons sprouting from the injured CST; however, very few collaterals were observed 6 weeks after the injury. The NSCM treatment could not increase the number of the sprouting collaterals either. This indicates that the possible recovery mechanism in which the regenerated CST can bypass the lesion and reconnect the neural circuit does not currently exist. Next, we observed the CST axons in the cervical cord. To our surprise, there were significant increases in the number of collaterals sprouting in this region, even more collaterals in the NSCM-treated rats. The sprouting axons entered the gray matter and reached the ventral lateral horn where the LPSNs were located.
Propriospinal neurons (PNs) have been said to play an important role in the recovery from incomplete spinal cord injury (SCI). The CST–PN contacts project past the lesion and allow transmission of descending signals (for example, motor commands) below the lesion. The formation of de novo spinal circuits is now considered an important substrate for functional recovery after incomplete SCI (5,9). The stimulation of the PNs caudal to the spinal cord transection can promote locomotor function restoration (45). We also demonstrated that severed CST sprouting fibers form “new” contacts with surviving PNs following SCI in the present study. The NSCM could increase the number of contacts nearly threefold. The synaptophysin-positive staining between the sprouting fibers and the PNs indicated that regenerated axons formed effective contacts (Fig. 4). The new circuit connection was in accordance with functional recovery after the NSCM treatment. Although the histological analysis after the surgery indicated that the lesion depth was very good, the local contusion unavoidably affected the LPSN tracts. Just as the results showed, the interneurons died by apoptosis because many of the cell populations had been axotomized after the thoracic region was injured. We excluded the effect of the reduction in number of the retrogradely labeled neurons by normalizing with the number of PNs. We found that the CST–PN contacts were also higher in the NSCM-treated rats than the control rats but lower than the result without PN normalization. Given the fact that the CST–PN contacts were only affected by the CST numbers after PN normalization, we concluded that the effect of NSCM increasing CST–PN contacts was partly due to decreased neuronal loss and partly to increased collaterals after NSCM treatment.
The augmented CST–PN contacts reminded us that the NSCM may have an effect on the protection of neurons. Several studies provide strong evidence that cellular destruction contributes to the secondary damage after SCI (7,15). It is absolutely clear that the cellular destruction will affect PN-based neural circuit reconnection. We found that SCI could induce apoptosis and loss of neurons in the cervical spinal cord, which was also indicated by the decreased number of cervical LPSNs that were retrogradely labeled following tracer injections into the lumbar cord after SCI. PNs are a potential target in the quest to develop new SCI treatments. They have been shown to respond well to many different types of therapy. Growing evidence that apoptosis contributes to CNS pathology damage (25) raises the possibility that measures having a therapeutic effect may act by blocking apoptosis. Here we show that NSCM effectively prevents trauma-induced apoptosis of neurons in the proximal and distal degenerating segments within the transected dorsal columns. This was rarely reported from previous articles relevant to stem cells. The role of NSCM in preventing loss of neurons with a concomitant antiapoptotic effect mirrors the neuroprotective effects of NSCs. SCI is a very complex pathophysiological process that comprises the primary and secondary phases of injury. Serious impairment of neural functions can be caused by direct damage within the primary phase. The pathophysiological mechanisms involved in the secondary phase are also very important (32,47). Although NSCM can prevent loss of neurons and promote the reconnection of the LPSN relay neural circuit, it cannot prevent some secondary damage, such as the glial scarring, cyst and syrinx formation according to our experiments.
Our research demonstrated that NSCM has a critical supporting role in promoting recovery. Recently, NSCs were found to be able to create a more favorable environment for limiting damage and promoting regeneration via immunoregulation (1,31), expression of growth factors and cytokines (40), improved vascularization and provision of a permissive growth substrate (29), and/or suppressing cavity formation (18). Here we show that NSCM effectively protects the interneurons and their neural circuit connection. This function was rarely reported by previous relevant articles. These results further indicate that the positive effects of NSCs (improved functional outcome after SCI) result from their multifaceted impact on the injured spinal cord.
Footnotes
Acknowledgments
We are grateful to Larry Benowitz (Harvard Medical College) for suggestions of part of the study. This research was supported by a program of the National Natural Science Foundation of China (31040033), The Translational Research Program of Boston Children's Hospital of Harvard Medical College (to L.B.), Heilongjiang Natural Science Foundation (D201081), and Excellent Scientist Program of Academy Fellow Wei Han Yui. The authors declare no conflict of interests.
