Abstract
Experiments on spinal cord injury (SCI) have largely focused on the transplantation of stem cells into injured spinal cords for motor recovery while neglecting to investigate bladder dysfunction. The present study was performed to investigate the effect of B10 human mesenchymal stem cells (hMSCs) directly transplanted into the bladder wall of SCI rats and to determine whether they are capable of inhibiting collagen deposition and improving cystometric parameters in SCI rats. Forty 6-week-old female Sprague–Dawley rats were divided into four groups (group 1: control, group 2: sham operated, group 3: SCI, group 4: SCI rats that received B10 cells). B10 cells were labeled with fluorescent magnetic nanoparticles (MNPs). Four weeks after the onset of SCI, MNP-labeled B10 cells were injected to the bladder wall. Serial magnetic resonance (MR) images were taken immediately after MNP-B10 injection and at 4 weeks posttransplantation. Voiding function was assessed at 4 weeks posttransplantation, and the bladder was harvested. Improvements in bladder fibrosis and bladder function were monitored by molecular MR imaging. Transplantation of B10 cells into the SCI rats markedly reduced their weights and collagen deposition. MR images showed a clear hypointense signal induced by the MNP-labeled B10 cells at 4 weeks posttransplantation. Transplanted B10 cells were found to differentiate into smooth muscle cells. The intercontraction interval decreased, and the maximal voiding pressure increased after SCI but recovered after B10 cell transplantation. Survival of B10 cells was found at 4 weeks posttransplantation using anti-human mitochondria antibody staining and MR imaging. The transplanted B10 cells inhibited bladder fibrosis and ameliorated bladder dysfunction in the rat SCI model. MSC-based cell transplantation may be a novel therapeutic strategy for bladder dysfunction in patients with SCI.
Introduction
Patients with spinal cord injury (SCI) experience urological complications (2), and SCI causes severe lower urinary tract dysfunction, including overactive bladders and urinary retention, that results in increased bladder thickness and fibrosis (21, 31). The urinary bladder wall can accumulate connective tissue and become fibrotic following SCI, and this fibrosis may adversely affect smooth muscle function and the capacity of the bladder to empty properly, resulting in low capacity, high pressure, and noncompliance. These changes can be treated with augmentation enterocystoplasty, but potential surgical complications such as dysbolism, urolithiasis, and absorptive disadvantages related to the usage of bowel segments and cancer induction are unavoidable (1).
The therapeutic potential of bone marrow-derived human mesenchymal stem cells (hMSCs) on cardiac or liver fibrosis has been previously reported (6, 19). Recently, we have reported that transplantation of hMSCs into the bladder wall in a rat bladder outlet obstruction model inhibits bladder fibrosis and improves bladder function (9). Inhibition of collagen deposition using hMSCs could also lead to functional restoration and provide an effective treatment for bladder fibrosis in SCI.
Hepatocyte growth factor (HGF), initially identified as a potent mitogen for hepatocytes, secreted by hMSCs, plays an essential role in tissue angiogenesis and regeneration and acts as a potent antifibrotic agent (17, 22).
Reliable in vivo monitoring of transplanted cells should enable a systematic investigation of cell therapies (25), and newly available fluorescent magnetic nanoparticles (MNPs) (7, 34) should improve the imaging of transplanted cells in experimental bladder fibrosis.
Previous SCI studies addressing bladder dysfunction have dealt with grafting stem cells directly into injured spinal cord (5, 11–13, 29). In the present study, hMSCs labeled with MNPs were transplanted directly into the bladder wall to repair bladder fibrosis and restore bladder dysfunction in SCI, and the status of the transplanted hMSCs was monitored by molecular magnetic resonance (MR) imaging.
Materials and Methods
Human MSCs
BM3.B10 (B10), a stable immortalized bone marrow hMSC line, was generated via transfection of human fetal bone marrow stem cells with amphotropic replication-incompetent retroviral vector encoding v-myc (15). B10 cells were grown in Dulbecco's modified Eagle medium with high glucose (Welzene, Seoul, South Korea) containing 10% fetal bovine serum (GenDEPOT, Barker, TX, USA) and 20 μg/ml gentamicin (Life Technologies, Grand Island, NY, USA). B10 cells used in the present study were cultures at the 10th to 13th passage.
Labeling of B10 Cells with Fluorescent MNPs
Fluorescent silica MNPs (NEO-STEM TSR50; Biterials, Seoul, South Korea) were 50 nm in size and contained rhodamine B isothiocyanate (RITC) conjugated to terminal silanol groups. B10 cells were incubated with MNPs (100 μg/ml) overnight at 37°C. MNP labeling of B10 cells was confirmed using immunofluorescence microscopy.
Spinal Cord Injury and Transplantation of B10 Cells Into Rat Bladders
All procedures were conducted in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee of the Chung-Ang University. A total of 40 6-week-old female Sprague–Dawley rats (200 g,
The rats were anesthetized intraperitoneally with a 3:2 mixture of Zoletil (30 mg/ml; Bayer Korea, Seoul, South Korea) and Rompun (10 mg/ml; Bayer Korea). The rats were subjected to an injury at the T9 vertebral level of the spinal cord using the Chung-Ang University Hospital Model 2.0 (CAUH-2) pneumatic impactor (3 mm depth) (10, 32, 33) with a high intensity. Bladder compression was performed twice a day for emptying. Saline was injected into the muscle of the bladder of the rats in group 3 using a 500-μl syringe with a 26-gauge needle. MNP-labeled B10 cells (1 × 106) were transplanted into the bladder muscle of the rats in six areas 4 weeks after the SCI operation in group 4, and the bladder was harvested 4 weeks after cell transplantation.
Locomotor Behavior Test
Behavioral tests were performed in the treatment groups using a 300-s (15 rpm) rotarod test (Daejong Instrument, Seoul, South Korea). Locomotion scores were assessed using the Basso–Beattie–Bresnahan (BBB) scale. Testing began 1 week after injury and then continued for 8 weeks.
MRI of MNP-Labeled B10 Cells in Rat Bladder
MRI examinations were performed on 3.0T MR systems (Achieva Tx, Philips Healthcare, Amsterdam, the Netherlands, and Magnetom Verio, Siemens Healthcare, Berlin, Germany) by using a surface coil. Follow-up MR imaging was performed up to 4 weeks after transplantation of the MNP-labeled B10 cells.
Histological Examination of the Rat Bladder
At the end of the in vivo MR imaging experiments, each animal was perfused through the heart with 100 ml of cold saline and 100 ml of 4% paraformaldehyde (Sigma-Aldrich, St. Louis, MO, USA) in phosphate-buffered saline (Sigma-Aldrich). After 24 h of fixation in 4% paraformaldehyde, the spinal cord and bladder were paraffin embedded. Spinal cord and bladder sections 4 μm thick were processed for staining with Masson's trichrome (Sigma-Aldrich). To evaluate the results of the Masson's trichrome staining, representative portions of each slide were examined in a blinded fashion with a square micrometer, and the mean area of staining was expressed as a relative percentage. The mean percent collagen area was defined according to the formula: (collagen)/(collagen + muscle). These area calculations were determined as smooth muscle, which stained red, and connective tissue, which stained blue. Quantitative image analysis was performed using Optimas version 6.1 (Media-Cybernetics, Rockville, MD, USA). Histological sections of 4 μm obtained from the parts of the spinal cord with lesions were stained with 0.2% thionin (Nissl stain; Sigma-Aldrich). Nissl stain was used to observe the morphological changes in the neurons of the spinal cord. The spinal cord sections were deparaffinized in xylene, rehydrated in alcohol of different concentrations, immersed into 0.2% thionin solution for 20 min, and finally dehydrated with alcohol followed by clearing with xylene (Daelim CNS, Seoul, South Korea).
Recovery of Voiding Function After hMSC Transplantation
Voiding response was assessed at 4 weeks after transplantation. The rats were anesthetized with isoflurane (2% oxygen) for surgical insertion of a bladder catheter. The bladder was exposed via a midline abdominal incision. A catheter (PE-50; Harvard Clinical Technology, Inc., South Natick, MA, USA), the bladder end of which was heated to create a collar, was inserted through a small incision in the bladder dome, and a suture was tightened around the collar. The other end of the catheter was passed through the subcutaneous tissue and exited through the skin. After closing the abdominal incision by suturing the muscle and skin, rats that were to be studied without anesthesia were placed for 5–6 h (including 2 h of recovery from isoflurane anesthesia) in a restraining cage (Jeung DO Instrument, Seoul, South Korea) that was large enough to permit them to adopt a normal crouching posture, but narrow enough to prevent them from turning around. The rats were subsequently allowed to recover from the isoflurane anesthesia (JW Pharmaceutical, Seoul, South Korea). The bladder catheter was connected via a T-stopcock (Daihan Scientific, Seoul, South Korea) to a pump (Harvard Clinical Technology, Inc.) for continuous infusion of physiological saline and to a pressure transducer. Physiological saline was infused at room temperature into the bladder at a constant rate of 0.04 ml/min to elicit repeated voiding responses. The intravesical pressure to induce micturition, pressure threshold (PT), maximal voiding pressure (MVP), and intercontraction interval (ICI; the interval between voids or reflex bladder contractions) were measured.
Immunofluorescence to Detect Differentiation of Transplanted hMSCs
Tissues were embedded in paraffin, sectioned (4 μm thick), and mounted onto glass slides (Dongseo Science, Seongnam, South Korea). Sections were deparaffinized in xylene and rehydrated in a graded series of alcohol solutions, followed by dH2O. Antigen retrieval was performed in a dH2O steam bath for 30 min. Tissues were immuno stained with monoclonal human mitochondria antibody (1:200; Millipore, Billerica, CA, USA) to identify the presence of transplanted B10 cells. Antibodies specific for smooth muscle actin (SMA; 1:1,000; Abcam, Cambridge, MA, USA) were used for the identification of smooth muscle cell types. Bladder sections were incubated in specific primary antibodies overnight at 4°C. The bladder sections were washed three times in Tris-buffered saline-Tween solution (Sigma-Aldrich), and then incubated with Alexa Fluor 488-conjugated anti-rabbit IgG and Alexa Fluor 594-conjugated anti-mouse IgG (1:500; Molecular Probes, Eugene, OR, USA). Negative control sections from each animal were prepared for immunohistochemical staining in an identical manner, except that the primary antibodies were omitted. Stained sections were then examined under an Olympus fluorescence microscope (IX71; Tokyo, Japan).
Statistical Analysis
One-way analysis of variance (ANOVA) and Tukey's post hoc test were used for analysis of stem cell transplantation. Statistical analysis was performed by using SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA). Data are presented as means ± SE. A value of
Results
Phenotype of Human MSCs
The cloned B10 cells are 8–10 μM in size and tripolar or multipolar in morphology. Cytogenetic analysis of B10 cells showed that the normal karyotype of B10 was 46, which is a normal human karyotype (data not shown). The doubling time of B10 cells was determined to be 36 h.
Locomotor Behavior
The hindlimbs of all the animals with SCI were completely paralyzed 1 day after contusion injury. Both of the rotarod and BBB scores were 0, and the development of SCI was confirmed and still preserved until 8 weeks after SCI (Fig. 1). Although the bladders were compressed twice a day, distention of the bladders in all the animals was noticed during the phase of manual expression.

Functional test of the SCI model after B10 cell transplantation. (A) Maintenance time on the rotarod. (B) BBB open-field locomotor scores for the sham-operated, injury, injury + B10 cells groups tested at 4 weeks after transplantation.
In Vivo MR Imaging
Observation of MNP-labeled B10 cells by immunofluorescence microscopy demonstrated abundant RITC dots in the cytoplasm of the B10 cells (Fig. 2C, D). MR imaging of the MNP-labeled B10 cells in the bladder showed hypointense signals on T2*-weighted images until 4 weeks posttransplantation. B10 cells positive for human mitochondria antigen were found in the transplantation site, indicating that MNP-labeled B10 cells survived in the bladder at 4 weeks posttransplantation (Fig. 2).

In vivo MR imaging of MNP-labeled B10 cells in the bladder of SCI model rats. (A) Schematic drawing of the bladder, urethra, and transplanted hMSCs. (B–D) Confirmation of transplanted MNP-labeled B10 cells. (B) The area of hypointense signals 4 weeks posttransplantation as seen with MR imaging. (C) In vitro immunostaining of fluorescent MNP-labeled B10 cells. The abundant RITC dots in the cytoplasm of the cells are clearly visible. (D) B10 cells positive for human mitochondria antigen were found in the B10 cells transplanted into the bladder, indicating that MNP-labeled B10 cells are present in the bladder at 4 weeks posttransplantation. MR, magnetic resonance; MNP, magnetic nanoparticles; B, bladder wall; M, injected hMSC; U, urethra. Scale bar: 10 mm (B); 20 μm (C); 50 μm (D).
Body and Bladder Weight
There were no significant differences in body weights between the groups. The SCI group showed higher bladder weight than the sham group. Bladder weight was lower in the B10 cell transplantation group than in the SCI group (

Changes in body weight (A) and bladder (B) after B10 cell transplantation. There was no significant difference in body weight between the groups. The group with SCI showed an increased bladder weight compared with the sham-operated group (
Histological Examination Results of the Spinal Cord
Nissl staining of the spinal cord sections 2 and 8 weeks post-SCI demonstrated that the Nissl bodies of neurons were dissolved, and the nuclei shifted toward the periphery of the cells. Nonrecovery from SCI was still evident from Nissl staining at 8 weeks after SCI (Fig. 4).

Nissl staining of lesions in the spinal cord following SCI. The spinal cords of the rats were isolated at 2 (A) and 8 weeks (B) after SCI. Nissl staining was performed in coronal sections of the spinal cord to examine the regeneration of the injured CNS. There were no regenerated neurons around the lesions seen on any slides, indicating that the spinal cord was not regenerated. Scale bar: 200 μm.
Changes in Collagen Deposition After Transplantation
Collagen deposition in the bladder increased markedly in the SCI group compared with the control group, and collagen deposits were significantly decreased in the SCI group that received B10 cell transplantation into the bladder compared with the control SCI group without transplantation (Fig. 5). The SCI group showed a higher ratio than the sham-operated group (

Histological changes in collagen deposition after B10 cell transplantation. Collagen deposition increased in the group with SCI and decreased after B10 cell transplantation. (A–H) Masson's trichrome staining. Control, A (100×) and B (400×). Sham, C (100×) and D (400×). SCI, E (100×) and F (400×). SCI + B10 hMSCs, G (100×) and H (400×). Scale bar: 200 μm. (I) Changes in collagen deposition after B10 cell transplantation. The group with SCI showed an increase in percentage collagen area, which decreased after the transplantation of B10 cells. B10, human mesenchymal stem cells; Normal, control; SCI, rats with spinal cord injury; SCI + B10, B10 cell transplantation rats with spinal cord injury.
Differentiation of Xenografted Human MSCs in the Bladder
In the bladder transplanted with B10 cells, grafted B10 cells expressing immunoreactivity against SMA were found, indicating that B10 cells differentiated into smooth muscle cells. The merged cells expressing yellow fluorescence were doubly positive for human mitochondria antigen and SMA, indicating that they were from B10 hMSCs (Fig. 6).

Differentiation of B10 hMSCs into smooth muscle. (A) In transplanted bladder sections, B10 cells expressed immunoreactivity against human mitochondria. (B) In transplanted bladder sections, B10 cells expressed immunoreactivity against SMA. (C) Merged cells expressing yellow fluorescence were SMA-positive cells that were derived from B10 cells. (D) The magnified view clearly shows merged cells expressing yellow fluorescence. B10, human mesenchymal stem cells; SMA, smooth muscle actin. Scale bar: 50 μm (C) and 20 μm (D).
Changes in Voiding Function Following Transplantation
ICI was lower following SCI and increased after B10 cell transplantation (
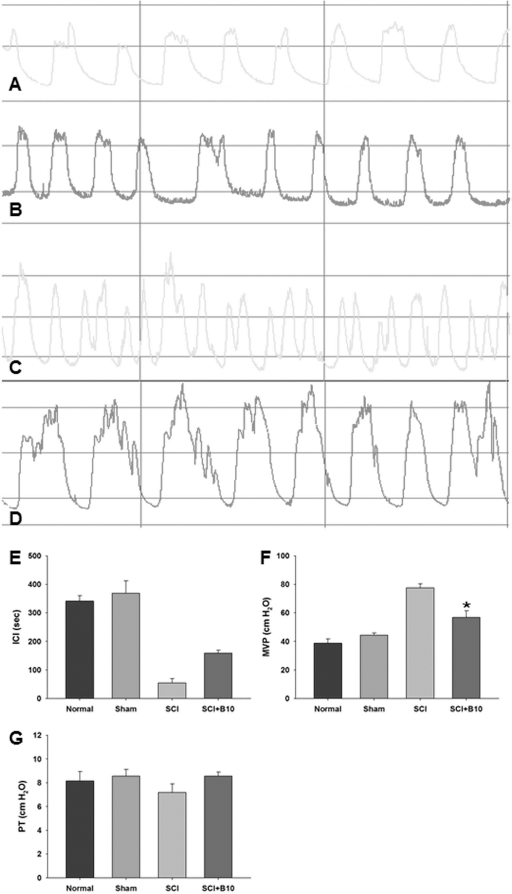
Improvement of bladder function after B10 cell transplantation. (A–D) CMG: A, control; B, sham; C, ICI decreased in SCI rats; D, ICI recovered in SCI rats + B10 cells transplantation. (E–F) Analysis of CMG. (E) The SCI rats showed decreased ICI compared with the sham-operated group, and this decrease was reversed after transplantation of B10 hMSCs (
Discussion
SCI weakened voluntary bladder and external urethral sphincter control in rats (18). Continuous cocontractions of the bladder and sphincter (bladder–sphincter dyssynergia) lead to inefficient voiding and large residual urine volumes (21). Continuous vesicular pressure and urine retention in spinalized rats might lead to complete deterioration of bladder compliance, function, infection, and other lower urinary tract complications (35). Augmented collagen deposition is considered an indicator of reduced bladder compliance and is consistent with reports of experimental animal models with SCI (16, 30).
Stem cell therapies for SCI have been evolving in their use in experimental models, showing promising recovery of bladder function. To date, direct stem cell injection in injured cord and systemic injection of stem cells in SCI animal models have been reported (5, 11–13, 29). However, no studies have dealt with the direct transplantation of MSCs into the bladder wall to influence bladder function following SCI.
The B10 stably immortalized hMSC line expressed cell type-specific markers for MSCs that were consistent with our previous study (26), and potential applications of B10 cells in fields of stroke, bladder fibrosis due to bladder outlet obstruction, and erectile dysfunction have been investigated (9, 26, 27).
MSCs are known to reduce fibrosis in the liver and lung (3, 20). In bladder outlet obstruction, transplantation of hMSCs into the bladder wall of a rat inhibits bladder fibrosis (26). In the present study, transplantation of B10 cells into the bladder wall reduced collagen deposits in the bladder following SCI.
In the spinal cords from the control SCI group, spinal neurons showed dissolved Nissl bodies, indicating that these neurons underwent degeneration and did not recover from the injury. Reduction of collagen deposition in the bladder was due to the transplantation of B10 cells into the bladder wall, rather than the repair of the SCI. hMSCs can secrete growth factors and contribute to the reduction of fibrosis through paracrine mechanisms, rather than by cell incorporation (8, 24). In a previous study, we showed that B10 cells secrete various growth factors including nerve growth factor, brain-derived neurotrophic factor, neurotrophin-3, insulinlike growth factor, HGF, glial-derived neurotrophic factor, ciliary neurotrophic factor, and vascular endothelial growth factor (15). We have also previously shown that the HGF-overexpressing B10 cells inhibit collagen deposits in bladder outlet obstructions in rat more than hMSCs alone does (26). HGF suppresses the expression of transforming growth factor-β1 (TGF-β1) and its type I receptor, TβR-1 (14). HGF release from B10 cells may be mediated by specific targeting and blocking of TGF-β1 expression and signaling (14). Thus, HGF could be a promising agent for treating bladder fibrosis.
We found that TGF-β protein expression also increased after bladder outlet obstruction and recovered after the transplantation of MSCs (26).
In contrast to the local injection of rat MSCs, there is concern that capillary clogging could occur when a larger number of cells, such as rat MSCs are infused. Such a potential complication could result in hemodynamic compromise, interference with pulmonary gas exchange, and respiratory distress (4). Indeed, intravenously injected hMSCs are found to localize mainly in the pulmonary capillary bed (23). Thus, local injection of hMSCs may be more beneficial than systemic intravenous injection. In the present study, the presence of B10 cells was verified using an anti-human mitochondria antibody in bladder sections transplanted with B10 cells into the bladder wall. In addition, the presence of MNP-labeled B10 cells was demonstrated using MRI.
SCI rats exhibit frequent urine expulsion (31). In the present study, aggravated ICI and MVP after SCI improved after B10 cell transplantation. B10 cell recruitment to the bladder after SCI appeared to be associated with increased regeneration of bladder smooth muscle cells, which may have contributed to an improvement in the bladder dysfunction (32). Together, the inhibition of bladder fibrosis also improved the cystometric parameters in SCI in rats.
Molecular imaging would be a valuable tool for the development and evaluation of cell-based treatment strategies (28). In the present study, the ability of in vivo MR imaging to detect labeled hMSCs was confirmed in the bladder of rats with SCI. The validation of MNP-labeled B10 cells was made using anti-human mitochondria antibody in rat bladder sections. Transplanted MNP-labeled B10 cells were observed in vivo at 4 weeks posttransplantation.
In conclusion, B10 cells directly transplanted into the bladder wall inhibited bladder fibrosis and mediated the recovery of bladder dysfunction in a rat SCI model. Transplantation of a MSC-based cell therapy could be a novel therapeutic strategy to reverse bladder dysfunction in patients with SCI.
Footnotes
Acknowledgments
