Abstract
Neonatal stroke is a major cause of mortality and long-term morbidity in infants and children. Currently, very limited therapeutic strategies are available to protect the developing brain against ischemic damage and promote brain repairs for pediatric patients. Moreover, children who experienced neonatal stroke often have developmental social behavior problems. Cellular therapy using bone marrow mesenchymal stem cells (BMSCs) has emerged as a regenerative therapy after stroke. In the present investigation, neonatal stroke of postnatal day 7 (P7) rat pups was treated with noninvasive and brain-specific intranasal delivery of BMSCs at 6 h and 3 days after stroke (1 × 106cells/animal). Prior to transplantation, BMSCs were subjected to hypoxic preconditioning to enhance their tolerance and regenerative properties. The effects on regenerative activities and stroke-induced sensorimotor and social behavioral deficits were specifically examined at P24 of juvenile age. The BMSC treatment significantly reduced infarct size and blood-brain barrier disruption, promoted angiogenesis, neurogenesis, neurovascular repair, and improved local cerebral blood flow in the ischemic cortex. BMSC-treated rats showed better sensorimotor and olfactory functional recovery than saline-treated animals, measured by the adhesive removal test and buried food finding test. In social behavioral tests, we observed functional and social behavioral deficits in P24 rats subjected to stroke at P7, while the BMSC treatment significantly improved the performance of stroke animals. Overall, intranasal BMSC transplantation after neonatal stroke shows neuroprotection and great potential as a regenerative therapy to enhance neurovascular regeneration and improve functional recovery observed at the juvenile stage of development.
Introduction
Neonatal stroke occurs in approximately 1 per 1,000 newborn babies and is a major cause of mortality and long-term morbidity (41,54). Current therapeutic strategies to protect the developing brain against ischemic damage in pediatric patients are very limited (28). Moreover, children with perinatal asphyxia develop problems in their social behaviors later in their lives (19). Similarly, mild and moderate neonatal encephalopathy may lead to social problems, anxiety and depression, cognitive dysfunction, attention deficits, and some other neurological/neuropsychiatric problems in juveniles and young adults (55,66).
Stem cell transplantation holds promise as a regenerative therapy for neonatal brain injury (45,52). Bone marrow-derived mesenchymal stem cells (BMSCs) have been considered as an appropriate cell source for treatment of some neonatal disease models, including perinatal asphyxia, hypoxic–ischemic brain injury, and cerebral palsy (17,43, 44,56,58). However, whether BMSC therapy may have a long-term benefit to prevent or reverse neurological and neuropsychiatric problems is not well established (12).
In the present investigation, we tested BMSC transplantation after neonatal stroke in postnatal day 7 (P7) rat pups. BMSCs were delivered intranasally at 6 h and 3 days after onset of neonatal stroke. The intranasal route is noninvasive and targets the brain tissue specifically. Our previous study had demonstrated the presence of the intranasally delivered cells in the ischemic hemisphere. Exogenous cells reached the ischemic cortex and deposited outside of vasculatures as early as 1.5 h after intranasal administration (65). The present investigation tested the feasibility of repeated intranasal administrations of BMSCs and examined its lasting effects on regenerative mechanisms and functional benefits in juvenile rats that had suffered neonatal stroke.
Materials and Methods
Neonatal Stroke Model
Barrel cortex ischemia in neonates was induced with modifications. Compared to our previous method (30), the modifications included the animal gender, anesthesia method, and silk suture size. Male P7 Wistar rats (Charles River Laboratories, Wilmington, MA, USA) were anesthetized by 1.5% isoflurane (Piramal Healthcare, Andhra Pradesh, India). Pups were placed in a noninvasive head holder (Smiths Medical, Arden Hills, MN, USA) to allow for a 2.5- to 3.0-mm-diameter craniectomy through the right parietal skull. The transparent dura was left intact over the whisker-barrel cortex area. Distal branches of the middle cerebral artery were permanently ligated using sterile 10 silk sutures (Surgical Specialties, Vancouver, BC, Canada) that were passed through the dura under a dissecting microscope (Olympus, Tokyo, Japan). This was combined with permanent cauterization of the common carotid artery. Animal procedures were in accordance with protocols approved by Emory University Institutional Animal Care and Use Committee. Cerebral ischemic surgeries were performed by the same animal surgeon.
Cell Preparation and Characterization
BMSCs were isolated and harvested from the fibias of Wistar rats. We have modified our previously published methods (63) using slightly different culture conditions and characterization methods. BMSCs were flushed from the fibias of postnatal 21-day rats (Charles River Laboratories). Cells were cultured in Dulbecco's modified Eagle's medium (Cellgro, Manassas, VA, USA) supplemented with 15% fetal bovine serum (Sigma-Aldrich, St. Louis, MO, USA). Cultures were maintained at 37°C incubator containing 5% carbon dioxide. We performed fluorescence-activated cell sorting to characterize the MSC population using cluster of differentiation 105 (CD105)–phycoerythrin (PE), CD73–fluorescein isothiocyanate (FITC), CD34–FITC, and CD45–FITC markers (1:20; eBioscience, San Diego, CA, USA). All cells used in this study were harvested for transplantation during passages 3 to 5 when they were 80–90% confluent.
Cell Hypoxic Preconditioning and Intranasal Administration
Hypoxia preconditioning (HP) treatment of BMSCs prior to transplantation was performed to enhance their survival and homing to the ischemic lesion sites that would contribute to the regeneration process (25,51,68). For HP, the oxygen concentration in the chamber was maintained at 0.1–0.3% with a residual gas mixture composed of 5% carbon dioxide balanced with nitrogen for 24 h followed by 1 h of reoxygenation before harvesting. Control BMSCs were cultured in normoxic conditions for 24 h, while the experimental group was subjected to HP treatment. In this report, the term “BMSCs” refers to HP-treated BMSCs. BMSCs were rinsed with phosphate-buffered saline (PBS; Sigma-Aldrich) and harvested by trypsinization using Trypsin-EDTA (Life Technologies, Carlsbad, CA, USA) for intranasal delivery. Intranasal delivery was performed as previously described (8,65) with modifications for neonatal rats. For neonatal rats, the modifications included nonanesthesia procedures and delivery time points. To increase the barrier permeability of nasopharyngeal mucosa and facilitate the cell entry into the brain, all animals received 100 U hyaluronidase (Sigma-Aldrich) dissolved in sterile PBS, 30 min prior to the administration of cells. Five-microliter drops per nostril were given with 1-min intervals until a total volume of 100 μl of cell suspension (containing 1 × 106 cells) or vehicle was administered. The BMSC transplantation was first applied 6 h after stroke; a second transplantation was performed 3 days after stroke.
BrdU Administration
For labeling of newly generated cells, 5-bromo-2′-deoxyuridine (BrdU; Sigma-Aldrich) was administered to all animals by intraperitoneal (IP) injection. The dosage was 25 mg/kg/day before weaning and 50 mg/kg/day after weaning at P21. Injections began on day 3 after neonatal stroke (the same day of the second BMSC transplantation or saline administration) and continued once every 2 days until the animals were sacrificed at 17 days after stroke.
Assessment of Ischemic Infarct Volume
Ischemic infarct size was assessed 72 h following stroke. The brain was sliced into 1-mm coronal sections using a rodent brain matrix (Harvard Bioscience, South Natick, MA, USA) and incubated in 2% triphenyltetrazolium chloride (TTC; Sigma-Aldrich) at 37°C for 5 min. Brain sections were scanned, and the unstained versus stained TTC area was determined using NIH ImageJ (Bethesda, MD, USA) on the ventral side of five brain slices per animal. Infarct, ipsilateral hemisphere, and contralateral hemisphere areas were measured and infarct volume (mm3) and infarct ratio were calculated using the indirect method.
Extravasation of Evans Blue (E) Dye
Sterilized 2% Evans Blue (EB; Sigma-Aldrich) solution was administered intravenously at a dosage of 0.08 ml per rat at 6 h before sacrifice to visualize the blood-brain barrier (BBB) leakage. BBB permeability was investigated by monitoring extravasation of EB dye. The red autofluorescence of EB dye-albumin conjugate is evident in tissue sections examined by fluorescence microscope. Six hours after EB injection, and 72 h after stroke, rats were sacrificed with an overdose of pentobarbital (100 mg/kg; Sigma-Aldrich) and decapitated (n = 6–8 per group). Brains were removed. Coronal sections were used to examine the EB extravasation by photographing under the tetrarhodamine isothiocyanate (red) excitation wavelength at 1.25× for fluorescent microscopy (BX51, Olympus). Photoshop Professional was used to make an image mosaic (Adobe Photoshop CS5, San Jose, CA, USA).
Immunohistochemical Staining
Frozen brain sections were sliced into coronal sections 10-μm thick using a cryostat vibratome (Ultrapro 5000, St. Louis, MO, USA). The sections were completely air dried and fixed with 10% buffered formalin (Thermo Fisher Scientific, Waltham, MA, USA). Brain sections were then submerged in an ethanol (Sigma-Aldrich)/acetic acid (VWR International, Suwanee, GA, USA) solution (2:1) for 10 min, washed three times with PBS solution, and incubated with 0.2% Triton X-100 (G-Biosciences, St. Louis, MO, USA) for 45 min. Slides were then blocked with 1% fish gelatin (Sigma-Aldrich) in PBS for 1 h at room temperature. Incubation with neuronal nuclei (NeuN) anti-mouse (1:400; Millipore, Billerica, MA, USA), collagen IV (Col IV) anti-rabbit (1:400; Millipore), glucose transporter 1 (Glut-1) anti-rabbit (1:1,000; Millipore), and glial fibrillary acidic protein (GFAP) anti-mouse (1:50; Thermo Fisher Scientific, Norcross, GA, USA) primary antibodies were diluted in PBS at 4°C overnight. For double-labeled slides with BrdU (Sigma-Aldrich), brain sections were postfixed with formalin in 10% buffered formalin and washed in PBS. Slides were then treated with methanol (–20°C) and allowed to air dry. After rewetting in PBS, sections were treated with 0.1 mol/L borate buffer (Sigma-Aldrich) at pH 8.4. Slides were incubated with BrdU anti-rat antibody (1:400; Bio-Rad Laboratories, Hercules, CA, USA) overnight at 4°C. Primary antibodies were removed by rinsing with PBS and replaced with secondary antibodies conjugated to Alexa Fluor 488 (1:200), cyanine 3 (Cy3; 1:1,000) or Cy5 (1:500) (all Life Technologies) for 1 h at room temperature before rinsing again with PBS. Hoechst 33342 (Life Technologies) was used to stain nuclei. After a final rinse, slides were mounted with ProLong Antifade mounting medium (Life Technologies).
Behavior Tests
Adhesive Removal Test
We tested adhesive removal of a sticky dot at days 10, 17, and 24 after stroke by an investigator blinded to the treatment groups. The mean times (seconds, averaged from three to four trials) required to sense and remove stimuli from the left paw (time to contact and time to remove) were recorded. All testing trials were conducted during the daytime.
Olfactory Function Test
Olfactory function was assessed by using a modified buried food finding test at days 17 and 24 after stroke. The tasks rely on the sense of smell to locate food. After fasting for 24 h, the rats were placed in a 25.9 × 47.6 × 20.9-cm rat cage (Allentown, Inc., Allentown, NJ, USA) containing a pellet of rodent food buried approximately 1-cm deep around the center of the cage. The time that the mouse required to uncover the food and grab it with its forepaws or teeth was recorded.
Social Interaction Test
Animals were placed into a test cage, and the time spent in social exploration was recorded at days 17 and 24 after stroke. Social sniffing was defined by the test animal sniffing any part of the body of the test partner. Social grooming was defined by the animal licking and chewing the fur of the test partner, while placing its forepaws on its back or its neck. Following/chasing behaviors were recorded when the rat walked or ran in the direction of the test partner, which stayed where it was or moved away. Total time of active social interactions in this investigation was obtained as the sum of interaction times per 10 min.
Home Cage Behavioral Tests
Behavioral changes of rats were monitored and analyzed using the automated behavior recognition software (Clever Sys Inc., Reston, VA, USA). The monitoring system had four cameras that monitored four cages, with each cage (191 mm × 292 mm × 127 mm) containing one neonatal rat. The behavior patterns were continuously recorded for 24 h. For this behavioral study, we used 12–16 animals per group. Activities were monitored at days 17 and 24 after stroke, and videos were analyzed by the HomeCageScan Software 3.0 (Clever Sys Inc.).
Statistical Analysis
For comparison between two groups, we used the Student two-tailed t-test. Graph Prism version 5.0 (San Diego, CA, USA) was used to make graphs and to perform statistical analysis. One-way analysis of variance (ANOVA) was used for data analysis and multiple comparisons (for three-group data) were corrected with Bonferroni's test for pairwise comparisons. Blind counting for the number of marker-positive cells was performed using ImageJ software. Significance was assumed at a value of p = 0.05 in all statistical analyses. Randomization was performed, and the sample size was further determined using Power analysis (Power and Precision 4; Biostat, Inc., Englewood, NJ, USA).
Results
BMSC Transplantation Decreased Infarct Size and Blood–Brain Barrier Injury After Neonatal Stroke
We showed in previous reports that HP BMSCs and neural progenitor cells exhibited elevated survival rates in vitro and after transplantation (65). Based on these observations, HP was adopted as a routine cell culture procedure in the present investigation. After transplantation into neonatal rats at 6 h after stroke, BMSCs significantly reduced the infarct volume measured by TTC staining at 3 days after stroke (p < 0.05) (Fig. 1A–C). EB assay also showed significantly decreased extravascular leakage after BMSC treatment (p < 0.05) (Fig. 1D–F). This suggested that BMSC delivery could attenuate brain edema.

Neuroprotection by BMSCs after transplantation into neonatal rats. (A–C) Brain infarct formation was measured 3 days after focal ischemia using TTC staining. Hypoxic preconditioned BMSCs were intranasally delivered 6 h after the onset of stroke. Mice that received BMSCs showed smaller infarct size compared to stroke mice with saline control. Scale bar: 2 mm. (D, E) EB assay showed the decreased extravascular leakage after BMSC treatment. Scale bar: 2 mm. (F) There were significantly lower ratios of leakage area in the BMSC treatment group. Mean ± SEM; n = 6–8; *p < 0.05 compared to saline.
BMSC Transplantation Enhanced Angiogenesis and Neurogenesis in the Developing Brain After Neonatal Stroke
Angiogenesis and neurogenesis are long-term regenerative processes during neurovascular remodeling and repair after ischemic injury (16). To label the newly formed cells, BrdU was injected 3 days after stroke and continued for 14 days. In stroke control animals at 17 days after stroke, there were Glut-1 (marker for endothelial cells) and BrdU-positive cells in the peri-infarct region, suggesting formation of new vessels (Fig. 2A). GFAP-positive astrocytes were observed around new vessels (Fig. 2A). This angiogenic activity was significantly higher in BMSC-treated animals indicated by Col IV and BrdU colabeling using a stereological counting method, as we previously described (p < 0.05) (64) (Fig. 2B–E). In addition, NeuN and BrdU double-positive cells were also dramatically increased compared to both sham control and stroke control animals (Fig. 2B–D, F). Consistent with increased angiogenic activity, local blood flow in BMSC-treated animals recovered much better at 17 days after stroke compared to stroke controls (Fig. 3).

Neurovasculature in penumbra regions of the developing brain after neonatal ischemia. Immunohistochemical fluorescent imaging was performed 17 days after neonatal stroke. (A) Representative three-dimensional image showing neurovasculature in penumbra regions. Red: BrdU; green: Glut-1 stained for endothelial cells; blue: GFAP stained for astroglia; purple: Hoechst 33342. (B–D) Representative images of BrdU+/Col IV+/NeuN+ staining. (E, F) Quantified data for BrdU-colabeling vessels or neurons. There were significantly higher proportions of BrdU+ vessels and BrdU+ neurons in the BMSC treatment group. Scale bar: 20 mm. Mean ± SEM; n = 6–8; *p < 0.05 compared to sham; #p < 0.05 compared to saline.

Blood flow measurement in the penumbra of the developing brain after neonatal ischemic stroke. Cerebral blood flow was measured in sham, vehicle (saline)-treated stroke control, and BMSC-treated stroke rat pups using laser Doppler imaging. The assay was performed in P24 rats. Blood flow was measured in both contralateral and ipsilateral hemispheres. The local blood flow in the peri-infarct region was significantly decreased in stroke animals but improved in BMSC-treated rats. Mean ± SEM; n = 6–8; *p < 0.05 compared to sham; #p < 0.05 compared to saline.
BMSC Transplantation Improved Sensorimotor and Olfactory Function After Neonatal Stroke
The focal ischemic stroke specifically damaged the sensorimotor cortex in the ipsilateral hemisphere. The functional deficit can be accurately detected using the adhesive removal test (5). In stroke control animals, the time to sense/ contact the sticky dot attached to the affected forepaw and the time to remove the sticky dot were both significantly increased. At 17 days after stroke, BMSC-treated stroke rats acted significantly faster in both activities compared to stroke controls (p < 0.05) (Fig. 4A, B). In our stroke model, we also observed the development of an olfactory function deficit at 17 days after stroke, resulting in an increased time for stroke animals to locate hidden food. Stroke animals that received BMSC transplantation, however, needed significantly less time to find the food (p < 0.05) (Fig. 4C).

Sensorimotor and olfactory functional recovery after neonatal stroke. The adhesive removal test was performed at 17 days after ischemia. The time to contact (A) and remove the sticky dot from the left paw (B) was significantly reduced in rat pups receiving BMSCs compared with saline-treated rats. (C) Intranasal BMSC treatment improved olfactory functions measured at 17 days after stroke compared to both sham control and saline-treated stroke animals. Mean ± SEM; n = 12–16; *p < 0.05 compared to sham; #p < 0.05 compared to saline.
BMSC Transplantation Increased Social Behaviors in Juvenile Rats After Neonatal Stroke
Neonatal stroke may cause long-lasting neuropsychiatric disorders (1). The social interaction behavior was tested in the neonatal stroke model. Seventeen days after stroke, stroke animals spent significantly less time grooming each other and had less total social time compared to sham animals of the same age (p < 0.05) (Fig. 5). On the other hand, stroke animals that received BMSC transplantation showed significantly higher interest in social activities. They spent much longer time in social grooming, following, and social sniffing compared to saline-treated stroke animals (p < 0.05) (Fig. 5).
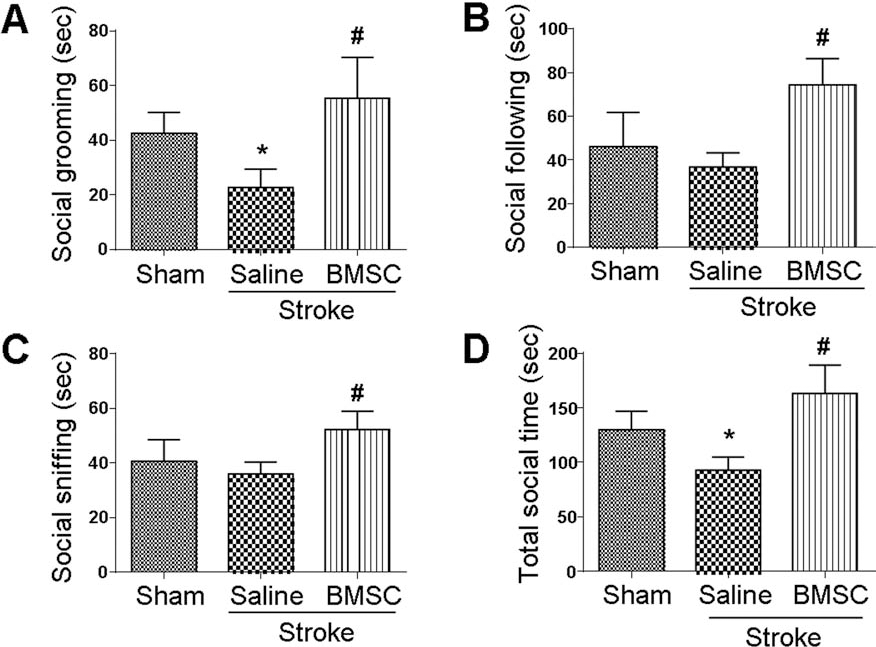
Social behavior alterations after neonatal stroke. The social interaction test was performed at postnatal day 24. Intranasal BMSC treatment improved social behaviors [social grooming (A), social following (B), and social sniffing (C)] in rat pups measured by calculating social behavior activity time (D). Mean ± SEM; n = 12–16; *p < 0.05 compared to sham; #p < 0.05 compared to saline.
BMSC Transplantation Increased Home Cage Activities After Neonatal Stroke
Using a home cage monitoring system and automated behavior recognition software (Clever Sys Inc.), we measured some natural activities of stroke and normal rats in an environment without human intervention. At age P24, 17 days after stroke, the animals showed significantly lower active behaviors compared to controls, including traveling, coming down, rearing up, jumping, grooming, and hanging behaviors (p < 0.05). Stroke rats that received BMSC treatment improved in these activities (Fig. 6A–F). We also observed increased sleep time after stroke, while BMSC-treated stroke rats did not increase their sleep time (Fig. 6G).
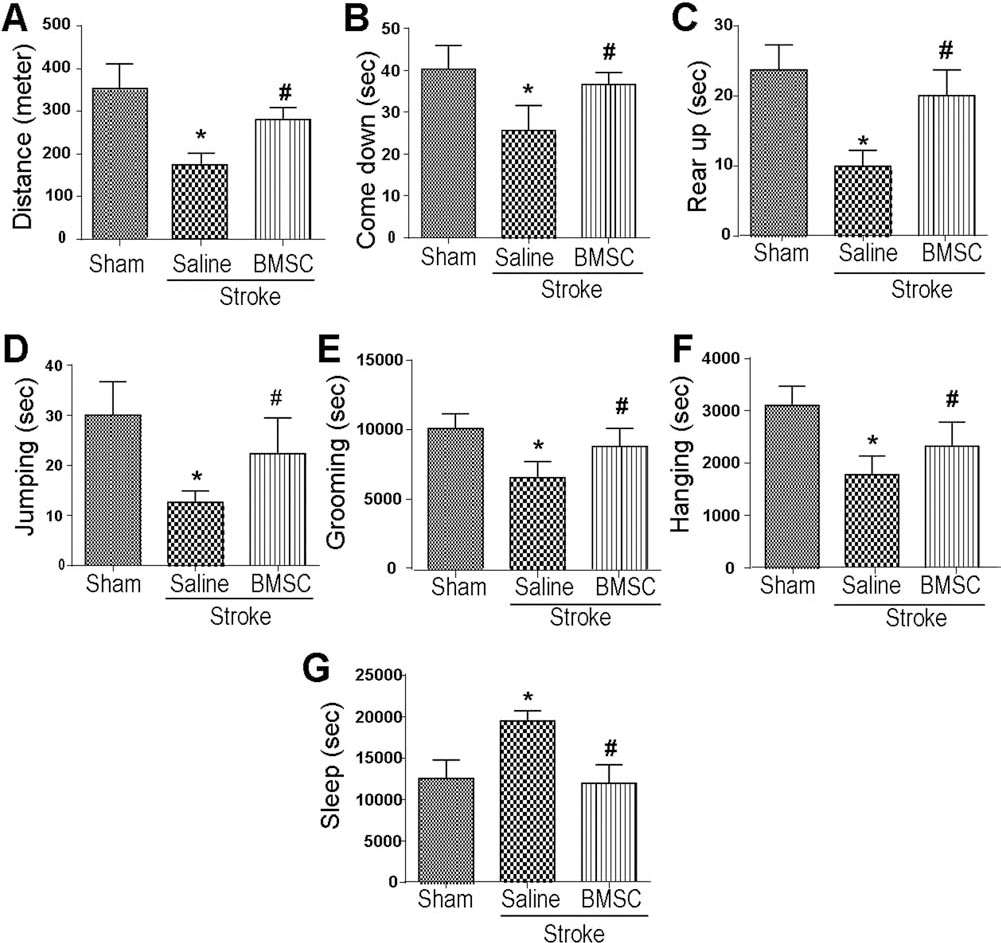
Home cage activities after neonatal stroke. In P24 rats, the home cage monitoring system was used to monitor the animal's behaviors in their home cage environment without human intervention. Some active behaviors (A–F) and sleep time (G) were measured. Mean ± SEM; n = 12–16; *p < 0.05 compared to sham; #p < 0.05 compared to saline.
Discussion
Neonatal stroke and perinatal hypoxic–ischemic brain injury cause high mortality and long-term morbidity. Although current understanding of the underlying mechanisms has improved during recent years, few effective treatments and therapeutic options have been developed. Prenatal or perinatal injury such as inflammation and perinatal asphyxia to the developing brain can generate behavioral problems and neuropsychiatric deficits in infants and children (3,38). Previous investigations using perinatal asphyxia P1 rat model revealed some neurological alterations at P15 such as social withdrawal, neophobia, and stereotyped behaviors (36). In our model, we used a P7 rat pup, which is believed to be equivalent to the full-term human infant in terms of the developmental stage of the CNS (9,70). The present investigation of the neonatal stroke model and long-term observation further reveals the neurological and neuropsychiatric alterations after the focal cerebral ischemia. More importantly, we show that transplantation of BMSCs via the intranasal route is effective in attenuating the functional deficits.
Recently, BMSCs have been used in clinical trials to treat stroke patients (23,35). The protocol has been tested in nonhuman primates and human patients who receive intravenous injections of their own BMSCs (34,40,46). The results show neuroprotection, neurotrophic effects, and anti-inflammatory responses after transplantation therapy (20,22). In preclinical laboratories, BMSCs have been considered a promising cell source for treatment of many diseases, including ischemic stroke (15,63), traumatic brain injury (60), intracerebral hemorrhage (62), subarachnoid hemorrhage (31), spinal cord injury (33), and other neurodegenerative disorders (2,29,39,42). However, most preclinical and all clinical studies have been performed on adult animals or humans, and only a few reports have studied stem cell therapy in neonatal stroke models (52,56, 59,61). Those laboratories show great promise for cellular therapy using core blood stem cells or BMSCs to provide enhanced trophic support and an enriched microenvironment in the damaged developing brain (11,32,57).
It was shown that BMSCs released cytokines, chemokines, and growth factors such as brain-derived neurotrophic factor, epidermal growth factor, fibroblast growth factor 2, glial-derived neurotrophic factor, hepatocyte growth factor, insulin-like growth factor 1, nerve growth factor, placental growth factor, stromal cell-derived factor 1, transforming growth factor, and vascular endothelial growth factor (VEGF) (13). Pretreated or genetically modified BMSCs targeting the elevation of these factors showed increased cell viability and enhanced migration and homing capacity to lesion sites (21,24,48,50,53,69). BMSCs can stimulate endogenous neuronal differentiation and synaptic connection, thus improving neuronal activity (26). BMSC transplantation can promote the proliferation of neural stem cells in the subventricular zone and neurogenesis in the ischemic region (37).
Demonstration of the survival or death of transplanted BMSCs and the migration route after intranasal administration of these cells were reported in our previous investigation (65). Specifically, we were one of the first groups to develop the novel strategy of HP of stem cells and neural progenitor cells in order to improve their survival and regenerative property after transplantation. We previously showed that HP BMSCs expressed high levels of CXC chemokine receptor type 4, matrix metalloproteinase-2 (MMP-2), and MMP-9. These genes helped the transplanted cells reach the brain through nasal routes (10,27) to the peri-infarct regions. In the present investigation, HP BMSCs with higher cell viability were used as a routine in vitro procedure before transplantation.
We and many other groups have also identified the BMSC-stimulated microvessel reconstruction in the ischemic cortex, indicating an angiogenesis/neurovasculaturetargeting therapy for functional recovery in neonates with stroke (7,14,18,30). To improve the effectiveness of the stem cell therapy, we administered BMSCs twice at 6 h and 3 days after stroke. It was presumed that early delivery may show a neuroprotective and an anti-inflammatory action, while the delayed administration targeted an intermediate phase of angiogenesis, neurogenesis, axonal sprouting, neurovascular repair, and remyelination. As we predicted, transplantation of BMSCs at 6 h after stroke showed significant protective effects of reducing infarct formation and protecting the BBB.
Immune-modulatory benefits of transplanted BMSCs and a global downregulation of proinflammatory cytokines can be found at 4 weeks after transplantation, which may contribute to long-term benefits (47,49). Owing to these therapeutic potentials, secondary damages can be effectively prevented by postischemia transplantation of BMSCs into ischemic brain through integrated mechanisms involving host neurons, astrocytes, and microglia (4,67). We show here that this transplantation method has noticeable impacts many days after stroke and has significantly improved regeneration originating from either or both endogenous and exogenous.
In a neonatal stroke model, administration of BMSCs can reduce white matter injury, improve motor deficits, and stimulate forebrain repairs (6). Our data have demonstrated that intranasally delivered HP BMSCs significantly improve sensorimotor and locomotor activity of juvenile rats subjected to a focal cortical ischemia at P7. Based on the previous results showing one possible route through the olfactory bulb, we detected enhanced olfactory functions of rat pups. Since very few studies have addressed the social behavioral changes after ischemic stroke in neonates, our work seems to be innovative in showing the social behavioral benefits of BMSC transplantation in the developing stages of rats. The recovery of sensorimotor functions and enhanced olfactory functions may help to promote social interactions in rat pups. Further investigation needs interpretation of the underlying mechanisms.
Footnotes
Acknowledgments
This work was supported by NIH grants NS085568 (L.W.), NS057255 (S.P.Y.), NS058710 (L.W.), NS073378 (S.P.Y.), AHA Grant-in-Aid 12GRNT12060222 (S.P.Y.), and AHA Established Investigator Award (L.W.). The authors declare no conflicts of interest.
