Abstract
In mammals, the two main types of adipose tissues, white and brown adipose tissues, exert different physiological functions. White adipose tissue (WAT) is for storing energy, while brown adipose tissue (BAT) is for energy consumption. Adipose-derived stem cells (ADSCs) are abundant in WAT and BAT, have multipotent characteristics, and are easily extracted. ADSCs can be differentiated into several cell lineages, including adipocytes, osteoblasts, chondrocytes (cartilage cells), myocytes, and neuronal cells. Therefore, ADSC could be considered as a strategy for future regenerative medicine and tissue engineering.
Keywords
Introduction
Recently, we have seen great progress in differentiating molecular regulation of specific cell types. Excessive energy would be stored in adipose tissue when energy intake exceeds energy expenditure for a long time. Adipose tissue is distributed across multiple locations. One of the largest stores are the superficial adipose tissue layers and deep adipose tissue layers, which account for approximately 80% of total body fat including subcutaneous fat; the other is internal adipose tissue (visceral and nonvisceral), which accounts for approximately 20% of the total body (22).
There are two types of adipose tissues in mammals, white and brown adipose tissues. White adipose tissues (WAT) can provide tissues with lipids acting as energetic fuel or lipid reconstructive material, and brown adipose tissue (BAT) can utilize fatty acids for thermogenesis (26). So far, the role of adipose tissue is crucial for energy homeostasis. However, the molecular mechanisms that regulate preadipocyte cell growth, adipose differentiation, and lipogenesis in adipocytes cells are unclear (27). Adipogenesis is the process whereby undifferentiated progenitor cells differentiate into fat cells (62). The transcriptional basis of the differentiation of fat cells, the expression of adipose genes, and promotable adipogenesis has been increasingly described in recent years (9,44,45). When appropriate hormonal agents, such as corticosteroids, cyclic adenosine monophosphate (cAMP), insulin-like growth factor-1 (IGF-1), and other cellular growth factors, stimulate the culture of preadipocytes, the differentiation of adipocytes can be promoted compared to nonstimulated cells (46). There is a large amount evidence that the differentiation of preadipocytes faithfully imitates almost all of the biochemical and morphological characteristics of fat cells to the cell body (46). Following hormonal agent induction, confluenced preadipocytes proceed through mitotic clonal expansion, arrest cell growth, and then coordinate the expression of fat cells (45).
Adipose Tissues and Adult Stem Cells
Recently, pluripotent stem cells have been tested to induce adipose tissue, as well as several other lineages. Adipose tissues, by providing substantial energy reserves of vertebrate organisms, may be mobilized to demand energy economy and so play an important function. Adult stem cells possess self-renewal properties and multipotency (multidifferentiative potential) (5). Recently, stem cell research has advanced greatly and rapidly with the application of human bone marrow and embryonic stem cells for regenerative medicine and repair. A growing focus on the therapeutic use of stromal cells derived from adipose tissue, so-called adipose-derived stem cells (ADSCs), also plays an important role in the regenerative application. Unfortunately, the naming scheme for ADSCs is very inconsistent across studies including: adipose stem cells (ASCs), adipose-derived stem cells (ADSCs), adipose-derived adult stromal cells (ADASCs), adipose-derived adherent stromal (ASAS) cells, adipose mesenchymal stem cells (AdMSCs), preadipocyte, processed lipoaspirate (PLA) cells, and others (3,21,25). It has been reported that the extraction rate of stem cells derived from adipose tissue is generally higher than that of stem cells derived from bone marrow-derived mesenchymal stem cells (BMSCs) (10). Adipose tissue provides a rich and easy supply of stem cells that resemble MSCs with their general differentiation potential and plasticity. These cells display cell surface marker profile characteristics similar to other adult stem cells and differentiation, such as BMSCs. ADSCs express multiple stem cell-associated genes that are expressed by both BMSCs and embryonic stem cells (ESCs). Additionally, ADSCs and BMSCs share many constitutively expressed genes responsible for angiogenesis, matrix remodeling, mitogenesis, and differentiation signaling pathways including fibroblast growth factor (FGF; FGF-2, -6, -7), metalloproteinases (MMPs; MMP-3, -12, -14, -23), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), stromal cell-derived factor-1 (SDF-1), and tissue inhibitor of metalloproteinases (TIMPs; TIMP-1, -2, -3) (55). On the other hand, genes functioning on extracellular matrix remodeling, inflammation morphogenesis, and tissues repair, such as fibroblast growth factor receptor-3 (FGFR3), fibronectin, neurophilin-1, osteonectin, VEGF-D, and integrins (α5, α11, and β1), are highly transcripted by ADSCs (14,37,55).
Isolation and Culture of Adipose-Derived Stem Cells
MSCs have now been found in several tissues, including adipose tissue. In addition, adipose tissue can provide many more stem cells than bone marrow, with a yield of 5,000 cells per gram of tissue versus 100–1,000 cells (71). ADSCs can be found in any type of white adipose tissue, including the internal (visceral and nonvisceral) and subcutaneous fat. Methods to culture ADSCs have been extensively detailed in the literature (35,89), although it is worth noting the variable effects of plating density, serum level, passage, and oxygen tension during culture. Such morphology of mouse ADSCs was digested with type I or VIII collagenase and was observed in our laboratory (Fig. 1). Type I collagenase is commonly used in, and is highly recommended for the isolation of adipocytes, epithelial cells, hepatocytes, lung cells, and adrenal cells. Type VIII collagenase is generally used in adipocyte and hepatocyte isolation (35,66). Interestingly, the quantity of stromal vascular fraction (SVF) cells that we can acquire may depend on which types of collagenase we used. As shown in Figure 1, type VIII collagenase exhibited greater performance than type I collagenase.
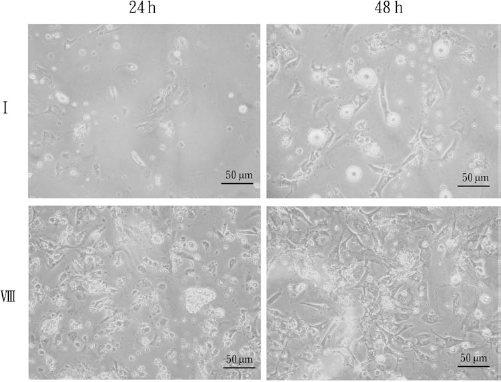
Culture containing adipose-derived stems cells 24 and 48 h after isolation of the stromal vascular fraction (SVF) with type I or type VIII collagenase from mouse adipose tissues. The fat pads of white adipose tissue were isolated from mice. Each gram of the isolated fat pad was digested with 2 mg/ml of type I or type VIII collagenase (Sigma) in 4 ml of Dulbecco's modified Eagle's medium (DMEM) at 37°C for 1 h. Morphology of adipose-derived SVF in culture observed at x40 magnification on an inverted phase-contrast microscope.
Cell Surface Marker of Adipose-Derived Stem Cells
The typical categories of MSC surface marker proteins are as follows: adhesion molecules, such as CD29 (integrin β1) and CD49e (integrin α5); activated lymphocyte cell adhesion molecule (CD166; ALCAM); receptor molecules, such as CD44 (hyaluronate) and CD144 (cadherin-5); surface enzymes, such as CD13 (aminopeptidase) and CD73 (5′ecto-nucleotidase); extracellular matrix proteins, such as CD90 (Thy-1), CD105 (endoglin), and CD146 (Muc18); intercellular adhesion molecules [CD54 (ICAM-1)]; vascular adhesion molecules [CD106 (VCAM)]; complement regulatory proteins, such as CD55 (decay acceleration factor); and histocompatibility antigens [human leukocyte antigen (HLA)-ABC] (11,14,24,71). Markers in common with BMSCs include CD13 (aminopeptidase), CD29, CD44, CD58 [lymphocyte function associated antigen-3 (LFA-3)], CD90, CD105, and CD166 (11,12).
Adipose tissue is constituted by multiple types of cells including adipocytes, endothelial cells, fibroblasts, immune cells, and vascular smooth muscle cells, as well as stem cells. ADSCs can express several traits similar to BMSCs including a fibroblast-like morphology, as well as a series of detectable cell-specific proteins and CD markers (13,20,81,85). Surface antigen expression is widely used in immunology by examining the expression of surface protein molecular in ADSCs. The ADSCs were examined for surface protein molecule expression by flow cytometry. Human ADSCs express the mesenchymal stem cell markers CD9 [tetraspanin 29 (TSPAN29)], CD10 [common acute lymphoblastic leukemia antigen (CALLA) or neprilysin or neutral endopeptidase (NEP)], CD13, CD29, CD44, CD49d (integrin α4), CD49e, CD54, CD55, CD73, CD90, CD105, CD106, CD146, CD166, and STRO-1 (stromal precursor antigen-1), but they are negative for the hematopoietic cell lineage markers CD14 [lipopolysaccharide receptor (LPS-R)], CD19 (B4), CD34 (Mucosialin), CD45 [leukocyte common antigen (LCA), Ly-5], CD16 (FcγRIII), CD56 (NCAM), CD61 (integrin β3), CD62E (E-selectin), CD104 (integrin β4), and CD106 and are also negative for the endothelial cell (EC) markers CD31 [platelet endothelial cell adhesion molecule-1 (PECAM-1)] and CD144 (13,20,28,31,53,80,81,85).
Cells in the SVF (freshly isolated) and early-passage human ADSCs have relatively higher expression of CD117 (c-kit), HLA-DR, and stem cell-associated markers, such as CD34, and low levels of stromal cell markers, such as CD13, CD29, CD44, CD63 [lysosome-associated membrane glycoprotein 3 (LAMP-3)], CD73, CD90, CD166, and CD105 (53,80,86). However, in human ADSCs, SVF (fresh), or early-passage ADSCs, the following cell markers are differentially expressed: CD14, CD31, CD34, CD105, CD106, CD117, CD166, HLA-DR, and STRO-1 (Table 1). Being WAT separating the initial fresh SVF, the passage and expanded human ADSCs show that these cells appear with stem cell markers, such as CD29, CD44, CD73, CD90, CD105, and CD166 (28,76,91). When the passage number increased, the hematopoietic stem cell (HSC) markers, such as CD11, CD14, CD45, and CD34, expressed on SVF will decrease or be lost because SVF cells consist of multiple-cell populations in comparison with ADSCs that were selected and subsequently expended from adipose tissue as a single-cell population (53).
Cell Surface Markers of ADSC, SVF, and BMSC in Human and Mouse

ADSC, adipose-derived stem cell; SVF, stromal vascular fraction, isolated from white adipose tissue; BMSC, bone marrow-derived stem cell. +, positive in all cited references; +/-, both positive and negative references found; -, negative in all cited reference.
Differentiation Potential of Adipose-Derived Stem Cells
Compared with BMSCs, the classic example of an adult stem cell population, it was believed that the major value of ADSCs was primarily related to their differentiation into various mature lineages. ADSCs display multi-lineage plasticity (multipotency) in that they possess the ability to differentiate along multiple cell lineages (from three germ layers: mesoderm, endoderm, and ectoderm) (51). Upon completion in the last few years of basic and clinical research, MSCs can differentiate into muscle, cartilage, and fat cells, depending on the selection conditions. ADSCs express the mesenchymal cell-specific markers and molecular typical for the embryonic stem cell phenotype (embryonic stem cell markers): OCT4 (octamer-binding transcription factor 4; POU5F1), Nanog (homeobox family of DNA binding transcription factors), and Sox2 [SRY (sex determining region Y)-box 2] (59). ADSCs are capable of secreting a large number of cytokines and growth factors that support angiogenesis, tissue remodeling, and antiapoptotic events, such as hepatocyte growth factor (HGF), interleukin-6 (IL-6), macrophage colony-stimulating factor (M-CSF), transforming growth factor-β1 (TGF-β1), tumor necrosis factor-α (TNF-α), and vascular endothelial growth factor (VEGF) (38). Cytokines and growth factors may affect cell differentiation in cultured cells (other than those added externally to the culture) and might control and manage the neighboring cells.
Recent research reports show that when ADSCs obtained from many donors were cultured with appropriate medium supplementation, they differentiated into not only adipogenic lineages but also osteogenic, chondrogenic, and myogenic lineages (Table 2). Up until now, whether a single ADSC could differentiate into all of the cell lineages remains unclear; however, in recent studies, it has been shown that pluripotent ADSCs can be differentiated into most mesenchymal cell types, including adipocytes (8,24,28,48,93,94), chondrocytes (18,24,28,93,94), osteoblasts (24,28,30,32,93,94), neuronal cells (1,63,64), myocytes (24,93,94), cardiomyocytes (57,69), hepatocytes (4,67,68,75), pancreatic cells (78), and endothelial cells (52,57,82,87). Even with these phenotypic differences, tissue repair using freshly isolated special SVF cell is possible (80).
Differentiation Potential of ADSCs

Adipogenic Differentiation (Adipogenesis)
Under adipogenic conditions (medium supplemented with insulin, isobutyl-methylxanthine, dexamethasone, rosiglitazone, and indomethacin), ADSCs are observed to express genes and proteins associated with the adipocyte phenotype, including leptin, peroxisome-proliferating activated receptor γ (PPARγ), glucose transporter type 4 (GLUT4), and glycerol-3-phosphate dehydrogenase (GPDH). In adipose differentiation of human ADSCs, the efficiencies of early (4) and late (25) passage cells of adipogenic lineage are different. ADSCs of passage 4 show multiple intracellular lipid droplets in ~90% cells of adipogenic medium. ADSCs of passage 25 showed multiple lipids in the remaining intracellular droplets within 45–50% of cells in adipogenic media (91). Insulin is strictly required in the conversion of preadipocytes to adipocytes. Several transcriptional factors in the adipogenesis process have been identified, which act cooperatively to trigger the terminal differentiation program (9,44,45). Two transcription factor families, the CCAAT/enhancer binding protein (C/EBP) and PPAR families, in the adipose differentiation program are very important (62,70).
Osteogenic Differentiation (Osteogenesis)
This type of differentiation occurs in vitro under the influence of dexamethasone, β-glycerophosphate, L-glutamine, ascorbic acid, and/or vitamin D3 (30,56,93). The osteoblast-like cells by stimulation of ADSCs are observed to express genes and proteins associated with the osteoblast phenotype, including alkaline phosphatase, type I collagen, osteopontin, osteonectin, and Runx2 (Runt-related transcription factor 2) (17,30,93).
Runx2, a key transcription factor, is a DNA-binding protein that acts as a scaffold-regulating factor and is involved in osteoblastic differentiation and skeletal morphogenesis. Members of the TGF-β superfamily, such as the bone morphogenic proteins (BMPs), stimulate osteogenesis. Transcriptional mechanisms of osteogenesis involve Runx2 to execute signals from the BMP and WNT (wingless-type MMTV integration site family) pathways to promote phenotype commitment and osteogenesis as a central mediator (6,23).
Chondrogenic Differentiation (Chondrogenesis)
ADSCs possess the capacity to differentiate and display a chondrogenic phenotype in vitro. The chondrogenic potential of human ADSCs in 3D culture has been demonstrated by assessing their ability to seed onto high-density micromasses (33,34), agarose, alginate, or gelatin 3D scaffolds (2,41,49), or by exposure to specific growth factors. The main factors activating the expression of genes relevant to chondrogenesis in vitro are insulin growth factor (IGF), BMPs, and TGF-β in the medium. ADSCs successfully differentiate into chondrocytes and mainly express genes and cartilage matrix proteins such as SOX-9 (SRY-box containing gene 9), aggrecan (large aggregating proteoglycan), biglycan [small leucine-rich protein 1A2 (SLRR1A)], decorin (bone proteoglycan II), cartilage oligomeric matrix protein (COMP), chondroitin sulfate, and type II collagen (7,76).
Myogenic Differentiation (Myogenesis)
Adipose-derived stem cells (ADSCs), which differentiate into putative myogenic cells, exhibit a myogenic morphology and express several proteins consistent with the myogenic phenotype [heavy chain of myosin, myogenic determination factor 1 (MyoD1)] in a medium containing 5% horse serum and glucocorticoid, such as dexamethasone and/or hydrocortisone (24,93,94). The differentiation of myocytes occurs with the lowest yield and reproducibility of all the differentiated cell types reported in the literature (77).
Other Differentiations
Recently, ADSCs have been shown to differentiate into functional, pancreatic-like cells following exposure to nicotinamide, activin-A, exedin-4, HGF, and petagastrin. ADSCs have been successfully differentiated into cells that were able to synthesize and secrete insulin, glucagon, and somatostatin (78). It is difficult to interpret the utility of ADSCs in supporting angiogenesis because of the mixed-cell populations present in the cell preparations derived from adipose tissue by different groups (52,57,82,87). Several in vitro studies have shown that ADSCs could be differentiated into neuron using a growth factor medium supplemented with valproic acid (VPA), insulin, hydroxyanisole, epidermal growth factor (EGF), dexamethasone, and fibroblast growth factor (FGF) (1,63). ADSCs were induced to differentiate into functional hepatocyte-like ADSCs, which were stimulated with hepatocyte growth factor (HGF), FGF-1, and FGF-4. These cells were capable of synthesizing urea, maintained glycogen stores, and incorporated into the liver microarchitecture in vivo (4,68,75).
Conclusion and Future Challenges
In most mammals, there is a large amount of adipose tissue from which the multipotent stem cells, ADSCs, can be extracted. ADSCs have the potential to differentiate along the mesodermal lineages of myoblast, chondroblast, osteoblast, and adipocyte (61,71). Currently, ADSCs are one of the most popular targets in adult stem cell research because ADSCs could be isolated from patients at any age, and proposed uses of these cells in tissue regeneration are truly impressive. Analysis of ADSC characteristics and detailed extensive understanding could lead to successful use of these stem cells in regenerative medicine therapies. Current regenerative medicine treatments mainly focus on the biological characteristics and the application potential of adult stem cells. Induced pluripotent stem (iPS) cells can be artificially manufactured from nonpluripotent cells, such as typical adult somatic cells or other multipotent stem cells, through induction of transcription factors, such as Oct4, Sox2, Nanog, and c-MYC, and they resemble but are not identical to undifferentiated embryonic stem cells (42,43,54,74,88). In the field of reprogramming somatic research, the application of ADSCs may be more useful than other types of cells. Impressively, Sun and his collaborators generated human iPS cells from adult ADSCs freshly isolated from patients (73). The use of ADSCs in cell-based therapies may be a viable treatment for numerous diseases and injuries (25,50,79,83). The most important features of stem cells from adipose tissue might be to contain cells with the ability to act as functional and vascular building blocks for several tissues. In conclusion, ADSCs hold great promise in translational research on tissue repair and have potential therapeutic uses in clinical tissue regeneration (29,39,50,65,79).
Footnotes
Acknowledgments
This study is supported in part by the National Science Council (Taiwan) (NSC100-2321-B-039-002 and NSC100-2314-B-039-006-MY3) and the Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH101-TD-B-111-004). The authors declare no conflict of interest.
