Abstract
Transplantation of bone marrow mesenchymal stem cells (BMSCs) provides a promising regenerative medicine for stroke. Whether BMSC therapy could repair ischemia-damaged neuronal circuits and recover electrophysiological activity has largely been unknown. To address this issue, BMSCs were implanted into the ischemic barrel cortex of adult mice 1 and 7 days after focal barrel cortex stroke. Two days after the first transplantation (3 days after stroke), the infarct volume determined by TTC staining was significantly smaller in BMSC-treated compared to vehicle-treated stroke mice. The behavioral corner test showed better long-term recovery of sensorimotor function in BMSC-treated mice. Six weeks poststroke, thalamocortical slices were prepared and neuronal circuit activity in the peri-infarct region of the barrel cortex was determined by extracellular recordings of evoked field potentials. In BMSC-transplanted brain slices, the ischemia-disrupted intracortical activity from layer 4 to layer 2/3 was noticeably recovered, and the thalamocortical circuit connection was also partially restored. In contrast, much less and slower recovery was seen in control animals of barrel cortex stroke. Immunohistochemical staining disclosed that the density of neurons, axons, and blood vessels in the peri-infarct region was significantly higher in BMSC-treated mice, accompanied with enhanced local blood flow recovery. Western blotting showed that BMSC treatment increased the expression of stromal cell-derived factor-1 (SDF-1), vascular endothelial growth factor (VEGF), and brain-derived neurotrophic factor (BDNF) in the peri-infarct region. Moreover, the expression of the axonal growth associated protein-43 (GAP-43) was markedly increased, whereas the axonal growth inhibiting proteins ROCK II and NG2 were suppressed in the BMSC-treated brains. BMSC transplantation also promoted directional migration and survival of doublecortin (DCX)-positive neuroblasts in the peri-infarct region. The present investigation thus provides novel evidence that BMSC transplantation has the potential to repair the ischemia-damaged neural networks and restore lost neuronal connections. The recovered circuit activity likely contributes to the improved sensorimotor function after focal ischemic stroke and BMSC transplantation.
Keywords
Introduction
Stroke is a leading cause of human death and disability worldwide. Among all stroke cases, ischemic stroke accounts for about 80% of stroke patients (15). Cerebral ischemia initially causes oxygen and glucose deprivation, excitotoxicity, and cell death in the affected cortex, resulting in an “ischemic core” area and a peri-infarct or penumbra region. Without therapeutic intervention, the ischemic core may show time-dependent expansion into the peri-infarct region, leading to enlarged infarction and additional cell death several days or even weeks after stroke (17,18). On the other hand, accumulating evidence suggests that neurons and vessels within the peri-infarct area are salvageable therapeutic targets that may undergo postischemic remodeling and contribute to recovery of sensorimotor function (9,10). To achieve functional recovery, repair of ischemia-damaged neuronal networks and connections in the peri-infarct region is necessary. A clear demonstration of these therapeutic effects in stem cell therapy, however, has been challenging and rarely reported before.
Currently, very few therapeutic approaches can ameliorate ischemic injury and restore the lost function. Recent evidence suggests that transplantation of bone marrow mesenchymal stem cells (BMSCs) can improve neurological and behavioral deficits in animal stroke models (6,32,33). Transplantation of BMSCs can increase axonal sprouting and axon density in the peri-infarct area (34,43). Many investigations highlight the notion that neurotrophic factors released from BMSC grafts play a vital role in the beneficial effect of BMSC transplantation therapy (7,32,38,47). So far, functional improvement is mostly shown using neurological and behavioral tests; the cellular and structural mechanism of BMSC-induced benefits in the ischemic brain is largely unknown. In hemispheric cerebral ischemia models, the large ischemic lesion destroys most parts of the gray and white matters. Thus, severe stroke models are not practical for the evaluation of specific neuronal pathways such as the thalamocortical connectivity that is critical for brain sensorimotor functions. Apparently, in the development of regenerative therapies, it is essential to evaluate the functional as well as anatomical integrity of neural infrastructures in the postischemic brain.
Over the past 10 years, the rodent somatosensory barrel cortex has received increasing attention due to its well-defined structure–function relationships and its suitability for neural plasticity investigations (19,21,51). The barrel cortex is characterized by the discrete clustering of its layer 4 neurons collectively called “barrels” (53). Sensory information from a single whisker is precisely transmitted via the thalamus to each corresponding layer 4 barrel, which in turn produces excitatory input into layer 2/3 of the cortex via vertical axonal projections. The circuit from layer 4 to layer 2/3 in each column responds most strongly to the stimulation of the corresponding whiskers. This precise structure–function relationship has been studied in brain slices by extracellular field potential recordings. The location of the recording and stimulating electrodes was found to coincide well with the physiologically defined circuit pathway (2–4,44). In the present study, we took the advantage of the barrel cortex ischemic stroke model of rodents developed by Wei et al. (51). The barrel cortex stroke model has a relatively smaller infarct volume and a well-defined ischemic edge and periinfarct region. We show in this investigation that this stroke model is especially suitable for electrophysiological evaluation of circuit connectivity from the thalamus to the barrel cortex and layer 2/3 neurons.
The present investigation tested the hypothesis that the neuronal activity of intracortical connections and the thalamocortical circuit in peri-infarct cortex could be restored or enhanced by BMSC transplantation therapy. The cellular and molecular mechanisms of the neural network repair promoted by BMSCs were also explored by immunohistochemical staining and Western blotting analysis.
Materials and Methods
Focal Ischemia of the Barrel Cortex of Mice
Adult male C57BL/6 mice (National Cancer Institute, Bethesda, MD, USA) were randomly divided into three groups: 14 animals in sham control group, 29 animals in vehicle-treated stroke group, and 34 animals in BMSC-treated stroke group. The focal barrel cortex ischemic stroke was induced by permanent ligations of distal branches of the middle cerebral artery (MCA) plus 7-min bilateral occlusion of the common carotid arteries (CCAs) as previously described (51). Sham control mice only received surgery lesions on skull surface. The brain infarct size was identified by 2,3,5-triphenyltetrazolium chloride (TTC; Sigma-Aldrich, St. Louis, MO, USA) staining using routine procedures. The animal protocol was approved by the Emory University Institutional Animal Care and Use Committee (IACUC).
Bone Marrow Mesenchymal Stem Cell Cultures and Hypoxic Preconditioning
For transplantation experiments, BMSCs were isolated from the femoral and tibial bones of green fluorescent protein (GFP) transgenic Wistar rats (Charles River, Wilmington, MA, USA). Mononuclear cells were suspended in Dulbecco's modified Eagle's medium (DMEM; Cellgro, Manassas, VA, USA) supplemented with 15% fetal bovine serum (FBS; Sigma-Aldrich) and plated in 100-mm cell culture dishes (Corning Incorporated, Corning, NY, USA). The identification and characterization of cluster of differentiation 90 (CD90)-positive but CD34- and CD45-negative mesenchymal stem cells as well as their differentiation potential had previously been verified in our isolated cells (24,50). Passage 3–4 BMSCs were routinely used in our experiments. As shown in our previous investigations, a pretreatment with sublethal hypoxia significantly enhances the survival and regenerative capabilities of transplanted stem cells (20,25,48). Consequently, all BMSCs used in this investigation were subjected to the hypoxic preconditioning treatment. Cells were passaged and cultured for 3 days until they became confluent and then exposed to 0.5% O2 for 24 h.
Transplantation of BMSCs Into the Ischemic Brain
The cell injection procedures followed our previous reports with some modifications to fit the focal ischemic stroke model of the mouse (49). After hypoxic preconditioning, BMSCs were labeled with Hoechst 33342 (1 μg/ml; Molecular Probes, Carlsbad, CA, USA). Cell density was adjusted to 50,000 cells per 1 μl DMEM prior to transplantation. A total of 100,000 BMSCs in DMEM were infused into two different sites in the penumbra using a Hamilton 80330 701-μl removable needle syringe (Hamilton Company, Reno, NV, USA) under the guide of Kopf microstereotaxic injection system (David Kopf Instruments, Tujunga, CA, USA). BMSCs were injected into the same sites at 1 and 7 days after stroke. DMEM-containing cells was injected at 1 ml/min; before syringe removal, a 2-min waiting period was observed to allow BMSCs to diffuse completely into the peri-infarct region. Vehicle-treated mice received an equal amount of DMEM injection at the same sites. This injection procedure did not cause noticeable mechanical brain damage due to the small diameter (0.47 mm) of the needle and the well-controlled operation.
Functional/Behavioral Testing and Blood Flow Measurement
The functional deficit and recovery were evaluated using corner tests 3 days before and 6, 14, 21, 28, 35, and 42 days poststroke. A 30° corner with an open end was set up as previously described (54). Every mouse was trained prior to surgery and received 10 turning tests at every time point. The sham control mice turned either left or right at an equal rate. Mice with focal ischemia in the right barrel cortex preferentially turned towards the left side. Scores were calculated by the number of left turns subtracted from the number of right turns. After each behavioral test, cerebral blood flow was measured using a laser Doppler perfusion scanning imager (Perimed, Stockholm, Sweden).
Thalamocortical Slices Preparation
Mice were anesthetized, decapitated, and the brains were removed and placed into ice-cold artificial cerebrospinal fluid (ACSF) containing: 124 mM NaCl, 3 mM KCl, 1.25 mM NaH2PO4, 1.0 mM MgCl2, 26 mM NaHCO3, 2.0 mM CaCl2, and 10 mM glucose (all from Sigma), saturated with 95% O2 and 5% CO2, at pH 7.4. Thalamocortical slices were prepared following the description of Agmon and Connors (2). Slices (400 μm thick) were obtained and collected with a soft brush, then incubated at room temperature 23–24°C) in ACSF for 60–90 min before recording. In order to keep the intact circuit between the thalamus and the barrel cortex, usually only one slice can be obtained from each 2-month-old animal.
Electrophysiological Recording
Thalamocortical slices were transferred to a small recording chamber (RC-22C, Warner Instruments, Co., Hamden, CT, USA) on a fixed-stage microscope (Olympus CX-31, Olympus Co., Tokyo, Japan) and superfused at a rate of 3 ml/min with ACSF at 23°C. Under the microscope the barrel field was clearly visible. To record the excitatory activity of local circuit in the same barrel, a recording glass micro-electrode (World Precision Instruments, Inc., Sarasota, FL, USA) filled with ACSF containing 5 mM bicuculline methiodide (BMI; Tocris Bioscience, Minneapolis, MN, USA) with resistance of 3–4 MΩ was placed in layer 2/3 cortex, and a stimulating 0.1 MΩ tungsten monopolar electrode (World Precision Instruments, Inc.) was placed in layer 4 of the cortex. 2-Amino-5-phosphonopentanoic acid (AP-5) and 2,3-dioxo-6-nitro-1,2,3,4-tetrahydr obenzo[f]quinoxa-line-7-sulfonamide (NBQX) were purchased from Tocris Bioscience and dissolved in distilled water and dimethyl sulfoxide (DMSO; Sigma), respectively, to study the contribution of glutamate receptors. To record the excitatory activity of the distant circuit from the ventrobasal nucleus (VB) of the thalamus to the barrel cortex, the stimulation electrode was located within the VB region of the thalamus whereas the glass recording electrode was placed within layer 4 of the barrel cortex. Stimulation intensity ranged from 5 to 45 μA, and the duration was 0.1 ms. Recording and stimulation electrodes were placed 200 μm (located by Narishige Hydraulic, MO-103 Micromanipulator, Tokyo, Japan) below the slice surface. The stimulation output (Master-8; AMPI, Jerusalem, Israel) was controlled by the trigger function of the EPC9 amplifier (HEKA Elektronik, Lambrecht, Germany). The evoked field response was recorded in the current-clamp mode of EPC9 amplifier. Data were filtered at 3 KHz and digitized at sampling rates of 20 KHz using Pulse software (HEKA Elektronik).
Immunohistochemistry
After electrophysiological recording, thalamocortical slices were collected, fixed, and cut on a cryostat microtome (10 μm thickness, Vibratome 5040, MetriCore, Inc., St. Louis, MO, USA) at −20°C. Slides containing thalamocortical sections were stored at −80°C until use. Coronal brain sections were cut from the perfusion-fixed brains. After being dried and washed three times in PBS (3.2 mM Na2HPO4, 0.5 mM KH2PO4, 1.3 mM KCl, and 135 mM NaCl, pH 7.4; Sigma-Aldrich) for 5 min, slides were placed in a 2:1 ethanol/acetic acid solution for 10 min at room temperature, washed three times in PBS, treated with 0.2% Triton X 100 (G-Biosciences, St. Louis, MO, USA) in distilled water for 5 min, and then washed in PBS. After blocking in 1.0% fish gelatin (Sigma, St. Louis, MO, USA) diluted in water at room temperature for 1 h, primary antibodies against neurofilament (NF) (1:200; Chemicon International, Temecula, CA, USA), collagen IV (1:300; Biomeda Corp., Foster City, CA, USA), GFP (Abcam, Inc., Cambridge, MA, USA), stromal-derived factor-1 (SDF-1; R&D Systems, Minneapolis, MN, USA), vascular endothelial growth factor (VEGF), brain-derived neurotrophic factor (BDNF) (all 1:200), and doublecortin (DCX, 1:50; all from Santa Cruz Biotechnology, Dallas, TX, USA) in PBS were added overnight at 4°C. Slides were washed three times in PBS and incubated with secondary antibodies cyanine 3 (Cy3)-conjugated anti-mouse or rabbit, and fluorescein isothiocyanate (FITC) anti-chicken IgG (1:500, 1:400; Molecular Probes, Carlsbad, CA, USA) in PBS for 1 h. Slides were visualized by fluorescent microscopy (BX61, Olympus, Tokyo, Japan).
The positively stained cells were counted in the periinfarct region. Design-based stereology principles and systematic random sampling were employed to ensure accurate and nonredundant cell counting (52). The counting was performed on three 20× or six 40× randomly selected images from the peri-infarct region in each 10-μm-thick section. A total of six sections that spanned the entire infarct region of each animal brain were selected for cell counting. Cell counts were obtained by using the image processing program Image J (National Institutes of Health, Bethesda, MD, USA) and averaged by Excel (Microsoft, Redmond, WA, USA).
Western Blot Analysis
Brain tissues were taken from the peri-infarct region of the cortex, and proteins were extracted by homogenization in protein lysis buffer (25 mM Tris–HCl, 150 mM NaCl, 5mM EDTA, 0.1% SDS, 2 mM sodium orthovanadate, 100 mM NaF, 1% Triton, leupeptin, aprotinin, and pepstatin, pH 7.4; Sigma-Aldrich). Samples were centrifuged at 14,000×g for 15 min to pellet the insoluble fraction, and supernatant was collected. Protein concentration of each sample was determined using the bicinchoninic acid assay (Sigma-Aldrich). Proteins from each sample (30 mg) were separated by sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis in a Hoefer minigel system (Amersham Biosciences, Piscataway, NJ, USA) and transferred in the Hoefer transfer tank to a polyvinylidene difluoride (PVDF) membrane (BioRad, Hercules, CA, USA). Membranes were blocked in 5% bovine serum albumin (BSA; Santa Cruz Technology) diluted in Tris-buffered saline containing 0.1% Tween-20 (Bio-Rad Laboratories) (TBST) at room temperature for at least 1–2 h and then incubated overnight at 4°C with one of the following primary antibodies: VEGF, BDNF (1:200–1:500; both Santa Cruz Biotechnology), SDF-1, chemokine (C-X-C motif) receptor 4 (CXCR4) (1:500 both R&D Systems), growth-associated protein 43 (GAP-43, 1:500; Boehringer Mannheim Biochemicals, Mannheim, Germany), chondroitin sulfate proteoglycan (NG2, 1:500; Chemicon), and Rho-associated, coiled coil-containing protein kinase 2 (ROCK II, 1:1,000; BD Biosciences, Franklin Lakes, NJ, USA). Mouse β-actin antibody (1:2,000; Sigma-Aldrich) was used as protein loading control. After primary antibody incubation, membranes were washed with TBST and incubated with alkaline phosphatase-conjugated anti-mouse or anti-rabbit IgG antibodies (Promega, Madison, WI, USA) for 1–2 h at room temperature. Finally, membranes were washed with TBST followed by three washes with TBS. The signal was detected by the addition of 5-bromo-4-chloro-3-indolylphosphate/nitroblue tetrazolium (BCIP/NBT) solution (Sigma-Aldrich) quantified and analyzed using the imaging software Image J (NIH, Bethesda, MD, USA). The intensity of each band was measured and then normalized against β-actin.
Statistical Analysis
Data from the corner test, blood flow, and histological measures were analyzed using one- or two-way ANOVA followed by a post hoc Tukey's test for differences between two or more groups. Electrophysiological data were compared by Student's two-tailed t test. Changes were identified as significant if p < 0.05. Data were graphed using GraphPad Prism, version 4 (GraphPad Software, Inc., San Diego, CA, USA). Values are presented as mean±SD or mean±SEM as specified.
Results
Subacute Effect of BMSC Treatment on Ischemic Damage
In the BMSC transplantation group, we performed two administrations of BMSCs at 1 and 7 days after barrel cortex stroke (100,000 cells per injection into the peri-infarct region) (Fig. 1A). BMSCs were pretreated with hypoxia preconditioning (0.5% O2 for 24 h) before transplantation. Our previous investigations have demonstrated that hypoxic preconditioning increases expression of trophic factors, and prosurvival and regenerative genes such as BDNF, VEGF, erythropoietin (EPO), and B-cell CLL/lymphoma 2 (bcl-2) in stem cells and progenitor cells, and thus promotes cell survival and regenerative potential of transplanted cells in the ischemic environment (20,25,48). Based on these data, hypoxic preconditioning was used in this investigation as a routine pretreatment of BMSCs.
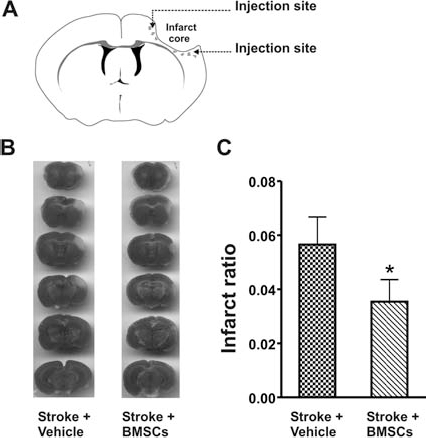
Cell transplantation and neuroprotective effect after focal ischemia. (A) Bone marrow mesenchymal stem cells (BMSCs) were injected into the peri-infarct cortex at 1 and 7 days after the onset of focal ischemia. (B) 2,3,5-Triphenyltetra-zolium chloride (TTC) staining of coronal brain sections from vehicle- and BMSC-treated barrel cortex stroke mice reveals the infarct area (white) in the cortex. (C) Quantified data show that the infarct volume (mean±SD) of BMSC-treated brains was significantly reduced compared with the vehicle-treated brain. n = 5 in each group, *p < 0.05.
The subacute neuroprotective effect of BMSCs against ischemic injury was first evaluated by measuring the infarct volume. Two days after BMSC transplantation (3 days poststroke), five mice in either vehicle- or BMSC-treated group were sacrificed, and brains were sliced into 1-mm coronal sections and stained with TTC. In the BMSC-treated mice, the infarct volume (TTC negatively stained area) was much smaller than that in vehicle-treated mice (Fig. 1B, C). Infarct volume was calculated using the indirect method and determined by summation of the infarct size of all stereotaxic levels in the brain (30). Compared to stroke control mice, the ratio of infarct volume was significantly reduced in BMSC-treated mice (0.053±0.027 in stroke mice vs. 0.035±0.022 in stroke plus BMSC mice; mean±SD, p < 0.05, n = 5) (Fig. 1C).
Chronic Effect of BMSC Treatment on Functional Outcomes and Local Cerebral Blood Flow
The remaining mice received the second BMSC transplantation at 7 days poststroke and were subjected to behavioral tests and blood flow measurements weekly for up to 42 days after stroke. In the corner test specifically designed to assess the sensorimotor activity of the whisker barrel cortex, normal control animals used both sides of their whiskers equally for exploratory activities. Since the numbers of left and right turns cancel each other out, the test score is normally around 0 in controls. In stroke control and stroke plus BMSC groups, the initial testing scores were close to nine, indicative of preferable use of one side of whiskers. A decline in the score was observed in both groups during the 6-week monitoring period due to gradual recovery of sensorimotor function (Fig. 2A). BMSC-treated animals, however, showed much faster functional improvement in the corner test compared to stroke controls. At 5 weeks posttransplantation, BMSC-treated mice showed significantly better functional improvement (Fig. 2A).

Behavioral test and blood flow measurement. (A) Corner test was performed 3 days before stroke and repeated every week up to 6 weeks after stroke. The testing score of BMSC-treated mice was significantly better than the score of vehicle-treated stroke mice. n = 10 in each group, *p < 0.05. (B) Cerebral blood flow was measured in vehicle control and BMSC-treated stroke mice using laser Doppler imaging. (C) Blood flow imaging was quantified 3 days before and weekly poststroke. The local blood flow in peri-infarct region was significantly improved in BMSCs-treated mice 5 and 6 weeks poststroke. Mean±SEM; n = 10 in each group, *p < 0.05.
Cerebral blood flow supplied to the barrel cortex was also measured using a laser Doppler perfusion imager immediately after stroke and at several delayed time points (Fig. 2B). The quantified blood flow changes demonstrate a rapid reduction in flow level immediately after MCA occlusions and a slow recovery throughout the ensuing 6 weeks. At 35 and 42 days poststroke, the local blood flow restoration in the BMSC group was significantly better than that in the vehicle-treated group (Fig. 2C).
Effect of BMSC Treatment on Circuit Activities in the Peri-infarct Cortex
To correlate the improved sensorimotor function with the neuronal activity in the ischemic barrel cortex, electrophysiological activities of neocortical circuit were measured in the thalamocortical slices 2 days and 5 weeks after BMSC transplantation. At 2 days after the first BMSC transplantation (3 days after the onset of focal ischemia), extracellular recordings were performed in brain slices from vehicle- and BMSC-treated stroke animals. When the recording electrode was placed 50 or 100 μm from the ischemic edge, no evoked field potentials were detected at this time (data not shown). This was most likely due to the energy disturbance, disruption of ionic homeostasis and silencing of electrical activity shortly after ischemia in vivo and acutely after brain dissection in our recordings even if the majority of the cells in the penumbra were alive (16). We thus focused on the delayed time points after BMSC therapy.
Five weeks after BMSC transplantation, we evaluated the intracortical activity within the same cortical barrel column by recording field potentials in layer 2/3 cells in response to stimulation in layer 4 (Fig. 3A). In vehicle- and BMSC-treated brain slices, the evoked field potential could now be observed in the peri-infarct cortex 50 or 100 μm from the ischemic edge. The field potential normally has two components; the slow component is sensitive to N-methyl-d-aspartate (NMDA) receptor antagonists such as AP-5 and the fast component is sensitive to α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor antagonists such as NBQX (Fig. 3B). The amplitude of the fast component was used to evaluate functional efficacy of excitatory inputs from layer 4 to layer 2/3. In brain slices from sham control mice, the field potential amplitude in layer 2/3 elevated with increasing stimulation intensity (Fig. 3C). In brain slices from stroke control mice, when the recording electrode was placed 50 μm from the ischemic edge, 5 and 10 μA stimuli in layer 4 did not induce any response in layer 2/3. Stimulation of 15 μA only induced a very weak response in stroke control (Fig. 3D) and BMSC-treated groups (Fig. 3E), with no amplitude difference between the two groups (Table 1). When the recording site moved to 100 μm from the ischemic edge (Fig. 3F, G), 10 and 15 μA stimuli in the layer 4 induced clear responses in the two groups. The amplitude of evoked responses in the BMSC-treated group was significantly larger than that in vehicle group (Table 1).

Field potential recording of local circuit in the barrel cortex. (A) Left and middle, recording at layer 2/3 and stimulation at layer 4 barrel in the same barrel column. Right, recording and stimulation sites at 50 or 100 μm from ischemic edge in stroke slices. (B) The fast (arrow 1) and slow (arrow 2) component of the field potential was suppressed by 2,3-dioxo-6-nitro-1,2,3,4-tetrahydr obenzo[f] quinoxaline-7-sulfonamide (NBQX; 20 μM) and 2-amino-5-phosphonopentanoic acid (AP-5; 100 μM), respectively. (C) Field potential recording in sham control slice and their amplitude responding to stimulation of 5, 10, and 15 μA. (D, E) The recording at peri-infarct cortex 50 μm from ischemic edge in vehicle- and BMSC-treated slices. Only 15 μA stimulation elicited a weak response in the two groups (arrows). (F, G) The recoding at 100 μm from ischemic edge, 10 and 15 μA stimuli elicited visible responses (arrows) in vehicle-treated slices, but significantly bigger responses (arrows) were seen in BMSC-treated slices.
Amplitude of the Field Potential in Layer 2/3 Evoked by Stimulation at Layer 4 of the Barrel Cortex

Five weeks after the second bone marrow mesenchymal stem cell (BMSC) transplantation (6 weeks poststroke). Values are means±SEM. NR, no response. Number of brain slices: n = 10 in sham control group, n = 9 in stroke + vehicle group, n = 8 in stroke + BMSCs group. *p < 0.05 compared with stroke + vehicle group.
Effect of BMSC Treatment on Thalamocortical Connections After Barrel Cortex Stroke
Restoration of electrical signal transduction along the thalamocortical connections is critical for the recovery of sensorimotor behavioral functions. The functional connectivity between the barrel cortex and the thalamus was demonstrated by recording field potentials in cortical layer 4 in response to stimuli in the VB of the thalamus (Fig. 4A). In vehicle- and BMSC-treated slices, the evoked field potential was recorded in the peri-infarct cortex 100 μm from ischemic edge (Fig. 4A). A previous study showed that stimulation within VB of the thalamus could induce a typical negativity wave in the corresponding barrel (2). In our experiment, a 45-μA stimulus was applied in the VB to induce the field potential response in the corresponding barrel. The AMPA receptor antagonist NBQX (20 μM) but not NMDA receptor antagonist AP-5 (100 μM) abolished the induction of this response (Fig. 4B), suggesting that AMPA receptors are the key postsynaptic elements that meditate signal transmission from the thalamus to the barrel cortex. The current stimulation at 45 μA was repeated every 30 s, and a stable field potential recording was obtained in sham control slices (Fig. 4C). Consistent with the intracortical recording, there was no evoked field potential in the peri-infarct cortex of vehicle-treated slices 100 μm from the ischemic edge (Fig. 4D). However, an identifiable evoked response could be induced in the same region of BMSC-treated slices when the VB was stimulated (Fig. 4E). Different from sham control slices, the response decreased when the second stimulation was applied 30 s later but recovered upon the third stimulation after another 30 s. The amplitude of the field potentials and the latency between stimulation artifact and the immediate onset of field potential were measured and showed improvements only in BMSC-treated brain slices, suggesting partial restorations of axonal connections (Table 2).

Field potential recording of thalamocortical connections. (A) Left and middle, stimulation at ventrobasal nucleus (VB) of the thalamus and recording at layer 4 of the barrel cortex. Right, recording site at 100 μm from ischemic edge in stroke slices. (B) The field potential (arrows) responding to VB stimulation was suppressed by NBQX (20 μM) but not AP-5 (100 μM). (C) In sham control slices, field potential could be stably recorded in layer 4 responding to 45 μA stimuli at the VB every 30 s. (D) In the vehicle-treated slice, no visible response was recorded in peri-infarct cortex 100 μm from ischemic edge. (E) In the BMSC-treated slice, visible responses were evoked by subsequent stimuli at the VB (arrow).
Characteristics of the Field Potential in Layer 4 Evoked by Stimulation at VB of the Thalamus
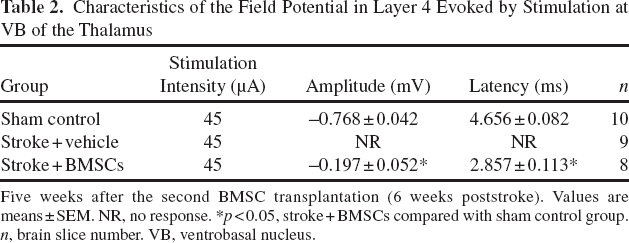
Five weeks after the second BMSC transplantation (6 weeks poststroke). Values are means±SEM. NR, no response. *p < 0.05, stroke + BMSCs compared with sham control group. n, brain slice number. VB, ventrobasal nucleus.
BMSC Therapy Increased the Density of Axon and Blood Vessel in the Peri-infarct Cortex
Immediately after electrophysiological recordings, brain slices were fixed with buffered formalin and stained to determine the density of axon (neurofilament) and blood vessels (collagen IV) in the peri-infarct cortex (Fig. 5A). Staining with anti-neurofilament and anti-collagen IV antibodies showed that ischemia caused significant decreases in the density of neurons, axons and blood vessels. The normalized densities of neurons, axons, and blood vessels were markedly decreased to 11.0±6.1%, 11.7±8.7%, and 22.2±13.1% of control level in the peri-infarct region. Transplantation of BMSCs significantly restored the densities of neurons, axons, and blood vessels to 31.1±17.9%, 37.2±16.5%, and 40.3±19.9% of control level (mean±SD, p < 0.05, compared with vehicle-treated controls; n = 8–10 in each group) (Fig. 5B–D).

Immunofluorescence staining of axons, neurons and blood vessels in the peri-infarct cortex. (A) Neurofilament staining (red) was used to reveal neuronal cell body and axons, while blood vessels were labeled with collagen IV (red). BMSCs were prelabeled with Hoechst 33342 (blue) before transplantation; the blue color indicated transplanted cells in the ischemic region. The white dotted lines illustrate the ischemic edge. (B–D) Positive staining of neurofilament and collagen IV in peri-infarct region was counted and density was calculated as the counts per field. The density of neurons, axons, and blood vessels were dramatically decreased in vehicle-treated mice, but significant higher density was seen in BMSC-treated mice. Mean±SD; *p < 0.01, vehicle-treated (n = 9) versus sham control group (n = 10). #p < 0.05, BMSC-treated (n = 8) versus vehicle-treated group (n = 9).
Effect of BMSC Transplantation on the Expression of Regenerative Proteins
To explore the possible mechanism that might mediate the BMSC effect on promoting tissue repair, four mice in each group were decapitated 2 weeks poststroke and the brain tissues from the peri-infarct region were collected and analyzed using Western blotting assay. The proregenerative trophic factors VEGF and BDNF were significantly elevated by BMSC treatment compared to their expression in vehicle-treated and/or sham control mice (Fig. 6A, B). SDF-1, a chemokine whose main function is stimulation and regulation of stem cell/progenitor directional migration towards the injured region (29), was significantly elevated in the BMSC-treated cortex (Fig. 6A, B). The SDF-1 receptor CXCR4 was not significantly changed (Fig. 6A, B). Aside from those regenerative factors, we measured levels of the axonal GAP-43 and growth-inhibiting proteins NG2 and ROCK II. The expression of GAP-43 was significantly increased by BMSC treatment compared to the vehicle-treated stroke and sham control brain (Fig. 6C, D). On the other hand, the expression of NG2 was significantly decreased in BMSC-treated stroke brain (Fig. 6C, D). ROCK II expression was suppressed in both the vehicle- and BMSC-treated stroke brain (Fig. 6C, D).

Expression of trophic factors, axon growth-promoting and inhibiting proteins in the peri-infarct cortex. Brain tissues in the peri-infarct region were collected 2 weeks poststroke and analyzed with Western blotting. (A, B) Representative bands showing the expression levels of vascular endothelial growth factor (VEGF), brain-derived neurotrophic factor (BDNF), stromal-derived factor-1 (SDF-1), and chemokine (C-X-C motif) receptor 4 (CXCR4). Gray density was analyzed with the imaging software Image J and normalized against β-actin. Statistical analysis in bar graphs shows significant upregulation of VEGF, BDNF, and SDF-1 in the stroke brain treated with BMSC transplantation compared to sham control (*) or stroke vehicle control (#). (C, D) Representative bands of growth associated protein-43 (GAP-43), Rho-associated, coiled coil-containing protein kinase 2 (ROCK II), and chondroitin sulfate proteoglycan (NG2) expression levels. BMSC treatment significantly elevated the expression of GAP-43 compared to both sham control (*) and stroke vehicle control (#). The expression of ROCK II and NG2 proteins was significantly inhibited by BMSC treatment compared to sham control (*) or stroke vehicle control (#). Stroke alone also suppressed ROCK II expression. n = 4 in each group; mean±SEM; *,#p < 0.05 versus sham control and stroke vehicle control, respectively.
In immunohistochemical staining experiments performed at 2 weeks after stroke, GFP-positive cells, indicative of transplanted BMSCs, were abundant in and around the peri-infarct region. These cells showed double-labeling with VEGF, BDNF, or SDF-1, implicating significant expression of these trophic factors in these cells in vivo (Fig. 7).

Expression of trophic factors in transplanted BMSCs. Immunohistochemical staining using specific antibodies against VEGF, BDNF, and SDF-1 was performed at 2 weeks after stroke. Double labeling with green fluorescent protein (GFP)-positive BMSCs was examined in brain sections. The green color of GFP expression on transplanted BMSCs was enhanced by staining with primary anti-GFP antibodies and secondary fluorescein isothiocyanate (FITC)-conjugated antibodies. In the peri-infarct cortex, most of the GFP-positive BMSCs were also colocalized with the positive VEGF, BDNF, and SDF-1 immunoreactivity. n = 5 mice in each group.
Effect of BMSC Transplantation on Neuroblast Migration and Survival in the Poststroke Brain
To further understand the effect of transplanted BMSCs on endogenous cells especially newly generated neuroblasts and their migration to the ischemic cortex, we performed DCX staining in brain sections 2 weeks after stroke. Many DCX-positive cells or neuroblasts were easily detectable in the subventricular zone (SVZ) area, implying active regenerative activity in this region (Fig. 8). Moreover, DCX-positive neuroblasts formed a migration stream from the ipsilateral SVZ to the ischemic cortex (Fig. 8). The number or density of DCX-positive cells in the peri-infarct cortex was noticeably greater in BMSC-treated mice than that in stroke control mice (32.1±11.7/field vs. 15.5±6.4/field; mean±SD, p < 0.05; n = 5 mice) (Fig. 8). This finding suggests that transplanted BMSCs support the directional migration of neuroblasts to the ischemic cortex and increase their survival in the peri-infarct region.

Transplanted BMSCs support migration and survival of DCX-positive neural progenitors in the peri-infarct region. (A) The drawing illustrates the ischemic infarct region in the right cortex and the migration route from subventricular zone (SVZ) to the ischemic cortex (inserted frame) measured in this experiments. (B) The low power images show the migration route of doublecortin (DCX)-positive cells (red) from SVZ to the ipsilateral cortex 2 weeks after stroke. The vertical dotted line marks the penumbra region. Many GFP-positive green cells and DCX-positive red cells can be seen in penumbra region in BMSC-treated stroke brain (second panel). (C) Enlarged images one and two marked in (B). (D) The bar graph shows the density of DCX-positive cells in penumbra of each group. Mean±SD; *p < 0.05, n = 5.
Discussion
The present investigation tested BMSC transplantation therapy in the barrel cortex stroke model of mice for electrophysiological, morphological, and functional assessments. We provide new evidence that BMSCs implanted directly into the ischemic cerebral cortex can show significant protective and repairing potentials in the periinfarct region. Transplanted BMSCs survive and produce trophic factors in the peri-infarct region, which in turn support migration and survival of neuroblasts, promote neurovascular remodeling and neocortical circuit restoration. All these therapeutic effects likely contributed to the improvement of sensorimotor functions several weeks after BMSC transplantation in the stroke animals.
BMSCs tested in this investigation were subjected to hypoxic preconditioning treatment prior to transplantation. This was based on the widely recognized benefits of hypoxic/ischemic preconditioning in a variety of cell types and tissues/organs reported over many years of investigations (14,22,41). In the last few years, we and a few other groups have explored the novel application of hypoxic preconditioning into stem cell transplantation therapy (20,25,40,48). We demonstrated that, compared to non-hypoxia-treated cells, hypoxic preconditioning is an effective strategy to promote survival, migration, and regenerative/repair capabilities of transplanted stem cells or progenitor cells via autocrine and paracrine effects (20,25,40,48). These beneficial effects of hypoxic preconditioning have been shown in transplantation therapies comparing normoxic controls and hypoxia-treated BMSCs and other types of stem cells (12,20,25,48). Thus, hypoxic preconditioning was adopted as a routine procedure in our investigation, and we suggest that the preconditioning approach should be applied in all cell-based therapies.
The two-step BMSC transplantation approach at 1 and 7 days poststroke was applied in the present investigation. The 1 day delayed administration of BMSCs resulted in a significantly reduced infarct volume 3 days post stroke, suggesting a neuroprotective effect as shown in previous investigations (11). However, noticeable sensorimotor function recovery was not obtained with transplantation at this early stage (data not shown). This subacute neuroprotective effect helps to preserve some cells and neurovascular structures that otherwise might perish in the next few days. It is expected that acute or subacute neuroprotection may not be necessarily accompanied with concurrent functional preservation at this time because preserved neurons can be in a dysfunctional state due to ischemia-induced disruption of ionic and molecular processes, changes in membrane protein, and ion channel activities. Moreover, neural networks are more or less damaged from the ischemic insult. For example, spreading depression of persistent depolarization takes place in vivo in the ischemic and penumbra regions and even expands into healthy brain areas and silences electrical activities and disrupts normal function days after the onset of ischemic stroke (16). It is not a surprise that acutely dissected brain slices during this period of time exhibited abnormal electrophysiological properties. The second administration of BMSCs 7 days poststroke was used to boost neuroprotection and regeneration and improve functional restoration. Score analysis of the corner test disclosed a slow recovery of sensorimotor function in vehicle-treated stroke mice over several weeks post stroke. The functional restoration in BMSC-treated mice developed faster than in vehicle mice and was significantly better 6 weeks poststroke (5 weeks post-second BMSC transplantation), suggesting a significant functional benefit of the BMSC therapy.
We tested the intracortical circuit activity within the barrel cortex by recording field potentials in layer 2/3 when stimulation was applied in layer 4. The normal field potential has two standard components; the slow and fast components are mediated by the NMDA and AMPA receptors, respectively (28). Their amplitudes were governed by stimulation intensity and the number of functional nerve fibers. Field potential recording was not practical in the ischemic core because of massive cell death in the core region. We selected two recording sites in the peri-infarct cortex with respective distances of 50 and 100 μm from the ischemic edge. At the 50-μm site, a tiny response was obtained responding to the maximum stimulation, without significant difference in amplitude between vehicle- and BMSC-treated stroke mice. However, at the 100-μm location, the elicited response became more visible and its amplitude in BMSC-treated slices was significantly larger than that in vehicle controls. These data suggest that more neuronal connections in the peri-infarct region of the cortex can be preserved or regenerated 5 weeks after BMSC treatment.
The enhancement of local circuit activity encouraged us to explore the restoration of distant circuit from the thalamus to peri-infarct cortex. The relatively long distance connection was demonstrated by recording the cortical field potentials in layer 4 that receives evoked signals from the VB nucleus of the thalamus. This field potential depended on AMPA receptors since it was blocked by NBQX but not AP-5. In peri-infarct cortex 100 μm from the ischemic edge, a weak but identifiable field potential could be detected in BMSC-treated slices but not in vehicle-treated slices, suggesting an improved connectivity from the thalamus to the ischemic barrel cortex. Interestingly, the latency of the field potential was significantly shorter in BMSC-treated slices than that in ischemic controls, indicating a much faster transmission of neurotransmitter from the presynaptic to postsynaptic membrane. This may suggest that the synaptic cleft in BMSC-repaired ischemic brain is narrower than that in vehicle-treated ischemic brain. This property is highly consistent with the notion that newly formed synapses have much smaller gaps between pre- and postsynaptic membranes, significantly narrower than the mature synaptic cleft and that the synaptic delay increases with age (31,45). The anatomical basis of neural circuit restoration was further revealed by immunohistochemical staining of axons and neurons in the same brain slices for which we performed electrophysiological recordings. We found that the density of axons and neurons in peri-infarct cortex was significantly higher in BMSC-treated slices than vehicle-treated slices, consistent with the differences observed in functional activities.
To dissect the endogenous repair mechanism promoted by the BMSC therapy, brain samples from peri-infarct region were collected 2 weeks after stroke and analyzed using Western blotting and immunohistochemical staining. BMSC therapy induced elevation of VEGF, BDNF, and SDF-1, suggesting a chronic and persistent trophic action present in the peri-infarct region. Double label immunohistochemical staining of GFP-positive BMSCs with VEGF, BDNF, and SDF-1 showed that BMSCs produced trophic factors in the peri-infarct region after transplantation. The expression level of GAP-43, ROCK II, and NG2 was also assessed in the peri-infarct region. GAP-43 is an indicator of axon sprouting and plasticity and is considered as a crucial component responding to regenerative activity in the nervous system during development and recovery from ischemic injury (8,36,46). ROCK II is one isoform of Rho Kinases (ROCKs), which has been implicated in growth cone collapse and neurite retraction (27,39). ROCKs and NG2 are important growth-inhibiting components participating in the generation of growth-inhibiting environment after nerve injury. The elevated expression of GAP-43 and the suppression of the growth-inhibiting factors ROCK II and NG2 in the BMSC-treated brain suggest that BMSC treatment may enhance axonal growth providing essential wiring infrastructure for neural network remodeling and circuit restoration. Further research will be necessary to understand the mechanism by which BMSCs inhibit ROCK II and NG2 expression.
Ischemia usually can stimulate endogenous neurogenesis in the SVZ of the adult brain. This is characterized by increased cell proliferation of DCX-positive neural progenitors in the SVZ and migration of these cells from SVZ towards the ischemic region (5,26). However, without treatment most DCX-positive neuroblasts undergo cell death in the ischemic region because of the hostile environment. Since transplanted BMSCs produce a number of prosurvival factors and suppress ROCKs and NG2, BMSC treatment improves the environment in the peri-infarct region for enhanced migration and survival of DCX-positive cells. The better survival of DCX-positive neuroblasts should ultimately contribute to the higher density of neurons and axons in BMSC-treated stroke mice.
The therapeutic action of BMSC transplantation, of course, is not limited to the mechanism described above. Current consensus agrees that multiple mechanisms may explain the therapeutic benefits of BMSC transplantation therapy. These include, but are not limited to, modulation of inflammatory and apoptotic responses (1,37), secretion of neurotrophic factors (7,32,38,47), and replacement of lost cells (13,23,35). In addition to trophic support and stimulation of endogenous regenerative activities, BMSCs have the potential of becoming neuronal and nonneuronal cells (42). Whether cell replacement from the BMSC-derived neurons contributes to tissue repair has been a controversial issue. We showed in a recent report that intravenously administrated BMSCs can differentiate into neuronal cells after homing to the ischemic brain and hypoxic preconditioning exhibits a promoting effect on the neuronal differentiation (50). The present investigation focused on the functional integrity and electrophysiological characterization of neuronal connectivity. The differentiation potential of BMSCs and its contribution to tissue repair after intracerebral injection remain to be specifically verified in a future investigation.
Footnotes
Acknowledgments
This work was supported by NIH grants NS045810, NS062097, NS058710, NS057255, NS075338, R41NS073378, the American Heart Association (AHA) Estab-lished Investigator Award and AHA Postdoctoral Fellowship (12POST12080252). It was also supported by the NIH grant NS055077 to the ENNCF (Emory Neurology-NINDS Core Facility). The authors declare no conflict of interest.
