Abstract
Allogeneic bone marrow mesenchymal stem cell (allo-BMSC)-based tissue-engineered bone (TEB) has great potential for bone defect repair. However, the immunogenicities and biological roles of allo-BMSCs are still controversial. In this study, we established an animal model of critical-sized mandibular defect in beagle dogs and compared the repairing effects of allo-BMSC-based TEB with autogenic BMSC (auto-BMSC)-based TEB without the administration of immunosuppressants. During the first 2 weeks postimplantation, a transient immune response in the allo-BMSC group was detected with an increase in proinflammation cytokines TNF-α, IFN-γ, and IL-2, a declination of anti-inflammation cytokine IL-10, and an increase in percentages of CD4+ and CD8+ T-cell subsets in peripheral blood. Nevertheless, there was no significant difference in bone union achievement, bone mineral density, and biomechanical properties between the two groups at 12 and 24 weeks postimplantation. Further subcutaneous implantation of allo-BMSCs/scaffold also exhibited the similar transient immune responses in the first 2 weeks postimplantation but followed by a decreased bone formation at 4 and 8 weeks postimplantation. These findings indicate that allo-BMSCs can induce a transient immunoreaction, which may temporally delay the osteogenesis of allo-BMSC/scaffold complex in early stage of in vivo implantation, whereas the long-term engineered bone formation was not affected.
Keywords
Introduction
Bone marrow mesenchymal stem cell (BMSC)-based bone tissue engineering has been proven as a promising approach to generate bone tissue and repair bone defects caused by trauma, infection, cancer, or congenital malformation (10). Even the critical-sized defects of mandibular bone, which were characterized by the complex contour and intraoral environment of mastication, deglutition, and oral secretions (16), were repaired using BMSC-based tissue-engineered bone (TEB) with a satisfactory bone mineral density in large animal models (34,45,46). The common strategy of bone tissue engineering employs a small amount of autologous BMSCs (auto-BMSCs) to expand and seed them onto a highly porous biodegradable scaffold in vitro, then the auto-BMSC/scaffold construct is transplanted into the defect to regenerate bone tissue (21). Auto-BMSC-based bone tissue engineering has gone from the bench to the bedside and shown effectiveness in the clinical practice to regenerate bone tissue (31,41,42). However, BMSCs isolated from elderly donors exhibit decreased proliferation and differentiation abilities ex vivo (28,36), making the treatment of aged patients with auto-BMSCs less applicable. The same concerns also apply to patients in conditions of chemotherapy, myelofibrosis, or lipid storage disease (2,39). In addition, the delay from extraction of auto-BMSCs to the implantation of the engineered tissue further limits the clinical applications of auto-BMSCs (30). Therefore, the approach of using allogeneic BMSCs (allo-BMSCs) as seed cells for bone tissue engineering has been considered as an alternative due to the low immunogenicity and immunomodulatory properties of BMSCs (3,11,15). Allo-BMSCs can be obtained from healthy donors or even embryonic tissues without eliciting donor site morbidity of an autogenous bone graft, and the application of allo-BMSCs could further facilitate the development of a bank of “off-the-shelf” cells for bone tissue engineering (17). However, some controversies still exist over the immunogenicity and biological roles of allo-BMSCs in bone tissue engineering in animal studies. On the one hand, allo-BMSC-based TEB has been reported to successfully repair critical-sized bone defects in dogs, pigs, and rabbits (3,11,30,44); on the other hand, allo-BMSCs from rats and mice were found to be immunogenic and result in rejection of the graft in vivo (7,37), and an additional study showed that critical-sized bone defects in rats were repaired by allo-BMSC-based TEB only in an initial short-term administration of immunosuppressant (2,19,39). Thus, it is crucial to clarify the immunogenicity and the correlation with the osteogenic capacity of allo-BMSCs in bone tissue engineering, especially in large animal models, in which model the immune reactions are more violent than in inbred rodents (4).
In the current study, we established an animal model of critical-sized mandibular defect in beagle dogs and compared the effects of orthotopic-engineered bone repair between auto-BMSC- and allo-BMSC-based groups without administration of immunosuppressive drugs. The immune responses, including the expression of inflammatory cytokines and the changes of T-cell subsets in peripheral blood, were evaluated as well. To further characterize the impacts of immunogenicity of allo-BMSCs at an early stage of implantation on the engineered bone formation, the allo-BMSC- and auto-BMSC-based TEB were implanted subcutaneously, and their bone formations during the 3 months postimplantation were evaluated and compared. This study gives insights on the immunogenicity and osteogenic capacities of allo-BMSC-based TEB and their correlations in vivo.
Materials and Methods
Construction of BMSCs/Scaffold Complex In Vitro
Animal experiments were approved by the Institutional Animal Care and Use Committee of Plastic Surgery Hospital (Institute). Male beagle dogs, 12 months old with an average weight of 16.7 kg, were obtained from Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). Bone marrow aspirates were harvested from the iliac bone of beagle dogs, and BMSCs were isolated by a Ficoll-Paque Plus solution (GE Healthcare, Uppsala, Sweden) and then cultured in a complete medium consisting of Dulbecco's modified Eagle medium-low glucose (DMEM-LG; Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; HyClone, South Logan, UT, USA). BMSCs were characterized through adhesive growth and trilineage differentiation into osteoblasts, chondrocytes, and adipocytes as previously described (5).
Natural corals were obtained from HaiNan Marine Development Plan and Design Institute (Hainan, China) and shaped into 30 mm × 13 mm × 9 mm with a 3-mm-diameter longitudinal central tunnel for implantation in orthotopic mandibular defect reconstruction site. From analysis by micro-CT system (μCT-1076; Skyscan, Kontich, Belgium), the coral exhibited a reasonably homogenous structure with a porosity of 57 ± 6.5% and mean pore diameter of 195 ± 75 mm as previously described (9,46). The β-TCP scaffolds were purchased from Shanghai Bio-Lu Biomaterials Co., Ltd. (Shanghai, China) and shaped into 8 mm × 6 mm × 3 mm for implantation in an ectopic subcutaneous site. The BMSCs (passage 2) were seeded on both the outer surface and inner axial tunnels of the sterilized scaffolds at a concentration of 20 × 106 cells/ml, and the cell/scaffold complex was rotated every 2 h for the first 6 h of seeding to reach a homogenous distribution of the cells (26) and further cultured in osteogenic induction media for 7 days before implantation.
Scanning electron microscopy was used to assess the growth of BMSCs on coral scaffolds.
Allogeneic Pair Determination Prior to Implantation by PCR-SSP Analysis
Mismatched donor–recipient pairs were predetermined by PCR sequence-specific primer (PCR-SSP) analysis (40). Beagle dogs' DNAs were isolated from peripheral blood samples by using a Nucleic Acid Isolation and Purification kit (Watson, ShangHai, China). PCR for polymorphic satellite markers, which were located in the class I region (C.2200) near DLA-53, was conducted by using sense primer 5′-ATCAAGTCCCATGTCGGGCT CCCGGTGC-3′ and antisense primer 5′-TGTCCTATT CATTCTCAGGATGACCACAT-3′. PCR products were analyzed by agarose gel electrophoresis; photograph and DLA type interpretation was documented.
Peripheral blood mononuclear cells (PBMCs) were isolated from heparinized blood by density gradient centrifugation with Ficoll-Paque Plus (GE Healthcare). PBMCs from donor or recipient dogs were inactivated by 10 μg/ml mitomycin C (UBIO Biotechnology Co., Ltd., Beijing, China) and used as allogeneic or autologous stimulators. Untreated PBMCs from recipient dogs were used as responders, and 1 × 105 stimulator PBMCs were cocultured with 1 × 106 responder PBMCs in RPMI-1640 medium (HyClone) in triplicate for 5 days. The proliferation of responder cells was assessed by the CellTiter 96 Aqueous Nonradioactive Cell Proliferation Assay kit (Promega, Madison, WI, USA). Absorbance was measured by Spectrophotometer (Ultrospec 2100 pro UV/Visible; GE Healthcare) at 490 nm to get optical density of allogeneic and autologous donor–recipient combinations (ODalloproliferation and ODautoproliferation). The stimulation index (SI) was calculated as ODalloproliferation/ODautoproliferation. Donor and recipient pairs were considered to be mismatched for DLA when different class I antigens were detected in PCR-SSP analysis, and the mixed leukocyte culture SI was more than three (SI > 3).
Implantation of Engineered Constructs
Repair of Mandibular Defects with TEB and Auto-Bone
Beagle dogs from predetermined auto-TEB, allo-TEB, auto-bone, and scaffold control groups (
Engineered Construct Implantation in Ectopic Sites
Beagle dogs from predetermined auto-TEB, allo-TEB, and control groups (
Evaluation of Bone Formation Postimplantation
Radiographic and CT Analyses
Mandible radiograms were taken under general anesthesia at 4, 12, and 24 weeks postimplantation. Maxillofacial CT images were obtained, and the three-dimensional images were reconstructed using a multislice spiral CT (GE Lightspeed Ultra 64; GE Healthcare, Milwaukee, WI, USA) at 12 and 24 weeks postimplantation. The mean CT value of each construct was derived from 10 serial CT images (0.6 mm section thickness, 0.4 mm interval).
Micro-CT Analyses
All animals were sacrificed at 24 weeks of mandibular defect repair, and both sides of the mandibles were harvested. A micro-CT system (μCT-1076; Skyscan) was used to visualize cross-sectional images of the mandibular specimens. The micro-CT settings were used as follows: voltage: 70 kV; current: 141 μA; exposure: 580 ms; filter: Al 1.0 mm; pixel matrix: 2,000 × 2,000; pixel size: 18 μm; rotation step: 0.6°. Images were reconstructed with the manufacturer's proprietary NRecon software. The bone tissue mineral density (TMD), tissue volume (TV), and tissue mineral content (TMC = TMD × TV) were calculated by micro-CT CTAn program.
Biomechanical Analysis
Mandibles were harvested at 24 weeks postimplantation and placed in a three-point bending system, providing an unsupported length of 30 mm. Load was applied in the occlusal-to-apical direction to the midpoint of the unsupported length, approximately in the middle of the repaired defect. The measurement was conducted using a hydraulic materials testing machine (858 Mini Bionix II; MTS, Eden Prairie, MN, USA) and stopped after bone fracture. Deformation was measured using the strain gauge at a loading rate of 0.5 mm/min. The bending stress was calculated with the equation: bending stress δf = 3
Histology Assays
The engineered constructs were retrieved, divided, fixed in 10% phosphate buffer formalin (Yili Fine Chemicals Co., Ltd., Beijing, China), and decalcified, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E; Yili Fine Chemicals Co., Ltd). For mandibular defect repair, the images of proximal, middle, and distal portions of longitudinal section and cross-section were captured. For engineered constructs implanted in the ectopic site, five images representing different areas were randomly captured under a high-power lens (200× magnification) of Leica DM3000 light microscope and analyzed using Image Pro Plus image analysis software (version 6.0) as previously reported (8).
ELISA Analysis for Osteogenic Markers ALP and OCN
The ectopic engineered constructs were collected at the time points of 3, 7, 14, 28, 56, and 84 days postimplantation and minced. The concentrations of ALP and OCN were measured using ELISA kits (DRE-9001, 9002; UBIO Biotechnology Co., Ltd.).
Evaluation of Peripheral Blood Cytokines and T-Lymphocyte Subsets
Peripheral blood was obtained from beagle dogs and centrifuged to extract serum. ELISA kits were used to detect the concentration of tumor necrosis factor-α (TNF-α) and interferon-γ (INF-γ) (EK0525, EK0373, Boster Bio-Engineering Limited Company, Wuhan, Hubei, China), IL-2 and IL-10 (1039-9, 1057-9; GBD, San Diego, CA, USA) in the serum. T-lymphocyte subsets (CD4+, CD8+) were determined by flow cytometry (FCM) through McAb direct immunofluorescence technique (MCA1038PE, MCA1039F; AbD Serotec, Kidlington, UK; BD Accuri C6 flow cytometer).
Statistical Analysis
Data were presented as mean ± standard error of the mean (SEM). Statistical analysis was performed by one-way ANOVA Newman–Keuls multiple comparison for three or more groups and Student's
Results
Construction and Implantation of BMSC/Coral Constructs
All beagle dogs tolerated the surgical procedures well without any complication. The multilineage differentiation potential of BMSCs isolated from bone marrow of beagle dogs was demonstrated using specific histology and immunohistochemical staining after being induced to osteogenic, chondrogenic, and adipogenic lineages (Fig. 1A). SEM analysis showed that BMSCs attached and proliferated well on the coral scaffold after 7 days of 3D culture in vitro.
Construction of BMSC/coral complex in vitro and determination of allogeneic donor–recipient pairs. (A) Trilineage differentiation of BMSCs and cell/coral constructs in vitro. BMSCs were identified by Alizarin red staining (AZR) after osteogenesis, type II collagen immunohistochemical staining after chondrogenic induction, and Oil red O staining after adipogenic induction. Scanning electron microscopy (SEM) was used for evaluation of the three-dimensional porous structures of coral scaffold. Scale bars: 100 μm. (B) Beagle dogs were mismatched for major histocompatibility complex class I molecules by dog leukocyte antigen (DLA) typing and class II molecules by mixed lymphocyte reaction assay (MLR). PCR using sequence-specific primers (PCR-SSP) was used for DLA typing of six dogs. Bands with the same patterns represent the same allele. Allogeneic mismatch pairing was predetermined according to the results and validated by one-way MLR assay. SI is stimulation index. (C) Surgical procedure of allo-TEB transplantation. After adjustment of the titanium plate, an osteoperiosteal segmental mandibular defect of 30 mm length was made at the right side. The defect was filled with allo-TEB graft.
Donor and recipient dogs were paired on the basis of mismatched DLA typing, and a significant increase (SI > 3) in proliferation of recipient PBMCs when mixed with the donor PBMCs in one-way MLR assays confirmed the DLA mismatched allogeneic donor–recipient combination (Fig. 1B). Four groups of auto-TEB, allo-TEB, auto-bone, and coral scaffold control were used to repair the critical-size mandibular bone defects (Fig. 1C).
Repair of Mandibular Defects After BMSC/Coral Implantation
At 4 weeks postimplantation, X-ray detection showed the distinct implant–recipient junction in all experimental groups (Fig. 2A). At 12 weeks postimplantation, the decreasing radiodensity of the implant in TEB and coral scaffold control groups indicated a rapid degradation of the scaffold. At 24 weeks postimplantation, the implant–recipient junction appeared radiopaque, and the radiodensity of the implant prominently increased in TEB and auto-bone groups; on the contrary, complete radiolucency was observed at the cut ends in coral scaffold group. CT evaluation showed that the CT value of the engineered bone in TEB groups, which was calculated by the mean CT value of cell/scaffold minus that of scaffold control group, increased from 35.6% and 31.24% of auto-bone group at 12 weeks to the same high level of auto-bone group at 24 weeks postimplantation (Fig. 2B). This indicated the similar growth trends of new bone formation in both TEB groups. In the end, the mandibular continuity was achieved in the auto-TEB group, allo-TEB group, and auto-bone group but not in scaffold control group (Fig. 2C).
X-ray detection for the repair of mandibular defects. (A) Radiographs of treated defects taken at different time points postimplantation. (B) Diagram of CT evaluation of auto-TEB, allo-TEB, and auto-bone group 12 and 24 weeks postoperation. The CT value of the engineered bone in auto-TEB and allo-TEB groups postoperation was calculated by the mean CT value of tissue- engineered bone minus that of the scaffold-only group. The auto-bone group served as a positive indicator. (C) 3D-CT image reconstruction for analysis of bony union and the continuity of mandibular body shape in auto-TEB, allo-TEB, auto-bone, and scaffold control groups. White arrows indicate the junction sites.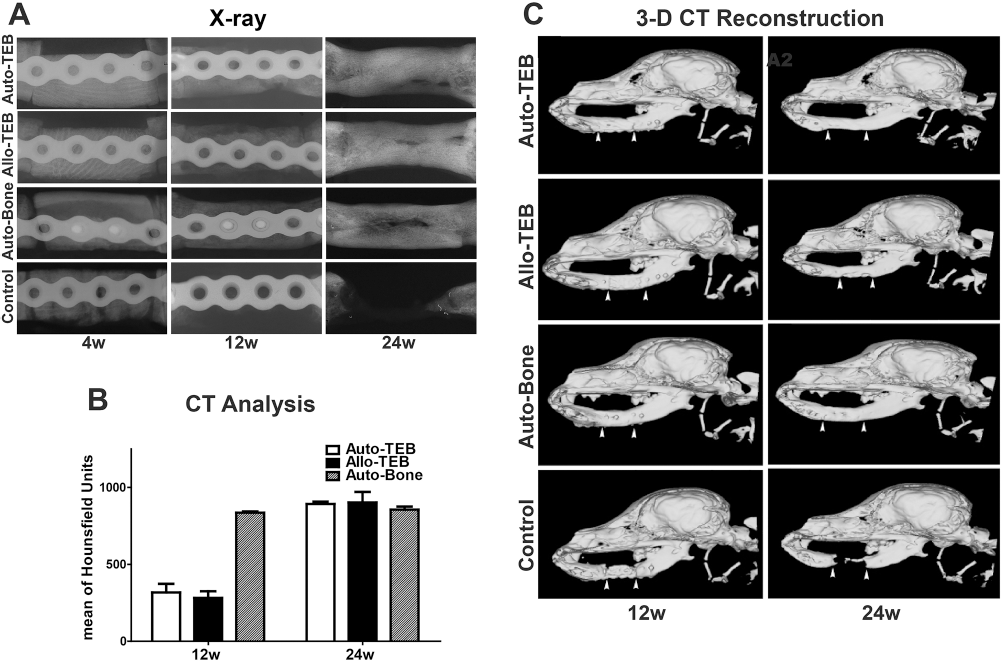
The gross view of the retrieved mandibles after 24 weeks postimplantation showed a smaller volume in TEB groups than in the auto-bone group (Fig. 3A1 to C1). Micro-CT 3D reconstruction exhibited the continuity of repaired mandibles in TEB and auto-bone groups (Fig. 3A2 to C2), with larger medullary cavity in the auto-bone group and very little residual granular radiodense material (accounts for only 1% of complex by volume) in TEB groups on the cross-sectional view (Fig. 3A3 to C3). No bony union was observed in the coral scaffold control group (Fig. 3D1, D2). Quantitative analysis showed that there was no significant difference in bone TMD, TV, and TMC between the auto-TEB and allo-TEB groups; however, the bone TV and TMC in both groups were lower than that in the auto-bone group (Fig. 3E–G). The micro-CT analysis of control group could not be conducted at 24 weeks postoperation due to the complete degeneration of the coral scaffold.
Morphology, micro-CT, and biomechanical analysis of repaired mandibles 24 weeks postimplantation ex vivo. The gross view, side, and cross view of micro-CT reconstruction images of auto-TEB group (A1-3), allo-TEB group (B1-3), auto-bone group (C1-3), and scaffold-alone group (D1-2) were presented. White arrows indicate the junction sites. Quantitative analysis of bone TMD (tissue mineral density) (E), TV (tissue volume) (F), and TMC (tissue mineral content) (G) of auto-TEB and allo-TEB. *
Biomechanical and Histological Properties of Repaired Mandibles After 24 Weeks
Measurement of mechanical strength of retrieved mandibles demonstrated a remarkably lower ultimate load in the TEB groups than that in the auto-bone group and contralateral normal edentulous mandibles (Fig. 4A) and similar bending strength in TEB and auto-bone groups but lower than in normal mandibles (Fig. 4B). For Young's modulus, no difference was found in all four groups (Fig. 4C).
Biomechanical analyses of orthotopically repaired mandibles 24 weeks postimplantation. There was no significant difference on ultimate load (A), bending strength (B), and Young's modulus (C) between the auto-TEB and allo-TEB groups. *
H&E staining images of the middle part, proximal, and distal ends, and cross-section of the implants confirmed that bone union was achieved in TEB and auto-bone groups, as well as good incorporation with both ends of the host bone (Fig. 5A1-C4). In both TEB groups, engineered bone did not show obvious differences in the structures of osteocyte, lacunae, vascular, and Haversian canals. More empty lacunae indicating the dead bone were observed in the auto-bone group around the implant–host junction (Fig. 5C1, C3). Only fibrous tissue and minimal new bone formation at the cut end were observed in the scaffold control group (Fig. 5D1, D2).
Histological examination of the bone defect sites in allo-TEB, auto-TEB, auto-bone, and coral scaffold control groups 24 weeks postoperation. White arrowheads indicate the new bone formation in the form of osteocyte, lacunae, and lamellar bone structure in the implants of auto-TEB (a2) and allo-TEB (b3). White arrows in a1, a4, b1, b4 indicate “Haversian canal”-like structure with small blood vessel and osteoblast inside it. Black arrows in a3, b3 indicate the hollow cavity that replaced the radiodense residual material after decalcification. In the auto-bone group, the “live” lacuna (white arrowhead in c1) and vacant lacuna (black arrow head in c1) were coexisting around the Haversian canal (white arrow in c1, c4) in accordance with “creeping substitution theory,” and the most part of the graft was presented by “dead bone” structure (black arrow in c3). Only fibrous tissue and minimal new bone formation at the cut end was observed in the scaffold-alone group (D, d). Scale bars: 100 μm.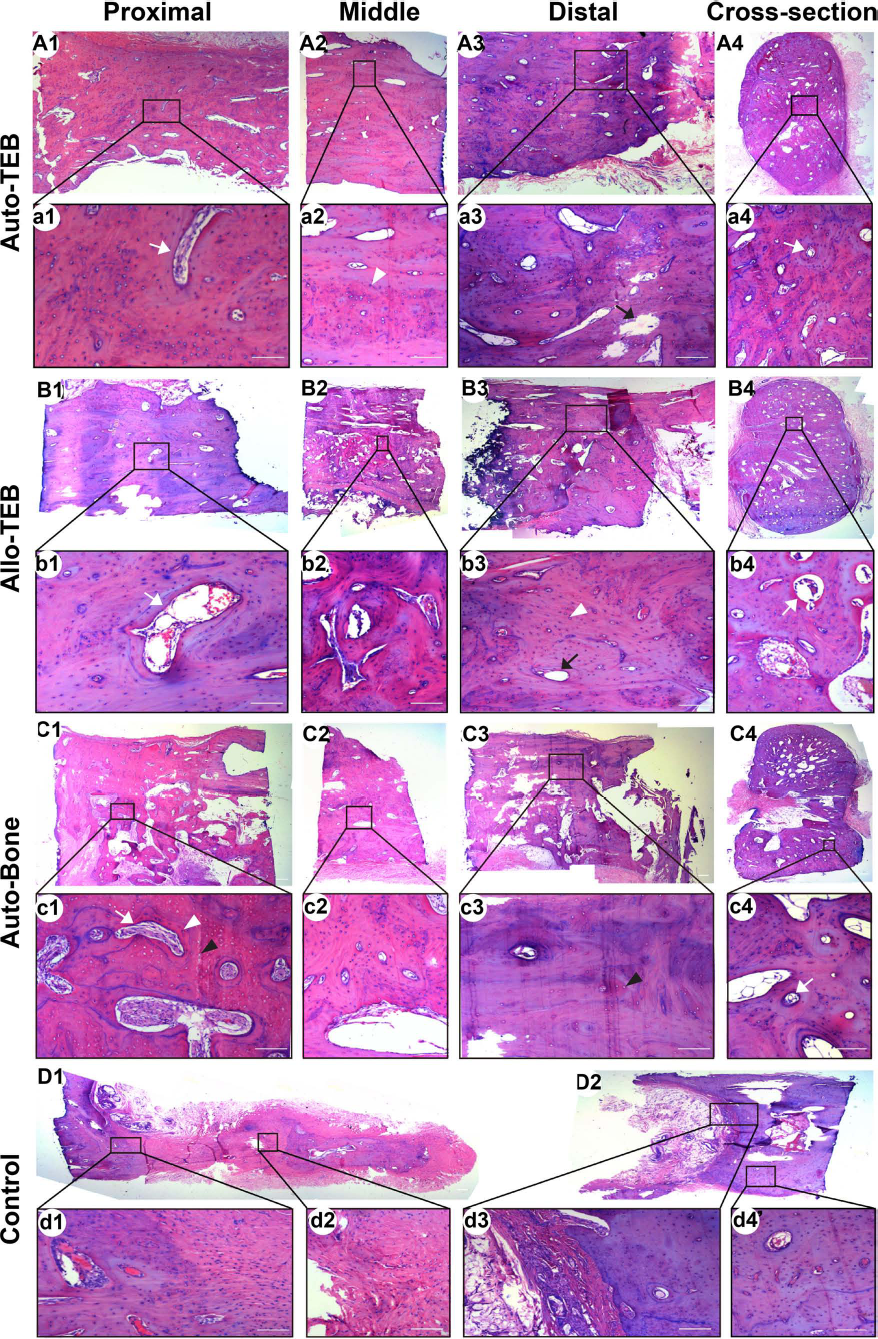
Immune Responses in Recipient Dogs After BMSC/Coral Implantation for Mandible Repair
Secretion patterns of IFN-γ, TNF-α, IL-2, and IL-10 by PBMCs in beagle dogs were measured by ELISA assay at different time points after implantation surgery (Fig. 6). The results showed that the secretions of the cytokines were restored to the preimplantation level after transient changes. The secretion of IFN-γ was higher in the allo-TEB group (95.16 ± 5.77 pg/ml) and auto-TEB group (59.28 ± 5.18 pg/ml) than in the scaffold control group (38.53 ± 4.62 pg/ml) at 3 days postimplantation with significant differences in three groups (Fig. 6A). The secretion of TNF-α reached the highest level (161.58 ± 8.46 pg/ml) in the allo-TEB group at 3 days postimplantation and decreased gradually to a level similar to the other two groups after 2 weeks of implantation (Fig. 6B). On the contrary, the concentration of TNF-α in the auto-TEB group (40.49 ± 5.51 pg/ml) was lower than in the scaffold control group (64.47 ± 7.14 pg/ml) at 7 days postimplantation. The secretion of IL-2 transiently increased in all three groups; the peak concentration of IL-2 was observed at day 7 (724.88 ± 10.71 pg/ml) in the allo-TEB group and at day 14 in the auto-TEB group (620.79 ± 11.61 pg/ml) (Fig. 6C). The IL-10 levels in these three groups declined temporarily (Fig. 6D), and the lowest concentration of IL-10 appeared at day 7 in the allo-TEB group (137.66 ± 6.34 pg/ml) and postponed to day 14 in auto-TEB group (180.43 ± 5.93 pg/ml).
Immunological analysis of peripheral blood in the auto-TEB, allo-TEB, and scaffold alone groups after orthotopic mandibular defect reconstruction and TEB transplantation. The levels of IFN-γ (A), TNF-α (B), IL-2 (C), and IL-10 (D) in the auto-TEB, allo-TEB, and control groups were evaluated by ELISA analysis. The percentages of CD4+ T-lymphocyte subset (E) and CD8+ T-lymphocyte subset (F) were evaluated by flow cytometry analysis in the auto-TEB, allo-TEB, and control groups. *
The percentages of CD4+ and CD8+ T-cell subsets in peripheral blood were calculated by FCM (Fig. 6E, F). The results showed that the percentage of CD4+ T lymphocytes in the allo-TEB group reached a peak after 2 weeks of transplantation (53.10 ± 1.74 %) with a significant difference compared to the other two groups (auto-TEB: 45.70 ± 1.23%; control: 43.47 ± 1.82%). The percentage of CD8+ T lymphocytes in the allo-TEB group was increased and significantly higher at day 7 (25.80 ± 0.93%) posttransplantation than in the auto-TEB and control groups.
Engineered Bone Formation and Immune Responses in Recipient Dogs After BMSC/β-TCP Ectopic Implantation
To further observe the impacts of host immune responses to allo-BMSC-based TEB formation during the early stage, BMSCs/β-TCPs were implanted subcutaneously in beagle dogs, and engineered bone formation was assessed by histological examination and ELISA analysis. H&E staining showed that at 4 weeks postimplantation, BMSCs proliferated along the inner surface of the scaffold and deposited the unmineralized collagen fibers and bone matrix–osteoid more obviously in the auto-TEB group than in the allo-TEB group (Fig. 7A). After 8 weeks of implantation, although increased osteoid was observed in both TEB groups, the auto-TEB group presented much more osteoid formation than the allo-TEB group. After 12 weeks of implantation, mature bone tissue was found in both TEB groups without a noticeable difference between the two groups. For the scaffold control group, the pores of the scaffold were filled with fibrous connective tissue only. ELISA analyses showed the expressions of osteogenic markers ALP and OCN gradually increased in both TEB groups but were significantly higher in the auto-TEB group during 28-56 days postimplantation for ALP and 14-48 days for OCN (Fig. 7B).
Histology and immunogenicity analysis of engineered bone formation after ectopic implantation with auto-TEB, allo-TEB, and β-TCP. (A) Histological examination of ectopic bone formation at 4, 8, and 12 weeks postimplantation (H&E staining, scale bars: 100 μm). Os, osteoid matrix; Sc, scaffold; Tb, mature trabecular bone structure; Ft, fibrous tissue. Quantitative comparison of bone formation by image analysis of H&E staining. *
Serum levels of IL-2 and IL-10 displayed a similar tendency as in previous experiments of orthotopic mandibular repair (Fig. 7C). During 4 weeks postimplantation, in all three groups, the concentrations of IL-2 transiently increased, and IL-10 temporarily decreased with significant differences between groups. The peak concentration of IL-2 and the lowest concentration of IL-10 in the allo-TEB group was observed at day 7 (702.00 ± 29.64 pg/ml, 133.49 ± 5.73 pg/ml) and postponed to day 14 in the auto-TEB group (597.98 ± 34.79 pg/ml, 164.83 ± 8.70 pg/ml).
Discussion
Although auto-BMSCs have been recognized as the optimal cell sources for bone tissue engineering, the alternative cell sources of allo-BMSCs will allow prefabrication of TEB readily available for implantation without causing donor site morbidity in patients. However, the immunogenicity and bone formation ability of allo-BMSCs in vivo is still surrounded by controversy (2,3,7,11,19,30,37,39,44). Here we presented that in beagle dogs a transient immune response was elicited at the early stage of allo-BMSC-based TEB transplantation for the repair of critical-sized mandibular defects and was confirmed after the subcutaneous implantation. Our study also demonstrated that the short-term immunoreaction of the recipient could delay the subcutaneous osteogenesis of allo-BMSC/scaffold construct during 8 weeks postimplantation, whereas long-term orthotopic and ectopic engineered bone formation was not affected.
MSCs are known to express a median level of histocompatibility complex class I (MHC-I), undetectable levels of MHC-II and costimulatory molecules such as CD80, CD86, CD40, and CD40L in vitro, which may make MSCs nonimmunogenic when administered in MHC-mismatched recipients (22,32,38). However, under the influence of a proinflammatory cytokine interferon-γ (IFN-γ), the expressions of MHC-II and CD80 in MSCs could be induced in vitro and in vivo (6,24,35), indicating that allogeneic MSCs may not be intrinsically immunoprivileged (29) and could be direct, indirect, or semidirect allo-recognized by host-specific CD8+ and CD4+ T cells. The subsequent antigen-specific cytotoxic T lymphocyte and delayed-type hypersensitivity response in vivo could facilitate a cell-mediated allograft acute rejection (23,35). In our study, compared to the auto-BMSC group, a transient cell-mediated immune response in the allo-BMSCs group was detected with an increase in Th1 proinflammatory cytokines IFN-γ, TNF-α, IL-2, a declination of Th2 anti-inflammatory cytokine IL-10, and an increase in percentages of CD4+ and CD8+ T-cell subsets in the peripheral blood in the first 2 weeks posttransplantation. Our study is the first to identify a transient immune response in the early stage of orthotopic or ectopic allo-BMSC-based TEB transplantation in the large animal model; moreover, we demonstrate that the short-term immunoreaction of the recipient could delay the subsequent osteogenesis of the subcutaneous allo-BMSC/scaffold construct until 3 months postimplantation. These findings were consistent with the reports in mice that serum IL-2 increased at 3 days until 14 days and resulted in a loss of engineered bone formation when allo-BMSC-based TEB was transplanted subcutaneously (37), and an increased proportion of host-derived lymphoid CD8+ infiltration at 14 days postimplantation was accompanied by detrimental functions of allo-BMSC TEB implants within 3 weeks (13). Studies in rat further revealed that the allogeneic BMSC TEB graft could obtain successful osteogenesis only by using adjuvant systemic immunosuppressant FK506 in the initial stage of transplantation (2). These studies, combined with our findings, strongly suggest that BMSCs are immunogenic upon implantation in immunocompetent MHC-mismatched recipients, no matter in inbred rodents or in large animals.
We also observed that during the first week of implantation, the auto-BMSC group exhibited significantly lower serum levels of proinflammatory cytokines TNF-α and IL-2 but higher anti-inflammatory cytokine IL-10 than the scaffold control group. The findings provided evidence for the immunosuppressive properties of auto-BMSCs, which could suppress the immunoreactions induced by the coral scaffold at the early stage of implantation. Additionally, the serum level of another proinflammatory cytokine IFN-γ in the auto-BMSC group was elevated promptly and significantly higher than in the scaffold control group at 3 days postimplantation. Previous studies also suggested that the immunosuppressive abilities of BMSCs require preliminary activation by IFN-γ alone or together with other chemokines (20,33), and IFN-γ-primed BMSCs were better suppressors to MLRs by stimulating the secretion of immune suppressive factors, such as indoleamine 2,3-dioxygenase activity (27), hepatocyte growth factor (12), and prostaglandin E2 levels (1). Therefore, the elevated IFN-γ in the auto-BMSC group might play a role in the immunomodulatory properties of BMSCs, which was upstream to the downregulation of TNF-α and IL-2 and upregulation of IL-10.
As to the osteogenic capacity of allo-BMSC-based TEB, in agreement with previous studies (3,11,30,44), our results of radiology, micro-CT, biomechanics, and histology examinations showed that the repair of mandibular defects for beagle dogs by allo-BMSC-based TEB were comparable to auto-BMSC-based TEB after 24 weeks of orthotopic transplantation. Nevertheless, a slower osteogenesis was observed in subcutaneous transplantation of allo-BMSC group at 4 and 8 weeks postimplantation and followed by a similar bone formation at 12 weeks, revealing that the short-term immune responses delayed the subsequent bone formation at the early stage of allo-BMSC/scaffold graft transplantation. Our data is supported by Liu et al.'s report showing that secreting of IFN-γ by proinflammatory T lymphocytes inhibited BMSC-based bone regeneration by downregulation of Runx-2 pathway and enhancement of TNF-α signaling in the stem cells (25). On the other hand, the delayed osteogenesis in the allo-BMSC group might also be due to the decreased number of BMSCs residing in the allo-BMSC/scaffold complex after implantation. It was reported that MHC-mismatched allogenic stem cells would be eliminated by natural killer cells (43) or by TNF-α and cytotoxic T-cell-mediated cell death during the immune rejection (14). Therefore, in our study, the significant elevation of TNF-α levels and CD8+ T-cell number at the early stage might cause cell death of allo-BMSCs in the TEB construct and result in the slower osteogenesis in the first 8 weeks after implantation. However, the higher expression of IFN-γ in the allo-BMSC group might also provide beneficial effects on the acquisition of the immunosuppressive properties of BMSCs and contribute to the succeeding survival of partial allo-BMSCs.
Conclusion
We presented that allo-BMSC-based TEB could successfully repair the critical-sized segmental mandibular defects in canines even though a transient cell-mediated immune response occurred in the early phase of transplantation, which indicated that allo-BMSCs were not completely immunoprivileged. Further subcutaneous implantation of allo-BMSC-based TEB showed a slower osteogenesis in the first 8 weeks postimplantation following the similar trend of immunoreactions, whereas engineered bone formation after 3 months postimplantation was not affected. Our study gives insights on bone regeneration abilities and immune properties of allo-BMSCs in the large animal model and may provide instructions for the standardization and optimization of bone tissue engineering using allo-BMSCs in future clinical practice.
Footnotes
Acknowledgments
