Abstract
Culture of human pancreatic islets is now routinely carried out prior to clinical islet allotransplantation, using conditions that have been developed empirically. One of the major causes of early islet destruction after transplantation is the process termed instant blood-mediated inflammatory reaction (IBMIR). The aim of this study was to develop in vitro methods to investigate IBMIR and apply them to the culture conditions used routinely in our human islet isolation laboratory. Freshly isolated or precultured (24 h, 48 h) human islets were incubated in either ABO-compatible allogeneic human blood or Hank's buffered salt solution (HBSS) for 1 h at 37°C. Tissue factor (TF) expression and leukocyte migration were assessed by light microscopy. TF was also quantified by ELISA. To assess β-cell function, glucose-stimulated insulin secretion (GSIS) assay was carried out. The extent of islet β-cell damage was quantified using a proinsulin assay. Islets cultured for 24 h had higher GSIS when compared to freshly isolated or 48-h precultured islets. Freshly isolated islets had significantly higher TF content than 24-h and 48-h precultured islets. Incubation of freshly isolated human islets in allogeneic human blood released 6.5-fold higher level of proinsulin in comparison to freshly isolated human islets in HBSS. The high level of proinsulin released was significantly attenuated when precultured islets (24 h or 48 h) were exposed to fresh blood. Histological examination of fresh islets in blood clot showed that some islets were fragmented, showing signs of extraislet insulin leakage and extensive neutrophil infiltration and necrosis. These features were markedly reduced when the islets were cultured for 24 h. These results suggest that our standard 24-h islet culture is markedly beneficial in attenuating IBMIR, as evidenced by increased GSIS, lower content of TF, decrease islet fragmentation, and proinsulin release.
Keywords
Introduction
Allogeneic islet cell transplantation is now a successful therapy for certain selected patients diagnosed as having type 1 diabetes mellitus. However, the success of the procedure is limited by the profound islet attrition that occurs in the immediate posttransplant period (17). This limits the functional capacity to regulate glucose levels even when four donors are used, corresponding to around 20-30% of that in a healthy nondiabetic subject (16).
One of the major causes of islet attrition at the early stages of transplantation has been termed instant blood-mediated inflammatory reaction (IBMIR). It occurs rapidly when isolated islets are exposed to human blood following infusion in the portal vein. It is thought to be an innate inflammatory response, triggering activation of complement and infiltration of polymorphonuclear cells into “foreign” material. Investigation of IBMIR is made difficult by the lack of a murine allograft model, at least not a model of similar severity to that found in human islet allografts. Investigation has implicated activated tissue factor (TF) leading to the generation of thrombin, a potent inflammatory mediator, resulting in activation of platelets and neutrophils. This could activate the coagulation cascade in an islet recipient (2,12). Thrombin inhibition abrogates the IBMIR response to human islets (14).
Culture of human islets prior to transplantation is beneficial to clinical islet allotransplantation in terms of its logistical advantages (7). It provides a window of opportunity to complete quality assurance tests, to mobilize the patients, or to transport the islets to another center for transplantation. Islet culture may lessen islet immunogenicity by depleting viable hematogenous and lymphoid cells (9). However, since there is some reduction of islet equivalent number with culture, there is an ongoing debate as to whether cultured islets are superior to freshly isolated islets in transplantation (5,10).
We have previously confirmed that exposure of freshly isolated human islets to allogeneic blood results in significant destruction of islet tissue with activation of the coagulation cascade and consumption of neutrophils and monocytes (18). We hypothesized that culture of islets improved the subsequent islet graft function. Hence, the aim of the present study was to assess the IBMIR effect produced by freshly isolated islets compared to islets cultured for 1 or 2 days.
Materials and Methods
Following appropriate consent and ethical approval from local and national committees, human pancreata (n=5) were retrieved from heart-beating donors (age range 35-61 years; gender three females, two males; BMI range 21-36; cold ischemia times <10 h).
Human Islet Isolation
Isolation was performed according to a modified Edmonton protocol (17). In brief, the pancreatic duct was securely cannulated, and cold collagenase type NB1 and neutral protease (Serva Electrophoresis, Heidelberg, Germany) were injected slowly, ensuring no leaks. Digestion of the pancreas at 37°C was allowed to proceed in the digestion chamber (Biorep Technologie, Miami, FL, USA), and the circuit fluid was sampled until free-cleaved islet tissue was seen [using 0.25% dithizone (Sigma-Aldrich, Dorset, UK) to identify the islets]. Once cleaved islets were seen, the digest was then collected in conical flasks (Corning plastics; Fisher Scientific, Leicestershire, UK) containing 8-10 ml of 20% human albumin (Blood Products Laboratory, Elstree, UK). Islets were purified using a continuous Biocoll density gradient (Autogen Bioclear, Wiltshire, UK) on a COBE 2991 cell separator (Cobe Instruments, Lakewood, CO, USA). The volume and purity of islets in the final sample was assessed by microscopic sizing on a grid after staining with dithizone. Viability and functionality were assessed by fluorescein diacetate/ethidium bromide stain (Sigma-Aldrich) (4) and glucose-stimulated insulin secretion (GSIS), respectively. All preparations used demonstrated islet viability around 80-95% immediately after isolation.
Islet Culture
Islet preparations were maintained in standard flat-bottomed 175-ml gas-permeable flasks (Sarstedt, Leicester, UK), each containing 30 ml of our standard culture medium [CMRL (PAA, Somerset, UK): 5.5 mM glucose (Sigma-Aldrich), with added 1% L-glutamine (PAA), 1% penicillin/streptomycin (PAA), 50 ml of 20% human albumin, and 1% HEPES (Lonza, Basel, Switzerland)] incubated at 37°C, 5% CO2 in air for 24 and 48 h. Islets aliquoted at 30,000 IEQ per flask. These volumes were enough to completely cover the tissue at the gas–fluid interface yet minimize hypoxia.
Blood Samples
Following informed consent, blood was collected from healthy volunteers (n=3; age range 25-60 years; gender one female, two males). All the blood samples used in the experiments were blood group matched with islets and collected from healthy individuals who had not undergone previous blood transfusion, pregnancy, or transplantation and whose panel reactive antibody screen was negative. Fresh blood was used instantly, whereas citrated blood was used between 30 and 60 min after collection.
Histology
The clots containing the islets were fixed in 4% paraformaldehyde (Sigma-Aldrich), dehydrated, and embedded in paraffin wax, serial sectioned (5 μm) to extinction, and stained with hematoxylin and eosin (H&E; Sigma-Aldrich). For cryosectioning, clots containing the islets were accumulated in a conical receiver (Corning, Scientific Laboratory Supplies Limited, Nottingham, UK), flash frozen, and cryostat (Bright Instrument Company Limited, Cambridgeshire, UK) mounted, then serial sectioned (5 μm) to extinction.
Immunohistochemistry
The sections were incubated with murine monoclonal anti-human TF (CD142) (No. 4509; 1:1,000; American Diagnostica, Greenwich, CT, USA), an in-house guinea pig anti-human insulin antibody (1:5,000), murine antihuman insulin antibody (1:1,000; Sigma-Aldrich), murine monoclonal anti-human neutrophil elastase NP57 (1:1,000; DakoCytomation, Denmark) with appropriate controls. The peroxidase or alkaline phosphatase-conjugated goat anti-mouse (Sigma-Aldrich) and rabbit anti-guinea pig (DAKO, Cambridgeshire, UK) secondary antibodies were diluted at 1:50. Positive binding was revealed by addition of diaminobenzidine substrate chromogen (Sigma-Aldrich) and counterstained with hematoxylin. The distribution of neutrophils in the clots and adjacent to the islets was assessed by semiautomatic morphometry (Axiovision, Zeiss, Germany), the degree of leukocyte infiltration expressed as pixels per neutrophil in three zones (within islet, peri-islet, rest of field). Image analysis software was used to count the number of neutrophils, and statistical confirmation was reached only if islets with no neutrophils were excluded. Log transformation was used to normalize the remaining data, and Bonferroni post hoc test for multiple comparisons was performed.
TF Assay
Fifty handpicked human islets cultured for 0 h, 24 h, or 48 h were used to measure TF content. The islets were lysed at each time point by repeated freeze–thaw in buffer [50 mM Tris-HCl (Sigma-Aldrich), 100 mM NaCl (Baxter, Northampton, UK), 0.1% Triton X-100 (Sigma-Aldrich), pH 7.4] and allowed to incubate for 30 min at 37°C. Samples were immediately assayed without dilution (Actichrome Tissue Factor; American Diagnostica).
GSIS
Ten handpicked human islets from each preparation (fresh or cultured for 24 h, 48 h) were incubated for 45 min at 37°C in 2 ml RPMI-1640 (Gibco, Invitrogen, UK) media containing 3 mmol/L D-glucose, 1% human albumin, 1% L-glutamine, and 0.3% penicillin/streptomycin followed by stimulation with 16 mmol/L D-glucose for 45 min. Human insulin was measured in aliquots of the media using a Mercodia human insulin enzyme-linked immunosorbent assay (ELISA) (Uppsala, Sweden).
Proinsulin Assay
Fifteen size-matched handpicked human islets (freshly isolated or following culture for 0 h, 24 h, 48 h) were incubated for 1 h at 37°C in 360 μl Hank's balanced salt solution (HBSS; Lonza) (control), 360 μl fresh human allogeneic blood, or 360 μl citrated-recalcified allogenic blood. Allocation of islets was protected from observer bias by final step random selection of islets for each condition. Human blood without islets was used as a negative control. After incubation for 1 h, blood serum was collected from the islets. The proinsulin released in the blood/serum suggests β-cell destruction as previously described (1,18). To ascertain that, prior to the experiments, the islets were not disrupted thus leaking proinsulin, we determine the proinsulin content after the experiment. The islets in the clots were lysed by repeated freeze–thaw to allow extraction of proinsulin content. The samples were then centrifuged at 211 × g for 5 min, and the supernatants were stored at −20°C. Human proinsulin was assayed by Mercodia Proinsulin ELISA. This assay cross-reacts with proinsulin Des and proinsulin Split but does not cross-react with human insulin.
Statistical Analysis
The significance of changes in level of TF or proinsulin was tested by analysis of variance (ANOVA). If ANOVA indicated a significant difference, the data were analyzed by using Bonferroni method for multiple comparisons as a post hoc test for the difference between groups. A value of p ≤ 0.05 was considered statistically significant. The statistical analysis of infiltrating leukocytes was altered, and this is discussed in the relevant section.
Results
Effect of Culture on Human Pancreatic Islet Functionality
The functionality of human islets was assessed by GSIS (Fig. 1). Freshly isolated islets stimulated with 3 mM glucose released 5.3 ± 1.5 mU/L/islet insulin, and when stimulated with 16 mM glucose released 6.7 ± 1.4 mU/L/islet insulin. Islets cultured for 24 h secreted 5.2 ± 1.0 mU/L/ islet insulin when stimulated with 3 mM glucose when compared to 9.7 ± 1.2 mU/L/islet insulin when stimulated with 16 mM glucose. Islets cultured for 48 h secreted 4.0 ± 0.5 mU/L/islet insulin when stimulated with 3 mM glucose when compared to 5.7 ± 0.9 mU/L/islet insulin when stimulated with 16 mM glucose. Human islets cultured for 24 h secreted significantly higher insulin concentration in response to 16 mM glucose when compared to stimulation with 3 mM glucose and was significantly higher compared to freshly isolated islets.

Effect of culture on human islet functionality. The functionality of human pancreatic islets was assessed by GSIS. Islets cultured for 24 h secreted significantly higher insulin when compared to freshly isolated islets (*p ≤ 0.05; n = 5 independent experiments were carried out).
Effect of Culture on Human Islet TF Production
Human islets in normal pancreas were strongly positive for TF (Fig. 2Ai), when compared to the exocrine tissue, which was virtually negative. Insulin-expressing isolated islets in blood clot were also strongly positive for TF (Fig. 2Aiii, iv). Freshly isolated human pancreatic islets had significantly higher TF content than 24-h and 48-h precultured islets. Islets cultured for 48-h had significantly lower TF content when compared to freshly isolated and 24-h cultured islets (Fig. 2B).

Effect of culture on human islet TF production. (A) (i) Human pancreatic tissue shows islet cells immunolabeled for TF (arrow). (ii) Negative control showing expression of IgG. (iii) Islet expressing TF. (iv) Insulin-producing islet in blood clot expressed both TF and insulin. Scale bar: 100 μm. (B) TF content was measured in freshly isolated or precultured (24 h, 48 h) islets. The concentration of TF per islet decreased significantly after 24 h and 48 h in culture (*p < 0.05). At 48 h in culture, the TF concentration per islet was significantly lower when compared to 24 h in culture (+p < 0.05).
Effect of Culture on Human Islet Proinsulin Release
Freshly isolated islets incubated in HBSS for 1 h released more proinsulin than 24-h and 48-h precultured islets, which were both below the lowest detectable limit (shown as DL in Fig. 3). Incubation of freshly isolated or precultured (24 h, 48 h) human islets in fresh ABO-compatible allogeneic blood for 1 h significantly augmented the release of proinsulin when compared to their corresponding controls (freshly isolated or precultured islets in HBSS). Freshly isolated islets in blood released significantly higher proinsulin when compared to precultured islets (24 h or 48 h) incubated in fresh blood. A similar trend was observed when fresh blood was substituted with citrated-recalcified blood. To determine the proinsulin release is not due to leakage of proinsulin as a result of islet disruption prior to the experiment, we determine the islet proinsulin content. At the end of the experiment, freshly isolated islets in citrated-recalcified blood were disrupted, and the proinsulin content (129.5 ± 27.7 pmol/L/15 islets) was not significantly different from that found in islets cultured for 24 h exposed to the citrated-recalcified blood (127.6 ± 18.4 pmol/L/15 islets). Blood on its own had minimal proinsulin well below the DL.

Effect of culture on human islet proinsulin release. (A) Islets in HBSS: Proinsulin concentration above lowest detectable limit (DL) was found in freshly isolated islets in HBSS but was below the limit following culture (24 h, 48 h). (B) Islets in fresh blood: Proinsulin concentration, when freshly isolated islets were exposed to fresh blood, was 6.5-fold more than that found in control. These values were significantly decreased when islets were precultured for 24 h, 48 h. (C) Islets in citrated-recalcified blood: Similar high levels of proinsulin were found when islets were placed in citrated-recalcified blood. These values were significantly decreased when islets were precultured for 24 h, 48 h. Normal blood contains no detectable proinsulin. n=4 independent experiments were carried out. Results shown are the means + SEM. *p ≤ 0.05 when compared to corresponding controls (islet + HBSS). +p ≤ 0.05 when compared to fresh blood or citrated blood. *p ≤ 0.05 when compared to islet with fresh blood or citrated blood at 0 h.
Histological Assessment of Human Islets (Fresh and Precultured) After Incubation in Allogenic Blood
Histological examination of fresh islets incubated in fresh blood for 1 h showed that the islet structure was generally retained, but some of the islets were fragmented, showing pycnotic nuclei (Fig. 4A) and signs of extraislet insulin leakage (Fig. 4B). These features were markedly reduced when the islets were cultured for 24 h (Fig. 4D, E) before exposure to fresh blood.
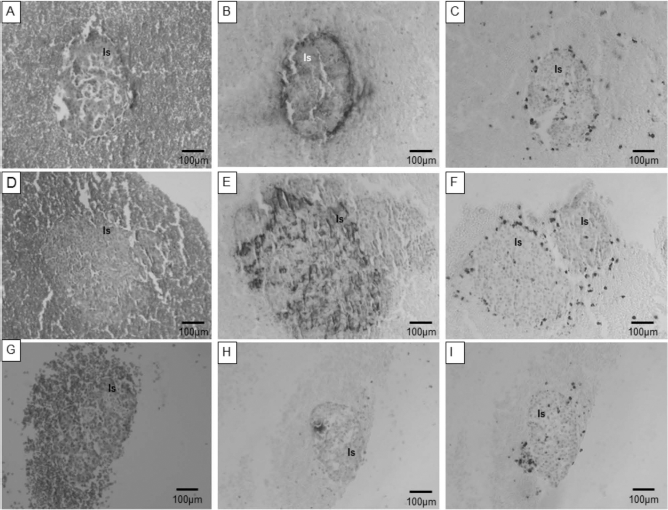
Histological assessment of human islets (fresh and precultured) after incubation in allogenic blood. Morphology of human islets (Is) in blood clots. Adjacent columns show representative serial sections. (A, B, C) Freshly isolated islets. (D, E, F) Islets precultured for 24 h and (G, H, I) precultured for 48 h. (A, D, G) H&E staining; (B, E, H) immunolabeled for insulin (brown); (C, F, I) immunolabeled for neutrophils. Scale bar: 100 μm.
Islets cultured for 48 h before exposure to fresh blood showed β-cell necrosis with insulin leakage, individual islet degranulation of β-cells, and vacuolation (Fig. 4G, 1H).
Neutrophils were easily identified in the blood clot and appeared to migrate to the vicinity of the islet within 1 h. To discover whether there was specific attraction of neutrophils to the islet, the number of neutrophils infiltrating the peri-islet areas was compared to the neutrophils found in the nonislet areas. When freshly isolated islets were incubated with fresh allogeneic blood for 1 h (Fig. 4C), there was a significant (p < 0.05) accumulation of neutrophils into the islets, when compared to precultured islets (24 h, 48 h) mixed with fresh blood (Fig. 4F, I). However, the remaining data were too sparse to achieve statistical significance for any other comparison of conditions.
Discussion
Several studies have demonstrated that IBMIR occurs in vitro or in vivo during islet infusion into the hepatic portal vein (1,2,18). IBMIR has been shown to cause disruption of islet morphology (2,12), and it is likely to cause loss or primary nonfunctional transplanted islets (19). Previously, we have demonstrated that within 1 h of incubation in allogeneic blood, 37% of islets showed features of severe damage on histologic evaluation, confirmed by an increase in proinsulin release compared with control islets. Cell death was predominantly through necrosis with mononuclear and neutrophil infiltration (18).
To determine whether culture of islets improved subsequent islet graft function by attenuating IBMIR, we first assessed changes in functionality of human islets cultured for 0 h, 24 h, 48 h by GSIS. At the onset of the experiment, all the islets were viable, assessed by fluorescein diacetate/ethidium bromide staining. We demonstrated that cultured islets especially after 24 h were more functional when compared to freshly isolated islets or to 48-h precultured islets. These results were substantiated by the insulin staining in Figure 4B, E, and F. Freshly isolated islets (Fig. 4B) leaked insulin, potentially due to islet disruption. Precultured islet (24 h) expressed insulin within the cells, suggesting the islet was intact. Precultured islets (48 h) expressed considerably less insulin when compared to freshly isolated or 24-h precultured islets, possibly due to islet degranulation of β-cells and vacuolation.
We then determined the expression of TF, a mediator of IBMIR, on human islets cultured for 0 h, 24 h, and 48 h. TF is a transmembrane glycoprotein of the cytokine receptor superfamily type 1 and when activated is a major cellular trigger of blood coagulation. We have shown that human islets in vivo (pancreatic section) and in vitro express the proinflammatory mediator TF, as previously reported (8). Anti-TF and TF-inhibitory drug site-inactivated factor VIIa have been shown to inhibit IBMIR in vitro (18). A recent report has described the use of antisense TF RNA to switch off TF production in a porcine model of neonatal islets, thus suppressing IBMIR (11). These findings suggested that IBMIR is triggered by TF. Moreover, it has also been demonstrated that TF released by human islets affects islet engraftment in type 1 diabetic patients (3). It has been shown that TF expression is significantly lowered by culturing the islets in the antioxidant nicotinamide (13). However, we found that even without nicotinamide, 24-h and 48-h precultured islets produce significantly less TF compared to freshly isolated islets. Cultured islets have been shown to express lower MAP kinase activation and heat shock protein when compared to freshly isolated islets (6). A decrease in signaling molecule MAP kinases and heat shock protein could suggest a reduction in inflammation, including a decrease in TF and therefore increased viability.
We also investigated ATP content of cultured islets as another parameter of islet viability (data not shown). The ATP content was used in the previous report of culture (6), and indeed, we found it was marginally higher in cultured islets than that of freshly isolated islets, suggesting recovery from stress and maintenance of cellular homeostasis and function. However, the overlap of data from the two groups was too great to make it a useful addition to the tests.
To determine the effects of cultured islets on IBMIR, proinsulin assay was carried out to measure the ongoing islet disintegration. Incubation of freshly isolated islets in allogeneic blood for 1 h released significantly more proinsulin compared to islets in HBSS. The proinsulin concentration was considerably reduced in precultured islets incubated in blood, suggesting that culture attenuates blood-induced islet destruction. Islets cultured for 24 h released less proinsulin, but had high proinsulin content, confirming that these islets were largely unaffected by the blood. IBMIR is also known to trigger activation of polymorphonuclear cells in blood (18). In our study, we have showed that 24-h culture was also beneficial in attenuating neutrophil infiltration when compared to freshly isolated islets. Mice were not used as a bioassay in this study, mainly because the mouse model is flawed by the incompatibility of mouse receptors for human insulin (15).
In the present study, we have shown that the standard operating protocol for 24-h human islet culture used in our islet transplantation program is clearly beneficial in attenuating IBMIR. It decreases islet fragmentation, as evidenced by a reduction in proinsulin release and the histological assessment of the islet morphology. Moreover, 24-h human islet culture secreted more insulin, high GSIS, and had lower content of the proinflammatory mediator TF when compared to freshly isolated islets. Islets precultured for 48 h had reduced functionality, lower GSIS, and very low insulin expression despite lowered TF expression and proinsulin levels. These findings would suggest that 24-h culture may be beneficial but not 48 h. The increase formation of vacuoles within the 48-h preculture could potentially be responsible for the low GSIS and low insulin expression observed. More work needs to be done to fully understand the beneficial effect of 24-h culture on IBMIR.
Footnotes
Acknowledgments
This study was supported by grants from Diabetes Wellness & Research Foundation, Diabetes UK, and National Institute for Health Research (NHS, UK). The authors declare no conflicts of interest.
