Abstract
Tissue factor (TF) expression on islets has been shown to trigger instant blood-mediated inflammatory reaction (IBMIR), leading to rapid islet loss in portal vein islet transplantation. This study investigated whether antisense RNA-mediated TF gene knockdown in islets could suppress IBMIR as a strategy to overcome IBMIR. Neonatal porcine islet cell clusters (NICCs) were transfected with or without TF-specific antisense RNA or a nonspecific RNA by a lipid-based method. Expression of both TF gene and protein in NICCs was analyzed after transfection by real-time PCR, Western blot, and FACS, respectively. The impact of antisense RNA transfection on NICC viability and in vitro function was examined by FACS and insulin release test, respectively. The effect of TF knockdown in NICCs on IBMIR was assessed with an in vitro tubing loop assay using human blood. A significant reduction in TF gene and protein expression was achieved in TF antisense RNA but not control RNA transfected NICCs, which did not affect NICCs' viability or their insulin secreting capacity. Incubation of TF antisense RNA transfected with human blood resulted in a considerable reduction in blood clot formation, platelet consumption, and complement and coagulation activation compared to that observed in the loops containing human blood and untreated or control RNA transfected NICCs. Consistent with these findings, infiltrating neutrophils in the blood clots with entrapped TF antisense RNA transfected NICCs was also reduced substantially compared to that seen in the clots containing untreated or control RNA transfected NICCs. This study presents a nontoxic TF antisense RNA-mediated TF knockdown in porcine islets that leads to an effective suppression of IBMIR, suggesting a potentially new strategy to improve islet transplantation outcomes.
Keywords
Introduction
Clinical islet transplantation has been shown to be a promising therapy for type 1 diabetes. However, poor engraftment of the transplanted islets limits its application. A significant proportion of islets transplanted intraportally are destroyed in the early posttransplantation period by the instant blood-mediated inflammatory reaction (IBMIR) (2,19), which is characterized by coagulation and complement activation that leads to platelet consumption and neutrophil infiltration of the islets (25,37), and may even be stronger in the xenograft setting due to species barrier (4,33). IBMIR is triggered immediately by several inflammatory mediators when islets come in contact with human blood in the portal vein (2). Tissue factor (TF) has been shown to one of the mediators to initiate IBMIR, although the precise signaling molecules that are critical in triggering this response remain to be identified. TF is a 47-kDa transmembrane glycoprotein that is essential for the initiation of the extrinsic coagulation system and is pivotal for the activation of the intrinsic pathway (8,22). It has been reported that islets express TF, and IBMIR is suppressed in vivo by using an anti-TF monoclonal antibody (5). Infusion of islet cells expressing TF into the portal vein triggers IBMIR, leading to the activation of coagulation pathway and the production of thrombin (16,24,29,31). Collectively, these data suggest the importance of TF expression by islets in regulating IBMIR.
In this study we present a novel approach, TF-specific antisense RNA-mediated gene knockdown to silence TF expression by neonatal porcine islet cell clusters (NICCs) as a means of effectively inhibiting IBMIR. This gene targeting approach allowed us to specifically target TF production by NICCs and to study its role in initiating IBMIR in vitro. Unlike some strategies that have been developed for overcoming IBMIR, this technique does not affect the viability and function of porcine islets, and it circumvents the side effects associated with gene therapy and bleeding risk of systemic IBMIR inhibitors.
Materials and Methods
Animals
Three- to 5-day-old newborn pigs of either sex were purchased from local farms and were used for the preparation of porcine islets. The animal study was approved by the animal Ethics Committee of the Third Xiangya Hospital.
Preparation of Porcine Islets
NICCs were isolated from donor pancreata as described previously (18), and were cultured at 37°C, 5% CO2 in RPMI-1640 medium (Invitrogen) supplemented with 15% porcine serum (Invitrogen), 10 mM nicotinamide (Sigma), 2 mM L-glutamine (Invitrogen), 50 mM isobutylmethylxanthine (Sigma), 100 U/ml penicillin, and 100 U/ml streptomycin (Invitrogen). Culture medium was changed the first day after islet isolation and every second day thereafter.
Antisense RNA Transfection
Antisense RNA specific for TF (sequence: 5′- ACA GUG CUU CCU UUA UGA-3′) was designed to target the coding region of porcine TF mRNA (GenBank ID 396677) and a size-matched nonspecific oligonucleotide (control RNA) (sequence: 5′-CUC AUC GAG CCA UUG GUG-3′) was used as control RNA for transfection of NICCs. Both TF-specific antisense RNA and control RNA were 2′-O-methyl modified with a 3′-butanol tag to increase the stability for resilient to the nuclease digestion (20,40), and were synthesized by Oligos Etc Inc. (Wilsonville, OR, USA). After 24 h in culture nondissociated NICCs were transfected with TF antisense RNA or control RNA, using oligofectamine reagent (Invitrogen) according to the manufacturer's instruction. The conditions for the antisense RNA transfection were optimized, which resulted in the highest transfection efficiency in NICCs selected for the following transfection. In brief, oligofectamine reagent was incubated with RPMI-1640 for 5 min at room temperature prior to being mixed with antisense RNA or control oligonucleotide for an additional 20-min incubation at room temperature. One thousand NICC islet equivalents (IEQ) in RPMI-1640 (no porcine serum and antibiotics) were then incubated with the oligofectamine complex containing 8 μM TF antisense RNA or control RNA/ well and 16 ja.l oligofectamine reagent/well in six-well plates for 6 h at 37°C in a humidified 5% CO2 incubator. After transfection, NICCs were washed and cultured in complement RPMI-1640 before being used in the subsequent experiments.
Real-Time PCR
Total RNA was isolated from NICCs with the TRIzol reagent (Invitrogen) according to the manufacturer's instructions, followed by cDNA synthesis using the SuperScript™ IIRT (Invitrogen). Real-time PCR was performed 48 h after transfection with Stratagene Mx3000P (Agilent Technologies, Redwood City, CA, USA) using SYBR Green qPCR Supermix-UDG universal PCR Master Mix (Invitrogen). cDNA was subjected to PCR with PCR primers specific for porcine TF (sense: 5′-GGAGCCTCTGTATGAGAACT-3′ and antisense: 5′-CGGAGGCTTAGGAAAGTGTT-3′). The reaction was amplified as follows: 40 cycles of 95°C for 15 s, 61°C for 20 s, and 72°C for 30 s followed by an extension at 72°C for 10 min. PCR reaction with porcine β-actin primers (sense: 5′-CACGCCATCCTGCGTCTGGA-3′ and antisense: 5′-AGCACCGTGTTGGCGTAGAG-3′) was used as an internal control.
Fluorescence Activated Cell Sorting (FACS) Analysis
A sheep anti-human TF antibody (Affinity Biologicals, Ancaster, Canada), which was reported to react with porcine TF protein (21), was used for FACS analysis of TF protein expressing NICCs. NICCs collected 4 days after transfection were separated into single cells by accutase (Millipore, Billerica, MA, USA). NICC cells were both surface and intracellular stained with the anti-human TF antibody by incubation with the antibody for 30 min on ice in staining buffer [1% bovine serum albumin (BSA) in phosphate-buffered saline (PBS)] followed by staining with fluorescein isothiocyanate (FITC)-conjugated donkey anti-sheep antibody (Sigma) on ice for an additional 30 min. The surface stained cells were then fixed and permeabilized with fixation/permeabilization buffer (BD Biosciences, San Jose, CA, USA) prior to intracellular staining with the anti-TF antibody in permeabilization buffer (BD Biosciences) for 30 min on ice. Cells were collected on FACScan equipped with CellQuest software (BD Biosciences), and gated to exclude nonviable cells. TF expression level in NICCs was presented as mean fluorescence intensity (MFI) of TF-positive staining cells. For analysis of NICC viability, dissociated NICCs were stained with Annexin V-PE (phycoerythrin) and 7-aminoactinomycin D (7-AAD) using Annexin V-PE apoptosis detection kit (BD Biosciences) according to the manufacturer's instructions. The viability was calculated as the percentage of Annexin V and 7-AAD double negative staining cells.
Western Blotting
Western blotting was performed by lysing equal number of NICC samples (4 days after transfection) using Tris glycine sodium dodecyl sulfate (SDS) lysis buffer (Cell Signaling Technology, Danvers, MA, USA). Homogenized cell lysates were then separated by 10% SDS-polyacrylamide gel electrophoresis and transferred onto nitrocellulose membranes (Hybond-C; Amersham, Bucks, UK). The membranes were blocked for 1 h at room temperature with 0.01% Tween 20 and 5% skim milk in PBS followed by incubation with sheep anti-human TF antibody (Affinity Biologicals, Ancaster, Canada) or mouse anti-human β-actin antibody (Santa Cruz, Santa Cruz, CA, USA) overnight at 4°C. The membranes were then incubated for 1 h at room temperature with horseradish peroxidase (HRP)-conjugated rabbit anti-sheep IgG Santa Cruz (Santa Cruz Biotechnology) or anti-mouse IgG (Santa Cruz) secondary antibody, respectively. Immunoreactive bands were visualized using enhanced chemiluminescence detection kit (ECL; Thermo Scientific, USA).
Insulin Content Assay
Four days after transfection 10,000 NICC IEQ were washed with and resuspended in cold HBSS medium in a final volume of 2 ml. Cell suspensions (200 μl) of each NICC sample were homogenized by ultrasonication on ice prior to analyses of DNA content using an Quanti-IT PicoGreen ds DNA kit (Invitrogen) and insulin content with an insulin immunoradiometric assay kit (Biosource, Nivelles, Belgium), respectively. Insulin capacity was calculated as the ratio of the insulin content to the DNA content in microgram in 1,000 NICC IEQ.
In Vitro Test of Islet Function
Four days after transfection (day 6 of NICC isolation) glucose-stimulated insulin release was measured and expressed as the stimulation index (34), calculated as the ratio of insulin release in high (25.0 mM) glucose in the presence of 1 mM 3-isobutyl-1-methylxanthine (IBMX) to low (2.5 mM) glucose during 60 min of static incubation in Krebs' Ringer bicarbonate buffer. Insulin levels were determined with an insulin immunoradiometric assay kit (Biosource, Nivelles, Belgium).
In Vitro Tubing Loop System
Human blood donors were healthy volunteers and the protocol was approved by the Human Research and Ethics Committee of the Third Xiangya Hospital. Three pig donors were used for each of nine independent loop experiments. A tubing loop model (24,31) was used for in vitro simulation of portal flow. At day 6 of isolation (4 days after transfection) 1,000 NICC IEQ were suspended in 100 μl of PBS and transferred to a heparin-boned PVC tube (inner diameter 6.3 mm, length 450 mm) (Medtronic Inc., Minneapolis, MN, USA) followed by addition of 7 ml of fresh human blood to each loop containing untreated, control RNA, or TF antisense RNA transfected NICCs. The loops were then placed in a shaking incubator at 37°C for 60 min prior to collecting samples for the subsequent assessments. Blood clots were measured for size and weight and remaining blood was collected for hematological analysis using Coulter ACT-diff analyzer (Beckman Coulter, Miami, FL, USA), and coagulation activation [thrombin–antithrombin (TAT)] using commercially available EIA kits (Enzygnost TAT kit, Behringwerke, Marburg, Germany), and assays of complement activation (C3a and sC5b-9) using ELISA assay kits (C3a OptEIA kit, BD Biosciences; MicroVue SC5b-9 Plus EIA Kit, Quidel, San Diego, CA, USA).
Immunohistochemistry
To determine whether the endocrine structures of NICCs were affected by antisense RNA transfection, NICC samples with or without transfection were fixed in agar and embedded in paraffin 4 days after transfection. Sections (5 μm) of NICC samples were incubated with guinea pig anti-porcine insulin antibody (Dako Laboratories, Mississauga, Ontario, Canada), guinea pig antiporcine glucagon antibody (Linco Research, Inc., St. Charles, MO, USA), goat anti-human somatostatin antibody (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA), and rabbit anti-human chromogranin (Dako Laboratories), respectively, for 60 min. After washing, the sections were incubated with a corresponding HRP-conjugated secondary antibody dilution (goat anti-guinea pig, rabbit anti-goat, or goat anti-rabbit, respectively; Dako Laboratories, Mississauga, Ontario, Canada) for 30 min.
For analysis of neutrophil infiltration in blood clots, 6-to 8-μm tissue sections of paraffin-imbedded blood clots were used for neutrophil staining. Briefly, after blocking endogenous peroxidase with 0.3% H2O2 (Kebo Laboratory AB, Stockholm, Sweden) in PBS for 15 min at room temperature, clot sections were incubated with mouse anti-human neutrophil elastase (Dako, Cytomation) for 30 min at room temperature followed by incubation with HRP-conjugated goat anti-mouse IgG (Dako) for a further 30 min at room temperature. The reaction was then developed using diaminobenzine (DAB) (Dako) as substrate.
Statistical Analysis
Results were analyzed by one-way ANOVA followed by post Turkey's HSD test (SPSS, version 18.0, software) and were presented as mean ± SEM. A value of p < 0.05 was considered as statistically significant.
Results
Antisense RNA Transfection Leads to Efficient TF Knockdown in Porcine Islets
To examine whether neonatal pig islet tissue expresses TF, we performed immunohistochemical analysis of TF expression by neonatal pancreas, showing TF-positive staining of neonatal pancreas in situ, which indicates a constitutive TF expression by neonatal pig islets (data not shown). We then used lipid-based antisense RNA transfection to silence TF expression to determine the importance of TF expression on neonatal pig islets in regulating IBMIR. A 55% (range of 50–60%) reduction in TF gene expression was detected in TF antisense RNA but not control RNA transfected NICCs by real-time PCR when compared to their untreated counterparts 48 h after antisense RNA transfection (Fig. 1A). The reduced level of TF gene expression in TF antisense RNA transfected NICCs remained significant for at least 8 days after antisense RNA transfection compared to that detected in untreated or control RNA transfected NICCs (Fig. 1B). The TF knockdown was confirmed by Western blotting analysis of TF protein expression, showing a significant reduction in TF protein expression in TF antisense RNA but not control RNA transfected NICCs compared to their untreated counterparts (2.2-fold reduction in intensity of immunoreactive TF band) (Fig. 1C). The reduced level of TF protein expression was further detected by FACS, demonstrating markedly reduced MFI of TF expressing cells in TF antisense RNA transfected NICC (21.1 of MFI for TF expressing cells in TF antisense transfected NICC vs. 38.7 or 35.4 of MFI for TF expressing cells in untreated or control RNA transfected NICC, respectively) (Fig. 1D). Taken together, these data showed a specific and efficient TF knockdown in porcine islets by TF antisense RNA transfection.

Tissue factor (TF) knockdown in porcine islets. (A) Quantitative analysis of TF gene expression in neonatal porcine islet cell clusters (NICCs) by real-time PCR 48 h after transfection with or without TF antisense RNA or control RNA. TF gene expression is shown as relative expression compared to the internal control gene β-actin. Data represent the mean ± SEM of one representative of three independent experiments. *p < 0.05, compared with untreated NICC. (B) TF gene expression in NICC at different time points after transfection with or without TF antisense RNA or control RNA was analyzed by quantitative PCR and data is shown as the mean ± SEM of one representative of three independent experiments as described above. *p < 0.05, compared with untreated and control RNA transfected NICC. (C) A representative analysis of TF protein expression in NICC by Western blot. (D) Fluorescence-activated cell sorting (FACS) analysis of TF protein expressing cells in NICC. Gate was set on live cells and the level of TF protein expression in TF-positive staining NICC cells was assessed by the mean fluorescence intensity (MFI) of each staining. Data are representative of three independent experiments.
NICCs Retain Intact Morphology and Endocrine Structures After Antisense RNA Transfection
The effect of antisense RNA transfection on NICC integrity and their endocrine structures was examined 4 days after transfection by the microscopy and the immunohistochemistry, respectively, showing a similarly intact NICC membrane structure observed in all three NICC samples (untreated, TF antisense RNA transfected, and control RNA transfected NICCs) under the microscope (data not shown), and no changes in the images of endocrine-secreting positive cells (insulin-, glucagon-, somatostatin-, and chromoganin-secreting cells) within NICCs, regardless of being transfected with or without TF antisense or control RNA transfection, detected by immunohistochemistry (Fig. 2). Taken together, these data indicate that both NICCs' integrity and their endocrine structures were not affected by TF antisense RNA transfection.

Endocrine structures of NICCs. A representative of immunohistochemical analysis of insulin-, glucagon-, somatostatin-, and chromoganin-secreting cells, respectively, within NICCs transfected with or without TF antisense RNA or control RNA (400× magnification).
Antisense RNA Transfection Does Not Affect NICC Viability and In Vitro Function
The viability of NICCs was analyzed 4 days after antisense RNA transfection by FACS. A similar viability was detected in all NICC samples examined, as shown by a 93.3% of total live and viable cells (negative for both Annexin V-PE and 7-AAD staining) in untreated NICCs versus 92.7% or 90.2% of total live and viable cells in TF antisense RNA or control RNA transfected NICCs, respectively (Fig. 3A). The results indicate that transfection with antisense RNA did not affect the viability of NICCs.

Assays of NICC viability and in vitro function. (A) FACS analysis of live NICCs. The percentage of viable cells in untreated, TF antisense, or control RNA NICCs, respectively, was calculated as the percentage of Annexin V and 7-aminoactinomycin D (7-AAD) double negative staining cells. Data shown are representative of three independent experiments. (B) Assessment of insulin capacity of NICCs. Insulin capacity was calculated as the ratio of the insulin content to the DNA content in micrograms in 1,000 NICC islet equivalents (IEQ). Data are shown as mean ± SEM from one of three independent experiments. (C) Glucose stimulation test was used to assess NICC function. Insulin stimulation index was calculated as the ratio of insulin release in high (25.0 mM) glucose in the presence of 1 mM 3-isobutyl-1-methylxanthine (IBMX) to low (2.5 mM) glucose. Data are shown as mean ± SEM from one of three independent experiments.
To evaluate the effect of antisense RNA transfection on insulin capacity of NICC, the contents of DNA and insuln in individual NICC samples were assessed prior to the tubing loop experiments. TF antisense RNA transfected NICCs demonstrated a similar value of insulin/DNA ratio (0.171 ± 0.03) to that shown by untreated (0.164 ± 0.04) or control RNA transfected NICC (0.163 ± 0.04) (Fig. 3B), suggesting an intact insulin capacity of TF antisense transfected NICCs. The unaffected NICC function characteristic by antisense RNA transfection was further evidenced by a similar stimulation index revealed by all three NICC samples (1.91 ± 0.12 of TF antisense RNA transfected NICCs vs. 2.02 ± 0.24 of untreated NICCs or 1.89 ± 0.11 of control RNA transfected NICCs) in the insulin release test (Fig. 3C). Taken together, these results indicate no harmful effect of antisense RNA transfection on both insulin content and insulin releasing capacity of NICCs.
TF Knockdown Results in Suppressed IBMIR In Vitro
We next analyzed the effect of TF knockdown on IBMIR with an in vitro tubing loop model. Blood clot formation as a result of IBMIR was observed after 60-min incubation of untreated NICCs or control RNA transfected NICCs with ABO-compatible human blood in tubing loops (Fig. 4A), which was not seen in the loops with human blood alone. However, both the size and weight (0.12 ± 0.03 g in the loops with TF antisense RNA transfected NICCs vs. 0.29 ± 0.05 or 0.28 ± 0.04 g in the loops with untreated NICCs or control RNA transfected NICCs, respectively, p < 0.001) of the clots formed were reduced substantially when TF antisense RNA transfected NICC were incubated with ABO-compatible human blood compared to that seen in the loops with untreated NICC or control RNA transfected NICCs (Fig. 4A, B).
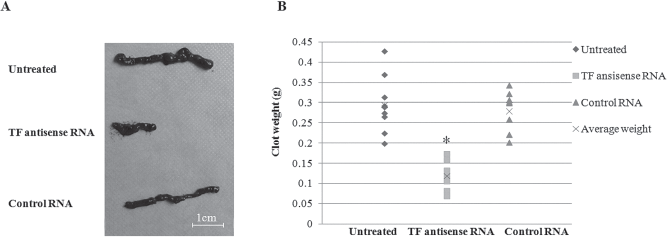
Formation of blood clots in tubing loops. (A) The size of clots formed in the loops with human blood and untreated, TF antisense RNA, or control RNA transfected NICCs. Data are representative of one of nine independent experiments. (B) The weight of clots collected from the loops as described above. Data from nine independent experiments are shown with the average weight depicted by “X.” *p < 0.05, compared with untreated NICC group.
Clot formation in the loops with untreated NICCs was correlated with exhausted platelets, white blood cells, monocytes, and neutrophils when compared to that detected in the loops without NICCs (Table 1). All data were normally distributed. The Tukey HSD ANOVA tests showed that the consumption of platelets (135 ± 8 × 109/L in the loops with TF antisense transfected NICCs vs. 45 ± 4 × 109/L in the loops with untreated NICCs, p < 0.05), white blood cells (WBC) (2.80 ± 0.22 × 109/L in the loops with TF antisense transfected NICCs vs. 1.90 ± 0.13 × 109/L in the loops with untreated NICCs, p < 0.05), monocytes (0.35 ± 0.04 × 109/L in the loops with TF antisense transfected NICCs vs. 0.19 ± 0.03 × 109/L in the loops with untreated NICCs, p < 0.05), and neutrophils (1.30 ± 0.22 × 109/L in the loops with TF antisense transfected NICCs vs. 0.60 ± 0.31 × 109/L in the loops with untreated NICCs, p < 0.05) was reduced significantly when TF antisense RNA transfected NICCs were incubated with human blood compared to that seen in the loops with untreated or control RNA transfected NICCs (Table 1). Consistent with these findings, significantly decreased activation of coagulation (TAT) (646 ± 113 vs. 12731 ± 2327 μg/L, p < 0.05) and complement (C3a and sC5b-9) (367 ± 8 vs. 559 ± 5 ng/ml of C3a, p < 0.05; and 299 ± 9 vs. 381 ± 15 AU/ml of sC5b-9, p < 0.05) was also detected in the loops with TF antisense RNA but not control RNA transfected NICCs compared to that with untreated NICCs (Table 1).
Blood Cell Counts, Complement, and Coagulation Analysis
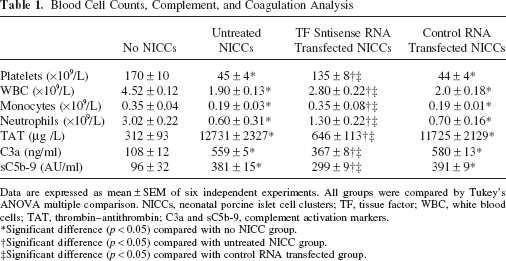
Data are expressed as mean ± SEM of six independent experiments. All groups were compared by Tukey's ANOVA multiple comparison. NICCs, neonatal porcine islet cell clusters; TF, tissue factor; WBC, white blood cells; TAT, thrombin–antithrombin; C3a and sC5b-9, complement activation markers.
Significant difference (p < 0.05) compared with no NICC group.
Significant difference (p < 0.05) compared with untreated NICC group.
Significant difference (p < 0.05) compared with control RNA transfected group.
TF Knockdown Inhibits Neutrophil Infiltration in Blood Clots
To assess the effect of TF knockdown on neutrophil infiltration of NICCs, blood clots collected from loops with NICCs were examined by immunohistochemically staining with neutrophil elastase. A large number of neutrophils were found infiltrating the clots with entrapped NICCs collected from the loops with untreated or control RNA transfected NICCs (Fig. 5A, C). Incubation of TF antisense RNA transfected NICCs with human blood resulted in a remarkable reduction in neutraphil infiltration within the clots in their loops compared to that seen with untreated NICCs (18 ± 8 neutrophils/NICCs in the clots from the loops with TF antisense RNA transfected NICCs vs. 82 ± 19 neutrophils/NICCs in the clots from the loops with untreated NICCs, p < 0.001) (Figs. 5A, 4B, 5C), indicating the requirement of TF expression on NICCs for neutrophil infiltration in this simulated model of IBMIR.

Neutrophil infiltration in porcine islets. Neutrophil elastase-positive staining cells surrounding NICCs entrapped within the clots from tubing loops with untreated NICCs (A), TF antisense RNA transfected NICCs (B), and control RNA transfected NICCs (C), respectively (400x magnification). (D) Infiltrating neutrophils/NICCs in the clots from each group are shown as mean ± SEM of one representative of three independent experiments. *p < 0.001, compared with untreated NICCs and control RNA transfected group.
Discussion
IBMIR that causes early loss of islets transplanted intraportally represents a key obstacle to successful islet transplantation. TF expressed and produced by islets has been shown to be a major trigger for IBMIR (3,4,24,31), which results in an immediate clotting event (activation of coagulation and complement cascades and aggregation of platelets) and a rapid infiltration of neutrophils and monocytes into islets, leading to tissue injury by disrupting islet morphology and triggering islet cell death by direct cytotoxicity or apoptotic induction (15,39). Thus, it is our hypothesis that the development of strategies to target TF expression on islets could provide protection of islets from IBMIR-mediated cell death in islet transplantation.
In this study, TF-specific antisense RNA was used to suppress TF gene expression in porcine islets by lipid-based transfection. The antisense RNA-mediated TF knockdown had no harmful effect on NICCs' viability and their insulin-secreting capacity. Thus, this allowed us to investigate the precise role of TF expression by islets in mediating IBMIR and provide useful information for the development of TF-targeting strategies to minimize IBMIR. TF gene knockdown in NICCs resulted in reduction in all components of xenogeneic IBMIR, namely decreased clot formation, reduced platelet consumption, inhibited activation of coagulation and complement, and suppressed neutrophil infiltration. In line with these findings, previous studies have shown that blockade of TF with a monoclonal anti-TF antibody or inhibition of TF production with nicotinomide reduced IBMIR in vivo and in vitro (5,26). Hence, our study further confirms the requirement of TF expression on NICCs for triggering IBMIR, although platelets and neutrophils are known to secrete TF in response to thrombin (11,28).
Suppression of IBMIR in pre-clinical islet transplant models has been achieved using anticoagulants such as heparin and melagatran, and/or by ex vivo pretransplant treatment with antioxidants including low molecular weight dextran sulfate (12,14,31). However, these strategies may cause systemic complications such as bleeding (38). Several other strategies to prevent or suppress IBMIR have been investigated. For example, addition of tirofiban and activated protein C (APC) to the coculture of human blood with allogeneic islets synergistically inhibited IBMIR (1). Coating of pig islets with human endothelial cells reduced all components of xenogeneic IBMIR in a tubing loop model with human blood (17). Liposomal formulations of thrombomodulin could increase engraftment after intraportal islet transplantation (7). Using islets from transgenic animals with human CD39, an inhibitory molecule of IBMIR could also be considered as an alternative approach to reducing coagulation in contact with human blood (9). The antisense RNA-mediated gene knockdown applied in the present study has been proved previously as an effective gene targeting approach to halting production of a specific protein due to its high efficiency in silencing target genes (6,23,36). The antisense technology has been widely used in gene therapy studies, which hold therapeutic potential in treatment of human diseases, such as cancer, Parkinson's disease, and high cholesterol (13,27,32,43). Indeed, Vitravene, a drug that is developed based on antisense technology, has been used clinically to treat an eye disorder associated with AIDS (30). Hence, the antisense RNA-mediated gene knockdown provides an attractive mean to specifically silence TF expression on islets to achieve a local suppression or prevention of IBMIR. This approach involves in a simple procedure without any notable signs of toxicity in islets or the potential to induce systemic side effects, and using clinically applicable reagents. The antisense RNA approach is distinctly differ from coating of islets with human endothelial cells, which involves multiple steps including endothelial cell isolation and in vitro culture prior to the coating procedure, nor is it likely to risk inflammatory or even adaptive immune responses, which has been seen in all existing procedures involving in introduction of new DNA into the cells, including islets (41,42). One major concern for using the antisense technology in gene targeting studies is its relatively short stability in the cells transfected, leading to a non-sustainable gene silencing (35). However, given the facts that IBMIR occurs in the first hours (3–6 h) after the infusion of islets into the portal vein (14), that a significant reduction in TF expression on NICCs could be achieved by TF antisense RNA transfection, and the low level of TF expression remained unchanged at least up to 8 days, which led to a suppressed IBMIR within 60 min observed in this study, one could presume that TF knockdown in NICCs by antisense RNA transfection may also be effective to reduce IBMIR in early hours after islet transplantation, provided that this technique becomes feasible. However, this hypothesis remains to be tested in an in vivo animal model.
In summary, our study confirms the requirement of TF expression by NICCs for initiating IBMIR, and demonstrates that TF knockdown by transfectoin of NICCs with TF antisense RNA may provide a new approach to suppressing or minimizing IBMIR. In addition, this technique is readily used to not only suppress gene expression of other molecules such as MCP-1 (10), which are produced by islets and serve as mediators of IBMIR, but also to mediate TF knockdown in human islets, thereby holding promise for improving outcome of clinical islet transplantation.
Footnotes
Acknowledgments
This work was supported by a grant from the Key Clinical Program of the Chinese Ministry of Health (2007-353) and the National Natural Sciences Foundation of China (30900359). The authors declare no conflicts of interest.
