Abstract
The instant blood-mediated inflammatory reaction (IBMIR) has been studied in whole blood models of human allo-islet transplantation for short periods (< 6 h). Beyond this time frame the innate response to intraportally transplanted islets is less well described. A novel whole blood model was applied to study blood–islet–graft interactions up to 48 h. Heparinized polyvinyl chloride tubing was sealed into small bags containing venous blood together with allogeneic human islets and exocrine tissue, respectively. The bags were attached to a rotating wheel (37°C). Concentrated glucose and sodium hydrogen carbonate were added every 12 h to maintain physiological limits for sustained immune cell functions. Plasma was collected at repeated time points for analyses of coagulation/complement activation and chemokine/cytokine production. Immune cell infiltration was analyzed using immunohistochemistry. Coagulation and platelet activation markers, thrombin–antithrombin complex (TAT) and soluble CD40 ligand (sCD40L) showed early high concentrations (at 6–12 h). sC5b-9 steadily increased over 48 h. At 6 h neutrophils and monocytes surrounded the clotted cellular grafts with a following massive infiltration of neutrophils. High and increasing concentrations of CXCR1/2 ligands [IL-8 and growth-regulated oncogene α/β/γ (Gro-α/β/γ)] and IL-6 were produced in response to human islets and exocrine tissue. The CCR2 ligand monocyte chemoattractant protein 1 (MCP-1) exhibited increasing concentrations in response to exocrine tissue. The CXCR3 ligand interferon-inducible T cell a chemoattractant (I-TAC) was produced in response to both human islets and exocrine tissue from 6 h. Monokine induced by g interferon (Mig) and interferon γ-induced protein 10 (IP-10) showed a later response, preferentially to exocrine tissue and with larger variations among preparations. An extended blood model of clinical islet transplantation allowed characterization of early immune activation in response to human islets and exocrine tissue. Increased production of chemokines targeting CXCR1/2, CCR2, and CXCR3 was observed, accompanied by massive intraislet neutrophil infiltration over 48 h. The model proved to be useful in exploring early blood-mediated reactions to cellular transplants and has relevance for evaluation of pharmacological interventions to prevent graft loss.
Keywords
Introduction
Clinical intraportal islet transplantation has over the last decade been established as an alternative treatment for a selected group of type 1 diabetes patients with hypoglycemia unawareness (6,52). Even so, improvement of β-cell survival and function in the early posttransplant period, to enhance intrahepatic engraftment, remains a major area of amendment (13,21). Originally defined in whole blood models of intraportal allo- and xenoislet transplantation, the instant blood-mediated inflammatory reaction (IBMIR) was later reported for hepatocytes, mesenchymal stromal cells, and recently in autologous islet transplantation (24,40,42). This immediate thrombotic and inflammatory reaction (7) is believed to cause a deleterious loss of transplanted islets prior to engraftment (18,59), initiate innate cellular immunity, and enhance adaptive alloimmunity (43). Recent promising results, using CXCR1/CXCR2 blockage in clinical islet transplantation, have underscored the importance of early chemokine-mediated inflammation posttransplant (12).
IBMIR has predominantly been explored in whole blood models running for short periods (< 6 h) (7,30,31,39). Importantly, this innate immune reaction accelerates and shifts into further phases over several days, as demonstrated in blood samples retrieved from patients undergoing clinical islet transplantation (8,37,41). Unfortunately, due to the lack of proper in vitro models, detailed knowledge of the triggered immune responses is still to a large extent missing, thereby limiting our means to intervene and counteract IBMIR. With this aim in mind, a comprehensive human in vitro whole blood model to study biocompatibility and immune response during prolonged incubations, using small volumes, was established (27). Using this novel model we here describe the timing of IBMIR initiated by pancreatic cells in contact with ABO-compatible blood for up to 48 h, with emphasis on identifying chemokine pathways involved in early immune cell recruitment. Addition of purified human islets and acinar tissue in parallel experiments showed that the exocrine cells also had an important role in triggering IBMIR, a finding of clinical relevance.
Materials and Methods
Islet Isolation
Islets of Langerhans were isolated from human pancreata, received from brain-dead donors, using a semi-automated method (22). Islet preparations with a purity of 66–77% and exocrine tissue were used. The exocrine tissue was assessed from impure, high-density fractions from the isolation procedure, predominantly containing acinar tissue. Purity of islet preparation was estimated with digital image analysis (19). Freshly isolated human islets and exocrine tissue were cultured free floating in islet medium composed of CMRL 1066 culture medium (Mediatech Inc., Manassas, VA, USA) with 10% ABO serum and supplements, for about 48 h at 37°C (5% CO2) prior to experiments [for a complete list of supplements see Stahle et al. (54)].
The Rotating Tubing Bag Model
A detailed characterization of the novel blood model including physiological and viability data, emphasizing the relevance of the model, was recently presented (27). In short, equipment in contact with blood was coated with a Corline heparin surface (Corline AB, Uppsala, Sweden) (3). Venous whole blood was drawn from healthy volunteers, and no anticoagulant was added (16). Heparinized polyvinyl chloride (PVC) tubing (Tygon R-3630; Saint-Gobain, France) was cut to suitable lengths and sealed at one end. One milliliter of blood was applied and the tube sealed at the other end, leaving one small hole for additives. Multiple tubing bags were attached to a rotating wheel (10 rpm) placed in a 37°C heating cabinet. Incubations were performed for up to 48 h. Concentrated glucose (833 mmol/L) and sodium hydrogen carbonate (NaHCO3; 1 mol/L) were added every 12 h to keep blood glucose at 3–10 mmol/L and pH ≥ 6.8, which are estimated physiological limits for sustained immune cell functions (27). Addition of cells resulted in higher glucose consumption; however, no systematic difference was seen between equal volumes of human islets and exocrine tissue. Physiological conditions during long-term incubations of whole blood were thoroughly evaluated using blood gas analysis (including pH, lactate, and ion concentrations) and osmolality (27). Blood cell viability was monitored by using phycoerythrin (PE) annexin V apoptosis detection kit I (BD Pharmingen, Erembodegem, Belgium) and measurements of hemolysis (lactate dehydrogenase) (27).
Study Design
The ethical review board in Uppsala approved the drawing of blood (Dnr 2008/264) and the use of tissue for islet isolation from human pancreas (Dnr 2009/371/2). Four major experiments with human islets and exocrine tissue, using four donor pancreata and four different ABO-compatible blood donors, were performed. After being washed two times in phosphate-buffered saline (PBS), 13–15 μl human islets or exocrine tissue were added to each tube containing 1 ml of ABO-compatible whole blood. Negative control consisted of whole blood with addition of 13–15 μl PBS (also referred to as control blood). Positive coagulation control was run for three of four experiments consisting of 5 μl rabbit brain thromboplastin (TP)-S (Biopool International, Ventura, CA, USA) prediluted 1:30 in PBS, added to 1 ml of whole blood. All treatments and controls were run in duplicates for each time point in all experiments. Duplicate tubing bags for each control/treatment were cut open after 6-, 12-, 24-, 36-, and 48-h incubation; blood and clots were collected for further analyses.
Coagulation
Coagulation was evaluated visually by recording the presence and estimated size of the clots with addition of human islets, exocrine tissue, or thromboplastin: 0 = no clots; 1 = small clots (<2 mm); 2 = medium-sized clots (2–5 mm); 3 = big clots (>5 mm).
Protein Analyses
After addition of ethylenediaminetetraacetic acid (EDTA; final concentration 0.10 M; Alfa Aeser, Karlsruhe, Germany), blood was centrifuged at 4,500 × g for 10 min, plasma collected and stored at −80°C for protein analysis. Thrombin–antithrombin (TAT), C3a, and sC5b-9 [terminal complement complex (TCC)] were detected using enzyme-linked immunosorbent assays (ELISAs). TAT was measured using a commercial ELISA kit (Nordic Diagnostica, Billdal, Sweden). C3a was detected using a monoclonal antibody 4SD17.3 as capture antibody and detected with biotinylated anti-C3a, followed by horseradish peroxidise (HRP)-conjugated streptavidin (Amersham, Little Chalfort, Denmark). sC5b-9 was detected using a monoclonal antibody aE11 (Diatec, Oslo, Norway) as a capture antibody and a polyclonal rabbit anti-human C5 followed by HRP-conjugated polyclonal swine-rabbit Ig (both Dako, Glostrup, Denmark) for detection. A standard curve was used to assess protein concentration.
Multiplex protein analyses using multiplex Luminex xMAP Technology (Millipore Corporation, Billerica, MA, USA) were performed for 18 cytokines/chemokines: interleukin-8 (IL-8), growth-regulated oncogene-α/β/γ (Gro-α/β/γ), monocyte chemoattractant protein-1 (MCP-1), interferon-inducible T cell α chemoattractant (I-TAC), monokine induced by γ interferon (Mig), interferon γ-induced protein-10 (IP-10), macrophage inflammatory protein 1α (MIP-1α), macrophage inflammatory protein 1β (MIP-1β), IL-6, IL-1β, tumor necrosis factor-α (TNF-α), interferon-γ (IFN-γ), fractalkine, IL-2, IL-12p70, IL-4, IL-5, IL-13, IL-10, and soluble CD40 ligand (sCD40L). Plasma was incubated with agent-specific colored magnetic beads, thereafter with detection antibodies and streptavidin–phycoerythrin (Millipore Corporation). Multiplates were read using a MAGPIX instrument (Luminex Corporation, Austin, TX, USA). Raw data [median fluorescence intensity (MFI)] was translated into protein concentration (pg/ml) using a standard curve. A few chemokines (IL-8, Gro-α/β/γ, MCP-1, IP-10) were expressed above the Luminex standard curve. For Gro-α/β/γ and IP-10, extrapolated values were used above 10,000 pg/ml. The highly expressed IL-8 and MCP-1 were also analyzed using an established GyroLab assay in our lab and exhibited the same pattern using this detection method.
Immunoassays for protein analysis using a GyroLab workstation (Gyros, Uppsala, Sweden) were performed for detection of tissue factor (TF), IL-8, MCP-1, and IL-6 expressions. For each protein, two monoclonal anti-human antibodies were loaded into a Gyrolab Bioaffy 200 CD micro-laboratory disk to detect TF (capture: Mab TFE; Enzyme Research Laboratories, South Bend, IN, USA; detection: CD142; BD Pharmingen, Stockholm, Sweden), IL-8 (capture: MAB208; detection: AF-208-NA; R&D Systems, Minneapolis, MN, USA), MCP-1 (capture: MAB279; detection: AF-279-NA; R&D Systems), and IL-6 (capture: 554543; BD Pharmingen; detection: MAB206; R&D Systems). A standard curve was run for each analyte and used to calculate protein concentration.
Immunohistochemistry
EDTA-treated blood samples with pancreatic cells imbedded in clots (and control clots without cells) were spun down, as previously described. After plasma was removed, the clots were washed and left in 4% neutral-buffered formalin for 24 h. Formalin fixated clots were rinsed with water and put in 70% ethanol, thereafter imbedded in paraffin. Formalin-fixed paraffin-embedded clots were cut in serial sections. Immunohistochemical staining was carried out manually using a standard immuno peroxidase technique. Primary antibodies were directed against endocrine tissue (chromogranin A), neutrophils [myeloperoxidase (MPO)], monocytes (CD68, CD14, CD163), and T cells (CD3). Antibodies were purchased from Neomarkers/Thermo Scientific (Fremont, CA, USA) (chromogranin A: MS-324-PO, clone LK2H10; diluted 1:1,200), Dako (Glostrup, Denmark) (MPO: A0398 diluted 1:1,200, CD68: M0814 clone KP1 diluted 1:2,000 and CD3: A0452 diluted 1:200), and Leica (Newcast, UK) (CD14: CD14-223-S clone 7 diluted 1:100 and CD163: CD163-S clone 10D6 diluted 1:75). All antibodies were diluted in PBS with 0.05% Tween-20 (Dako, Carpinteria, CA, USA). With the exception of chromogranin A and MPO, all antigens were unmasked by heat-induced epitope retrieval (pH 9.0). Counterstaining was performed with hematoxylin (Histolab, Göteborg, Sweden). Duplicate slides, with clotted islets and exocrine tissue, respectively, were stained and examined for each time point for chromogranin A, MPO, CD68, CD14, and CD163; for CD3, single slides were examined at early time points. Immunohistochemical findings were evaluated using an arbitrary scale, estimating the occurrence of immune cells in the close surrounding of the cellular graft (in the graft vicinity), infiltrating the graft tissue and in the rest of the surrounding clot: 0 = no cells, 1 = few cells, 2 = moderate number of cells, 3–4 = more than moderate number of cells/massive infiltration.
Statistical Analyses
Means of duplicates from each experiment were used for presentation and statistical analyses. Repeated measurement two-way ANOVA followed by Dunnett's test for multiple comparisons were performed on log2-transformed protein data. Analyses were performed using GraphPad Prism Version 6 (GraphPad Software, Inc., San Diego, CA, USA). Treatment effects were verified by nonparametric statistics (the Friedmans test) of “area under the curve” (analyses not shown). Statistics in figures refer to differences in response to human islets and exocrine tissue compared to control blood (*) and differences in response between human islets and exocrine tissue (#). Statistics for thromboplastin compared to control blood are given in the text. Hierarchical cluster analyses, using average linkage method with Euclidian distance measurement, were performed on log2-transformed protein expression data after normalizing by each cytokine/chemokine and displayed as dendrograms and heat maps using the PermutMatrix software (LIRMM, Montpellier, France) (10).
Results
Coagulation and Complement Activation
Addition of human islets, exocrine tissue, or thromboplastin resulted in coagulation and thrombocyte activation demonstrated by clotting together with TAT and sCD40L expression (Figs. 1 and 2). Noticeable clots formed in response to human islets and tended to dissolve after 12 h (Fig. 1). TAT and sCD40L exhibited early high concentrations in response to cells and thromboplastin (6–12 h), whereafter sCD40L decreased to similar concentrations as baseline and TAT remained at high concentrations throughout the study period (Fig. 2A, B). TF gradually increased in response to human islets and exocrine tissue compared to control blood (p < 0.05), whereas addition of thromboplastin kept an unchanged baseline expression (Fig. 2C). C3a exhibited high concentrations in response to cells and thromboplastin already at 6 h, and the levels remained high throughout 48 h (p = 0.069) (Fig. 2D). sC5b-9 production responded highly to the islets, exocrine tissue, and thromboplastin already from 6 h, successively increasing over time (p < 0.0001) (Fig. 2E).
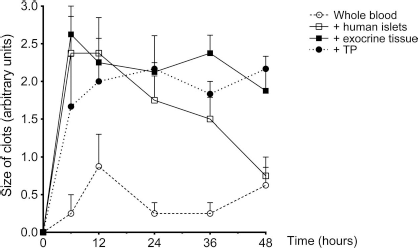
Visual evaluation of coagulation. Visual evaluation of clots in the tubing bags during 48-h incubation in whole blood only (control), or with addition of human islets, exocrine tissue, or thromboplastin (TP): 0 = no clots; 1 = small clots (< 2 mm); 2 = medium-sized clots (2–5 mm); 3 = big clots (>5 mm). Data presented as means ± SEM from four experiments except for TP (N = 3).

Coagulation and complement activation in response to human islets and exocrine tissue. Coagulation [TAT, sCD40L, tissue factor (TF)] and complement (C3a, sC5b-9) activation over 48 h when incubating whole blood only (control), or with addition of human islets, exocrine tissue, or thromoplastin (TP). Protein concentrations in plasma presented as means ± SEM from four experiments except for TP (N = 3). (A) TAT, (B) sCD40L, (C) TF, (D) C3a, (E) sC5b-9. p Values give the main treatment effect of addition of human islets and exocrine tissue compared to whole blood (control) from repeated measurements two-way ANOVA. p Values for post hoc tests given as asterisks for comparisons whole blood versus islets and exocrine tissue, respectively (*p < 0.05, **p < 0.01).
Immune Cell Infiltration
Immune cell infiltration was analyzed using immunohistochemistry (Figs. 3 and 4). Human islets and exocrine tissue within clots (also referred to as graft tissue) were identified throughout the 48-h incubation period by chromogranin A (islets) and hematoxylin staining. At later time points some clotted graft tissue demonstrated pyknotic nuclei (Fig. 3B) and central necrosis. The degree of immune cell infiltration varied among preparations, time points, and also among different parts of the cellular graft within the same clot. MPO+ neutrophils appeared in high numbers already at 6 h, mainly gathered around and with only single cells infiltrating the human islets and exocrine tissue (Figs. 3A and 4). Increasing numbers of MPO+ cells were infiltrating the graft tissue over time, and at 48 h, the infiltrating cells by number exceeded the closest surrounding clot (Figs. 3A and 4). Anti-CD68 antibody stained monocytes/macrophages and also ductal structures (Fig. 3C). Anti-CD14 and anti-CD163 antibodies were therefore used to identify monocyte/macrophage infiltration. CD14/CD163+ cells were gathered around the human islets and exocrine tissue already at 6 h, and the number increased only slightly over the following 42 h (Fig. 4). A few to moderate number of CD14/CD163+ cells infiltrated the grafts already at 6 h, but this infiltration did not increase over 48 h. CD163+ cells, often oval and falcate-shaped in appearance (Fig. 3D), tended to infiltrate the human islets and exocrine tissue to a greater extent than CD14+ cells (Fig. 4). CD3+ T cells were in general present only in small numbers equally distributed within the islets/exocrine graft tissue, in the graft vicinity, and in the surrounding clot for all samples and time points (Fig. 4). However, at 48 h clustering of CD3+ cells around exocrine grafts was noticed for two of the preparations (Fig. 3E).
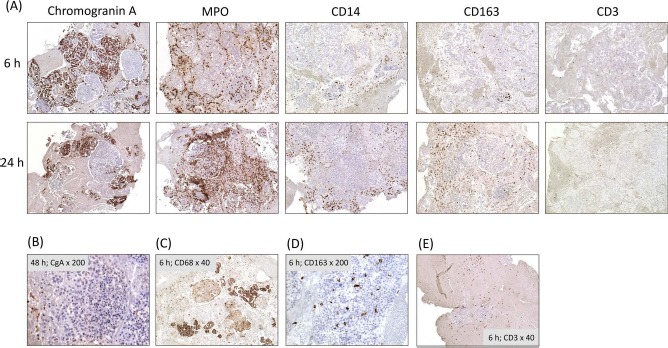
Immunohistochemical staining of immune cells, surrounding and infiltrating clotted pancreatic cellular grafts. (A) Representative photos (magnification: ×40) of formalin-fixed, paraffin-embedded sections of clots containing human islets after 6-and 24-h incubation in ABO-compatible whole blood. Chromogranin A+, MPO+, CD14+, CD163+, and CD3+ cells are presented, counterstaining with hematoxylin. (B) Pycnotic nuclei after 48-h incubation of exocrine tissue; chromogranin (CgA) and hematoxylin staining. (C) CD68 staining of an islet graft after 6-h incubation. (D) CD163 staining of monocytes infiltrating an islet graft. (E) CD3+ cells surrounding exocrine tissue after 6-h incubation.

Immunohistochemical evaluation of immune cell infiltration. Human islets and exocrine tissue embedded in clots, collected and stained after 6- to 48-h incubation in ABO-compatible whole blood. Graphs presenting arbitrary evaluation of MPO+, CD14+, CD163+, and CD3+ cells closely surrounding the cellular grafts—in the graft vicinity (solid lines; filled symbols), infiltrating the graft tissue (crosshatched lines; empty symbols) and in the rest of the clot (dotted lines; half-filled). Human islets and exocrine tissue presented as circles and squares, respectively. The arbitrary scale used: 0 = no cells, 1 = few cells, 2 = moderate number of cells, 3-4 = more than moderate number of cells/massive infiltration. Data presented as means ± SEM from four experiments.
Chemokine Expression
CXCR1/CXCR2-Binding Chemokines.
High concentrations of IL-8 (CXCL8) and Gro-α/β/γ (CXCL1/2/3) were measured in response to human islets and exocrine tissue compared to control blood (p < 0.01) (Fig. 5A). IL-8 showed the highest concentrations of all chemokines analyzed in response to pancreatic cells, with a significant increase already at 6 h. Gro-α/β/γ production was significantly elevated in response to exocrine tissue from 24 h and for human islets from 36 h compared to the control (Fig. 5A). Thromboplastin provoked high concentrations of IL-8 and Gro-α/β/γ in one experiment, while in the two other experiments the levels were similar to control blood (ns compared to control) (Fig. 5A).

Cytokine and chemokine response to human islets and exocrine tissue. Cytokine and chemokine expression in whole blood only (control), or with addition of human islets, exocrine tissue, or thromoplastin (TP), over 48-h incubation. Protein concentration in plasma (pg/ml) analyzed by multiplex Luminex technology (Gro-α/β/γ, Mig, IP-10, I-TAC) and immunoassays from GyroLab (IL-8, MCP-1, IL-6). (A) CXCR1-binding chemokines: IL-8 and Gro-α/β/γ. (B) CCR2-binding chemokine: MCP-1. (C) CXCR3-binding chemokines: I-TAC, Mig, and IP-10 and (D) IL-6. Data presented by boxplots (median, 25th and 75th percentile, min, and max) from four experiments; thromboplastin data from three of these experiments. For Gro-α/β/γ and IP-10 extrapolated values were used above 10,000 pg/ml. p Values give main treatment effect of addition of human islets and exocrine tissue compared to whole blood (control) from repeated measurements two-way ANOVA. p Values for post hoc tests given as asterisks for comparisons control versus islets and exocrine tissue, respectively (*p < 0.05, **p < 0.01) and bars for comparisons islets versus exocrine tissue (p < 0.05, p < 0.01).
CCR2-Binding Chemokine.
MCP-1 (CCL2) production exhibited an immediate (6 h) rise in response to both human islets and exocrine tissue (p < 0.05) (Fig. 5B). Exocrine tissue provoked increasing MCP-1 concentrations compared to control blood and also compared to human islets at 24–48 h (post hoc analyses; p < 0.01–0.05). The response to human islets was not significantly different from that in control blood from 12 h. MCP-1 response to thromboplastin was similar or lower than concentrations in control blood except for one experiment with higher levels than the control at 36–48 h (ns compared to control) (Fig. 5B).
CXCR3-Binding Chemokines.
I-TAC (CXCL11) demonstrated similar patterns for all experiments with increasing concentrations in response to both cell types compared to control blood already from 6 h (p < 0.001) (Fig. 5C). Exocrine tissue provoked significant higher I-TAC response than human islets at 48 h (post hoc analyses; p < 0.05). Production of Mig (CXCL9) and IP-10 (CXCL10) exhibited high variation among individuals but followed each other in expression (Figs. 5C and 6). Exocrine tissue induced high expression of Mig and IP-10 at later time points for predominantly two preparations. I-TAC, Mig, and IP-10 production in response to thromboplastin was overall close to, or even lower than, the control (ns compared to control; Fig. 5C).
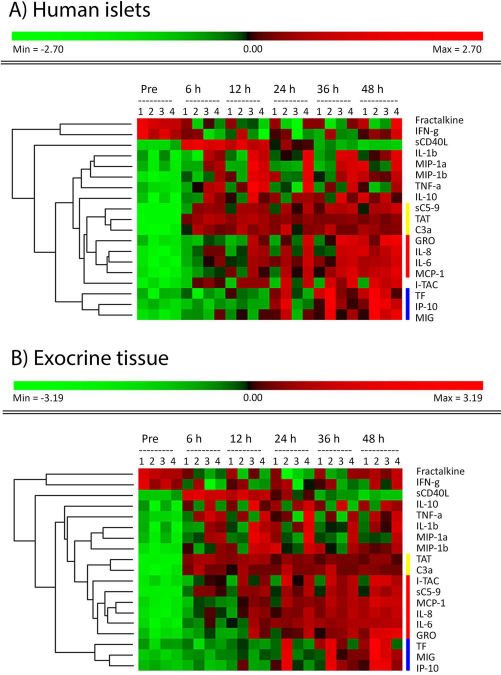
Visualization of protein expression using heat maps. Cluster analyses of protein concentrations in plasma in response to human islets (A) and exocrine tissue (B) illustrated by dendrograms and heat maps. These analyses illustrate changes/differences in expression over time and among experiments for each protein; not in comparison with control blood. The different experiments are given as numbers (1, 2, 3, and 4). Hierarchical cluster analyses were performed on log-transformed data after normalization by each analyte. Elevated concentrations, compared to mean for each protein, are shown with increasing intensities of red and reduced concentrations shown in green; black indicates no change. The dendrogram cluster proteins with similar expression patterns. Proteins with high (red) concentrations throughout the study period (TAT, C3a) were identified as well as those successively increasing from 6–12 h (IL-8, IL-6, MCP-1, Gro-α/β/γ, I-TAC) and those increasing later (TF, Mig, IP-10). sCD40L exhibited high concentrations at 6–12 h. Cytokines/chemokines with high variation among preparations exhibited a striped pattern (MIP-1α, MIP-1β, IL-1β, TNF-α, IL-10). Cytokines expressed at or below detection limit at all time points were not included (IL-2, IL-12p70, IL-4, Il-5, IL-13) in the analyses.
CCR5-Binding Chemokines.
MIP-1α (CCL3) and MIP-1β (CCL4) showed moderate levels of expression from 6 h, however with high variation among experiments and time points. There were altogether no significant elevations in response to added pancreatic cells compared to control blood. In two experiments, high MIP-1α and MIP-1β concentrations were found in response to cells and also in control blood, with high levels at 12–24 h (Fig. 6). For the same donor–recipient pairs the production of IL-1β and TNF-α was also upregulated at the same time points (Fig. 6).
Altogether, thromboplastin provoked similar levels of platelet consumption and TAT/sC5b-9 formation as pancreatic cells. However, the concentrations of CXCR1/2-ligands, MCP-1, IL-6, and CXCR3-ligands in response to thromboplastin were for most samples close to, or lower than, those measured in control blood.
Cytokine Expression
Human islets and exocrine tissue provoked a high production of IL-6 already within 6 h, increasing over the 48-h observation period (p < 0.05) (Fig. 5D). IL-6 response to thromboplastin was lower or similar to control blood (Fig. 5D). The inflammatory cytokines IL-1β and TNF-α showed overall low concentrations with high variation and no significant elevations in response to human islets and exocrine tissue. Soluble fractalkine (CX3CL1) and IFN-γ exhibited similar patterns with concentrations in untreated presamples exceeding those after incubation for 6–48 h in most samples; overall low concentrations with several values at or below the limit of detection (Fig. 6). IL-2, IL-12p70, IL-4, IL-5, and IL-13 were expressed below or at the limit of detection throughout the study (data not shown). IL-10 was expressed at low detectable concentrations at all time points, however with high variation among experiments and no significant response to added cells (Fig. 6).
Discussion
Thrombi were formed around the human islets within the first 6 h in our model. The initial coagulation activation was supposedly driven by platelet activation and the intrinsic coagulation pathway, triggered by exposure of extracellular matrix components (e.g., collagen) and cells on the islet surface (45,48,55). Platelet and coagulation activation peaked within 6–12 h, demonstrated by sCD40L and TAT expression, basically concurrent with kinetics in previous clinical data (30). This event was followed by increasing concentrations of TF, likely produced by the pancreatic tissue and leukocytes, including neutrophils (38,46). An equilibrium was reached between thrombosis and fibrinolysis within 24–48 h; clots surrounding the islets partly resolved over the study period. Complement cascades accumulated terminal complement complexes (sC5b-9) simultaneously with increased chemokine production and attraction of immune cells to the graft site. Over the observation time of 48 h there was an ongoing massive neutrophil infiltration, whereas monocytes/macrophages were found infiltrating the clotted graft tissue only in few to moderate numbers. CD163+ cells dominated the graft infiltrating monocytes suggesting a dominance of the M2 phenotype, which has been associated with cell proliferation, tissue repair, and a TH2 profile (36). Cells initially recruited to the graft vicinity (neutrophils and monocytes) are critical in shaping the innate immune response together with activated platelets. The activated platelets form complexes with leukocytes (granulocytes and monocytes) and trigger coagulation/complement activation by various means, providing a surface for coagulation factors and releasing chondroitin sulfate (26). High amounts of sCD40L are released, a key factor in several immune-modulating interactions (15). Through CD40L, platelets interact with and activate CD40-expressing endothelial cells (47). Also pancreatic islets express CD40; CD40-CD40L interaction leading to islet secretion of IL-8, MCP-1, IL-6, and MIP-1β (5,32). Accompanying the early cell recruitment, IL-8, MCP-1, and IL-6 were already produced at high levels by 6 h in our model, with even further increasing concentrations over the 48 h. These chemokines/cytokines have previously shown to be secreted by human islets, as well as by several types of innate immune cells (14,29,44). Concentrations of other chemokines were built up from 6 to 12 h (Gro-α/β/γ and I-TAC) or later (Mig and IP-10). The timing of chemokine production in our study essentially agrees with previous results from ischemia–reperfusion injury after organ transplantation (17).
Three chemokine–chemokine receptor axes were identified: CXCR1/2-, CCR2-, and CXCR3-mediated chemotaxis. High concentrations of IL-8 and the structurally related Gro-α/β/γ were produced in response to both human islets and exocrine tissue. Together with massive neutrophil infiltration, these results support recent promising preclinical and clinical findings of improved posttransplant graft function targeting CXCR1/2 in islet transplantation (12). CXCR1 and CXCR2 are predominantly expressed on myeloid cells, including neutrophils and monocytes (56). CXCR1 binds with high affinity to IL-8, and CXCR2 binds to both IL-8 and Gro-α/β/γ. Known from tumor biology, IL-8 and Gro-α/β/γ are mediators of angiogenesis (25,57). Therefore, administration of CXCR1/2 blockade beyond the induction period should be carefully applied, considering potential impairment of angiogenesis of a cellular graft critically dependent on revascularization.
MCP-1 was produced in response to human islets and exocrine tissue at 6 h, at later time points increasing concentrations in response to exocrine tissue. MCP-1, solely binding the CCR2 receptor, is mainly known as a monocyte attractant and activator, but also attracts memory T cells and NK cells. MCP-1 can be secreted by several pancreatic cells of epithelial origin, for example, islets, acinar cells, and ductal cells, and has been suggested to impair islet transplant outcome (9,28,44). The finding that high-density layers of ductal cells generate higher concentrations of MCP-1 agrees with our excessive response when incubating high-density fractions, although a major source of MCP-1 in the IBMIR will be activated recipient monocytes/macrophages (28). Beneficial effects on islet graft survival have been presented using CCR2-deficient recipients and CCR2 blockade in murine models of islet allo- and xenograft transplantation (1,35,51,53); however no clinical market drug is currently available (34).
CXCR3 (ligands I-TAC, Mig and IP-10) and CCR5 (ligands MIP-1α, MIP-1β and RANTES) are both predominantly expressed by T helper 1 (Th1) and NK cells. Experimental models of islet allotransplantation have demonstrated diminished immune cell recruitment and/or prolonged islet graft survival in CXCR3- and CCR5-deficient recipients or by using CXCR3- and IP-10-directed antibodies (2,4,58). Interestingly, for two exocrine preparations in our study, signs of T-cell accumulation in the vicinity of exocrine tissue coincided with high peaks in Mig, IP-10, and I-TAC expression. I-TAC was consistently produced in response to both human islets and exocrine tissue. Increased concentrations of Mig and IP-10 in peripheral blood following clinical islet transplantation were previously reported (12). The chemokine/cytokine pattern here presented is concordant with measurements in peripheral blood up to 7 days after clinical islet transplantation, further validating our in vitro model (12). Both CXCR3 and corresponding ligands (I-TAC, Mig, and IP-10) seem to be interesting future targets counteracting the IBMIR, potentially in combination with CXCR1/2 and CCR2 blockage. Unfortunately, today no CXCR3 blocker has reached the clinical market despite interest in several treatment areas.
Pancreatic cells provoked high and increasing concentrations of IL-6. IL-6 has proven to have both pro- and anti-inflammatory traits (50), also suggested by contradictive results concerning IL-6 exposure and blockade during islet culture prior to transplantation (11,49). The burst of the triad of proinflammatory cytokines (IL-1β, TNF-α, IFN-γ) in response to islets/exocrine tissue was not evident in our study. Interestingly, these inflammatory cytokines were not significantly changed in peripheral blood after clinical islet transplantation up to 7 days (12).
Acinar tissue, triggered an innate immune response of similar magnitude as an equal volume of human islets. Notably, production of TF and some of the early immune cell chemoattractants (MCP-1 and CXCR3-ligands) were higher in response to exocrine tissue than to human islets. Initiation of a potentially harmful innate immune response by cotransplanted exocrine tissue is in line with our previous clinical islet transplantation data, showing a negative impact of contaminating exocrine tissue on transplant outcome at 1 month (20).
This whole blood model was developed to mimic the microenvironment in the portal vein liver branches, where islets are trapped early after intraportal islet transplantation (33). Blood circulation around the trapped, thrombotic islets in the portal vein branches is most likely limited, as in our model. There are obvious drawbacks with a closed whole blood system, for example, lack of blood cell exchange and the loss of interaction with immunologically active endothelial cells and lymphoid tissue. However, the concordance with previous data from short-term blood models (7,39), preclinical animal models (17,23), as well as clinical studies (12,30), strengthens the relevance of our model for evaluation of pharmacological interventions of blood-mediated reactions to cellular transplants. This study, together with recent promising preclinical and clinical data (12), suggests that initial shielding of the transplanted islets from chemokine-mediated inflammation and cell recruitment could add to prevention of islet graft loss and enhance engraftment.
Footnotes
Acknowledgments
The authors thank the islet isolation team at the Rudbeck Lab, Uppsala, for their skilled work and the blood donors for kindly contributing with their blood to the experiments. We also thank Margareta Engquist and Helena Hermelin for valuable help with immunohistochemical analyses. This study was supported by grants from the Swedish Medical Research Council, the Nordic Insulin Fund, the Ernfors Family Fund, the Swedish Diabetes Association, Diabetes Wellness—Sweden, the Juvenile Diabetes Research Foundation International, AFA Insurances, and the Seventh Framework Programme of the European Union. M.H. was supported by grants from the Center for Clinical Research (CKF) Dalarna-Uppsala University. O.K.'s and B.N.'s positions are in part supported by the National Institutes of Health. Human pancreatic islets were obtained from The Nordic Network for Clinical Islet Transplantation, supported by the Swedish National Strategic Research Initiative EXODIAB (Excellence of Diabetes Research in Sweden) and the Juvenile Diabetes Research Foundation. The authors declare no conflicts of interest.
