Abstract
Islet allotransplantation results in increasing success in treating type 1 diabetes, but the shortage of deceased human donor pancreata limits progress. Islet xenotransplantation, using pigs as a source of islets, is a promising approach to overcome this limitation. The greatest obstacle is the primate immune/inflammatory response to the porcine (pig) islets, which may take the form of rapid early graft rejection (the instant blood-mediated inflammatory reaction) or T-cell-mediated rejection. These problems are being resolved by the genetic engineering of the source pigs combined with improved immunosuppressive therapy. The results of pig-to-diabetic nonhuman primate islet xenotransplantation are steadily improving, with insulin independence being achieved for periods >1 year. An alternative approach is to isolate islets within a micro- or macroencapsulation device aimed at protecting them from the human recipient's immune response. Clinical trials using this approach are currently underway. This review focuses on the major aspects of pig-to-primate islet xenotransplantation and its potential for treatment of type 1 diabetes.
Keywords
Introduction
Islet allotransplantation provides a potential cure for type 1 diabetes (T1D) 1 , but the shortage of islets from deceased human donors limits progress. Currently, diabetic patients may require islets from two or more donors to become normoglycemic2,3. A significant number of islets (perhaps up to 70%) may be lost when transplanted into the portal vein as a result of what is known as the instant blood-mediated inflammatory reaction (IBMIR) 4 and/or from a delay in revascularization of the graft5–11.
Because of these limitations, alternative sources of insulin-producing cells are being investigated. Developments in stem cell research have allowed the transformation of embryonic stem cells (ESCs) into pancreatic β-cells 12 . The in vitro generation of functional β-cells from human induced pluripotent stem cells (hiPSCs) derived from patients with T1D can correct hyperglycemia in mice 13 . In adult mice, exocrine cells have been directly reprogrammed into cells that closely resemble β-cells 14 . However, stem cells may possibly continue to proliferate in an uncontrolled manner after implantation in patients. Furthermore, stem cell-derived insulin-secreting cells have not yet been demonstrated to produce long-term normoglycemia in diabetic nonhuman primates (NHPs) or patients.
Islet xenotransplantation is an alternative promising approach to the treatment of T1D15,16. Pigs have anatomical and physiological similarities to humans. Additionally, the porcine pancreas can be easily excised, and successful islet isolation procedures have been developed 17 . Furthermore, in contrast to when a deceased human is the source, unlimited neonatal as well as adult porcine (pig) islets are obtainable. When transplanted into streptozotocin-induced diabetic NHPs, several studies reported long-term normoglycemia18–26. Use of pigs also has the great advantage of being readily genetically modifiable to provide some protection against the human immune and inflammatory responses. Porcine islet xenotransplantation has therefore become an attractive option for treating patients with T1D.
Here we focus on the major aspects of pig-to-primate islet xenotransplantation, including islet-source pig choice (optimal age, strain, and genetic modification), porcine endogenous retrovirus potential risks, free or encapsulated (immunoisolated) islet transplantation, combined islet and mesenchymal stromal cell or Sertoli cell (SC) transplantation, immunological tolerance induction, previous clinical trials on islet xenotransplantation, criteria for future clinical trials, and future perspectives.
Choice of Islet-Source Pig
Pig age, strain, genetic modifications, insulin secretion, and porcine endogenous retroviruses (PERVs) need to be considered27,28.
Choice of Pig Based on Age
Adult pigs can provide a large number of islets of large size and mature structure and function (Table 1)17,27,29,30. Following transplantation, the porcine islets have the potential to secrete insulin within minutes or hours31,32. However, the cost of housing the pig for a long period before pancreas excision, fragility of islets (making isolation difficult), and high cost of islet isolation are significant disadvantages 31 . In contrast, neonatal islet-like cell clusters (NICCs) and fetal porcine islet-like cell clusters (FICCs) are easy and inexpensive to isolate. They also have the potential for islet proliferation following transplantation33,34. Embryonic porcine islets, with their reduced immunogenicity, proliferative potential, and revascularization by host endothelium might provide a further advantage 35 . The main disadvantages of embryonic, fetal, and neonatal islet cell clusters is their delay in in vivo functioning after transplantation and the high expression of oligosaccharide galactose-α1,3-galactose (Gal), the major antigenic target for primate anti-pig antibodies. Expression of this antigen is much lower in adult islets 36 .
Comparison of Islets Isolated From Pigs of Different Ages *

Gal, galactose-α1,3-galactose; N/A, not available.
Choice of Pig Based on Strain
Islet quantity and function may vary with pig breed 37 . Despite several studies on the yield of high-quality islets from different strains, there is no consensus regarding the optimal pig strain for preclinical/clinical islet xenotransplantation.
High expression of extracellular matrix (ECM) proteins in islet capsules may make islet isolation easier, thus retaining healthier islets for transplantation. Expression of these proteins is higher in German Landrace pigs than in Deutsches Edelschwein pigs 38 . Hampshire and Duroc pigs have lower expression than Landrace and Pietrain pigs 39 . Older pigs express more ECM proteins than younger pigs38,40. How important this would be in moving toward clinical trials remains uncertain. Since the number of potentially transplantable islets is unlimited, this could compensate for any loss of islets during isolation, though clearly this would be less efficient and more expensive.
A high yield of islets was obtained from Chicago Medical School miniature pigs (now Seoul National University miniature pigs, Seoul, South Korea); the yield was higher than from other miniature pigs 41 . Islet yields from Landrace pigs were higher than German Landrace and Pietrain pigs42,43. Wuzhishan miniature pigs were considered to be a feasible source of islets as a much higher yield was obtained from this strain than from some market pigs27,44.
Choice of Pig Based on Genetic Engineering
In an attempt to protect islets from IBMIR and the primate innate and adaptive immune responses, various genetically engineered pigs have been developed (Table 2)27,30,45,46. These modifications have included gene knockout or gene knockdown, for example, knockout of the gene for the enzyme α1,3-galactosyltransferase (which adds Gal to the underlying oligosaccharides on the surface of the pig vascular endothelium) (GTKO pigs) 47 , knockout of the enzyme cytidine monophosphate-N-acetylneuraminic acid hydroxylase (which adds N-glycolylneuraminic acid)48,49, and knockout of the enzyme β1,4 N-acetylgalactosaminyltransferase (which adds N-acetylgalactosamine)50,51. Whether knockout of all of these genes will prove beneficial in porcine islet transplantation remains uncertain. These modifications have also included insertion of a human transgene, for example, for a human complement-regulatory protein (e.g., CD4624,52, CD5553–57, CD5958–60) or a human coagulation-regulatory protein (e.g., thrombomodulin 61 , endothelial protein C receptor 62 , tissue factor pathway inhibitor 63 , asialoglycoprotein receptor-164,65, CD39 59 , CD73 66 ) to provide some protection from primate complement injury and coagulation dysfunction26,67. Manipulations have also aimed toward providing a local immunosuppressive effect by introducing a molecule that:
Experience With the Xenotransplantation of Islets From Wild-Type Pigs in Immunosuppressed NHPs *

Table modified from Park et al. 30 .
Experience With the Xenotransplantation of Islets From Wild-Type Pigs in Nonimmunosuppressed NHPs *

Table modified from Park et al. 30 .
Experience With the Xenotransplantation of Islets From Genetically Engineered Pigs in NHPs (±Immunosuppressive Therapy) *

Anti-LFA-1, anti-lymphocyte function-associated antigen-1 monoclonal antibody; ATG, antithymocyte globulin; CM, cynomolgus monkey; GnT-III, N-acetylglucosaminyltransferase III; hHT, human α(1,2)fucosyltransferase; MMF, mycophenolate mofetil
Table modified from Zhu et al. 27 .
provides a T-cell costimulation blockade [e.g., cytotoxic T-lymphocyte antigen-4 immunoglobulin (CTLA4-Ig), LEA29Y (belatacept, a high-affinity variant of CTLA4-Ig)]68,69;
suppresses the cellular immune response [e.g., major histocompatibility complex (MHC) class II transactivator knockdown (CIITA-DN)] 70 ; MHC class 1 or class 2 knockout or knockdown 71 ; insertion of human leukocyte antigen class I histocompatibility antigen, α chain E (HLA-E)72,73, HLA-G74,75, human leukocyte antigen Cw3 (HLA-Cw3) 76 , human β-D-mannoside β-1,4-N-acetylglucosaminyltransferase III (GnT-III) 77 , or human TNF-related apoptosis-inducing ligand (TRAIL) 78 ;
provides a local anti-inflammatory effect [e.g., by the introduction of a transgene for hemeoxygenase-1 (HO-1), A2079,80, or CD47 81 ].
Recently, pigs with one or more genetic manipulations were produced with transgene expression being driven by an insulin promoter to specifically target pancreatic β-cells 28 . In a humanized mouse model, islets from insLEA29Y transgenic pigs demonstrated the potential to normalize glucose homeostasis and inhibit cellular rejection 68 . Transgenic expression of human complement-regulatory proteins (e.g., hCD46, hCD59, hCD5524,26,52,55,56,58,59) has been shown to provide significant protection against the primate humoral response. Expression of HO-1 can reduce islet apoptosis 79 . Knockout of pig tissue factor or overexpression of the human “anti-thrombotic” gene, CD39, reduces the effect of IBMIR and coagulation dysfunction 82 . Genetically “humanized” pigs exclusively expressing human insulin have been generated 83 .
Genetic Engineering to Increase Pig Insulin Production
Casu et al. reported that the metabolic demands on porcine islets in their natural host are significantly less than after their transplantation into a primate, particularly if the new host is a monkey rather than a human 84 . Nondiabetic cynomolgus monkeys show lower levels of fasting and stimulated blood glucose but higher levels of C-peptide and insulin than nondiabetic pigs. The reported levels in humans lie between those of monkeys and pigs85–88. Graham et al. reported that species incompatibilities in the pig-to-macaque islet xenotransplant model affect the translational and predictive value of pig-to-NHP islet transplant studies with regard to pig-to-human islet transplantation86,87. Developing approaches to improve insulin secretion may be beneficial or even necessary.
Genetic modification of the pig, even if this involves transgenes with an insulin promoter, does not appear to reduce porcine islet function further89,90. Increased insulin production by genetic modification may be a direction for the future. In vitro perifusion assays have shown that porcine islets exhibit a biphasic pattern of glucose-stimulated insulin secretion91–94, but isolated porcine islets secrete six to three times less insulin than human islets during the first and second phases after stimulation with 15 mM glucose. Insulin granule exocytosis triggered by glucose metabolism, and the ensuing rise in cytosolic calcium concentration, is regulated by two major amplifying pathways95,96. The first is a cyclic adenosine monophosphate (cAMP)-dependent pathway, which is activated physiologically by binding of glucagon-like peptide-1 (GLP-1) to its G protein-coupled receptor on β-cells. The second is a cholinergic pathway, which is activated by binding of acetylcholine or cholecystokinin to a type 3 muscarinic receptor. Both of these pathways, if efficiently activated, increase the number of readily releasable insulin granules in β-cells 97 and result in a greater secretory response to glucose stimulation. Cooper et al. have demonstrated that islets coexpressing GLP-1 and activated muscarinic receptor type 3 have significantly improved insulin secretion 98 . The authors have suggested that permanently inducing these changes in porcine β-cells by means of genetic engineering might be a novel approach to increase insulin secretion from isolated porcine islets, bringing their secretory function closer to that of human islets and rendering them more efficient in controlling host glycemia in both preclinical and clinical trials without the need to transplant extremely high numbers of islets 98 .
The Potential Risk of Porcine Endogenous Retroviruses (PERVs)
The potential risk of the presence of PERVs in islets has long been discussed99–102. Even the use of islets from designated/specific pathogen-free (DPF) pigs will not eradicate PERVs, but their presence is currently not thought to be a major problem that would prevent clinical application99,100. No evidence of PERV transmission was found in patients with T1D after long-term follow-up after porcine islet transplantation27,103–106. The existence of PERV-C-free Auckland pigs that have been used in preclinical and clinical trials may possibly offer a solution 107 . If necessary, PERV activation could be suppressed by genetic manipulation108–110, and there is also the potential to knock out PERV 111 , though neither approach may be essential, as no PERV transmission has been documented in clinical trials of porcine islet transplantation112–117.
When all of these data are considered together, there is evidence that the ideal sources of islets might be those isolated from neonatal DPF pigs with specific genetic modifications to protect islets from IBMIR and innate and adaptive immune and inflammatory responses. However, exact choices of strain and genetic manipulation have not yet been conclusively identified.
Free or Encapsulated Islet Transplantation
Two major approaches have been explored in islet xenotransplantation, namely, (i) free (or naked) porcine islet transplantation, in which islets are transplanted without physical protection around them, and (ii) encapsulated islet transplantation, where islets are encased in some form of protective capsule or device. The purpose of encapsulation is to protect islets from the recipient's immune and inflammatory responses, yet allow insulin to be released. When free islets are transplanted, some form of exogenous pharmacologic immunosuppressive therapy is required to prevent rejection (unless this could be achieved entirely by genetic modification of islets, which is not currently possible).
Free Islet Transplantation
In current clinical practice, free allo-islets are delivered into the portal vasculature, and the liver has been proven to be a site associated with clinical success2,118. Islets are infused through a catheter placed into the portal vein under ultrasound or fluoroscopic guidance119,120. One disadvantage of the hepatic site is the low oxygen tension 121 , as hypoxia is an apoptosis-inducing signal in β-cells 122 . Furthermore, after infusion into the portal vein, IBMIR has been proven a major hurdle123,124.
IBMIR encompasses complement activation, coagulation activation, platelet activation and aggregation, proinflammatory cytokine/chemokine production, and infiltration of leukocytes (Fig. 1) 125 . Interventions directed against the various components of IBMIR reduce early graft loss but are far from completely successful in this respect124,126–128. Complement activation can be partially controlled by agents such as cobra venom factor or soluble complement receptor 1 (CR1)4,129 or C5a-blocking peptide130,131. Coagulation can be reduced by heparin infusion4,132,133, low-molecular-weight dextran134,135, melagatran 136 , or an anti-tissue factor antibody (CNTO859) 137 . Antiplatelet agents, such as tirofiban, can inhibit platelet activation/aggregation 138 . Developmental endothelial locus-1 (Del-1) downregulates the interaction between platelets and monocytes, thus reducing aggregation 139 . By decreasing tissue factor expression, pretreatment of por cine islets with nicotinamide can ameliorate IBMIR140,141.

Overview of instant blood-mediated inflammatory reaction (IBMIR). The contact between blood and islets triggers the activation of coagulation that is mediated through tissue factor (TF). As a result, thrombin is generated, leading to fibrinogen deposition. Attachment of platelets to islets further increases the procoagulant. Complement (iC3b) is deposited on the islet surface, and C3a and C5a are activated, attract leukocytes, and promote formation of the membrane attack complex (MAC), which mediates the lysis of islets. FVIIa, activated coagulation factor VII.
However, several of these experimental agents cannot be used clinically, and so alternative agents (e.g., agents specifically targeting complement, such as compstatin) are under investigation142,143. The combination of antiplatelet and anticoagulant agents can, of course, increase the risk of bleeding, and therefore the patients (or NHPs) would require close monitoring.
Other strategies include the transplantation of islets from pigs overexpressing CD39144,145, human tissue factor pathway inhibitor 146 , human thrombomodulin 147 , or the knockout (or knockdown) of tissue factor148,149. Hawthorne et al. prevented IBMIR by using pig NICCs transgenic for human complement-regulatory proteins transplanted into baboons 150 .
Site of Implantation of Free Islets
Because of the loss of islets after their transplantation into the portal vein, other potential sites are being explored, though a perfectly hospitable site remains elusive (Table 3) 151 . Because the native pancreatic bed is relatively inaccessible, attempts have been made to deliver islet grafts at several other sites. Transplantation into the splenic vasculature resulted in significant morbidity, including infarction, rupture, and gastric perforation 152 . Although transplantation into the renal subcapsular space has become the gold standard for experimental purposes and for islet quality control in rodents, this site has not proved entirely successful in large animals or clinical studies, possibly because of a relative ischemia until revascularization takes place153,154. The intramuscular transplantation of pig NICCs into diabetic mice has been successful but has not yet been proven successful in large animal models 155 .
Comparison of Different Sites for Islet Transplantation *
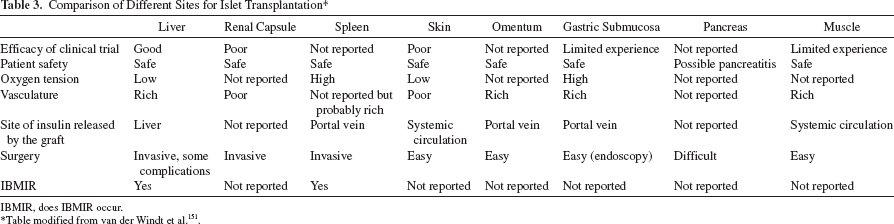
IBMIR, does IBMIR occur.
Table modified from van der Windt et al. 151 .
The omental pouch is a viable site that offers a safe, convenient, and efficacious alternative to islet transplantation into the renal subcapsular in rodents 156 . The omentum offers good vascularization and drainage of the produced insulin into the portal vein for direct utilization in the liver 151 . The results of allotransplantation in NHP models using the omental pouch have been reported 157 . There is a multicenter trial ongoing with the BioHub system, using omentum as an alternative transplant site 158 . Compared with islet transplantation into the portal vein, islets in the omentum have to secrete insulin and release it into the portal vein, but the omentum is anatomically more similar to the pancreas151,159.
Transplantation into the submucosal space of the gastrointestinal tract can be achieved by endoscopy and offers the advantage of subsequent biopsy160–162, but clinical testing has been very limited to date 163 .
Bone marrow is currently being considered as an alternative site for islet transplantation. Studies in mice demonstrated that syngeneic islets could survive in bone marrow indefinitely with greater success in inducing normoglycemia compared to islets transplanted into the liver164,165.
Immunosuppressive Therapy Following the Transplantation of Free Islets
Free islet transplantation requires the administration of significant exogenous immunosuppressive therapy to prevent rejection, especially in the xenotransplant setting. Modulation of the CD40–CD154 pathway has been associated with encouraging results19,20,23–26,166. However, anti-CD154mAb is currently unlikely to be administered clinically in view of the associated risk of thromboembolic complications. The anti-CD40mAb Chi-220 has been reported to be effective in pig-to-NHP islet xenotransplantation, but it is a depleting antibody 166 . Another anti-CD40mAb, 2C10R4, is a nondepleting antibody that has shown success in preclinical islet allotransplantation 167 and pig-to-NHP organ transplantation168–171, and so there is great potential for this mAb in clinical practice.
Other agents that might also contribute to successful suppression of the immune response are also being investigated, but efalizumab [anti-lymphocyte function-associated antigen-1 monoclonal antibody (anti-LFA-1mAb)] was also withdrawn from the market because of the occurrence of three cases of progressive multifocal leukoencephalopathy following a trial in patients with psoriasis 172 .
Transplantation of Immunoisolated Islets
Lifelong immunosuppressive therapy, as would be required after the transplantation of free porcine islets (unless immunological tolerance can be induced), might be accompanied by significant side effects or complications, and so islet transplantation that might not require such therapy is being explored. Cell immunoisolation by encapsulation in a semipermeable matrix to protect islets from immune cells is one such approach173,174. Encapsulated porcine islets have been transplanted to nonimmunosuppressed NHPs (Table 4)175,176.
Experience With the Xenotransplantation of Encapsulated Porcine Islets in Nonimmunosuppressed NHPs *

CM, cynomolgus monkey; MCD, monolayer cellular device; MSC, mesenchymal stromal cells; PLL, poly-L-lysine; PLO, poly-L-ornithine; NICCs, neonatal islet-like clusters.
Table modified from Zhu et al. 175 .
Encapsulation entails coating cells or tissue in a semipermeable biocompatible material that allows for the entry of nutrients, oxygen, and hormones while blocking the entry of immune cells and, ideally, immune molecules (e.g., antibody, complement, cytokines, chemokines) that might recognize and destroy the islets 177 . Islet encapsulation requires the encapsulating material to have several properties, including (i) biocompatibility, (ii) immunoprotection (yet allowing insulin to be released through the capsule wall), and (iii) the ability to allow oxygen and nutrient diffusion into the capsule for islet survival 178 .
There are three main types of encapsulation systems: intravascular devices, microcapsules, and macrocapsules 179 , and there is also a relatively new technique—conformal coating (Table 5)175,180.
Devices for Encapsulation of Porcine Islets *

APCN, amphiphilic conetwork; APF, arteria profunda femoris; CV, cubital vein; MCD, monolayer cellular device; NaCS, sodium cellulose sulfate; PAN-PVC, polyacrylonitrile-polyvinylchloride copolymer; PEG, polyethylene glycol; PLL, poly-L-lysine; PLO, poly-L-ornithine; PSU, polysulfone; PTFE, polytetrafluoroethylene.
Table modified from Zhu et al. 175 .
Intravascular and Extravascular Devices
Intravascular devices are islet-containing perfusion devices anastomosed to the vascular system as arteriovenous shunts179,181. Although this device ensures a rapid exchange of insulin and glucose, complications (e.g., thrombus formation, bleeding) associated with vascular prosthetic surgery potentially limit the therapeutic potential of this approach.
Extravascular devices can be categorized by their size into (i) microcapsules (150–1,000 μm) and (ii) macrocapsules (3 cm × 8 cm)182,183.
Microencapsulation
Microcapsules (Fig. 2A) offer better oxygen and nutrient transport because of higher surface area-to-volume ratio184,185. They require less complex manufacturing procedures and can be simply injected but are difficult to remove completely, if this becomes necessary 186 . Alginate is commonly used to entrap islets187,188.

Examples of encapsulation systems and modification of islets. (A) Overview of microencapsulation. Microcapsules incorporate individual or small groups of islets in a spherical hydrogel polymer with a stable structure. (B) Beta-O2 macroencapsulation system. Islets immobilized within the alginate compartment, which is covered by a polytetrafluoroethylene (PTFE) membrane. Alginate and PTFE provide immune protection and facilitate neovascularization. The double-chambered bioreactor is connected to subcutaneous refueling ports through which an oxygen–CO2 mixture is delivered by daily injection. (C) Monolayer cellular device. The collagen support is covered by a mono/bilayer of porcine islets and embedded with alginate. (D) Conformal coating. Cell surfaces can be modified with amphiphilic polymers that interact both with the lipid membrane and the bioactive substance.
Xenotransplantation of microencapsulated porcine islets in an alginate matrix confirmed their biocompatibility and safety and reduced the insulin requirement in NHPs189,190. There has been one report of long-term survival (>9.5years) of some alginate-based microencapsulated FICCs transplanted into the peritoneal cavity in a T1D patient in 1996, though the patient's glucose level was not controlled 191 . A clinical trial of a microencapsulated porcine islet system (“Diabecell”) conducted by Living Cell Technologies Limited (Auckland, New Zealand) has been carried out (see below)192,193.
A nationally regulated clinical trial of intraperitoneal alginate-poly-L-ornithine-alginate (APA)-encapsulated NICCs (obtained from Diatranz Otsuka) was carried out in 14 nonimmunosuppressed diabetic patients in New Zealand 194 . The results, though not yet fully reported, suggest only partial graft function, with little impact on the clinical status of the patients 98 . A second clinical trial conducted in Argentina, although again not fully reported yet, demonstrated improved diabetic status of patients for more than 2 years 98 (A. Abalovich and S. Matsumoto, unpublished data, personal communication). These pilot clinical trials have largely confirmed the safety of microencapsulated islets but have not yet convincingly confirmed their efficacy. Improvements in microcapsule design and fabrication, optimization of biomaterials and implantation site to facilitate oxygen transport, coupled with sufficient or renewable islets of high quality and low antigenicity may help to provide favorable results 175 .
Macroencapsulation
Macrocapsules can be implanted and removed with minimal risk, but oxygen and nutrient transport are limited185,195. Research on macrocapsules is focused on promoting neovascularization and providing sufficient oxygen and nutrition21,186,196–198. However, revascularization might be associated with the risk of rejection of islets.
A commercially available form of a macrocapsule is TheraCyte, which is made of bilayered polytetrafluoroethylene (PTFE)199–201. Either free or microencapsulated islets are placed in the membrane. NICCs in a TheraCyte system have reversed diabetes for up to 10 weeks in diabetic mice 189 . The TheraCyte system is impermeable to immune cells but permeable to antibodies and complement, which is a major limiting factor 220 . Furthermore, the beta-O2 implantable chamber has been created to offer an adequate oxygen supply to islets (Fig. 2B)196,202–203,204. Oxygen is supplied to islets via two ports connected to a gas reservoir integrated into the device 202 . Following structural improvements and successful application in large animals204,205, clinical evaluation of the beta-O2 device was initiated in eight patients by Beta-O2 Technologies in 2014. However, to ensure that adequate oxygen reaches the islets, the cell density within this device needs to be quite low and thus may be inadequate to sustain normoglycemia.
A monolayer made of alginate (adult porcine islets seeded as a monolayer on a human decellularized collagen matrix) (Fig. 2C) 174 implanted subcutaneously demonstrated an ability to correct hyperglycemia for up to 6 months in diabetic monkeys without the need for immunosuppressive therapy 21 . However, no recent studies appear to have been published by this group.
Conformal Coating
Conformal coating (Fig. 2D) 206 is a new approach to overcome the diffusion limitations associated with capsules of large size (>600 μm) by modification of the islet surface with polymerization [e.g., polyethylene glycol (PEG)] to form a thin coat (<50 μm)186,207–211. This approach allows transplantation into the portal vein, but the normoglycemia obtained has been very transient212–214. Improvements in the technology may allow coating of islets by molecules such as heparin, urokinase, or thrombomodulin132,186,209,215–219. The main drawback is the possible cytotoxicity of the compounds used. This strategy offers an opportunity to combine the inherent advantage of microencapsulation with conformal coating.
Site of Implantation of Encapsulated Islets
The microenvironment of the implant site plays a major role in the survival of encapsulated porcine islet xenografts. The intraperitoneal cavity has been the site of implantation most often, as there is less restriction of the volume of the grafts that can be implanted18,189,191,197,220–225. However, transplantation into the peritoneal cavity may aggravate hypoxia and inhibit the insulin-secretory response 226 . Furthermore, macrophages and lymphocytes in the peritoneum may be involved in the rapid degradation of the capsule223,227,228.
By contrast, the subcapsular kidney space (only suitable for microcapsules) and subcutaneous tissue (suitable for different encapsulation devices) have been associated with a weaker cellular response, better islet viability, and fewer broken capsules21,190,228. The subcutaneous space with prevascularization or cotransplantation of porcine islets and mesenchymal stem cells (MSCs) was reported to be associated with the promotion of neovascularization and reduced hypoxic stress198,229,230.
With the emergence of conformal coating and surface modification, the liver is being investigated as a potential site132,209,210,218. Other sites for microencapsulated or conformal-coated islet transplantation, such as muscle and bone marrow, have the advantage of good vascularization and relatively easy access164,231,232, but further studies are required.
Materials
The emergence of novel biocompatible encapsulation materials may promote progress in encapsulated islet transplantation. Such a membrane should be biocompatible and nondegradable and should allow the passage of insulin and glucose while preventing that of antibodies, lymphocytes, and toxic cytokines/chemokines. Materials such as alginate have resulted in various successful clinical applications and biocompatibility studies. A recent report indicated that a silicon nanopore membrane, designed with 7-nm-wide slit pores, protected encapsulated islets from cytokines, retained islet viability over 6 h, and islets remained responsive to changes in glucose levels 233 .
However, our opinion is that it will be difficult to develop a system that allows nutrients and oxygen to reach islets sufficiently and prevents islets from being damaged by antibodies, complement, and/or cytokines and chemokines.
Combined Islet and Mesenchymal Stem Cell And/Or Sertoli Cell Transplantation
Mesenchymal Stem Cells (MSCs)
MSCs are known to have regenerative, anti-inflammatory, and immunodulatory effects. There are extensive indicators that MSCs function satisfactorily across species 234 . MSCs might have considerable therapeutic potential in islet xenotransplantation. The cotransplantation of MSCs with adult porcine islets significantly improved islet vascularization and oxygenation 198 .
Porcine MSCs (pMSCs) express surface markers of MSCs but very low levels of swine leukocyte antigens (SLAs) and costimulatory molecules. pMSCs downregulate the human T-cell response to pig antigens as efficiently as do human MSCs235,236. They also have the ability to differentiate and help rebuild the vascular system after islet xenotransplantation237,238. In summary, pig or human MSCs have considerable potential in xenotransplantation. The ability to obtain pMSCs in very large numbers from adult pigs (from the adipose tissue or bone marrow) may prove a significant advantage over human MSCs.
Sertoli Cells (SCs)
Testicular SCs can secrete immunosuppressive factors, such as transforming growth factor-β1 (TGF-β1), which can inhibit lymphocyte proliferation239,240, prolong survival of transplanted islets 241 , promote β-cell replication242,243, and accelerate functional maturation and differentiation of neonatal porcine islets 244 . SCs exert a global immunosuppressive effect that extends across species barriers 245 . Cotransplantation of islets with SCs prolonged fish, rat, and porcine islet survival in mice246–248. Moreover, some islet survival might have been achieved following the cotransplantation of SCs with neonatal porcine islets in humans, though this study was not conclusive 249 .
In summary, SCs can prolong the survival time of islets during in vitro culture and promote vascularization of islets. The intravenous infusion of SCs can inhibit rejection of islet transplants. The combined transplantation of islets and SCs might attenuate both short-term and long-term loss of islet grafts 250 .
Induction of Immunological Tolerance
The induction of tolerance, if it could be achieved, would be particularly important after islet transplantation, as many young patients with T1D may require exogenous immunosuppressive drug therapy for decades if they are to remain normoglycemic. Tolerance induction could possibly have a dual protective effect, with the potential for inducing both tolerance to the islet graft and restoration of self-tolerance to prevent recurrence of autoimmunity251–254. Attempts have been made to induce a stable hematopoietic cell chimerism by the infusion of bone marrow to prolong islet graft survival252,255–257.
Previous Clinical Trials of Islet Xenotransplantation (Table 6)
Groth et al. first transplanted FICCs into the kidney subcapsular space of T1D patients in 1994 258 . NICCs were cotransplanted with SCs in a stainless steel chamber under the skin of patients in a study in Mexico; results showed some decrease in insulin requirement103,249. Some function of the transplanted cells, a low frequency of chronic complications, and no evidence of PERV activation were also reported by the same group in long-term follow-up of 23 patients with T1D after NICC transplantation in a device without exogenous immunosuppressive therapy104,105. The patient with long-term survival (>9.5 years) of microencapsulated FICCs into the peritoneal cavity, but without insulin independence, has been mentioned above 191 . In P.R. China, Wang et al. reported that NICCs were transplanted into the hepatic artery in 22 T1D patients who received clinically relevant immunosuppressive therapy; they provided evidence that some NICCs survived in 20 patients 259 .
Experience With Clinical Porcine Islet Transplantation *
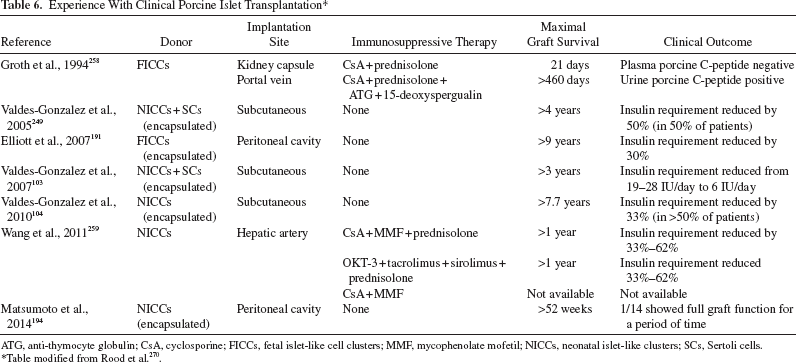
ATG, anti-thymocyte globulin; CsA, cyclosporine; FICCs, fetal islet-like cell clusters; MMF, mycophenolate mofetil; NICCs, neonatal islet-like clusters; SCs, Sertoli cells.
Table modified from Rood et al. 270 .
Studies have been undertaken by Living Cell Technologies, a company that has carried out phase I/II clinical trials in Russia, Argentina, and New Zealand under approval from the local government health authorities. NICCs encapsulated with alginate and ornithine were transplanted into various groups of diabetic patients in each of those countries, but reports of the results have been less than comprehensive.
At this time, there have been no clinical islet xenotransplantation trials in which the protocols have been proven to be effective. This underpins the urgent need for preclinical studies in NHPs to prove the effectiveness and safety of the porcine islets and the treatment protocols.
Criteria for Future Clinical Trials
Suggested criteria to be fulfilled in clinical trials of islet xenotransplantation have been published by the International Xenotransplantation Association260,261. The World Health Organization (WHO) urged its members to embark on clinical trials only when the national health authority, in the country where the trial takes place, establishes effective national regulatory control and surveillance mechanisms. Subsequently, the WHO convened a WHO Global Consultation on Regulatory Requirements for Xenotransplantation Clinical Trials in Changsha, P.R. China, in 2008, and again in Geneva, Switzerland, in 2011 262 .
It was suggested that the pigs should be DPF and PERV-C negative. The porcine islet products should be isolated under current good manufacturing practice (cGMP) conditions using standard operating procedures (SOPs) with strict quality control263–265. Successful reversal of diabetes in four of six (or five of eight) consecutive NHPs with a minimum follow-up of 6 months was considered to be sufficient to indicate potential success of a clinical trial 266 . Prior analyses of microorganisms, recipient monitoring, and a response plan for preventing disease transmission needed to be well organized 267 .
Patient selection for a pilot clinical trial should be restricted to those with T1D or T2D complicated by impaired awareness of hypoglycemia and/or end-stage renal failure 268 . Informed consent should be obtained after informing the patients of the benefit–risk determination and postprotocol subject responsibilities 261 . The absence to date of reported in vivo transmission of PERV provides some confidence that well-planned pilot clinical trials could be safely undertaken.
Perspectives and Conclusions
The availability of organs and cells from deceased humans for transplantation does not meet the demand. Even though several obstacles remain before clinical islet xenotransplantation can become a therapeutic reality, significant progress has been made in the development of genetically engineered pigs, effective immunosuppressive regimens, immunoisolation techniques, and the establishment of guidelines for the conduct of clinical trials. Diabetic monkeys receiving exogenous immunosuppressive therapy in the form of T-cell costimulation blockade have remained normoglycemic and insulin independent after transplantation with porcine islets for >1 year. Novel genetically engineered pigs including those with manipulations to increase insulin production, the identification of sites for islet implantation where loss from IBMIR is reduced, and the cotransplantation of MSCs and/or SCs may advance the field further. Porcine islet xenotransplantation has been demonstrated to be a potentially successful strategy to achieve normoglycemia and prevent some of the complications of diabetes and may open a new avenue for the treatment of T1D.
Footnotes
Acknowledgments
Some of the authors of this work were supported in part by grants from Sanming Projects of Medicine in Shenzhen, Fund for High Level Medical Discipline Construction of Shenzhen (2016031638), Shenzhen Foundation of Science and Technology (Grant No. JCYJ20160229204849975), the Project of Shenzhen Engineering Center (GCZX2015043017281705), Natural Science Foundation for Distinguished Young Scholars of Guangdong Province (2016A030306051), China Post doctoral Science Foundation (2015M580755), China Postdoctoral Science Special Foundation (2016T90813), Clinical Doctor-Basic Scientist Combination Foundation of Shenzhen Second People's Hospital, and Key Laboratory Project of Shenzhen Second People's Hospital. The authors declare no conflicts of interest.
