Abstract
Spinal cord injury (SCI) is a debilitating clinical condition, characterized by a complex of neurological dysfunctions. Neural stem cells from the subventricular zone of the forebrain have been considered a potential tool for cell replacement therapies. We recently isolated a subclass of neural progenitors from the cadaver of mouse donors. These cells, named postmortem neural precursor cells (PM-NPCs), express both erythropoietin (EPO) and its receptor. Their EPO-dependent differentiation abilities produce a significantly higher percentage of neurons than regular NSCs. The cholinergic yield is also higher. The aim of the present study was to evaluate the potential repair properties of PM-NPCs in a mouse model of traumatic SCI. Labeled PM-NPCs were administered intravenously; then the functional recovery and the fate of transplanted cells were studied. Animals transplanted with PM-NPCs showed a remarkable improved recovery of hindlimb function that was evaluated up to 90 days after lesion. This was accompanied by reduced myelin loss, counteraction of the invasion of the lesion site by the inflammatory cells, and an attenuation of secondary degeneration. PM-NPCs migrate mostly at the injury site, where they survive at a significantly higher extent than classical NSCs. These cells accumulate at the edges of the lesion, where a reach neuropile is formed by MAP2- and β-tubulin III-positive transplanted cells that are also mostly labeled by anti-ChAT antibodies.
Keywords
Introduction
Spinal cord injury (SCI) is most commonly caused by high-energy trauma, and the majority of patients are 10-10 years old. SCI results in enormous personal suffering and substantial costs to society (10, 33). There is currently no curative therapy, and the care in the acute phase is often limited to high-dose corticosteroid treatment, and surgical stabilization and decompression to possibly attenuate further damage (10, 11). In addition to axonal transection, the traumatic injury and the activation of secondary mechanisms of degeneration cause demyelination and death of multiple cell types (12, 32). The degree of recovery of function can be correlated to the extent of spared white matter at the injury site (43). A large number of studies have evaluated the effects of transplanting a variety of stem cells or stem cell-derived cells in SCI models, and remarkably, many studies using different strategies have indicated beneficial effects to a certain degree (30). Transplanted cells can improve the recovery of function either by replacing partially the lost cells or supplying a favorable environment that attenuates the effects of secondary degeneration. Therefore, they could enhance the amount of spared tissue at the injury site (i.e., transplanted stem cells can survive in such an unfavorable environment). We had previously reported that intravenously (IV) administered adult neural stem cells (NSCs), isolated from the subventricular zone (SVZ), accumulate at the site of injury and improve the early rate of hindlimb functional recovery. Their liability, however, lasts just about 12-20 days; then NSCs are phagocytized by macrophages, and the recovery process does not progress further (4). A similar fate had also been observed with embryonic stem cells (ESCs), although their anti-inflammatory action, accompanied by a good degree of myelin preservation, promoted an early improvement of hindlimb functional recovery (5). In view of such results and aiming at the isolation of adult NSCs capable of surviving in a highly unfavorable environment, we isolated adult NSCs from the SVZ several hours after death of the mouse donor (23). This procedure provides a population of NSCs, named postmortem neural precursor cells (PM-NPCs), which differentiate preferentially into neurons in vitro with a significant percentage of cells of cholinergic nature. Such a process is dependent on the autocrine erythropoietin (EPO) release and is prevented by exposure to EPO and EPOR antibodies (23). Here we report that IV-administered mouse PM-NPCs migrate to the spinal cord lesion site, differentiate mostly into cholinergic neuron-like cells, and form a reach neuropile. These differentiation events are preceded by the counteraction of secondary degeneration that is accompanied by the promotion of a stable recovery of hindlimb function.
Materials and Methods
Animal Care
For this study we used 71 adult CD1 male mice 25-30 g in weight (Charles River, Calco, Italy). All of the procedures were approved by the Review Committee of the University of Milan and met the Italian Guidelines for Laboratory Animals, which conform to the European Communities Directive of November 1986 (86/609/EEC). The animals were kept for at least 3 days before the experiments in standard conditions (22 ± 2°C, 65% humidity, and artificial light between 8:00 a.m. and 8:00 p.m.).
Postmortem-Neural Precursor Cell Derivation, Differentiation, and Labeling
PM-NPCs were obtained from 6-week-old CD-1 albino mice; their isolation, growth, and characterization were performed following methods described by Gritti et al. (17). Briefly, cells were isolated from the SVZ 6 h after sacrifice by cervical dislocation. Brains were removed, and tissues containing the SVZ region were dissected, transferred to Earl's balanced salt solution (Life Technologies, Monza, Italy) containing 1 mg/ml papain (27 U/mg; Sigma-Aldrich, Milan, Italy), 0.2 mg/ml cysteine (Sigma-Aldrich), and 0.2 mg/ml EDTA (Sigma-Aldrich). Tissue was incubated for 45 min at 37°C on a rocking platform. Tissues were then transferred to DMEM-F12 medium (Euroclone, Pero, Milan, Italy) and mechanically dissociated with a Pasteur pipette. Cells were collected by centrifugation (200 × g, 10 min, room temperature) and resuspended in DMEM-F12 medium (Euroclone) containing 2 mm l-glutamine (Euroclone), 0.6% glucose (Sigma-Aldrich), 9.6 mg/ml putrescine (Sigma-Aldrich), 6.3 ng/ ml progesterone (Sigma-Aldrich), 5.2 ng/ml sodium selenite (Sigma-Aldrich), 0.025 mg/ml insulin (Sigma-Aldrich), 0.1 mg/ml transferrin (Sigma-Aldrich), 2 μg/ml heparin (sodium salt, grade II; Sigma-Aldrich), basic fibroblast growth factor (bFGF; human recombinant, 10 ng/ml; Life Technologies), and epidermal growth factor (human recombinant, 20 ng/ml; Life Technologies) (5, 17). PM-NPC precursors were maintained in culture in the above-cited medium (5, 17). Differentiation of PM-NPCs was performed by plating the dissociated stem cells at a density of 40,000 cells/cm2 in the presence of adhesion molecules (Matrigel™; BD Biosciences, Buccinasco, MI, Italy) and bFGF (10 ng/ml) for 48 h. Cells were then exposed to the same medium without bFGF and the addition of fetal bovine serum (2% v/v; Euroclone) for the following 5 days as previously described (17, 23). Then, the extent of differentiation was determined by immunocytochemical staining (23). PM-NPCs were labeled with PKH26 (Sigma-Aldrich) just before transplantation according to the manufacturer's instructions. Briefly, cells were collected and washed thrice using HBSS (Life Technologies) in a 10-ml conical vial. After the last wash, the supernatant was aspirated, leaving in the tube no more than 25 μl of supernatant. A 2× cell suspension was prepared by adding 1 ml of Diluent C to the cell pellet followed by gentle pipetting. Immediately before staining, a 2× dye solution (4 × 10−6 M) in Diluent C was prepared by adding 4 μl of the PKH26 ethanol dye solution to 1 ml of Diluent C. The 1 ml of 2× cell suspension was added to 1 ml of 2× dye solution and incubated for 3 min at room temperature (final cell density was 1.2 × 107 cells/ml and 2 × 10−6 M PKH26). The staining was stopped by adding an equal volume (2 ml) of 1% bovine serum albumin (Sigma-Aldrich) solution in HBSS and incubated for 1 min. Cells were washed twice with HBSS to ensure removal of unbound dye. After the last centrifugation, the cell pellet was resuspended in a sterile saline solution. The final cell concentration was 3.3 × 105 cells/50 μl (15, 20, 41). PKH26 is an nontoxic cell dye characterized by long aliphatic tails (PKH26) that allow the dye incorporation in lipid regions of the cell membrane (20, 41).
Green Fluorescent Protein (GFP) PM-NPCs
PM-NPCs were obtained from 2-month-old C57 black male mice carrying the transgene for GFP under the control of ubiquitin promoter C [C57BL/6-Tg(UBC-GFP)30Scha/J; The Jackson Laboratories, Bar Harbor, ME, USA]. These precursors were characterized in vitro for neurosphere formation and differentiation capabilities, which resulted comparable to those of the PM-NPCs obtained from albino CD1 mice (5, 17, 23).
Dead Cell Administration
PM-NPCs (1 × 106 cells) were treated with 1 ml of 4% paraformaldehyde (Sigma-Aldrich) for 10 min at room temperature, 10 ml of PBS was added, cells were spun down at 123 × g, and suspended in PBS. This washing step was performed twice, and finally, the ready-to-use dead PM-NPCs were suspended at the final concentration of 3.3 × 105 cells/50 μl in saline solution.
Cell Quantification at the Lesion Site
Cells were considered PKH26 positive when fluorescence was visible by confocal microscope (Leica TSC2; Leica Microsystems, Heidelberg, Germany) and if the spots had an emission wave length of 567 nm, which corresponds to the emission of PKH26. We used sections obtained from animals transplanted with unlabeled PM-NPCs as a control. The counting of the cells was performed by assessing the PKH26-positive cells in the transversal sections in a region 4 mm from the center of the lesion site. The lesion epicenter was defined in cross-sections as the region with the minimum tissue sparing. The PKH26-positive cells present in a group of three consecutive sections (10 μm thick) were averaged, and the count was repeated every 400 μm. The total number of labeled cells was obtained integrating the curve obtained calculating the average of each section across a 4-mm span from the epicenter of the lesion (8).
Spinal Cord Injury, Experimental Groups, and Cell Administration
The traumatic SCI was performed using a commercially available Infinite Horizon (Precision Systems and Instrumentation, LLC, Lexington, KY, USA) SCI device (18, 31) at the T8 level. Surgery on the animals was performed with minor modifications of the method described elsewhere (18). Briefly, animals were anesthetized with 2.5% isoflurane in oxygen (1 L/min; Farmagricola, San Donato, Milan, Italy) for 5 min before surgery. A dorsal vertical incision was made through the skin from T7 to T12. The superficial fat pad was removed, and T7 and T10 bilateral paravertebral muscles were cut. Laminectomy was performed, and the spinal cord was exposed. The impactor tip was then positioned just above the cord following the manufacturer's instructions. A force of 70 Kdyne was applied for 1 s to the spinal cord. After the contusion, the muscles were sutured, and the skin closed by means of clips (2Biological Instruments, Besozzo, Varese, Italy). Experimental animals were divided into four groups: 1) laminectomy mice (n = 11), 2) lesioned mice treated by IV route with phosphate-buffered saline (PBS, n=22), 3) lesioned mice transplanted by IV route with PM-NPCs (n = 22), and 4) lesioned mice transplanted by IV route with dead PM-NPCs (n = 12). IV administration was performed by injections in the tail vein. PM-NPCs, PBS, or dead PM-NPCs were administered after spinal cord lesion. The first treatment was a slow IV injection of 50 μl in the tail vein performed within 30 min after injury, followed by a second injection 6 h later, and a third one 18 h after the lesion. Each cellular administration consisted of 3.3 × 105 cells in PBS for a total of 1 × 106 cells. The choice of a time limit of 18 h after SCI for administering PM-NPCs was determined by the optimal permeability of the blood-brain barrier at this time (42). PM-NPCs between the fifth and the ninth passage in culture were used for these experiments. The cultures were tested for proliferation and differentiation ability before being transplanted (23).
Behavioral Tests and Hindlimb Function
All outcome measures were assessed in a blinded fashion. Neurological function was evaluated first 24 h after injury and then twice a week for the first 4 weeks. The methods utilized are well known in the field of behavioral evaluation of recovery of function after SCI. Locomotor function and hindlimb recovery after contusion were evaluated with the open field test according to the Basso mouse rating scale (3). For behavioral experiments we used five animals in the laminectomy group and at least 12 animals for the other groups. Allodynia-like responses in the unaffected forepaw were assessed by means of standard hotplate test and cold stimulation. For hotplate testing, mice were placed on a hotplate, and the latency to licking was measured. Nonresponders were removed after 60 s. The response to cold was tested by the application of ethyl chloride spray (Gebauer Company, Cleveland, OH, USA) to the palm surface. The response was rated 1 (no response), 2 (brief withdrawal with licking), and 3 (vocalization, withdrawal with licking, and aversion) (19).
Fluorescence Imaging
An IVIS Lumina (Caliper LifeSciences Perkin Elmer, Monza, Italy) CCD camera was used to assess the fluorescent signal in organs collected from lesioned mice intravenously injected with PKH26-labeled cells and control mice injected with saline. Briefly, 48 h after injection, three animals per group were sacrificed, and organs (spinal cord, bowel, kidney, liver, spleen, and lungs) were collected. Fluorescence imaging was performed by using the DsRed and DsRed background excitation and emission passband filters (DsRed: excitation 500-550 nm, emission 575-650 nm; DsRed-background: excitation: 460-490 nm, emission: 575-650 nm). Image analyses were performed with Living Image® software (Caliper LifeSciences; Perkin Elmer) for background subtraction.
In Vivo MRI
MRI was performed using a 7T MR scanner (Bruker, Milan, Italy). Anesthetized animals (isofluorane gas anesthesia; Farmagricola) were positioned supine on the animal bed and inserted in the radiofrequency coil (diameter=38 mm) inside the magnet. Scout transverse images were acquired for correct positioning of the region of interest. Different MR sequences were tried in order to optimize spinal cord visualization and signal contrast:
Spin echo (MSME) sequence, (matrix 256 × 128; TR/ TE: 1,200/12 ms; two averages; acquisition time: 5′7″; fat suppression used)
Gradient echo (FLASH) sequence (matrix 256 × 128; TR/TE: 1,200/10 ms; two averages; acquisition time: 5′7″); not shown
Fast-spin echo (RARE) sequence (matrix 256 × 128; TR/TE: 2,000/56 ms; four averages; acquisition time: 4′16″).
All animals (n=4 for each group) were acquired in transverse (five slices of 0.7 mm thickness; FOV 4 × 4 and spatial resolution of 156 μm) and axial orientations (12 slices of 0.7 mm thickness; FOV 3 × 3 and spatial resolution of 117 μm) 10 months after lesion.
Tissue Collection and Processing, Histology, and Immunohistochemistry
At the end of the experimental period, animals were anesthetized by IP injection of cloralium hydrate (Sigma-Aldrich) 4% in distilled water and perfused with 4% paraformaldehyde in phosphate buffer 0.1 M pH 7.4 by transcardial perfusion. Spinal cords were postfixed overnight in the same fixative, cryoprotected with 30% sucrose (Sigma-Aldrich), quickly frozen, stored at −80°C, and sectioned by means of a cryostat (Leica). Every 20th section was stained with thionin (Sigma-Aldrich). Cross-sections containing the lesion epicenter and the complete T8 segment cavitations were analyzed by computerassisted image analysis. The percent of lesion was calculated as the area of the injured tissue divided by the area of the total cross-section at the level of the injury. Coronal sections (15 μm) were also collected onto glass slides and processed for immunocytochemistry. Sections were rinsed with PBS (Euroclone), treated with blocking solution (Life-Technologies), and incubated with primary antibodies overnight at 4°C. After treatment with primary antibodies, the sections were washed with PBS and incubated with appropriate secondary antibodies (Alexa Fluor® 546 or/and Alexa Fluor® 488, Molecular Probes®, Life Technologies) for 2 h at room temperature. Sections were washed in PBS, nuclei were stained with DAPI (1 μg/ml final concentration, 10 min at room temperature; Sigma-Aldrich), mounted using the FluorSave Reagent (Calbiochem, Merck Chemical, Darmstadt, Germany), and analyzed by confocal microscopy. In control determinations, primary antibodies were omitted and replaced with equivalent concentrations of unrelated IgG of the same subclass. The following primary antibodies were used: microtubule-associated protein 2 (MAP2; 1:300; Millipore, Vimodrone, Milan, Italy), glial fibrillary acidic protein (GFAP; 1:400; Covance, Rome, Italy), β-tubulin III (1:150; Covance), choline acetyltransferase (ChAT; 1:1,000; Millipore), and immunological response (ED1) rat anti-mouse macrophages/monocytes (1:25; Millipore). For immunofluorescence, the following secondary antibodies were used: 488 goat anti-mouse IgG (1:200; Alexa), 488 donkey anti-rabbit IgG (1:200; Alexa), 546 goat anti-rabbit IgG (1:200; Alexa), and 546 goat anti-chicken IgG (1:200; Alexa).
Assessment of Myelin Preservation
In order to perform a homogeneous analysis, the staining was carried out on sections of nonlesioned, lesion + saline, and lesion + cells animals placed on the same coverslip. Myelin preservation was evaluated comparing the levels of myelin in the ventral white matter at 0.4 mm (rostral and caudal) laterally from the lesion epicenter in healthy, saline, and cell-treated animals. The choice of the ventral white matter was based on the knowledge that the reticular spinal pathway descends mostly in the ipsilateral dorso- and ventrolateral funiculi and is directly involved in the regulation of the movement of the mouse foot (40). We previously reported that the quantification of the spared ventral myelin evaluated in a semithin section gave comparable results when fluoromyelin was used (FluoroMyelin Green, Molecular Probes; Invitrogen, Eugene, OR, USA) (40). The confocal microscope images for the laminectomy, saline, and cell-treated mice were obtained using the same intensity, pinhole, wavelength, and thickness of the acquisition. As reference, we used sections close to the ones analyzed and not treated with fluoromyelin. Briefly, the procedure of the staining was carried out by incubating the cryosections with fluoromyelin diluted 1:300 in PBS for 20 min; slides were then washed three times for 10 min each with PBS and mounted with FluorSave (Merck, Darmstadt, Germany), and qualitatively and quantitatively analyzed by confocal microscopy (Leica TSC2; Leica Microsystems).
Estimate of the Macrophage Number at the Site of the Lesion
ED-1-positive cells were counted in transversal sections made at the lesion epicenter (1 mm extension) and 0.4 mm rostral. As a negative reference, we used a consecutive section that was stained by omitting primary antibody anti-rat macrophages/monocytes and replacing it with equivalent concentrations of unrelated IgG of the same subclass. The zero level was adjusted on this reference and used for all the further analysis (we used a new zero reference for each new staining). The ED-1-positive cells present in a group of three consecutive sections (10 μm thick) were averaged, and we repeated this count each 100 μm. The total number of ED-1-positive cells was obtained integrating the average within volume analyzed, that is, the 1 mm around the epicenter of the lesion (8).
Estimate of Neutrophil Tissue Infiltration
Spinal cords, dissected 48 h after lesioning, were placed overnight in 4% formaldehyde, 50 mM phosphate buffer solution, pH 7.4, and then paraffin embedded. Sections of 5 μm thickness were cut through the epicenter (1 mm) and stained with Harris hematoxylin and eosin (Sigma-Aldrich, Milan, Italy). Neutrophils were identified by means of the Leder technique that employs naphthol AS-D chloroacetate (Sigma-Aldrich, Milan, Italy) dissolved in dimethyl sulfoxide (Sigma-Aldrich, Milan, Italy)/Triton X-100 (VWR, Milan, Italy) (9:1; v/v). Then pararosaniline hydrochloride (Sigma-Aldrich, Milan, Italy) dissolved in saline buffer solution was added. This was performed as described by Schlayer et al. (32). Red-stained polymor-phonucleated cells were counted in four nonconsecutive groups, randomly chosen around the lesion site of 400× histological fields. Results were expressed as number of neutrophils per histological power field (HPF).
RNA Isolation and Real-Time PCR Analysis
Mice (n=3 48 h postinjury group, n=3 1 week postinjury group) were anesthetized (5, 12, 13) and sacrificed by decapitation. Laminectomy was performed at the T5-T12 level. The spinal cord region corresponding to the lesion site was removed (we took 4 mm of tissue rostral and 4 mm of tissue caudal to the lesion epicenter). The tissue was put in 1 ml of Trizol® Reagent (Invitrogen), shock frozen, and kept at −80°C until performing the RNA isolation. Total RNA was isolated by using Trizol® Reagent (Invitrogen) in accordance with the manufacturer's instructions. The genomic DNA was removed by DNase I treatments (2 U/μg of RNA) (Ambion, Austin, TX, USA). The synthesis of single-strand cDNA was carried out on 1 μg of RNA, using U M-MLV Reverse Transcriptase III (Invitrogen) following the manufacturer's instructions.
Real-time (RT)-PCR was performed in an MJ Opticon 2 (Biorad, Segrate, Milan, Italy) using Brilliant SYBR Green qPCR Master Mix (Stratagene, La Jolla, CA, USA) following the manufacturer's instructions. The housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used for normalization of cytokine expression. The relative expression of cytokine genes, with GAPDH as reference gene, was determined using the 2-ΔΔCt method. We used this method because both target and reference genes were amplified with similar efficiencies near 100%. The primer design was performed using the DNASTAR Lasergene program. Primers used are the following: GAPDH (F: cgacttcaacagcaactcccactcttcc; R: gctccctaggcccctcctgttattatg), brain-derived neurotrophic factor (BDNF) (F: cattaccttcctgcatctgttgg; R: cgtggacgtttact tctttcatgg), interleukin-6 (IL-6) (F: gacaaccacggccttccctac; R: cgttgttcatacaatcagaattgcc), nerve growth factor (NGF) (F: tgggcccaataaaggttttgcc; R: tgggcttcagggacagagtctcc), tumor necrosis factor-α (TNF-α) (F: tctatggcccagaccctcacac; R: cagccactccagctgctcctc), macrophage inflammatory protein 2 (MIP-2) (F: acgcccccaggaccccactg; R: ggacagcag cccaggctcctcc), leukemia inhibitory factor (LIF) (F: aacgt ggaaaagctatgtgcg; R: gcgaccatccgatacagctc).
Statistical Analysis
Data were expressed as the mean ± SEM. Multiple group comparisons were made by ANOVA with Tukey's post hoc test. The analyses were performed using Prism 3.0 software (GraphPad Software, Inc., La Jolla, CA, USA). Statistical significance was accepted for a p < 0.05.
Results
PM-NPCs Improve Recovery of Hindlimb Function
The 70-Kdyne traumatic impact to the spinal cord caused a transient loss of ability in hindlimb function of saline-treated control mice, which was followed by a progressive gradual recovery reaching the maximum in 2-3 weeks (3.20 ± 0.08 points of the BMS scale; n = 12) (Fig. 1). This corresponds to plantar placing of the paw with or without weight support or occasional, frequent, or consistent dorsal stepping, but not plantar stepping (3). The recovery was earlier and reached a higher recovery extent up to 5.14 ± 0.06 at day 28 (n = 12, corresponding to frequent or consistent plantar stepping without coordination or frequent or consistent plantar stepping with some coordination) when injured mice were treated with adult PM-NPCs (see Materials and Methods). The behavioral improvement was particularly evident between days 7 and 14 after SCI; then it kept steadily improving thereafter (up to 90 days; n=6). The application of killed PM-NPCs failed to improve recovery of hindlimb function, and the rate of recovery was comparable to that of the saline group (Fig. 1). No signs of allodynia-like forelimb hypersensibility (19) were recorded at any time in any experimental group throughout the observational period of 90 days.

PM-NPCs improve recovery of hindlimb function. The open field locomotion was the test employed for the determination of motor function recovery. The score was determined according to the Basso Mouse Scale (BMS) (3). The experimental animals, tested the day prior to the injury, scored the maximum (nine points) in the BMS scale; then the BMS score fell to 0 following SCI and gradually recovered thereafter. The recovery of hindlimb function of lesioned mice showed a remarkable and long-lasting improvement when animals were treated with live PM-NPCs. The groups were randomized, and the analysis was performed in double-blind fashion. Values represent mean ± SEM. We determined the statistical differences by means of ANOVA test followed by Tukey's posttest. ***p < 0.001; **p < 0.01 versus saline; ○○○p < 0.001; ○○p < 0.01 versus killed PM-NPCs.
PM-NPC-Mediated Tissue Sparing
Spinal tissue preservation in the animals treated with PM-NPCs was strongly suggested by the in vivo NMR analysis performed in saline and cell-treated animals 10 months after lesioning (Fig. 2). The T2-weighted RARE images show that in saline-treated mice the spinal cord matter become very thin at the injury site surrounded by a very high hyperintense signal probably due to spinal fluid filling the empty space left by the reduced size of the surviving cord. Spinal cord shrinkage at the lesion site and fluid accumulation is a constant finding in all chronically lesioned untreated animals (Fig. 2). It was observed that the protective effect of PM-NPCs was maintained chronically, and the spinal cord matter was rather well preserved at site of injury even 10 months later. The hyperintensive signal was far less intense than in saline-treated animals (Fig. 2). The reduced tissue loss was likely due to tissue sparing. An important pathway in eliciting locomotion is the reticulospinal tract that descends mainly in the ipsilateral dorso- and ventrolateral funiculi. By electrophysiological studies, its function in the coordination of rhythmic stepping movements was shown (2, 25). In this region, myelin preservation was studied by means of FluoroMyelin™ staining followed by confocal quantitative analysis (39, 40) (details in Materials and Methods). Myelin preservation was quantified 28 days after lesioning, and sections were taken at the center of the lesion and 0.5 and 1.9 mm caudal to the lesion site. The quantitative assessment was performed on spared ventral and lateral white matter, and it is shown in Figure 3. Only intact myelin was assessed, and the rubble of degenerated myelin was obviously not considered. The traumatic lesion caused almost 50% loss of myelin in close proximity of the lesion and about 70% more caudally. The application of PM-NPCs reduced such a loss in a remarkable and highly significant manner in all three points of evaluation (Fig. 3).
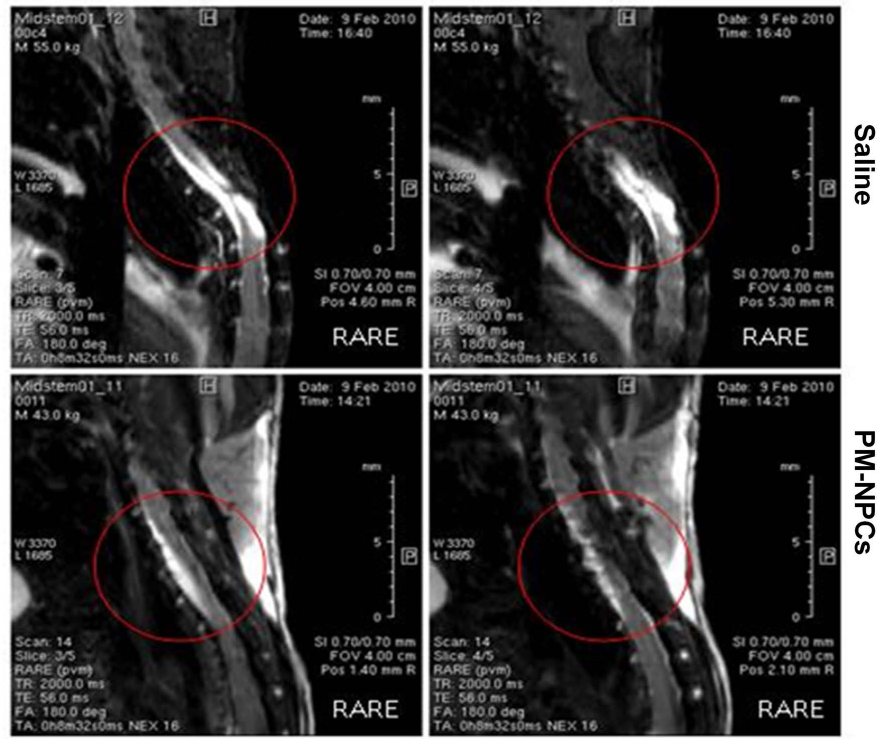
In vivo MRI 10 months after SCI. RARE sequences obtained at 10 months after injury and cell transplantation by means of a 7-tesla NMR (Bruker). A significantly better parenchymal preservation in PM-NPC-treated animals is shown, while there is only a thin residual tract in saline-treated mice that is accompanied by fluid accumulation. The images are representative for two mice, but similar images were obtained for at least four animals per group.

Sparing of myelin in the injured cord. The images and the relative quantification show the protective action of PM-NPCs on the myelin tracts in the injured cord. After animal perfusion, spinal cords were dissected, postfixed, and coronally sectioned by means of a cryostat. Myelin was quantified by means of Fluoromyelin™ staining (green) performed in sections at the lesion epicenter and 0.5 mm and 1.9 mm caudal to the lesion site through quantitative confocal analysis of ventral and lateral white matter as indicated in the graphs. The confocal microscope images for the saline and PM-NPC-treated mice were obtained using the same intensity, pinhole, wavelength, and thickness of the acquisition. As reference, we used sections close to the ones analyzed and not treated with fluoromyelin. For the quantification we considered sections from at least three animals per group. Values represent the mean ± SEM. We determined the statistical differences by means of ANOVA test followed by Tukey's posttest. ○○○p < 0.001, ○○p < 0.05, ○p < 0.01 versus PBS; ***p < 0.001, **p < 0.05, *p < 0.01 versus LAM.
PM-NPC Homing to Site of Injury, Survival, and Differentiation
After 48 h from IV injection of PKH26-labeled PM-NPCs, mice were sacrificed, and CCD camera analysis was performed in the following organs: spinal cord, bowel, kidney, spleen, lungs, and liver. Figure 4 shows a specific distribution of PM-NPCs at the lesion site in the spinal cord bearing the highest detected fluorescence peak. A smaller degree of positivity was observed in the spleen (Fig. 4A). The comparison of spinal cords from PM-NPC- and saline-treated animals showed a higher and specific signal of fluorescence relative to the presence of transplanted cells in the former and the absence of specific signal in the latter (Fig. 4B). We also serially sectioned seven cords of treated mice at 4 weeks after injury and detected PKH26- or GFP-positive PM-NPCs by means of confocal microscopy at the edge of the lesion. The total number of positive cells was 260,000 ± 45,000 per cord. This was 100-fold higher than surviving ESCs as reported previously and 10-fold higher than adult NSCs (4, 5).

Organ distribution of IV-administered PM-NPCs. PKH26-labeled PM-NPC distribution 48 h after injection was investigated by means of CCD camera. Animals were sacrificed and spinal cords and organs extracted to investigate organ distribution of PM-NPCs. (A) The picture shows the high accumulation of the labeled cells at the lesion site and a lower signal in the spleen. (B) Comparison of spinal cord fluorescence of PM-NPC-injected mice and placebo-treated animals showed a high and specific fluorescent signal relative to the presence of labeled transplanted cells only in the animals treated with PM-NPCs. Pictures were acquired from one PM-NPC treated mouse; however, we observed very similar results in at least three other animals.
Figure 5 shows that the density of postinjury reactive GFAP-positive cellular profiles was markedly reduced in the proximity of the PM-NPC clusters. The quantification confirmed the visual impression that GFAP-positive cells were significantly reduced around the areas of PM-NPC accumulation compared to other parts of the cord located where the GFAP-positive cells were more numerous (Fig. 5). None of the transplanted PM-NPCs differentiated into GFAP-positive cells. This result was quite different from in vitro differentiation, where about 40-50% of the cells became GFAP-positive cells. Also, no transplanted PM-NPCs showed positivity for oligodendrocyte markers (data not shown).

GFAP immunoreactive-positive profiles 28 days after lesioning. PM-NPCs reduce the extent of gliosis in their microenvironment in the injured spinal cord. Sections were taken at 1 mm caudal to the lesion site from cords of dead PM-NPC- (A) and PM-NPC-treated (B) mice. Quantification of GFAP-positive profiles in a 100-μm radius around PKH26-labeled PM-NPC clusters (C). GFAP staining is shown in green and PKH26-labeled PM-NPCs are shown in red. Values represent mean ± SEM. ***p < 0.001 versus saline.
Most engrafted PM-NPCs accumulated at the edges of the lesion (Figs. 6-8), where they formed clusters since the early days of their administration. The clusters dispersed along the lesion edges, and transplanted cells differentiated, assuming gradually the asymmetric cellular conformation of neurons. At 1 week after lesion and transplantation, small PM-NPCs were grouped along the irregular margins of the lesion, and almost 50% of the cells were already positive for MAP2 and ChAT (Figs. 6, 8). With time, the cell bodies of PM-NPCS increased in size, and most cells extended dendritic-like processes that were immunostained by specific antibodies to MAP2 and β-tubulin III. This was particularly evident at days 28 and 60 after lesioning (Figs. 6, 7). Immunohistochemical staining for ChAT revealed that most transplanted PM-NPCs were positive for ChAT and showed a marked increase in the formation of dendritic-like processes over time (Fig. 8). Such a differentiation into cholinergic neural cells in vivo confirmed the in vitro observations (23). The achievement of morphological complexity and the positivity to ChAT, MAP2, and β-tubulin III by transplanted PM-NPCs was not likely due to fusion with surviving host spinal cord neurons (Figs. 6-8). This is evident from their clearly different morphology and the absence of two nuclei in any single labeled cell. The quantification of such differentiation is shown in Table 1.

PM-NPCs MAP2 transdifferentiation in lesioned cords. GFP-labeled PM-NPCs (A) are clustered at the edges of lesion site at 7 days after their IV supplementation to the injured mice, some of them have differentiated into MAP2-positive cells (scale bar: 100 μm). A magnified detail is represented on the right site of the figure (scale bar: 50 μm). (B) PKH-labeled PM-NPCs (red) are present throughout the edges of lesion site at 90 days after their IV administration to SCI mice. Most of them differentiated into MAP2-positive cells (green; scale bar: 100 μm). A magnified image is represented on the right site of the figure (scale bar: 50 μm). Most PKH-labeled PM-NPCS acquired a neuronal-like shape with dendritic-like processes that intermingle with those of local spinal neurons that survived the lesion. Together they form a rich neuropil. All labeled cells have one nucleus. Nuclei are stained in blue (DAPI).

β-Tubulin III expression by PM-NPCs at the edges of the lesion. Pictures representing the edges of the lesion at 90 days after IV administration of PKH26-labeled PM-NPCs. The tissue was immunostained for β-tubulin III. Several PM-NPCs are present among the surviving neural profiles. Practically all PKH26-labeled cells were positive for β-tubulin III. We show two fields at different magnifications (scale bar: 75 or 20 μm), and an enlarged image shows clearly how each labeled cell has one nucleus and bears profiles positive for β-tubulin III.
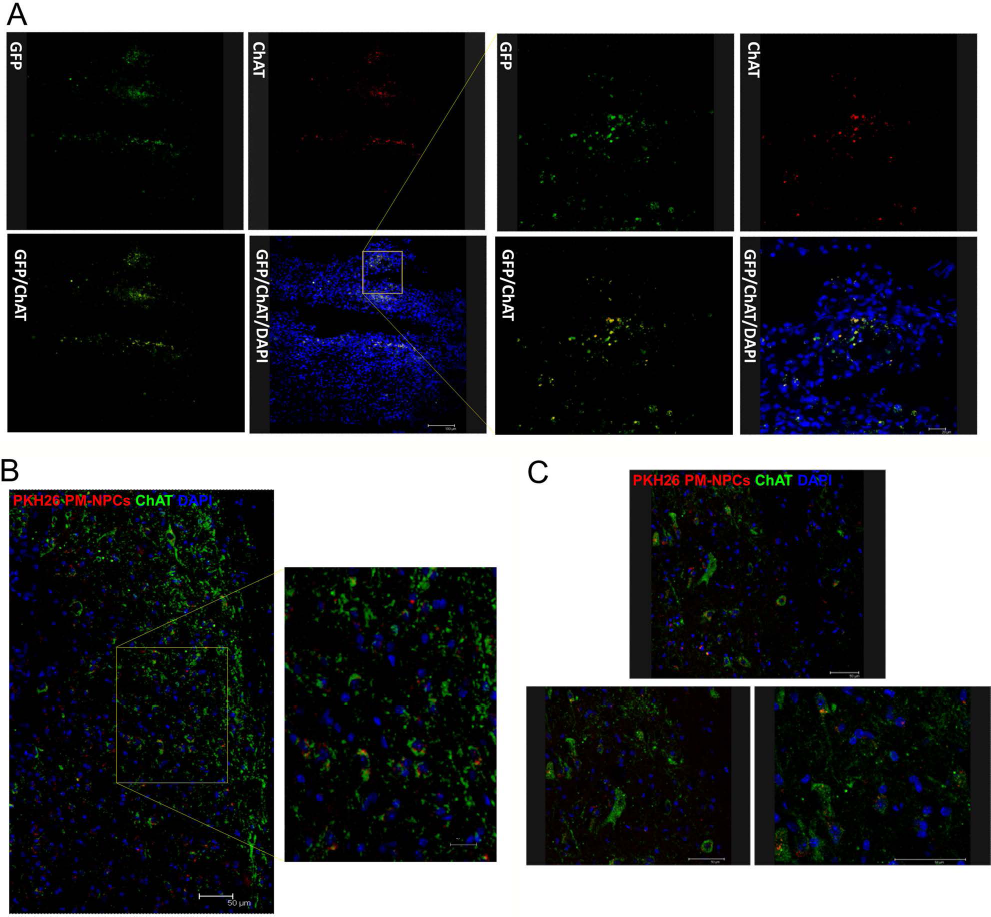
PM-NPC-transplanted cells differentiate into cholinergic neurons. (A). Some GFP-labeled PM-NPCs clustered at the edges of the lesion at 7 days after their IV administration. Several PM-NPCs were positively immunostained for ChAT (red). The pictures (scale bar: 100 μm) are enlarged in the right part of the panel (scale bar: 20 μm). (B) Several PKH26-labeled PM-NPCs (red) were accumulated at the edges of the lesion 28 days after their IV administration. Most PM-NPCs were positive for ChAT immunostaining (green) as shown clearly in the enlarged image on the right side. ChAT stain is dotted and in most cases perinuclear, although some positive processes are evident (scale bar: 75 and 20 μm). (C) PKH26-labeled PM-NPCs clustered at the edge of the lesion 60 days after their administration. All PM-NPCs were positive for ChAT immunostaining as clearly shown by the two consecutive enlarged images (scale bar: 50 μm). The labeling is now diffused throughout the neuron-like cell bodies and in the cellular processes.
Percentage of Transplanted PM-NPCs Positive for Neuronal Markers 60 Days After Intravenous Administration

For the quantification we considered at least four fields per marker. Values represent average ± SEM.
PM-NPC-Mediated Chemokine and Growth Factor Expression in the Injured Cord
The prompt action of PM-NPCs in promoting the recovery of function was suggestive of some additional effects by these cells beyond their cellular presence in the injured cord. Accordingly, we assayed the production of neurotrophic factors and inflammatory cytokines that represent the major targets of pharmacological agents aimed at repair in neurodegenerative disorders. We evaluated quantitatively the mRNA levels of six different factors (Fig. 9): BDNF, NGF, TNF-α, IL-6, MIP-2, and LIF. The comparison was made between the animals treated with saline or PM-NPCs. As additional controls, we assayed the chemokines in the T9 region of the spinal cord of both naive uninjured and laminectomized animals. As expected, all of the assayed chemokines were increased in the cord of the lesioned animals. We observed that MIP-2 expression was diminished by PM-NPCs at 48 h and 1 week after lesioning; differently, IL-6, TNF-α, NGF, and LIF were reduced at the earlier time, but significantly increased at 1 week compared to saline-treated mice. BDNF was increased by PM-NPCs in lesioned mice only at 48 h (Fig. 9).

PM-NPC-transplanted cells affect local expression of cytokines. Expression of cytokine mRNA was evaluated by real time RT-PCR. Each group was composed of six mice, and we performed three quantitative RT-PCRs in each experiment. We ran a duplicate for each sample and a parallel PCR for the gene of interest and the reference gene GAPDH. Values represent mean ± SEM. ***p < 0.001, **p < 0.01, *p < 0.05 versus control (CTRL); ○○○p < 0.001, ○○p < 0.01, ○p < 0.05 versus LAM; ###p < 0.001, ##p < 0.01, p < 0.05 versus saline.
Inflammatory Cell Migration to the Lesion Site
The administration of PM-NPCs counteracted the invasion of the lesioned spinal cord by neutrophils and macrophages. Neutrophil infiltration at the site of injury was investigated by means of Leder stain throughout the lesion site 48 h after lesioning. We chose this time interval since the number of neutrophils decreases with time as reported previously (5). In Figure 10A it is shown that in the better preserved tissue the number of neutrophils is reduced and the preventive effect PM-NPCs was quantitatively very significant. The effect of the transplanted cells was also observed when we monitored the number of ED-1-positive cells (macrophages) at the site of the lesion and rostral to the lesion (Fig. 10B) 1 week after injury. Both qualitatively and quantitatively, the counteracting action of PM-NPCs was evident and significant.
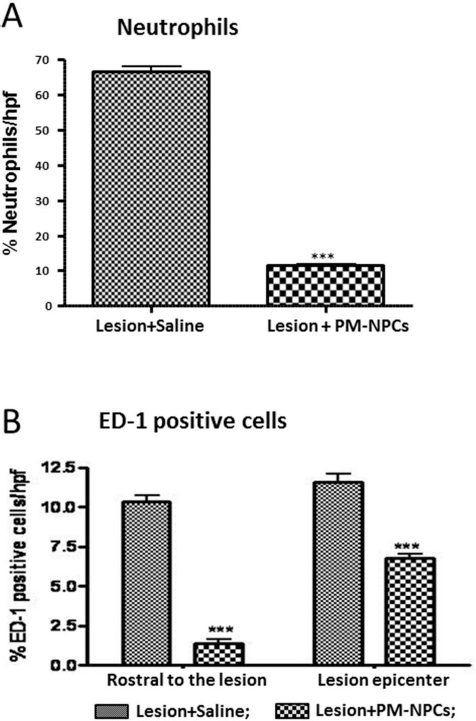
Tissue infiltration by neutrophils and macrophages. (A) Neutrophil infiltration was quantitatively assessed at lesion epicenter 48 h after lesioning by means of the naphtol AS-D chloroacetate (Sigma-Aldrich) technique for esterase. Results are expressed as number of neutrophils per HPF. The presence of transplanted PM-NPCs greatly reduced the invasion by neutrophils. (B) Coronal cryostat section of cord lesion epicenter and cord 0.4 mm rostral to the lesion were stained for ED-1, and the number of positive cells was quantitatively evaluated as described in Materials and Methods. Results are expressed as the number of ED-1-positive cells per HPF. For the quantification, we considered sections from at least six animals per group. Values represent average ± SEM. ***p < 0.001 versus saline.
Discussion
Considering their characteristic abilities to self-renew and differentiate into any cell type in the body, the therapeutic promise of stem cells justifies the intense efforts on investigating their application in cell therapy approaches (4, 5, 9, 12, 15, 24, 34, 37). Recently we reported the isolation and characterization of SVZ-derived neuronal precursors from mouse cadavers thus named PM-NPCs (23). Their differentiation ability leads to a higher proportion of β-tubulin III and MAP2-positive cells that can be as high as 45% compared to the 10–12% obtained from normal SVZ adult NSCs. These cells are also unique in that they produce EPO, and their autocrine EPO activation is responsible for their higher neural differentiation in vitro (23). In this study, it was shown that the treatment of SCI with PM-NPCs attenuates secondary degeneration and enhances significantly the amount of tissue sparing and myelin preservation, leading to a marked and long-lasting improvement in recovery of motor function. This is associated with a progressive and profound differentiation of transplanted PM-NPCs into neuronal-like cells with cellular dendritic-like processes positive for MAP2, β-tubulin III, and ChAT immunoreactivity.
Tissue Sparing, Reduced Invasion by Inflammatory Cells, and Recovery of Function
Transplanted PM-NPCs are located at the edges of the injury site and promote a faster and permanent recovery of function. This effect must be ascribed to the presence of the live transplanted PM-NPCs in the cord since the transplantation of killed PM-NPCs yields a recovery comparable to that obtained with saline. The long-lasting effects on the spinal cord were witnessed by the hindlimb recovery of function evaluated regularly up to 90 days after lesioning and also by the NMR in vivo assessment of cord integrity. This is in agreement with previous reports that indicated reduced gliosis in the spinal cord after injury when the animals were treated with EPO (12, 38, 40). A marked reduction of macrophage and neutrophil invasion of the lesion site was observed in PM-NPC-treated animals. This suggests a clear effect on the cellular components of the inflammatory reaction triggered by SCI; it may be that the reduction of the macrophage number contributes to a decrease in the retraction of injured axons through physical cell–cell interactions as recently shown (20, 21). Neutrophils and hematogenous macrophages invade the damaged spinal cord, peaking respectively at 12–24 h and 5–7 days after SCI (28, 36) and releasing proinflammatory cytokines and reactive oxygen and nitrogen species (27, 35, 36). It was shown that anti-inflammatory treatments selectively interrupted the early and destructive leukocyte-mediated actions in the injured cord, while leaving an opportunity for later regenerative interventions and wound healing responses (14, 16). The reduced cellular component of the inflammatory reaction promoted by PM-NPCs may be the basis of the observed myelin sparing.
Thus, the enhanced recovery of function might be due mainly to a major spinal cord tissue sparing after injury, as it was previously reported for agents such as EPO or reparixin (12–14). This may contribute to the creation of an environment favorable for PM-NPCs survival and differentiation.
Homing, Survival, and Differentiation of PM-NPCs
Transplanted PM-NPCs administered by tail vein injection of the injured animal are able to reach the lesion site and remnants of gray matter at its edges, where they survive and gradually differentiate in neuron-like cells. Analyses performed with both CCD camera and immunohistochemistry showed how transplanted PM-NPCs accumulate mostly in the injured region of the spinal cord with no signal perceived by the CCD camera in other organs. Previous reports had shown that NSCs transplanted into injured spinal cord may promote recovery of function, but possess a differentiation program generally restricted to glial lineages as dictated by the inflammatory–degenerative host spinal cord milieu that is not permissive for neural differentiation (1, 4, 6, 19, 33). The cellular differentiation process involving PM-NPCs in the lesioned spinal cord leads to a diminution of reactive gliosis in the areas of the cord where the transplanted cells are accumulated and to their almost total neuronal-like differentiation. This is supported by the progressive morphological changes and by the expression of neuronal markers such as β-tubulin III and MAP2, indicating that unlike other NPCs, PM-NPCs are able to differentiate in the lesion environment. We had previously reported that adult NSCs and ESCs mostly die or are phagocytized by macrophages within 10–20 days after their accumulation at the lesion site in the injured spinal cord (4, 5, 15). Thus, while in those studies we reported an acute rapid neuroprotective effect that did not lead to further improvement in the recovery at later time once the transplanted stem cells had died, here we show an early acute effect that gradually keeps improving with time. Owing to the small number of surviving transplanted cells compared to the massive neural tissue loss after SCI, the potential cell replacement suggested by the successful transdifferentiation into a MAP2 and β-tubulin III neuronal phenotype cannot be the predominant mechanism. However, the finding that almost all PM-NPCs at the injury site acquired features of cholinergic neural cells opens the possibility that the development of both proneuronal and procholinergic phenotype may be a contributing factor to the mechanisms mediating the neuroprotective effect of these cells. Neuroprotection by PM-NPCs might be mediated by complex paracrine actions in agreement with previous studies by our group that showed HIF activation in these cells. In this sense PM-NPCs produce and secrete EPO that is also the key factor for their neuronal differentiation in vitro (23). Thus, it is conceivable that PM-NPC preactivation of the HIF pathway and erythropoietin paracrine release has made these cells able to resist unfavorable conditions that take place after the SCI. Literature of the last decade has described through several different experimental paradigms how the supplementation of erythropoietin promotes neuroprotection and recovery of function in neurodegenerative conditions (26). It should be noted that PM-NPCs did not transdifferentiate into GFAP-positive cells nor into oligodendrocyte-like cells (data not reported).
Conclusions
The development of neuroprotective and restorative therapies remains a major unfulfilled medical need. In this regard, a stem cell-based therapy provides a promising therapeutic approach for preventing further neuronal damage through the attenuation of secondary degeneration and promoting repair and recovery of function after SCI. Hypothetically, cell-based therapeutics for SCI could affect tissue and/or functional outcomes in a number of ways through: (i) differentiation and functional integration of new cells into spared spinal cord circuitry (e.g., forming myelin and/or neurons); (ii) increased sparing of host neurons, myelin, and axons, and/or decreased host glial scaring; and (iii) increased host-mediated axonal regeneration or remyelination. Our study suggests that PM-NPCs could affect host hindlimb functional recovery after SCI by means of multiple mechanisms just summarized in the three points above. The ability of PM-NPCs to survive and differentiate in an unfavorable environment such as the site of SCI may derive from their postmortem origin and the consequent metabolic and genetic adaptation with activation of hypoxia-induced factor and EPO signaling (23, 29). The survival of PM-NPCS in the early 2–3 weeks postinjury is likely crucial for the creation of a healthier local milieu that favors neuronal differentiation and regeneration across the lesion of descending and ascending pathways (manuscript in preparation). Thus, our reported data show that PM-NPCs exert an early neuroprotective action not dissimilar from what was seen with NSCs and ESCs (4, 5), but due to their survival and possible EPO action, they differentiate into cholinergic neurons and, perhaps, reinforce the migration of host NSCs to the site of injury (7, 22) that is triggered by SCI (4). Thus, it is possible that the early survival of PM-NPCs is the basis for their neuronal differentiation and the creation of a more favorable environment that underlies the recovery of function.
Footnotes
Acknowledgments
We acknowledge Drs. R. Adami and L. Madaschi for the outstanding technical assistance, and Drs. A. Raspa and F. Marra for the scientific support. Authors acknowledge the economic support by FAIP (Federazione Associazioni Italiane Paraplegici), “Neurogel-en-March ” Foundation (France), and Fondazione “La Colonna. ” Author contributions. Stephana Carelli: conception and design, data analysis and interpretation, manuscript writing. Toniella Giallongo: performed experiments. Giovanni Marfia: data analysis and interpretation, manuscript writing. Davide Merli: performed experiments. Luisa Ottobrini: performed experiments. Michele Basso: data analysis and interpretation. Anna Maria Di Giulio: data analysis and interpretation, financial support, manuscript writing. Alfredo Gorio: conception and design, data analysis and interpretation, financial support, manuscript writing. The authors declare no conflicts of interest.
