Abstract
To investigate potential cures for spinal cord injury (SCI), several researchers have transplanted neural stem/progenitor cells (NS/PCs) into the injured spinal cord by different procedures, including intralesional (IL), intrathecal (IT), and intravenous (IV) injection. However, there are no reports quantifying or comparing the number of cells successfully transplanted to the lesion site by each procedure in vivo. The purpose of the present study was to determine the optimal method of cell transplantation to the SCI site in terms of grafted cell survival and safety. For this purpose, we developed mouse NS/PCs that expressed a novel Venus-luciferase fusion protein that enabled us to detect a minimum of 1,000 grafted cells in vivo by bioluminescence imaging (BLI). After inducing contusive SCI at the T10 level in mice, NS/PCs were transplanted into the injured animals three different ways: by IL, IT, or IV injection. Six weeks after the transplantation, BLI analysis showed that in the IL group, the luminescence intensity of the grafted cells had decreased to about 10% of its initial level, and appeared at the site of injury. In the IT group, the luminescence of the grafted cells, which was distributed throughout the entire subarachnoid space immediately after transplantation, was detected at the injured site 1 week later, and by 6 weeks had gradually decreased to about 0.3% of its initial level. In the IV group, no grafted cells were detected at the site of injury, but all of these mice showed luminescence in the bilateral chest, suggesting pulmonary embolism. In addition, one third of these mice died immediately after the IV injection. In terms of grafted cell survival and safety, we conclude that the IL application of NS/PCs is the most effective and feasible method for transplanting NS/PCs into the SCI site.
Keywords
Introduction
Because the adult central nervous system (CNS) has little potential for regeneration, spinal cord injury (SCI) usually results in severe damage, leading to paraplegia, tetraplegia, or worse. Several strategies have been used to develop new therapies that would allow patients to regain the use of their paralyzed limbs. Because cell transplantation is one of the potential therapies, various kinds of cells have been used as the transplantation source for SCI (14,18,32–34,37,38,46). Among them, neural stem/progenitor cell (NS/PC) transplantation is considered one of the most promising, because the transplantation of NS/PCs into rodent SCI models (13,15,27,42,44,48) and of human neural stem/progenitor cells (NS/PCs) into a marmoset SCI model (28) have been shown to promote functional recovery.
From the viewpoint of clinical trials, it is critical to determine how best to apply cells to the injured spinal cord. Three application methods for cell transplantation for SCI are used: intralesional (IL), intrathecal (IT), and intravenous (IV). Some previous studies have sought to compare different cell transplantation methods for treating SCI. For example, the IL application of NS/PCs was reported to be more effective than the IT application (43), and the IV application of NS/PCs more effective than the IL application (10). However, these reports are not directly comparable, because they did not use the same number of cells for the initial transplants. Previous reports using other kinds of cells for transplantation showed that the IL application was more effective than IT or IV for introducing cells to the SCI site (7,18,67), and that IT was more effective than IV (6,54). However, the number of cells transplanted by each method was not quantified or compared for the different methods in vivo, and the safety of the different methods was not compared. Moreover, some reports did not use uniform numbers of cells for the initial transplantation or examine the grafted cells in vivo over the long term. Therefore, it has remained unclear which application of NS/PCs is best for injured spinal cord in terms of cell survival and safety.
In the present study, to determine the best way to transplant NS/PCs into the SCI site, we applied NS/PCs to the mouse injured spinal cord by IL, IT, or IV injection, and quantitatively examined the survival and distribution of the grafted cells, as well as the complications associated with NS/PC transplantation, such as tumor formation and pulmonary embolism. For this purpose, we developed a new reporter gene for bioluminescence imaging (BLI), which enabled us to quantify the bioluminescent signals of small numbers of cells in vivo over time.
Materials and Methods
Lentiviral Vector Expressing the ffLuc Reporter Gene
A novel fusion HIV-1-based lentiviral vector, expressing ffLuc under the EF1α promoter (see Fig. 1A) (Hara et al., in preparation), was used in this study. ffLuc is composed of Venus and Luc2: Venus is a kind of yellow fluorescence protein (40), Luc2 is firefly (Photinus pyralis)-derived luciferase. The luciferase gene fragment was excised from the pGL4-Basic vector (Promega, Madison, WI). This vector enabled the detection of grafted cells as strong bioluminescent signals from ffLuc in live SCI mice and as fluorescent signals in spinal cord sections. Twenty-four hours before the transfection, 293T cells were seeded into poly-L-lysine-coated T175 flasks. The cells were transfected using the lipofection protocol for the FuGENE6 transfection reagent (Roche, Indianapolis, IN). Three days after transfection, the conditioned medium was collected, and the virus was concentrated by centrifugation at 25,000 rpm for 1.5 h at 4°C. The pelleted viral particles were resuspended and stored frozen at −80°C. The titer of the concentrated virus was 1 × 108 to 2 × 108 transducing units per milliliter (TU/ml) when assayed using 293T cells, and the infectivity was determined by fluorescence expression as analyzed on a FACS Calibur (Becton-Dickinson, Franklin, Lakes, NJ).

In vitro imaging and differentiation of lentivirally transduced NS/PCs. (A) Structure of the HIV-1-based lentivirus vector that expresses ffLuc under the elongation factor (EF)-1α promoter. (B) Fluorescent (left) and phase-contrast (right) images of neurospheres derived from ffLuc-expressing NS/PCs. (C) The ffLuc-expressing NS/PCs showed strong luciferase intensity. (D) There was a linear correlation between the number of labeled NS/PCs and their photon count in vitro. Data are the mean ± SEM; n = 10. (E-G) The ffLuc-expressing NS/PCs differentiated into Tuj1-positive (E), GFAP-positive (F), and CNPase-positive (G) cells. (H) Quantitative analysis of the ffLuc-expressing NS/PC phenotype after differentiation. Data are the mean ± SEM; n = 3. Scale bars: 200 μm (B), 20 μm (E-G).
NS/PC Labeling and Differentiation Assay
NS/PCs were cultured as reported previously (55). In brief, the striatum of a C57BL/6J mouse on embryonic day 14 was dissociated using a fire-polished glass pipette, and the dissociated cells were collected by centrifugation and resuspended in culture medium. The culture medium consisted of DMEM/F12 supplemented with a hormone mixture as described previously (55). Human recombinant fibroblast growth factor-2 (FGF-2) and epidermal growth factor (EGF) (20 ng/ml each) were added every 2 days. The cells formed floating cell clusters (neurospheres) within 2–3 days. The concentrated virus was added to the culture medium to infect primary NS/PCs (multiplicity of infection, MOI = 1.0). After being propagated for two passages, the neurospheres were used for in vivo BLI or dissociated into single cells and plated onto poly-L-ornithinecoated coverslips at a density of 1 × 105 cells/ml for in vitro analysis. The virally transduced NS/PCs were allowed to differentiate for 5 days and were fixed with 4% paraformaldehyde in 0.1 M phosphate-buffered saline (PBS) for immunocytochemistry.
SCI Model
Adult female C57BL/6J mice (20–22 g, n = 20; Clea, Tokyo, Japan) were anesthetized with an IP injection of ketamine (100 mg/kg) and xylazine (10 mg/kg). After laminectomy at the 10th thoracic spinal vertebra (T10), the dorsal surface of the dura matter was exposed, and SCI was induced using a commercially available SCI device (IH impactor, Precision Systems and Instrumentation, Lexington, KY), as described previously (47). This device creates a reliable contusion injury by rapidly applying a force-defined impact (60 kdyn) with a stainless steel-tipped impactor. All procedures were approved by the ethics committee of Keio University, and were in accordance with the Guide for the Care and Use of Laboratory Animals.
NS/PC Transplantation
The lentivirally transduced NS/PCs were transplanted into each mouse immediately after SCI using one of the three different methods: IL cell transplantation (IL group, n = 5), IT cell transplantation (IT group, n = 5), and IV cell transplantation (IV group, n = 10). For the IL group, the tip of a glass micropipette was inserted into the epicenter of the injured spinal cord, and the NS/PCs were injected through it. For the IT group, a laminectomy was performed at the L3 level, and the NS/PCs were injected into the subarachnoid space at the same level, through a 32-gauge catheter. For the IV group, the NS/PCs were injected into the tail vein or femoral vein through a 32-gauge catheter. For all the applications, the number and concentration of the transplanted cells were the same (5 × 105 cells/2 (0.l), and the NS/PCs were injected at a rate of 1 μl/min with a Hamilton syringe (25 μl) and stereotaxic microinjector (KDS 310, Muromachi-kikai, Tokyo, Japan). The number of viable cells in the suspensions was determined by cell counting using trypan blue dye and by the signal intensity of the NS/PCs determined by BLI prior to transplantation.
Bioluminescence Imaging
The Xenogen-IVIS 100 cooled CCD optical macroscopic imaging system (SC BioScience, Tokyo, Japan) (56) was used for BLI. Both in vitro and in vivo BLI was performed as previously reported (47). In brief, the signal intensity of the lentivirally transduced NS/PCs in vitro was examined on cells plated at various densities (approximate range 1 × 102 to 106 cells/10 μl and the bioluminescent images were captured immediately after the addition of D-luciferin [D-(-)-2-(6′-hydroxy-2′-benzothiazolyl) thiazone-4-carboxylic acid] (15 mg/ml). In vivo BLIs were captured 15 min after the IP injection of D-luciferin (0.3 mg/g body weight) with the field-of-view set at 10 cm, because the photon count was most stable with its peak intensity between 10 and 30 min after the IP injection of D-luciferin. The integration time was fixed at 5 min for each image. All images were analyzed with the Igor (WaveMetrics, Lake Oswego, OR) and Living Image software (Xenogen, Alameda, CA), and the optical signal intensity was expressed as photon-flux (photon count), in units of photons/s/cm2/steradian. Each image was displayed as a pseudocolored photon count image superimposed on a gray scale anatomic image. To quantify the measured light, we defined regions of interest (ROI) over the cell-implanted area and examined all values in the same ROI.
Immunohistochemistry
Animals were anesthetized and transcardially perfused with 4% paraformaldehyde in 0.1 M PBS 6 weeks after the transplantation. The spinal cord was removed, embedded in OCT compound, frozen in liquid nitrogen, and sagittally sectioned at 30 μm on a cryostat. For immunofluorescence staining, cultured cells or tissue sections were stained with primary antibodies, including anti-green fluorescence protein (GFP) (1:500; MBL, Woburn, MA), anti-glial fibrillary acid protein (GFAP) (1:500; AIC, Long Beach, CA), anti-β-III-tubulin (Tuj-1) (1:200; Sigma, St. Louis, MO), anti-2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase) (1:200; Sigma), anti-Hu (1:1000; a gift from Dr. Robert Darnell, The Rockefeller University), and anti-APC (CC-1) (1:200; Oncogene, Cambridge, MA). Hoechst 33342 was used to counterstain the nuclei (Molecular Probes, Eugene, OR) and then samples were incubated with Alexa Fluor-conjugated secondary antibodies (1:500; Molecular Probes, Eugene, OR). Images were obtained by fluorescence microscopy (Axiocam; Carl Zeiss, Munich, Germany) or confocal microscopy (LSM700; Carl Zeiss). For diaminobenzidine staining, the sections were incubated at 4°C with anti-GFP (1:500; MBL), followed by biotinylated secondary antibodies (1:500; Jackson Immunoresearch). Biotinylated antibodies were visualized using the Vectastain Elite ABC kit (Vector Laboratories, Burlingame, CA), followed by diaminobenzidine (Sigma). Quantitative analysis of the NS/PCs after their differentiation in vitro was performed as described previously (47).
Results
In Vitro Imaging and Differentiation Assay of Lentivirally Transduced NS/PCs
To identify and monitor the transplanted cells, NS/PCs were labeled by lentiviral infection with ffLuc expressed under the EF1α promoter (Fig. 1A). Owing to the stable and strong emission of ffLuc, which is a fusion protein of Venus and luciferase [(39), Hara et al., in preparation] we could observe the fluorescent signals of the Venus by fluorescence microscopy (Fig. 1B) and the bioluminescent signals of luciferase with BLI (Fig. 1C). FACS Calibur analysis revealed that >70% of these cells were positive for Venus (data not shown). The minimum number of cells that could be measured by their photon counts with BLI was approximately 100 cells in vitro. There was a linear correlation between the number of labeled cells and the photon count (Fig. 1D). The NS/PCs expressing ffLuc showed about 10 times stronger bioluminescence intensity in vitro than the reported intensity of NS/PCs expressing conventional luciferase with IRES-Venus (47). To confirm that the lentiviral transduction did not alter the properties of the NS/PCs, we performed differentiation assays on transduced NS/PCs. The transfected NS/PCs could be maintained and passaged using the conventional neurosphere method, and an in vitro differentiation assay revealed that the trasduced NS/PCs differentiated into Tuj1-positive (21.1 ± 2.1%), GFAP-positive (50.0 ± 2.8%), and CNPase-positive (17.9 ± 8.1%) cells (Fig. 1E-H), which are markers for neurons, astrocytes, and oligodendrocytes, respectively. These results showed the lentivirus-transduced NS/PCs had a similar multipotency to that reported for nontransduced NS/PCs (47).
Detection Limits for In Vivo Imaging of NS/PCs Transplanted Into Intact Spinal Cord
When transplanted into the intact spinal cord of adult C57BL/6 mice, the transfected NS/PCs showed one tenth of the photon count that was observed in vitro (Fig. 2A). There was a linear correlation between the numbers of grafted cells and the photon count in vivo (Fig. 2B), and the minimum number of grafted cells that could be measured with BLI was approximately 1,000 cells (Fig. 2C). As few as 100 grafted cells could be detected as bioluminescence in the normal spinal cord after opening the skin at the transplantation site (Fig. 2D).

In vivo imaging of the grafted NS/PCs in the intact spinal cord. (A) Luminescence of the grafted NS/PCs was detected in vivo. In vivo, their photon counts were approximately one tenth of those observed in vitro. (B) There was a linear correlation between the number of grafted NS/PCs and the photon count in vivo. Data are the mean ± SEM; n = 3. (C) The luminescence of 1,000 cells transplanted into the normal spinal cord was detectable by photon count. (D) The luminescence of 100 cells grafted into the normal spinal cord was undetectable without opening the skin.
In Vivo Imaging of NS/PCs Grafted Into the Injured Spinal Cord
To examine the survival and distribution of the transplanted cells in vivo, the photon counts of the grafted cells in each mouse were measured weekly by BLI for 6 weeks after transplantation. In the IL group, the photon counts of the grafted cells were detected at the lesion site for all 6 weeks, in spite of their decrease to approximately 30% of the initial level at 7 days and to 9.4 ± 0.03% at 6 weeks after the transplantation (Fig. 3A, B). In the IT group, the grafted cells were distributed along the entire subarachnoid space immediately after transplantation, and some were detected at the lesion site 7 day later. The bioluminescence of the grafted cells at the lesion site gradually disappeared thereafter. The photon counts of the grafted cells also decreased, to about 5% of the initial level at 1 week and 0.3 ± 0.0%, which is almost background measurement value, at 6 weeks after transplantation (Fig. 3C, D). In the IV group, no bioluminescence was detected at either the site of injury or in the uninjured spinal cord at any of the time points examined (Fig. 3E, F). In contrast, strong bioluminescence was observed in the bilateral chest immediately after the injection of the NS/PCs in all 10 animals of the IV group (Fig. 3G), and 3 of the 10 animals died immediately after the injection. In these mice, we confirmed the strong bioluminescence of the grafted cells in the lung (Fig. 3H). Although the luminescence in the bilateral chest became undetectable the next day in the surviving mice, two of the other seven mice died within 4 days of the transplantation. Five of the original 10 mice in the IV group survived for the full 6 weeks following the cell injection.
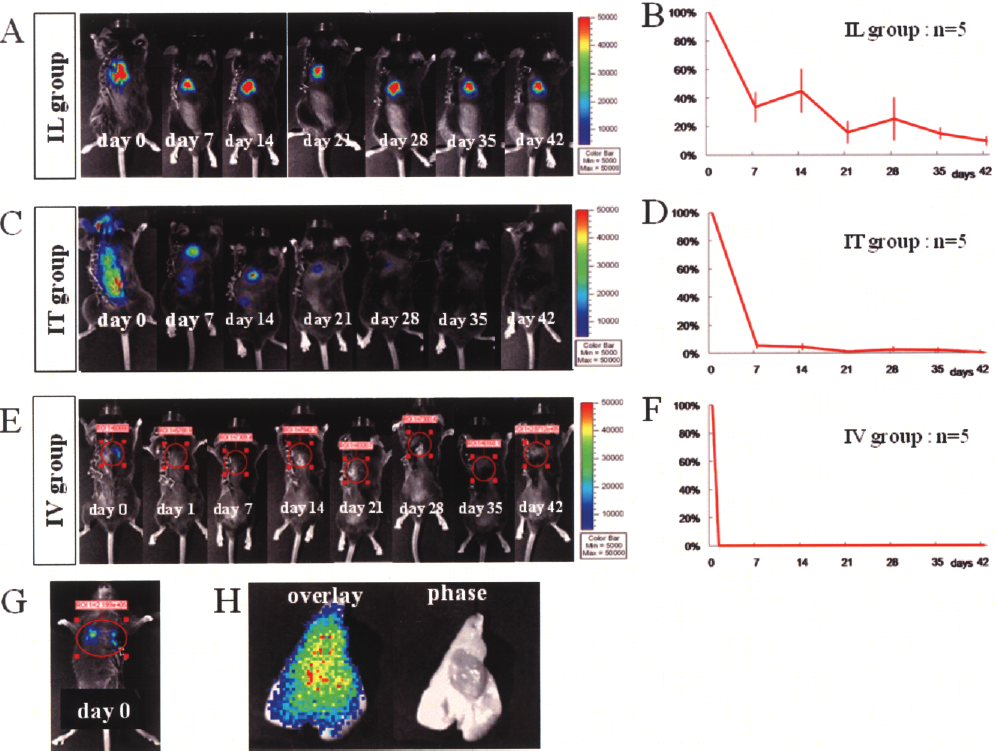
(A, C, E) Representative in vivo images of NS/PCs transplanted by the three application methods: (A) IL group, (C) IT group, and (E) IV group. (B, D, F) Quantitative analysis of the photon counts of the grafted NS/PCs in the IL group (B), IT group (D), and IV group (F). Data are the mean ± SEM; n = 5. (G) Strong luminescence was observed in the bilateral chest immediately after transplantation in all 10 animals of the IV group. (H) A resected lung from the IV group showed strong luminescence from the transplanted cells.
Immunohistochemistry
To examine the survival and distribution of the grafted cells histologically, immunohistochemistry with an anti-GFP antibody, which could detect ffLuc protein, was performed 6 weeks after transplantation to identify the ffLuc-positive grafted cells. Among the three groups, the IL group showed the best survival of ffLuc-positive cells at the lesion site (Fig. 4A, B). In the IT group, only a few grafted cells were detected at the lesion site (Fig. 4C, D). We could not detect any grafted cells at the lesion site in the IV group (Fig. 4E, F). In IL group, these grafted cells differentiated into neurons, astrocytes, and oligodendrocytes within the injured spinal cord (Fig. 4G–I), as previously reported (47). In the IT group, there were some ffLuc-positive cell clusters that lay on the surface of the spinal cord (Fig. 5A, C, D) and the cauda equina (Fig. 5B, E, F). These cells, which also differentiated into neurons, astrocytes, and oligodendrocytes (Fig. 5G–I), attached on the surface of pia matter of the uninjured spinal cord, but not invaded into the spinal cord tissue (Fig. 5C–F). In the IV group, ffLuc-positive cells were observed in the lung and spleen, and GFP-positive fragments were found in the glomeruli of the kidney (Fig. 6), although no ffLuc-positive cells were detected at the lesion site. There was no tumor formation, at least 6 weeks after transplantation, in any of the three groups.

Immunohistochemical analysis of the injured spinal cord 6 weeks after transplantation. The grafted cells were identified as ffLuc-positive cells by diaminobenzidine (A, C, E) and immunofluorescence (B, D, F) staining. (A, B) In the IL group, the grafted cells survived well at the lesion site. (C, D) In the IT group, a few grafted cells survived, and some were present at the surface of the lesion site. (E, F) In the IV group, no grafted cells could be detected at the lesion site. (G, H, I) In the IL group, the grafted cells differentiated into Hu-positive (G), GFAP-positive (H) and APC-positive (I) cells within the injured spinal cord. Scale bars: 500 μm at magnification 25×, inset at magnification 400× (A–F); 20 μm (G–I).

Immunohistochemical analysis of the IT group. (A, B) The grafted cells (arrow) were distributed on the surface of the uninjured spinal cord (A) and the cauda equina at 6 weeks after transplantation (B). (C, D) High magnification of the box in (A); the grafted cells were attached to the spinal cord surface, as shown by diaminobenzidine (C) and immunofluorescence (D) staining. (E, F) High magnification of the box in (B); the grafted cells were attached to the cauda equina, as shown by diaminobenzidine (E) and immunofluorescence (F) staining. (G, H, I) In the IT group, few grafted cells survived and differentiated into Hu-positive (G), GFAP-positive (H), and APC-positive (I) cells on the surface of the uninjured spinal cord and the cauda equina. Scale bars: 500 μm (A, B), 100 μm (C–F), 20 μm (G–I).

Immunohistochemical analysis of the several organs in the IV group. ffLuc-positive cells were found in the lung and spleen at 6 weeks after transplantation. Many ffLuc-positive fragments were also observed in the kidney, suggesting that degraded grafted cells had been trapped in the glomeruli. Scale bar: 50 μm.
Discussion
In the present study we used a new reporter gene, ffLuc, and BLI for the longitudinal tracking and quantification of grafted cells in vivo. Our findings demonstrated that the IL injection of NS/PCs resulted in the best survival of the grafted cells among the three applications, and no complications were seen in the recipient mouse. Our analysis also revealed the distribution of the cells transplanted by the three methods.
To analyze the kinetics of cells after their transplantation in vivo, several previous studies have used immunohistochemistry, gamma scintigraphic imaging (1,11,20, 24), and magnetic resonance imaging (MRI) (3,26,29,53). However, none of these methods permit the quantitative and continuous evaluation of the transplanted cells. Immunohistochemical analysis cannot track the fate of grafted cells in the same animal, because the animals need to be sacrificed at serial posttransplant time points to examine the grafted cells. Scintigraphic imaging using a radioactive tracer to label grafted cells enables the examination of their survival and distribution in vivo, but they can only be observed for a short time, owing to the tracer's short half-life and the dilution effect by cell division. MRI can detect the distribution of the transplanted cells at their initial deposition with high sensitivity, but the MRI signals from dead cells cannot be distinguished from those of live signals, which diminishes the usefulness of this method over time.
To overcome these problems, we used a BLI system, which enabled us to trace only living grafted cells in the same animal at multiple time points and to quantify the bioluminescence intensity of the grafted cells as a photon count (17,22,25,35,56,60,66). Although there are previous reports in which grafted cells were analyzed by BLI in injured spinal cord (33,47), the intensity of the bioluminescence in those studies was not strong enough to detect small numbers of cells. In the present study, we used a novel fusion protein, ffLuc. NS/PCs expressing ffLuc emitted bioluminescent signals almost 10 times stronger than those of NS/PCs expressing conventional luciferase with IRES-Venus (47), thus enabling the detection of the bioluminescent signals of transplanted NS/PCs, even though they were present in small number or deep inside the mouse body. Because the ffLuc that is transfected by lentivirus is expressed in the NS/PCs permanently, we can trace the grafted NS/PCs accurately for a long period after transplantation.
From the viewpoint of clinical trials for cell transplantation to treat SCI, there is concern about causing additional damage to the injured spinal cord during the cell transplantation procedure. However, several researchers who have performed IL cell transplantation for SCI demonstrated that motor function is not degraded by the IL application (16); rather, the transplantation of cells by IL injection promotes functional recovery (19,30,33,42,44,47).
The IT application is performed in two ways: one is by injection into the fourth ventricle (4,45,69,70) and the other is by lumbar puncture (5,21,36). Because the IT application via lumbar puncture is considered less invasive than transplantation by IL injection, this procedure has been used for cell transplantation into SCI patients in some hospitals (12,59,64). Previous studies also demonstrated that grafted NS/PCs transplanted through an IT application survive within the injured spinal cord (4,36,69). In the present study, however, cells transplanted by IT initially dispersed throughout the entire subarachnoid space, resulting in low survival at the lesion site. These findings suggested that the IT application is an uncertain method for delivering cells to the site of injury. Moreover, a small number of the grafted cells survived on the surface of the uninjured spinal cord and cauda equina. Although there was no evidence of tumor formation during the observation period of this study, it was previously reported that neural cells deposited at uninjured spinal cord sites had formed enlarging clusters of cells within a couple of weeks after transplantation (4). Furthermore, in a recent clinical procedure using an IT application, human NS/PCs caused a donor-derived brain tumor in a patient with ataxia telangiectasia, which was associated with comprised immunity (2). Therefore, it is highly likely that intrathecally applied cells adhering to the surface of the spinal cord could cause tumor formation in SCI therapy, too.
IV injection has been considered the most minimally invasive application procedure for several types of cells (31,61,63), and is already used for clinical therapies such as hematopoietic stem cell transplantation. However, in the present study, the intravenously injected NS/PCs were distributed throughout the body, and no grafted cells were detected within the injured spinal cord, although some grafted cells were trapped in the lung, spleen, and kidney. The ffLuc-positive fragments found in the kidney were broken grafted cells. Previous reports demonstrated that intravenously injected nonneural cells could be observed at a lesion site shortly after the transplantation, and the mechanisms for functional recovery in this situation are thought to be trophic support of the grafted cells and/or activation of the intrinsic neuronal cells, rather than the displacement of lost neurons and glia at the lesion site, especially for mesenchymal stem cell or bone-marrow-derived stem cell transplantation (52,62). However, the important mechanisms for functional recovery from SCI are not only trophic support, but also neural circuit reconstruction, axonal induction, and remyelination (8,50,65). The cells grafted into the host spinal cord play a role in maintaining the improved performance. Cummings et al. showed that selective ablation of the grafted cells in the host spinal cord causes the deterioration of motor function (19), indicating that the long-term survival of grafted cells in the host spinal cord is very important for functional recovery from SCI.
In the present study, we recognized the functional recovery in the IL group according to the behavioral analysis by BMS score (9) as reported previously (27,44,47), but not in the IT and IV groups (data not shown). However, there remained several possibilities that some mice technically might have received cauda equina impediment by IT injection and that some mice transplanted by femoral vein injection might also have suffered from postoperative scar formation producing hip joint contracture. Thus, further studies would be necessary to obtain a conclusive result in terms of functional recovery. The timing of cell transplantation is critical for the survival of grafted cells because microenvironments of injured spinal cord such as neurotrophic factors, proinflammatory cytokines, chemokines, and free radicals are different among the acute, subacute, and chronic phases of SCI (23,41,49,58). Previous studies indicated that the intralesionally grafted cells at the subacute phase showed better survival than at the acute phase (51). In the present study, we dared to choose the acute phase transplantation because the repair of blood–brain barrier and glial scar formation, which occur at the subacute and chronic phase, could prevent the grafted cells from infiltrating into the injured spinal cord after IT or IV injection at the subacute or chronic phase (57,68).
In the present study, all 10 of the animals in the IV group showed bioluminescence in the chest immediately after transplantation, three died immediately after the injection, and two more died within 4 days, due to pulmonary embolism. To reduce the risk of embolism, the grafted cells should be dissociated completely into single cells, or large cell clusters should be eliminated by filtration. However, complete dissociation of the grafted cells could decrease cell viability compared with the viability of small clusters. Furthermore, because both the IV and IT applications resulted in grafted cells homing to nonlesion sites, longer term observation is needed to evaluate tumor formation.
In conclusion, the transplantation of NS/PCs by IL injection resulted in the best survival of grafted cells among the three methods investigated, and no complications were seen afterwards. Transplantation by IT injection resulted in low survival of the grafted cells at the lesion site, and some cells were detected on the surface of the uninjured spinal cord, along the subarachnoid space. After the IV application, no grafted cells were detected at the SCI site, whereas luminescence was observed in the bilateral chest of all these mice, suggesting that the grafted cells caused pulmonary embolism immediately after the IV injection. Taken together, our findings indicate that the best method for the transplantation of NS/PCs into the SCI site is by IL injection, in terms of both grafted cell survival and safety of the recipient.
Footnotes
Acknowledgments
We thank T. Harada for expert animal care and S. Miyao for technical support. This work was supported by grants from the Key Technology Project for the Realization of Regenerative Medicine from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) Japan to M.N. and H.O.; the General Insurance Association of Japan to M.N. and Y.T.; and a Grant-in-Aid for the Global COE program from MEXT to Keio University.
