Abstract
Cerebral ischemic stroke is a very common condition that can cause death and disability. Studies have confirmed that stem cells have therapeutic effects if administered after a stroke. There is still a great deal of debate regarding the best route for cell transplantation. Intravascular delivery is the most commonly used one. In this study, the therapeutic effects of bone marrow stem cells (BMSCs) delivered by intra-arterial (IA) and intravenous (IV) injection in a rat transient middle cerebral artery occlusion model (MCAO) are compared. Histological analysis demonstrated that the IA route bypasses the pulmonary system and directs the cells to the ischemic parts of the brain more efficiently. The BMSCs delivered via the IA route promoted angiogenesis and improved functional recovery. The cerebral blood flow (CBF) of the rats was monitored during the IA injection process. No reduction in CBF or microstrokes was detected. Brain perfusion and metabolism, as evaluated by SPECT and PET, were better in rats treated with cells delivered via IA. Results showed that the IA route is a safe and effective way to transplant hBMSCs. This manuscript is published as part of the International Association of Neurorestoratology (IANR) special issue of Cell Transplantation.
Introduction
Cerebral ischemic stroke is a serious public health concern. It causes considerable death and disability (6,24). There is currently no ideal treatment for permanent focal cerebral ischemia, so exploring an effective therapy is of vital importance (14). Stem cell transplantation may be a useful option. Mesenchymal stem cells (MSCs) have been shown to have successful results in experimental animal models of ischemic stroke when administered systematically, reducing the severity of cerebral ischemic injury and promoting neurological function (3,32). Human bone marrow-derived stem cells (hBMSCs) are poorly immunogenic and are not detectably tumorigenic. Their use has also raised few ethical concerns (25,31). When hBMSCs are administered systematically, their curative effects are dependent on the homing of grafted cells to the target tissue (18,19).
Previous reports have demonstrated that intravenous (IV) injection of cells into rats after ischemic stroke causes large numbers of cells to accumulate in undesirable locations, primarily the lungs (13,21,28). Intra-arterial (IA) infusion bypasses the lungs and allows the cells to reach the brain. However, this technique may cause microocclusions (29). Recent studies have evaluated various IA infusion techniques and cell concentrations. The IA route was found to foster efficient homing to the brain without causing thromboembolism complications (4).
The aim of the present work was to study the biodistribution of IA-administered hBMSCs in the middle cerebral artery occlusion (MCAO) model of rats. The effects of IA injection of hBMSCs on cerebral blood blow (CBF) and the risk of microstrokes were also evaluated. Therapeutic effects were estimated by ethology score, molecular imaging technology, and immunohistochemistry.
Materials and Methods
Preparation of hBMSCs
The hBMSCs were isolated from the bone marrow aspirate, which was donated by a healthy person (male, 26 years old) who had provided informed consent. The mono-nuclear cells were separated by standard lymphocyte isolation techniques (1). After 24 h of incubation, the sample was decanted, and the adherent cells were collected and used to inoculate the flask. The cell culture system was comprised of 58% Dulbecco's modified Eagle's medium and a nutrient mixture of F12 plus 40% MCDB-201 medium. The cells were propagated for four to five generations and were subsequently digested, harvested, and frozen for later use. Isolation and culture were conducted in the Center for Excellence in Tissue Engineering (Institute of Basic Medical Sciences and School of Basic Medicine, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, P. R. China). The use of hBMSCs was approved by, and conforms with, the regulations of the Human Experimentation and Ethics Committee. Cell cultures were performed as described previously (11). hBMSCs were collected, counted, and analyzed by flow cytometry for anti-human cluster of differentiation 29 (CD29), CD105, CD44, fetal liver kinase 1 (Flk-1), CD34, CD44, and human leukocyte antigen (HLA)-DR expression (Becton Dickinson, Franklin Lakes, NJ, USA) conjugated with biotin. Biotinylated antibodies were detected by incubation with streptavidin phycoerythrin (PE) (Molecular Probes, Eugene, OR, USA). All analyses were performed using Accuri C6 Flow Cytometers with CFlow software (Accuri Cytometers, Inc., Ann Arbor, MI, USA).
Surgical Procedures and Use of hBMSCs
Adult male Sprague–Dawley rats (230–250 g, n = 27) were purchased from the Center for Experimental Animal Research of the Chinese Academy of Medical Sciences (Beijing, China). Research and animal care procedures were approved by, and in compliance with, the guidelines set by the committee on animal research of Peking Union Medical College Hospital, and all the procedures were consistent with the institutional ethics committee.
Cerebral ischemia was induced by a transient intraluminal vascular occlusion method using the intraluminal filament technique (16). In brief, under halothane (Lunan Pharmaceutical, Jinan, China) anesthesia, the right common carotid artery (CCA), external carotid artery (ECA), and internal carotid artery (ICA) were exposed. The ECA was cut with microscissors, and a monofilament nylon thread 0.25 mm in diameter (Sunbio Biotech, Beijing, China) was inserted into the stump of the ECA. The nylon thread was placed into the ICA until resistance was encountered (1.8–2.1 cm from the bifurcation of the carotid artery). After a period of occlusion (120 min), the nylon thread was withdrawn, and a plastic catheter (elongated subarachnoid tube with an outer diameter of 0.2 mm and inner diameter of 0.1 mm; Sunbio Biotech) was inserted into the ECA. The end of the catheter was sealed, and the wound was sutured (Johnson & Johnson, New Brunswick, NJ, USA) 24 h after ischemia. The modified neurological severity scores (mNSSs) test was then performed. Rats subjected to transient MCAO with mNSSs around 10–12 were randomly assigned to three groups (nine rats per group): 1) IA hBMSC-treated group. Catheters were connected to the ECA using a microsyringe. The catheter was implanted in the ICA, and cell suspensions [3 × 106 hBMSCs in 1 ml phosphate-buffered saline (PBS)] (Peking Union Medical College Hospital central laboratory, Beijing, China) were then injected over the course of 2 min. 2) IV hBMSC-treated group. Cell suspensions (3 × 106 hBMSCs in 1 ml PBS) were injected through the caudal vein over the course of 2 min. 3) Saline control group. The same volume of saline was injected through the caudal vein over the course of 2 min.
Laser Doppler Flow Measurements of CBF During Cell Transplantation
During cell transplantation, a laser Doppler probe (Moor LDI2-HR, Moor Instruments, Axminster, Devon, UK) was installed on the ipsilateral crania of three rats in the IA and IV groups using dental acrylic (Shanghai Dental Material Company, Shanghai, China). The probe was positioned on the thinned skull under the temporal muscle, near the middle cerebral artery (MCA). This probe was used to measure CBF in the MCA region. The values were recorded at the location with the highest sustained signal as a percentage of that baseline. Laser Doppler measurements were recorded before and after transplantation. Measurements were recorded for 15 min for all groups.
Functional Outcomes
The mNSSs were evaluated by observers 1, 3, 7, 14, and 28 days after transplantation. Observers were blind to the purpose of the experiment. The mNSSs are a composite of motor, sensory, balance, and reflex tests (23). This test is suitable for the evaluation of long-term neurological outcomes after unilateral brain injury.
SPECT Imaging for Perfusion of the Brain
99Tcm-ethyl cysteinate dimer (99Tcm-ECD) single-photon emission computed tomography SPECT imaging (Bioscan Nano Spect/CT, Poway, CA, USA) was performed 4 weeks after transplantation to determine the therapeutic effect of IV hBMSCs and IA hBMSCs on perfusion within the brain using semiquantitative analysis.
The anesthetic of 10% chloral hydrate (400 mg/kg; HaoChem, Shanghai, China) and 74–111 MBq (2–3 mCi in 0.5 ml) 99Tcm-ECD (Institute of Chinese Atomic Medicine, Beijing, China) was injected intravenously. After 5 min, imaging was started. The acquisition and reconstruction protocol were as described previously (8).
SPECT data were analyzed semiquantitatively. The regions of interest (3D ROIs) were drawn on the side without infarction. The contralateral ROI was mirrored in the infracted hemisphere. The activity inventory of each ROI was measured. The extent of hypoperfusion was expressed as a percentage obtained through the following formula: average radiation intensity right/average radiation intensity left × 100%.
PET Imaging for Metabolism in the Brain
18F-fludeoxyglucose-positron emission tomography (18F-FDG-PET) (micro PET Rodent; Siemens, Beijing, China) examination was also performed 4 weeks after transplantation to measure variations in glucose metabolic activity in the brain.
After anesthesia, 22.2 MBq (0.6 mCi in 0.5 ml) 18F-FDG (produced by the Department of Nuclear Medicine, Peking Union Medical College Hospital, Beijing, China) was injected intravenously. Acquisition and reconstruction protocols were as described previously (9). 3D ROIs were drawn on the side without infarction. The contralateral ROI was mirrored in the infracted hemisphere. The relative metabolic activity was measured in ROIs. The mean radioactivity uptakes (SUV) in ROIs were calculated. The extent of hypometabolism is expressed through the formula mean radioactivity uptake (SUV) of the infarcted brain/mean radioactivity uptake (SUV) of the normal brain × 100%.
Immunohistochemistry and Immunofluorescence
After PET imaging, rats were intracardially perfused with chilled saline followed by 4% paraformaldehyde (Peking Union Medical College Hospital central laboratory, Beijing, China). The brains and lungs were collected and fixed with 4% paraformaldehyde, embedded in paraffin, and cut into 8-μm serial sections. Coronal sections (8 μm) were taken within 2 mm of bregma and processed for immunohistochemical staining with monoclonal antibodies against human nuclei (1:100, MAB1281; Millipore, Billerica, MA, USA), vascular endothelial growth factor (VEGF; 1:500; Abcam, Cambridge, MA, USA), neuronal nuclei (NeuN; 1:500; Abcam), and von Willebrand factor (vWF; 1:300; Sigma-Aldrich, St. Louis, MO, USA). Secondary antibodies and visualization was performed using the ImmPRESS Universal (mouse/rabbit) Ig Kit (Vector Laboratories, Burlingame, CA, USA).
Transplanted hBMSCs were identified in the brains and lungs using the mouse anti-human nuclei monoclonal antibody MAB1281. VEGF was a meaningful parameter when measuring the angiogenesis in the peri-infarct zone. Additionally, vWF and NeuN could reflect the number of microvessels in the peri-infarct zone and the mature neurons left in the ischemic boundary zone, respectively.
Changes in microtubule-associated protein-2 (MAP-2) and myelin basic protein (MBP) were also monitored to assess microtubule assembly and hypomyelination. The sections were first treated with MAP-2 (1:100, Millipore) and cyanine 3 (CY3)-conjugated sheep anti-mouse IgG (1:500, Millipore) staining. Then they were treated with MBP antibodies (GalC, 1:500, Millipore) and fluorescein isothiocyanate (FITC)-conjugated sheep anti-rabbit IgG (1:500, Millipore) staining. To visualize the nuclei, the sections were counterstained with 6-diamidino-2-phenyl-indole (DAPI; 1:500, Sigma).
The sections were imaged using an image analysis system (Leica TCSSP2, Leica, Wetzlar, Germany). Immunoreactive cells were counted based on evaluation of an average of three slides (20 μm thick, 200-μm intervals, every 10th slide) from each animal using ImagePro Plus software (Media Cybernetics, Rockville, MD, USA). The NeuN- and VEGF-positive cells were counted with a 20× objective. vWF-positive microvessels were counted with a 10× objective. The three areas in each slice where positive cells were most dense were chosen for counting. Fluorescence signals of MAP-2 + MBP were also observed.
Statistical Analysis
All the data in this study are presented as means with standard deviations (SD). They were analyzed with SPSS 10.0 (SPSS, Inc., Chicago, IL, USA). One-way analysis of variance (one-way ANOVA), followed by post hoc Tukey's test, was used to analyze the differences between groups. Differences were considered significant at *p < 0.05 and **p < 0.01.
Results
IA Administration of hBMSCs Is Safe
Laser Doppler flow measurements of CBF showed that, during IA injection, CBF remained at baseline. There was no significant difference in CBF between the IA group and IV group (Fig. 1). These results suggested that, with these IA infusion techniques and at this cell concentration, the IA route does not affect the CBF.

Laser flow Doppler analysis of CBF after IV and IA stem cell injections. CBF was not affected by IA cell delivery.
IA Bypasses the Lungs and Directs hBMSCs to the Brain
Immunostaining with the mouse anti-human nuclei monoclonal antibody showed significantly more hBMSCs around the microarteries in the brains of rats subjected to IA compared with IV administration 4 weeks after transplantation (p < 0.05, n = 6) (Fig. 2A–C). In addition, far fewer hBMSCs were observed in the lungs of rats that had undergone IA injection compared with IV administration (p < 0.05, n = 6) (Fig. 2D–F). Fewer hBMSCs were observed in the brains of IV hBMSC-treated rats, while in the lungs, far more hBMSCs were observed in IV-treated rats than in those of rats that were IA treated.
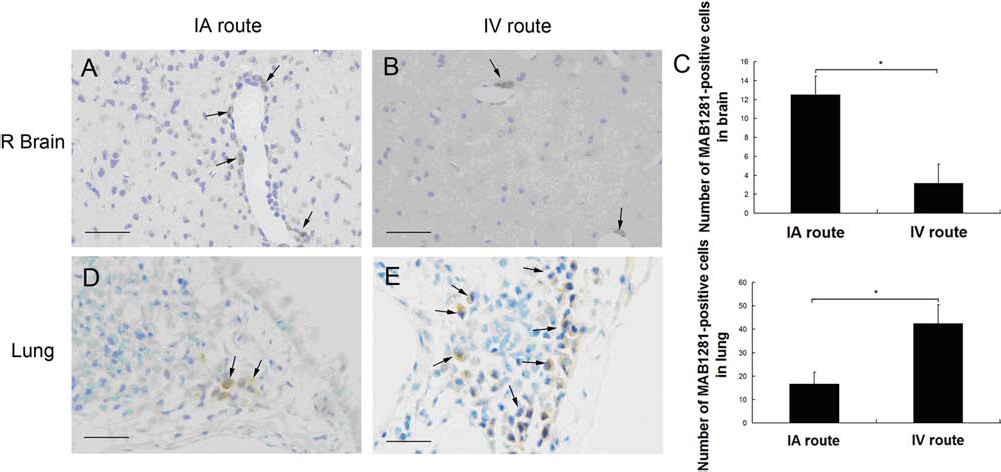
Histology of the brain and lungs of stroke rats. hBMSCs appear dark brown with anti-human mitochondria antibody (MAB1281; arrows). Scale bar: 200 mm. (A–C) hBMSCs were significantly more numerous around microarteries in the brains of IA hBMSC-treated rats at 4 weeks after transplantation than by the IV route (*p < 0.05, n = 6). (D–F) Far fewer hBMSCs were observed in the lungs of rats given IA injection than in those of rats given cells IV (*p < 0.05, n = 6).
hBMSC Grafting Improves Performance During Behavioral Tests
mNSSs were assessed during recovery time. mNSSs is a composite of motor, sensory, reflex, and balance tests. There was no difference in scores between groups at either the first day or the third day after MCAO. Significant functional improvement was observed in the IA treatment group starting 2 weeks after MCAO (p < 0.05, n = 6). Although there was no significant difference between the IV and IA groups at this point in time, the scores of the IA group were lower than those of the control group. The rats in the IA group could perform more tests and lacked fewer of the tested reflexes. Then, 4 weeks after MCAO, the IA group showed significantly more neurological recovery (lower mNSSs) than either the control group (p < 0.05, n = 6) or the IV group (p < 0.05, n = 6). At this point in time, the scores of both the IA group and IV group were lower than those of the control group, suggesting that both IA- and IV-transplanted hBMSCs have neurotrophic effects on the ischemic brain (Fig. 3).

mNSSs for each group from 1 to 28 days after MCAO.*p<0.05 versus the control group; #p < 0.05 versus the IV route group.
SPECT/CT and Perfusion
SPECT is a rapid, accurate, and noninvasive examination, which can be used to evaluate the hemodynamics of cerebral ischemia in live animal models. At 4 weeks after MCAO surgery, the extent of ischemia was recorded. It is here expressed relative to contralateral hemispheric area. The IA group (p < 0.01, n = 6) and IV group (p < 0.05, n = 6) showed significantly smaller radial rarefaction than the control group. The IA group had the smallest hypoperfusion area (Fig. 4A–D).
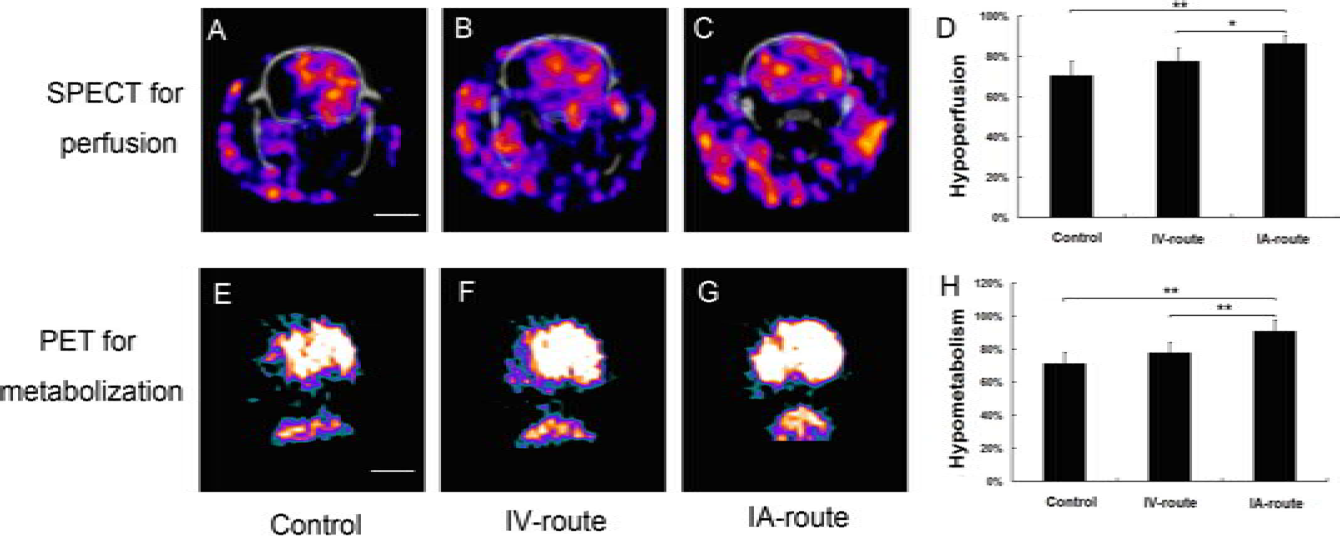
SPECT imaging for perfusion of the brain and PET imaging for metabolism in the brain. (A-D) The degree of cerebral ischemia as assessed by 99Tcm-ethyl cysteinate dimer (99mTc-ECD) perfusion imaging. (A-C) Representative SPECT images of each group obtained 4 weeks after transplantation. The radial rarefaction and defect area were significantly smaller in the IA group (C) and IV group (B) than in the control group (A). The IA-treated group had the smallest hypoperfusion area. (D) Statistical analysis showing the metabolic activity in each group (average signal intensity right/average signal intensity left x 100%). (E-H) The metabolic activity of glucose in the brain imaged using a microPET scanner. (E-G) Representative PET images of each group obtained 4 weeks after transplantation. There was less metabolic activity in the affected (right) hemisphere in all groups. The IA group (G) showed the least metabolic asymmetry. (H) Statistical analysis showing the metabolic activity in each group (average signal intensity right/average signal intensity left×100%). *p<0.05, **p<0.01. Scale bar: 1 cm.
PET and Metabolism
18F-FDG PET imaging can be used to assess ischemia-induced metabolic deficits and provide a sensitive and reliable means of evaluating cerebral ischemic lesions. At 4 weeks after MCAO surgery, all subjects showed reduced metabolic activity in the affected (right) hemisphere. The IA group showed the least metabolic asymmetry (Fig. 4E–G). The extent of ischemia is here expressed relative to the contralateral hemispheric area. The metabolic activity of the affected hemisphere was significantly higher in the IA group than in the IV group (p < 0.01, n = 6) or control group (p < 0.01, n = 6) (Fig 4H).
Angiogenesis and Metabolism as Indicated by Immunohistochemistry and Immunofluorescence
It is here hypothesized that the IA administration of hBMSCs can improve perfusion and metabolism in the ischemic brain. The neuroprotective mechanisms of this improvement were investigated using immunohistochemical testing. VEGF, vWF, and NeuN immunohistochemistry were performed 28 days after transplantation.
There were considerably more VEGF-positive cells in the peri-infarct zone in the IA group than in the IV group (p < 0.01, n = 6) and control group (p < 0.01, n = 6) 4 weeks after MCAO (Fig. 5 A–D). The number of mature NeuN-immunoreactive neurons was also higher in the IA group than in either the IV (p < 0.05, n = 6) group or control group (p < 0.01, n = 6) 4 weeks after ischemia (Fig. 5E–H). vWF is an established marker of microvessel proliferation (34). There were more microvessels in the cortical peri-infarct zones of the IA group than in those of the IV group (p < 0.05, n = 6) and control group (p < 0.01, n = 6) 4 weeks after MCAO (Fig. 5I–L). There was also more expression of MAP-2 + MBP protein, and the morphological structure of microtubules and myelination were more complete in the IA group than in either the IV group or control group 4 weeks after ischemia (Fig. 6).

VEGF, neurons, and microvessels in the ischemic boundary zone 4 weeks after transplantation. (A-C) Immunostaining of VEGF-positive cells in each group. There were far more VEGF-positive cells in the peri-infarct zone of the IA group rats (C) than in IV group (B) or control group rats (A) 4 weeks after transplantation. (D) Statistical analysis showing the number of VEGF-positive cells in each group. *p<0.05, **p<0.01. (E-G) Immunostaining of mature neuronal nuclei (NeuN)-positive neurons in each group. There were more mature NeuN-immunoreactive neurons in the IA group (G) than in the IV group (F) or control group (E). (H) Statistical analysis showing the number of NeuN-positive neurons in each group. *p<0.05, **p<0.01. (I-K) Immunostaining of vWF-positive microvessels in each group. More vWF-positive microvessels were present in IA (K) and IV (J) rats than in control rats (I). (L) Statistical analysis showing the number of vWF-positive microvessels in each group. *p<0.05, **p<0.01. Scale bar: 100 μm (A-C and E-G); 200 μm (I-K).

Double immunohistochemical responses of hBMSCs in the brains of rats in each group with respect to changes in MAP-2 and MBP. There was more MAP-2 + MBP protein expression in the IA group (I–L) than in either the IV group (E–H) or control group (A–D) at 4 weeks after transplantation. Scale bar: 100 μm. Nuclei were counterstained with 6-diamidino-2-phenyl-indole (DAPI).
The present results indicate that IA-transplanted cells promote angiogenesis in the peri-infarct zone (based on VEGF and vWF staining), promote the survival of mature neurons (based on NeuN/MAP-2 staining), and preserve the myelin sheath (based on MBP staining).
Discussion
Stroke is a common clinical condition, and there is currently only one effective treatment: tissue plasminogen activator (tPA) (14). Cell therapy, as a restorative strategy, holds broad prospects for development, but how to deliver the cells successfully is still unknown (3,17). IV delivery is the most common method because it is the least invasive. Many animal studies have shown the benefits of cells administered through IV routes, particularly rodent models of stroke (22,35). However, the efficiency of IV delivery is poor. The IV route may cause the cells to become trapped within peripheral organs, such as the lungs, liver, and spleen, leaving only a few cells to reach the brain (5,21). Intracerebral injection of cells is also common because it is the most direct route to the target organ (12). However, because the ischemic area is large and irregular, injection at multiple sites is required, causing tissue injury. The process of intracerebral injection is traumatic, resulting in postoperative complications, such as seizures and asymptomatic subdural hemorrhage (15). IA infusion may be the most suitable route for cerebral ischemia. In this way, cells can spread throughout the ischemic area with little injury (17). In the present study, the biodistribution of IA-administered hBMSCs and their therapeutic effects were studied in a MCAO model of rats. The risks of IA hBMSC injection on CBF were also recorded.
One potential adverse effect of IA administration is microstrokes, which can be caused by microvascular plugging. Some animal studies have indicated that the IA route of MSC delivery can reduce CBF. They also reported a high mortality rate. It was hypothesized that cells may cause ischemic injury by adhering to each other and blocking vessels (29). Some studies have shown that microstrokes attributable to the transplantation method are caused by interruptions in CCA flow that occur during cell injection (2). In the present study, a delivery method that could introduce cells to the body without interfering with blood flow within the CCA during injection was developed. This method involves transcranial Doppler, which is used to monitor the CBF during the transplantation process. With proper injection technique and a suitable cell concentration, the IA route did not result in any significant decrease in CBF.
The therapeutic effects of this treatment were evaluated from many different angles. The IA group showed the most favorable recovery of sensorimotor function (mNSS scores). The IV route was also associated with significantly more improvement in neural function than the control group.
Molecular imaging was also used to evaluate therapeutic effects. At 4 weeks after transplantation, 99mTc-ECD SPECT imaging was used to evaluate blood flow (20). These results demonstrated that the perfusion of the infarct side was markedly better in the IA route treatment group than in other groups. 18F-FDG PET imaging is a reliable method of assessing variations in metabolic performance (7). At 4 weeks after transplantation, 18F-FDG PET imaging showed that the rats that received hBMSCs through the IA route had significantly more glucose metabolism in their ischemic hemispheres than the rats in the other two groups.
All these results suggest that IA transplantation of hBMSCs is an effective means of delivering cells for the repair of neural injury after stroke. The mechanism underlying these therapeutic effects was examined. Immunohistochemical assessment was performed 4 weeks after transplantation. The results showed that, when hBMSCs were given intravenously, few reached the brain and survived. When hBMSCs were delivered intra-arterially, many more cells reached the brain and survived, and far fewer cells were trapped in the lungs in the IA group. Some studies have demonstrated that the production of trophic and angiogenic factors were one mechanism underlying the improvement in functional outcome (10,26). When hBMSCs were transplanted through the IA route, cell delivery and survival both improved significantly. The living cells produced trophic and angiogenic factors that promoted cell survival.
The results of immunostaining of VEGF showed there to be more VEGF-positive cells in rats in the IA route group. Immunostaining of vWF-positive capillaries in the peri-infarct zone showed there to be more functional vessels in rats treated via the IA route, which was consistent with the results of SPECT perfusion imaging. Cells transplanted via the IA route may improve perfusion through the promotion of endothelial cell survival and the induction of neoangiogenesis (1,30).
The results of immunostaining of NeuN showed there to be more neurons in rats in the IA group, suggesting that more cells survive in the peri-infarct zones of these rats than in those of other rats. The expression of MAP-2 + MBP was also higher, and the morphological structures of microtubule and myelination were more complete in the IA group than in other groups, suggesting that the microtubules and myelination process had been protected. All of these events promote the recovery sensorimotor function and spatial memory (27).
Recently, some scholars pointed out that IA delivery was not superior to IV delivery of autologous bone marrow mononuclear cells in acute ischemic stroke in rodents (33). Reasons for this discrepancy can be explained as follows. Firstly, in the low dose group, they chose a dose that would have had a minor effect, while in the high dose group, a dose 30 times higher was chosen in the experiments, which was much higher than that which we selected in the current study. Therefore, the dose in the experiments might occur below the minimum threshold, or perhaps they all arrived above the maximum threshold, which would lead to the same results. Secondly, the number of bone marrow-derived mononuclear cells selected on the previous study was 30 times higher than the MSCs at the pulmonary passage. So, in their experiments, the cell loss issues would be greatly diminished, while the advantages of the IA route would be lessened.
Conclusions
Ischemic stroke can be treated with IA hBMSCs, which we here found to exert therapeutic effects more efficiently than IV hBMSCs. IA hBMSC treatment is safe and can improve the posttransplantation engraftment and minimize cell diffusion to organs other than the brain. The functional recovery, perfusion, and metabolism of the brains of treated rats improved, suggesting that IA hBMSC treatment may be a suitable means of treating stroke patients.
Footnotes
Acknowledgments
This study was supported by grants from the National High Technology Research Project (2006AA02A115). The authors declare no conflict of interest.
