Abstract
Hematopoietic stem cell transplantation (HSCT) using umbilical cord blood (UCB) progenitors is increasingly being used. One of the problems that may arise after UCB transplantation is an impaired engraftment. Either intrabone (IB) injection of hematopoietic progenitors or mesenchymal stem cell (MSC) coadministration has been proposed among the strategies to improve engraftment. In the current study, we have assessed the effects of both approaches. Thus, NOD/SCID recipients were transplanted with human UCB CD34+ cells administered either intravenously (IV) or IB, receiving or not bone marrow (BM)-derived MSCs also IV or IB (in the right femur). Human HSC engraftment was measured 3 and 6 weeks after transplantation. Injected MSCs were tracked weekly by bioluminescence. Also, lodgment within the BM niche was assessed at the latter time point by immunofluorescence. Our study shows regarding HSC engraftment that the number of BM human CD45+ cells detected 3 weeks after transplantation was significantly higher in mice cotransplanted with human MSCs. Moreover, these mice had a higher myeloid (CD13+) engraftment and a faster B-cell (CD19+) chimerism. At the late time point evaluated (6 weeks), human engraftment was higher in the group in which both strategies were employed (IB injection of HSC and MSC coadministration). When assessing human MSC administration route, we were able to track MSCs only in the injected femurs, whereas they lost their signal in the contralateral bones. These human MSCs were mainly located around blood vessels in the subendosteal region. In summary, our study shows that MSC coadministration can enhance HSC engraftment in our xenogenic transplantation model, as well as IB administration of the CD34+ cells does. The combination of both strategies seems to be synergistic. Interestingly, MSCs were detected only where they were IB injected contributing to the vascular niche.
Keywords
Introduction
Hematopoietic stem cell transplantation (HSCT) using umbilical cord blood (UCB) is increasingly being used in order to treat malignant or nonmalignant hematologic diseases (24,31,32,41). UCB-HSCT is a suitable procedure in pediatric patients, but in adults, the hematopoietic recovery is slower than with bone marrow (BM) or peripheral blood (PB) transplantation (56). In a number of cases, engraftment is impaired or even absent, mainly because of the lower cellular dose, but other factors such as impaired microenvironment after intensive doses of chemoradiotherapy may also contribute to this (16,42). In order to increase hematopoietic recovery in the UCB transplantation setting, several approaches have been designed (16,42). Ex vivo expansion of hematopoietic stem cells (HSCs) has been proposed as a feasible method, but graft improvement has been hampered by HSC exhaustion in most experimental conditions (18,50). The use of a double cord blood unit transplantation (DCBT) has shown faster engraftment when compared to single-unit transplant, but limitations remain for a number of reasons (25,45). A third approach has been the infusion of third-party donor-mobilized HSCs, T-cells depleted, along with UCB transplantation as a “bridge engraftment” strategy till the engraftment of the latter, but their results are still very preliminary (10).
Two additional methods have been recently proposed: direct intrabone (IB) transplantation of UCB cells (22) or the coadministration of mesenchymal stem cells (MSCs) along with HSCs (30,37). The faster hematopoietic recovery with the first proposed method could be due to a better stem cell homing, preventing the cell loss observed when cells are infused intravenously (IV) (3).
The second approach is based on the fact that BM MSCs contribute to most cell types involved in BM HSC niches (36). They regulate BM homeostasis by cell-to-cell contact and through cytokine production (36). Also, these cells have shown immunomodulatory and anti-inflammatory properties (33,47). These properties make them an attractive therapeutic tool in UCB transplantation setting to enhance engraftment. Although several studies have been designed to investigate MSC capability to enhance hematopoietic engraftment as well as the mechanisms involved, controversial information has been published about their migration, engraftment capacity, and function after in vivo injection (17,37).
In this study, we have compared both procedures, IB administration of UCB cells and MSC injection in an animal model, in order to ascertain whether one of them or both could ameliorate hematopoietic engraftment.
Materials and Methods
MSC Isolation, Expansion, and Characterization
Human MSCs were isolated from BM cells of 20 healthy volunteer donors (11 males/9 females) with a median age of 43 years (range, 25–49 years) after informed consent had been obtained, in accordance with the local ethics committee of the University Hospital of Salamanca. Ten to 20 ml of BM were taken by iliac crest aspiration under local anesthesia according to standard institutional procedures. Mononuclear cells (MNCs) were isolated by Ficoll-Paque density gradient centrifugation (GE Healthcare Bio-Sciences, Uppsala, Sweden) and resuspended in Dulbecco's modified Eagle's medium–low glucose (DMEM) (Gibco, Invitrogen, Paisley, UK) supplemented with 10% fetal calf serum (FCS) (BioWhittaker, Lonza, Verviers, Belgium) and 1% penicillin/streptomycin (Gibco), as previously reported (5,46). Cells were seeded at an initial density of 106 cells/cm2 and incubated in a humidified atmosphere with 5% CO2. MSCs were expanded in T75 culture flasks (Corning, Corning, NY, USA) and expanded as previously described. After the third passage, MSCs were assessed following the minimal criteria recommended by the International Society for Cellular Therapy (ISCT) (9). Differentiation capacity to osteoblast, adipocytes, and chondrocytes was done using NH Osteodiff Medium, NH Adipodiff Medium, and NH Chondrodiff Medium, respectively (Miltenyi Biotec, Bergisch Gladbach, Germany), as previously described (5). For immunophenotypic analysis, MSCs were incubated for 15 min with fluorescein isothiocyanate (FITC)-conjugated cluster of differentiation 90 (CD90), FITC-conjugated CD44, FITC-conjugated CD34, phycoerythrin (PE)-conjugated CD73, PE-conjugated CD14, PE-conjugated CD166, phycoerythrin-cyanine 5 (PC5)-conjugated anti-human leukocyte antigen (HLA)-DR, PC5-conjugated CD45, and PC5-conjugated CD19 (all from Becton Dickinson Biosciences, San Jose, CA, USA) and allophycocyanine (APC)-conjugated CD105 (R&D Systems, Minneapolis, MN, USA). Measurements were made on a FACSCalibur flow cytometer (Becton Dickinson Biosciences). Data were analyzed using the Infinicyt program (Cytognos, Salamanca, Spain), as previously described (8).
CD34+ Cell Selection
A total of 20 fresh human UCB units (from 9 male and 11 female subjects) were obtained from the Barcelona Blood and Tissue Bank according to their ethical rules. Eighty to 100 ml of heparinized cord blood was diluted in phosphate-buffered saline (PBS; Gibco). MNCs were isolated by Ficoll-Paque density gradient centrifugation, and then CD34+ progenitor cells were isolated by magnetic labeling using the human CD34 MicroBead Kit (Milteny Biotec) in accordance with the manufacturer's protocol. After isolation, the purity of CD34+ cells was evaluated by flow cytometry using FITC-conjugated CD34 (Becton Dickinson Biosciences).
Lentiviral Vector
For the lentiviral vector (LV) used in this study, a 1.7-kb Luc2 fragment was isolated from pGL4.10(luc2) vector (Promega, Madison, WI, USA) using XhoI and XbaI restriction sites. Subsequently, this Luc2 fragment was cloned in the LV pRRL central polypurine tract cytomegalovirus internal ribosome entry site green fluorescent protein woodchuck hepatitis virus posttranscriptional regulatory element self-inactivating vector [pRRL-cPPT-CMV-IRES-GFPPRE-SIN, kindly provided by Prof. Hoeben, Leiden University Medical Center, Leiden, The Netherlands (4)] using the same restriction sites, downstream of the CMV promoter, to generate pRRL-cPPT-CMV-Luc2-IRESGFP-PRE-SIN. Expression of the luciferase gene driven by the CMV promoter was confirmed with in vitro assays (11,12).
Transfection of Virus-Producing Cells
For virus production, 2×106 human embryonic kidney (HEK) 293T cells (kindly provided by Prof. J. P. Bolaños, from the University of Salamanca, Spain) were seeded on a 15-cm diameter culture dishes (BD Falcon, San Jose, CA, USA) 1 day before transfection and maintained in DMEM supplemented with 10% FCS and the antibiotics penicillin (100 U/ml) and streptomycin (0.1 mg/ml). Briefly, 293T cells were transfected with the envelope vector pLV-vesicular stomatitis Indiana virus G protein (VSVG), the packaging vector psPax2, and the LV vector containing the luciferase (Luc2) gene ∗∗∗pRRL-cPPT-CMV-IRES-GFPPRE-SIN by using the calcium phosphate DNA precipitation method, as previously described (15). The medium containing lentiviral supernatant was collected at 48 h after transfection, filtered through a 0.45-mm pore filter (BD Falcon), pooled, and stored at −80°C until use. The titer of the virus supernatant was estimated by transducing 293T cells with different dilutions, and the transduction efficiency was determined by flow cytometry analysis of GFP. Titer was calculated using the following formula: (cell concentration at the day of transduction)×(virus supernatant dilution)×(% GFP-positive cells)/total volume.
MSC Transduction
Passage 2 MSCs were transduced when a 50% confluence of the stromal layer was observed. Transductions were carried out in the presence of 8 mg/ml of hexadimethrine bromide (polybrene; Sigma-Aldrich, Steinheim, Germany) and 1:8 LV supernatants, resulting in a multiplicity of infection of 1.5. After 24 h, the transduction medium was replaced with a fresh medium. At 90% confluence, cells were harvested, and GFP+ cells were analyzed by flow cytometry to determine transduction efficiency. The multiplicity of infection (MOI) used in this experiment was 1.5, resulting in a transduction efficiency of the MSCs of >70% on the basis of GFP expression analyzed by flow cytometry.
Transplantation of NOD/SCID Mice
Six-week-old nonobese diabetic severe combined immunodeficient (NOD/SCID; NOD.CB17-Prkdcscid/NcrCrl) mice were purchased from Charles River Laboratories (Barcelona, Spain), housed in microisolator cages, and maintained under sterile conditions in the animal facility of the University of Salamanca. All procedures followed the Spanish and European Union guidelines (RD 1201/05 and 86/609/CEE, respectively) and were approved by the Bioethics Committee of the University of Salamanca (reg. no. 201100007924). The NOD/SCID mouse transplant model was established as reported previously with modifications (20). A total of 100 female mice were used for these experiments (20 per experimental group). Six to 8 h before transplantation, animals were exposed to 300 cGy total body irradiation from a cesium (CS) source (Gammacell-200, Nordion International, Ottawa, ON, Canada). In this study, the IV or intrabone (IB) injection as well as the coadministration of MSCs were combined to test their effect on hematopoietic engraftment according to the following transplantation groups: (1) 105 IV CD34+ cells; (2) 105 IV CD34+ cells and 5×105 IB MSCs; (3) 105 IV CD34+ cells and 5×105 IV MSCs; (4) 105 IB CD34+ cells; (5) 105 IB CD34+ cells and 5×105 IB MSCs. Before transplantation, animals were anesthetized with a mixture of ketamine (90 mg/kg; Imalgene 500, Merial, Lyon, France) and xylazine (10 mg/kg; Rompun 2%, KVP Pharma, Bayer Healthcare, Kiel, Germany). For IV injection, cells were resuspended in a total volume of 200 ml of PBS and slowly injected by tail vein. For IB injection, cells were injected through the knee into the femur in a total volume of 20 ml of PBS. Animals were transplanted with MSCs from an adult donor and HSCs from a pool of three to four different UCB units, being all units from same sex donor for each pool. There were sex mismatches between HSC and MSC donors. Hematopoietic engraftment was evaluated by flow cytometry at weeks 3 and 6 after transplantation in peripheral blood (PB), injected femur (right femur), and contralateral femur (left femur) samples (52). Six weeks after transplantation, mice were killed, and their femurs were fixed in 4% formaldehyde (Panreac, Barcelona, Spain) for immunohistological analysis.
Analysis of Human Hematopoietic Engraftment by Flow Cytometry
For flow cytometry studies, PB was obtained from mice eye vein and BM samples from both femurs. All cell suspensions were collected in PBS, subjected to red cell lysis by treatment with ammonium chloride, washed once, and then resuspended again in PBS. To analyze human hematopoietic cells engraftment, samples were stained with FITC-conjugated anti-human CD45 (Miltenyi Biotec). For multilineage detection, cells were stained with APC-conjugated CD3, PE-conjugated CD13, PE-conjugated CD14, and APC-conjugated CD19 (all from Becton Dickinson Biosciences). In all cases, 10 min before acquisition, samples were stained with 7-amino-actinomycin D (7-AAD; Becton Dickinson Biosciences). An unstained control was also included for each sample. For data analysis, hematopoietic cells were gated by forward/side scatter to exclude potential contamination with GFP-transduced MSCs. Then, CD45 expression was used to gate human cells and 7AAD to exclude dead cells. Finally, human hematopoietic subpopulations were detected by CD3 (T-cells), CD13 (myeloid cells), CD14 (monocytes), and CD19 (B-cells) expression within the human CD45+ live gate. In addition, as previously indicated, a control unstained sample was acquired in each study of PB or BM, and rare unspecific positive event (if any) values were subtracted to the values of the positive events for each marker.
In Vivo Bioluminescence
In order to track the mesenchymal population in vivo, bioluminescence images (BLI) were acquired weekly using a Xenogen IVIS 50 (Caliper Life Sciences, Alameda, CA, USA) bioluminescence/fluorescence optical imaging system at different time points. A few minutes before BLI, the mice were anesthetized with isoflurane (1–3%) (Forane, Abbot Laboratories, Madrid, Spain). Mice were given the substrate d-luciferin (150 mg/kg in PBS) (Promega) by intraperitoneal injection. Ten minutes after administration of substrate, the anesthetized mice were placed onto the warmed stage inside the light tight camera box with continuous exposure to isoflurane (1–2%). After acquiring photographic images of mice, luminescent images were acquired with 2-min exposure times. Exposure conditions (time, aperture, stage position, binning, and time after injection) were kept identical in all measurements. For quantification, standard regions of interest (ROIs) were defined for femurs and lungs separately. Measurements were expressed as photons per second. The resulting gray scale photographic and pseudocolor luminescent images were automatically superimposed so that identification of any optical signal with location on the mouse was facilitated. Optical images were displayed and analyzed with IVIS Living Image (Caliper Life Sciences) software packages.
Immunohistochemistry
Femurs were fixed, decalcified for 24 h, and embedded in paraffin, and 5-μm-thick tissue sections were placed on glass slides. The sections were then incubated with 5% normal donkey serum and bovine serum albumin to block nonspecific binding. In order to detect cells from human origin, a primary antibody for human mitochondria detection (Millipore, Billerica, MA, USA) was used. For detection of human MSCs, endogenous GFP signals were amplified by staining sections with rabbit anti-GFP antibody (AbCam, Cambridge, UK). As secondary antibody, Alexa Fluor448-conjugated anti-rabbit IgG and Alexa555 anti-mouse IgG were used (both from Invitrogen). After washes, 1 μg/ml 4′,6-diamidine-2′-phenyl indole, dihydrochloride (DAPI; Sigma-Aldrich) in PBS was added, and the cells were incubated at room temperature for 5 min. Slides were viewed with a Leica DMI6000B fluorescence microscope (Leica Microsystems GmbH, Wetzlar, Germany) equipped with camera system.
Statistical Analysis
Median and ranges were calculated for each variable. To estimate the significance of the differences between the groups, the nonparametric Mann–Whitney U test was used. As a result of multiple subgroup assessments, a modified Bonferroni correction (α/5) was used to adjust the p value. Differences were considered to be significant for values of p < 0.05. All statistical analyses were done with SPSS 17.0 (Chicago, IL, USA).
Results
MSCs from BM samples were expanded in all cases. All of them adhered to plastic surfaces; were capable of differentiating into adipocytes, osteoblasts, and chondrocytes; expressed the antigens CD44, CD73, CD90, CD105, and CD166; and were negative for hematopoietic antigens following the criteria defined by the ISCT (9) for MSC use. In all cases, we were able to transduce MSCs. Transduction efficiency of the MSCs was >70% on the basis of GFP expression analyzed by flow cytometry (data not shown). CD34+ cells were isolated from all UCB samples. Cell purity was above 85% in all cases.
Human Hematopoietic Engraftment
In order to test the optimal transplantation combination, human hematopoietic engraftment was analyzed in NOD/SCID mice. Human cells were detected in all cases by flow cytometry. Mice receiving only IV CD34 were used as controls.
Human engraftment was expressed as percentage of human CD45+ cells present among the total number of BM or PB MNCs (Fig. 1). Regarding the injected femur (right), 3 weeks after transplantation (Fig. 1A), higher human hematopoietic cells were observed in all new combinations when compared with CD34+ cells IV injection. The highest percentage of human chimerism was observed in mice receiving IV infusion of both cell types (MSCs and CD34+ cells). Regarding left femurs only mice receiving IV, CD34+ cells together with MSCs showed a higher human hematopoietic chimerism than other groups, independent of the injection route of MSCs (Fig. 1B).

Human hematopoietic engraftment in nonobese diabetic/severe combined immunodeficient (NOD/SCID) bone marrow. Percentage of human CD45+ cells in bone marrow samples from both femurs, right (A, C) (injected site) and left (contralateral) (B, D), at 3 (A, B) and 6 (C, D) weeks after transplantation. Results expressed as medians (range). IV, intravenous injection; IB, intrabone injection in right femur; CD34, human CD34+ cells; MSC, mesenchymal stem cells.
Six weeks after transplantation (Fig. 1C, D), the highest BM human engraftment was observed in mice which had received MSCs and CD34+ cells by IB injection (Fig. 1C). In the left femur, no differences were observed at week 6 (Fig. 1D).
When percentage of human CD45+ cells was analyzed in PB (Fig. 2), no differences among the different study groups and time points were observed (Fig. 2A, B).

Human hematopoietic cells in NOD/SCID peripheral blood. Percentage of human CD45+ in total mononuclear cells in peripheral blood (A, B) as well as percentage of myeloid CD13+ cells (C, D) and lymphoid CD19+ cells (E, F) into the human population at 3 (A, C, E) and 6 (B, D, F) weeks after transplantation. Results are expressed as medians (range). IV, intravenous injection; IB, intrabone injection in right femur; CD34, human CD34+ cells; MSC, mesenchymal stem cells.
In order to analyze the engraftment of myeloid cells, monocytes, B- and T-cells, CD13+, CD14+, CD19+, and CD3+ cell subsets were determined, respectively, within the human CD45+ cell population.
At week 3, when PB was evaluated, only the group receiving both cell types intravenously showed higher level of human myeloid cells (Fig. 2C). Also, CD19+ cells were only detected in MSC-receiving groups (Fig. 2E). Six weeks after transplantation, in PB, the groups with HSCs IV and MSCs showed higher level of lymphoid cells (Fig. 2F).
At 3 weeks, in BM, the majority of human cells showed CD13 expression with similar number of positive cells in all groups (Fig. 3A, B). CD19+ cells were mainly detected in MSC-receiving groups (Fig. 3C, D) producing higher differences with no MSC groups.

Human hematopoietic subpopulations in NOD/SCID bone marrow at week 3. Percentage of hematopoietic subpopulation of human myeloid cells (CD13+) (A, B) and lymphoid cells (CD19+) (C, D) within the total human population in bone marrow from both femurs right (A, C) (injected site) and left (contralateral) (B, D) at week 3 after transplantation. Results expressed as medians (range). IV, intravenous injection; IB, intrabone injection in right femur; CD34, human CD34+ cells; MSC, mesenchymal stem cells.
In the latest period, in BM, the percentage of myeloid cells among human population was decreased, while the CD19+ population was increased (Fig. 4). In all groups, the percentage of CD19+ cells in both femurs was more than 30% with the highest level in the left femur of mice receiving both MSCs and HSCs by IV injection (Fig. 4D).
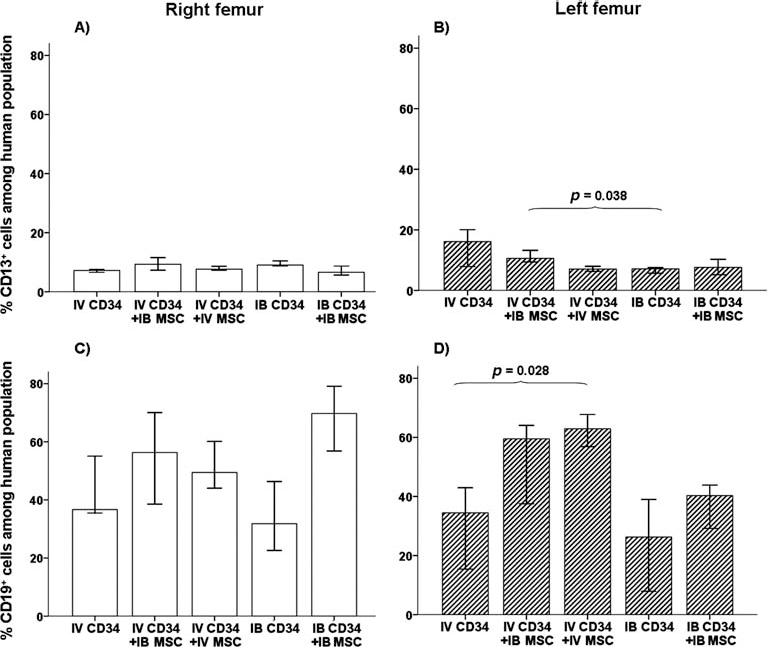
Human hematopoietic subpopulations in NOD/SCID bone marrow at week 6. Percentage of hematopoietic subpopulation of human myeloid cells (CD13+) (A, B) and lymphoid cells (CD19+) (C, D) within the total human population in bone marrow from both femurs, right (A, C) (injected site) and left (contralateral) (B, D), at week 6 after transplantation. Results expressed as medians (range). IV, intravenous injection; IB, intrabone injection in right femur; CD34, human CD34+ cells; MSC, mesenchymal stem cells.
Monitoring of Luciferase-Transduced MSCs in Mice
Human-transduced MSCs were injected IV or IB in mice at the time of hematopoietic transplantation. The day after IV injection of MSCs, cells were mainly detected in lungs (Fig. 5). In some cases, a focal spot of bioluminescence was observed at the injection site. After IB injection, cells were detected at two levels: lungs and right femurs (injection site). No differences were observed regarding the hematopoietic transplantation route; both groups receiving IB MSC injection with IV or IB CD34 cells had a similar distribution of MSCs.

In vivo bioluminescence imaging of NOD/SCID mice. In transplantation groups receiving human MSCs (which expressed luciferase), a weekly imaging of MSC distribution has been done. A representative animal is shown. The reflected light gray scale picture is overlaid by a pseudocolor bioluminescence image (BLI), which allows for allocating the BLI signal to the respective areas shown in the underlying reflected light picture.
In all cases, signal from lungs decreased and was not detectable 2 weeks after injection. By contrast, signal from injected femurs could be detected during the whole study period (Fig. 5).
For luciferase signal quantification, defined ROIs were used over the lung and right femur.
The mean peak BLI signal intensities of 1 day after transplantation was 3.1×109, 5.9×109, and 4.6×109 photons per second in IV CD34+ IB MSCs, IV CD34+ IV MSCs, and IB CD34+ IB MSCs, respectively. After 14 days, BLI signal intensities decreased in lungs to background signal intensity (Fig. 6A). However, BLI signal intensities of groups with IB MSC injection was maintained from day +1 to day +42: 4.8×1010 to 4.9×109 and 4.6×1010 to 3.1×109 in photons per second in IV CD34+ IB MSCs and IB CD34+ IB MSCs, respectively (Fig. 6B).

Quantitative analysis of BLI production. The in vivo presence of human MSCs was measured by bioluminescence photometry. In mice lungs, photon flux was going down, and it was not detectable after week 2 (A). By contrast, in previously injected femurs, the signal was reduced during the first 4 weeks, but then its photon emission was maintained (B).
Detection of Human Cells Within Murine BM
Six weeks after transplantation, mice were killed, and femurs were fixed for immunofluorescence detection of human cells. Human mitochondria staining allowed detecting human cells and GFP expression and the differentiation between human MSCs (GFP+) and human hematopoietic cells (GFP-).
Human hematopoietic cells in BM were mainly distributed in femur epiphysis showing higher numbers close to the injected site (epiphysis near to the knee) in groups receiving IB injection (Fig. 7A–C). These cells formed colonies of round-shaped cells and were growing together close to subendosteal region (Fig. 7D–F). Usually, cases with higher chimerism showed a similar trend of human cells by immunohistochemistry.
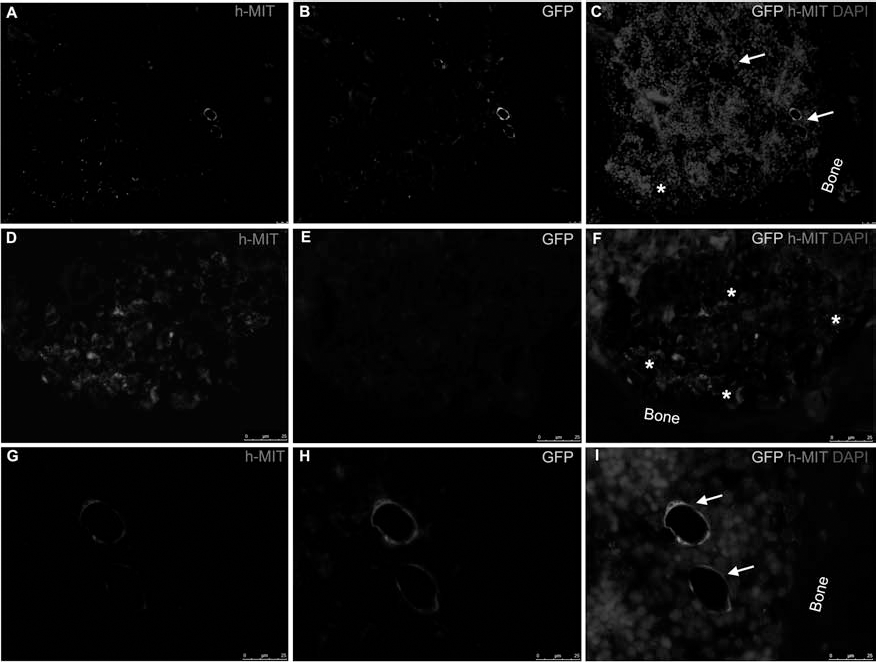
Immunohistochemical analyses of human mitochondria and GFP expression in mice femurs. With the human mitochondria staining, human cells could be detected and green fluorescent protein (GFP) expression and amplification allowed differentiating human MSCs (GFP+) and human hematopoietic cells (GFP-). Six weeks after transplantation, both human cells types, MSCs (→) and hematopoietic cells (∗), were mainly detected in femur epiphysis (A–C). Hematopoietic cells were forming cluster of human cells in subendosteal area (A–C, D–F). MSCs could be detected around blood vessels (A–C, G–I). Scale bars: 25 μm.
Human MSCs were only detected in femurs where they were previously injected and were mainly located in femur epiphysis (Fig. 7A–C), but few GFP-positive cells could be detected in each femur. These cells showed fibroblastic or polygonal shape and could be detected at two levels: some of them were distributed between hematopoietic cells or close to the endosteum, but others showed a clear perivascular distribution (Fig. 7G–I). Blood vessels surrounded by human MSC-derived cells were located in a subendosteal region but not associated with human hematopoietic cells.
Discussion
Although both strategies have been shown to be useful separately (6,35,40,55), in the current manuscript, we have assessed together the role of intrabone administration of HSC and MSC coadministration in the cord blood xenotransplantation model. To study their homing ability and functional dynamics, MSCs were administered via intrafemoral and were transduced with a luciferase-expressing vector and thus tracked by bioluminescence.
UCB has emerged as an alternative source of HSCs for transplantation, and several studies have been focused for strategies to enhance UCB engraftment in adult patients (16,42). A higher cryopreserved MNC dose has been associated with faster and higher probability of neutrophil recovery and a higher CD34+ cell dose with better disease-free survival (19). Since the low number of cells is critical, other strategies that could be used include IB transplantation of UCB cells in order to prevent cell losses (3,13). In a previously reported paper, Massollo et al. (34) demonstrated, using a rat model, that after IB injection, most HSCs escaped to circulating blood as soon as 30 min after their administration, but their first entry contact with the recipient BM deeply modifies the kinetic and migratory characteristics of donor HSCs increasing their capability to cross the filter of the pulmonary capillary network and improves their homing in BM sites remote from the injection. Our results comparing IV and IB UCB transplantation showed that after IB transplantation, human hematopoietic cell engraftment is not only higher in the injection site but also demonstrates their capability to migrate far from injection sites. This is in accordance with the previous data that suggest that IB injection permits engraftment even without previous irradiation (3).
An alternative to enhance HSC engraftment is to improve BM microenvironment function. The influence of BM stroma in hematopoiesis and the axis stromal-derived factor 1/chemokine (C-X-C motif), receptor 4 (SDF1/CXCR4) is a key regulator of both homing and lodgment of HSCs in the niche (38). In this line, improving BM stroma by the coadministration of MSCs is an attractive approach. Several studies have been designed to test this possibility by using MSCs from different sources (21,30,37). In this regard, using placental MSCs in a murine model, Hiwase et al. showed that MSC coinfusion could increase both single and double unit UCB transplantation (21). In the present study, the effect of MSC coinfusion in both transplantation settings, IV or IB injection of HSCs, was tested. We could show in accordance with previous data that when IV transplantation was performed, the addition of MSCs increased human chimerism. When MSC injection route was analyzed, the human engraftment was higher even in femurs not receiving MSCs, demonstrating the capability of MSCs to migrate or to act far from the injection site. The effect of MSCs was higher in the injected femur, and it was more evident 6 weeks after transplantation. Although it was not the aim of the current study, taken into account our results, it would be of interest to perform the xenotransplant experiments with limiting doses of hematopoietic stem cells and also with different doses of MSCs. This should be considered in the future to reinforce the results and to assess the effects of the cell dose in this setting.
It would be also of interest to assess in future works if the same strategy (intrabone administration with or without MSCs) also enhances engraftment of human PB or BM hematopoietic stem cells, although in PB or BM HSCT the cell dose is not as relevant as in the cord blood transplantation setting. Nevertheless, it could be useful in the autologous transplantation, especially in those cases of multitreated patients or poor mobilizers.
The next aim of the present work was to know how MSCs exert their effect. Data relating to their engrafting capability in recipient tissues are controversial (1,27,53). In the current study, we tracked the human MSCs until 6 weeks after transplantation by using BLI. The use of BLI for detection of spatial distribution of transplanted MSCs was validated by detecting emitted photons. Sequential evaluation of grafted cell viability was possible with the BLI system since the luciferin–luciferase reaction depends on ATP, and only living cells release photons (23). The first studies tried to find MSC in BM recipients after HSCT (27,43,53). Due to the low number of MSCs present in BM samples, only in cases of patients with osteogenesis imperfecta (27) or a disease with high stromal damage some MSCs from donor origin could be detected (53). It has been reported that, when MSCs have been IV injected, most of them were retained in lungs microvasculature, and they could not be detected in BM (54). These data are in accordance with our results. By bioluminescence, when cells were IV injected, they were only detected during 2 weeks after transplantation. Using a more sensitive technique as immunohistochemistry, 6 weeks after transplantation, human MSCs could not be detected either.
To improve MSC local effects, new trials searching for new routes of administration are being explored (49,51). Local injection of MSCs as intracoronary (57), intrautero (27), kidney (7), or skin injection (48) showed higher engraftment. Taking in account the results in IB injection of hematopoietic cells (22,44), we have considered that this route of delivery could be optimal for MSC administration. In fact, some groups are testing the use of this cells by IB injection in cases of bone damage as multiple myeloma (28). We have shown that, when MSCs were locally injected (in BM), they could engraft in recipient BM, and they could be followed for at least 6 weeks.
Their spatial distribution within the BM was also studied. For years, the main niche for HSCs in BM has been the osteoblastic niche (2,39). Due to their osteoblastic differentiation capability, endosteal region has been assumed as an optimal niche for MSCs (39). We were able to detect human MSCs near the BM endosteum. But recent studies have described another vascular niche, located in BM sinusoids, where HSCs could also be found (14). Some groups have described an MSC population located around these blood vessels (2,29). After the immunofluorescence analysis, we could also find human MSCs, GFP-positive, around blood vessels close to the subendosteal region. This fact is also in accordance with recent data suggesting that endothelial cells might form part of the subendosteal niche and together constitute a common niche where both endosteum and sinusoids contribute to hematopoiesis (2).
In summary, our results show that in our model MSCs could increase hematopoietic engraftment in both IB and IV hematopoietic transplantation. Nevertheless, they were only found in BM recipients after IB injection of MSCs showing a subendosteal and perivascular location within the BM.
Footnotes
Acknowledgments
We thank Miryam Santos for technical assistance. This study was supported in part by a grant from Gerencia Regional de Salud de Castilla y León (ref. GRS/222/A/08) and by a grant from Consejería de Educación de la Junta de Castilla y León (ref. HUS003A10-2). S.C. was supported by Junta de Castilla y Leon (FPI grant EDU/1878/2006). The authors declare no conflicts of interest.
