Abstract
Preclinical trials confirmed the potential of mesenchymal stromal cells (MSCs) to improve functional recovery after experimental stroke. Beneficial effects of MSCs are often attributed to their immunosuppressive/immunomodulatory functions. Surprisingly, the influence of MSCs on the immune system after stroke is poorly understood, but requires special consideration because cerebral ischemia is associated with stroke-induced immunodepression that predisposes to bacterial infections with increased mortality. In this study, we intravenously transplanted syngeneic murine bone marrow-derived MSCs (mMSCs) into C57BL/6 mice at 6 hours after transient middle cerebral artery occlusion (MCAo; 60 minutes) to investigate the impact of MSCs on stroke-induced immunodepression. Transplantation of syngeneic splenocytes or phosphate-buffered saline (PBS) served as controls. An immune status was determined by flow cytometry on days 3 and 14 after MCAo, which did not reveal any negative effects of cell transplantation on stroke-induced immunodepression. Although our mMSCs were found to exert immunosuppressive effects
Introduction
Mesenchymal stromal cells (MSCs) are a heterogeneous population of stem and progenitor cells with tissue-protective and regenerative capacities. Beneficial effects of MSCs have been demonstrated in rodent models of stroke, where intravenous administration of bone marrow-derived MSCs resulted in smaller infarct volumes and improved functional recovery (Chen et al, 2001; Kurozumi et al, 2005). The therapeutic capacities of MSCs were mainly attributed to their paracrine neuroprotective effects (Munoz et al, 2005; Scheibe et al, 2012). However, MSCs are also able to modulate adaptive and innate immune responses, which has been exploited therapeutically in animal models of immune disorders, such as multiple sclerosis (Constantin et al, 2009; Zappia et al, 2005) or graft-versus-host disease (GvHD) (Le Blanc et al, 2004). Among their immunosuppressive effects, MSCs were found to inhibit the proliferation of T-lymphocytes, B-lymphocytes, and natural killer-cells, dampen the respiratory burst of neutrophils, and reduce the antigen-presenting functions of dendritic cells (Singer and Caplan, 2011; Uccelli et al, 2008). This may be desired in ongoing clinical trials using MSCs for the treatment of autoimmune disorders like multiple sclerosis (Uccelli et al, 2011). However, focal cerebral ischemia (stroke) is associated with severe immunodepression and increased susceptibility to bacterial infections, which is the leading cause of stroke-related mortality (Dirnagl et al, 2007). Thus, additional immunosuppressive effects of MSCs may be detrimental. In fact, stroke-induced immunodepression is characterized by autonomic dysfunction and a transient depletion of T-, B-, and natural killer-cell populations, as well as suppression of their effector functions (Prass et al, 2003). Since none of the preclinical or clinical MSC stroke trials has addressed the effects of MSC treatment on stroke-induced immunodepression (Bang et al, 2005; Chen et al, 2001; Honmou et al, 2011; Kurozumi et al, 2005; Lee et al, 2010), we studied the impact of intravenous MSC transplantation on the peripheral immune responses to transient middle cerebral artery occlusion (MCAo) in adult mice.
Materials and methods
Animals and Experimental Model of Stroke (Middle Cerebral Artery Occlusion)
All animal experiments were performed in accordance with ARRIVE guidelines, national and institutional guidelines and were approved by the LAGeSo. Male C57BL/6 mice aged 8 to 14 weeks (Charles River, Sulzfeld, Germany) were exposed to a 60-minute filamentous MCAo as described (Katchanov et al, 2001). In sham-operated mice, an identical surgical procedure was performed, but the filament was directly withdrawn to allow immediate reperfusion.
Six hours after MCAo, mice were transplanted by tail vein injection with either 1 × 106 murine MSCs (mMSCs), 1 × 106 splenocytes from C57BL/6 mice, or with the vehicle PBS in a volume of 400
Infarct Volumetry
Histological determination of infarct lesion volume was performed at 24 hours or 3 days after MCAo or sham surgery. The brains were cut into 20
Cell Culture of Murine Mesenchymal Stromal Cells and PKH26 Labeling
Murine MSCs were obtained from the bone marrow of tibias and femurs of C57BL/6 mice aged 8 to 12 weeks (BfR, Berlin, Germany). Murine MSC isolation and culture techniques, their characterization by cell surface epitope expression and differentiation assays into fat, bone, and cartilage were previously described (Scheibe et al, 2011).
For the labeling of mMSCs with PKH26 (Sigma-Aldrich, Schnelldorf, Germany), cells were washed with serum-free alphaMEM (Biochrom, Berlin, Germany), centrifuged and resuspended in a 16 × 10−6 mol/L PKH26 staining solution. After incubation at 25°C for 5 minutes, the staining reaction was blocked by adding 1 mL fetal calf serum (Biochrom) for 1 minute. After three washing steps, the cells were counted and prepared for injection.
Mixed Lymphocyte Reaction and Mitogen-Stimulated Proliferation Assay
Immunosuppressive functions of mMSCs were investigated
Blood Samples and Preparation of Cell Suspensions
Mice were killed and blood specimens were collected into heparinized or citrate-filled syringes by puncture of the inferior vena cava. Whole blood samples were diluted 1:1 with PBS and peripheral blood mononuclear cells (PBMCs) were isolated by density gradient centrifugation with Histopaque 1083 at 400
Single-cell suspensions from spleen and lymph nodes (mediastinal, axillary, brachial, pancreatic, renal, mesenteric, lumbar, inguinal, and iliac) were prepared by forcing a donor spleen or pooled lymph nodes from one animal diluted in 5 mL PBS through a 100-
For the isolation of lung leukocytes, the right ventricles of mice were punctured and the lung circulation was perfused with PBS to remove blood cells. Thereafter, the lung tissue was digested with DNase I (Sigma-Aldrich) and collagenase D (Roche, Mannheim, Germany) following the protocol ‘Preparation of single-cell suspensions from mouse lung’ provided by Miltenyi Biotech (Bergisch Gladbach, Germany). Thereafter, pulmonary leukocytes were purified by MACS (magnetic-activated cell sorting) from single-cell suspensions of the lung after positive selection with mouse CD45-microbeads (Miltenyi Biotech) in accordance to the manufacturer's instructions. CD45-positive pulmonary leukocytes were washed with RPMI 1640 and counted using trypan blue. Magnetic-activated cell sorting separations were controlled by surface staining and FACS analysis revealed purity between 98.1% and 99.4% of CD45-positive selections.
Flow Cytometry
Immune status was obtained from PBMCs, mononuclear splenocytes, and lymph node cells by flow cytometric analysis of different leukocyte subsets. Cell suspensions were stained with the following antibodies: rat anti-mouse CD3 (17A2), rat anti-mouse CD4 (GK1.5), rat anti-mouse CD8a (53-6.7), rat anti-mouse CD11b (M1/70), rat anti-mouse CD19 (6D5), rat anti-mouse CD25 (3C7), rat anti-mouse CD45 (30-F11), rat anti-mouse CD86 (GL-1) and rat anti-mouse I-A/I-E (M5/114.15.2) (all BioLegend, Fell, Germany). Flow cytometric measurements were acquired on a MACSQuant Analyzer (Miltenyi Biotec) and analyzed using the FlowJo software (Tree Star, Ashland, OR, USA).
Since immunodepression after stroke correlates with infarct size, only MCAo animals with a direct infarct volume >35 mm3 were included in the immune status data analysis (excluded animals MCAo 3 days: PBS
Cytokine Secretion
Cytokine secretion was assessed in cell culture supernatants of
Ex-Vivo Stimulation Assays
Heparinized whole blood samples from animals killed 24 hours after MCAo were diluted 1:5 in RPMI 1640 (Biochrom) and were stimulated with 100 ng/mL lipopolysaccharide (LPS; endotoxin) from
Statistical Analysis
Statistical analysis was performed using the software GraphPad Prism 5.01 (GraphPad Software, La Jolla, CA, USA). Different statistical tests were applied: unpaired
Results
Murine Mesenchymal Stromal Cells Show Immunosuppressive Effects In Vitro
Immunosuppressive functions of mMSCs were assessed
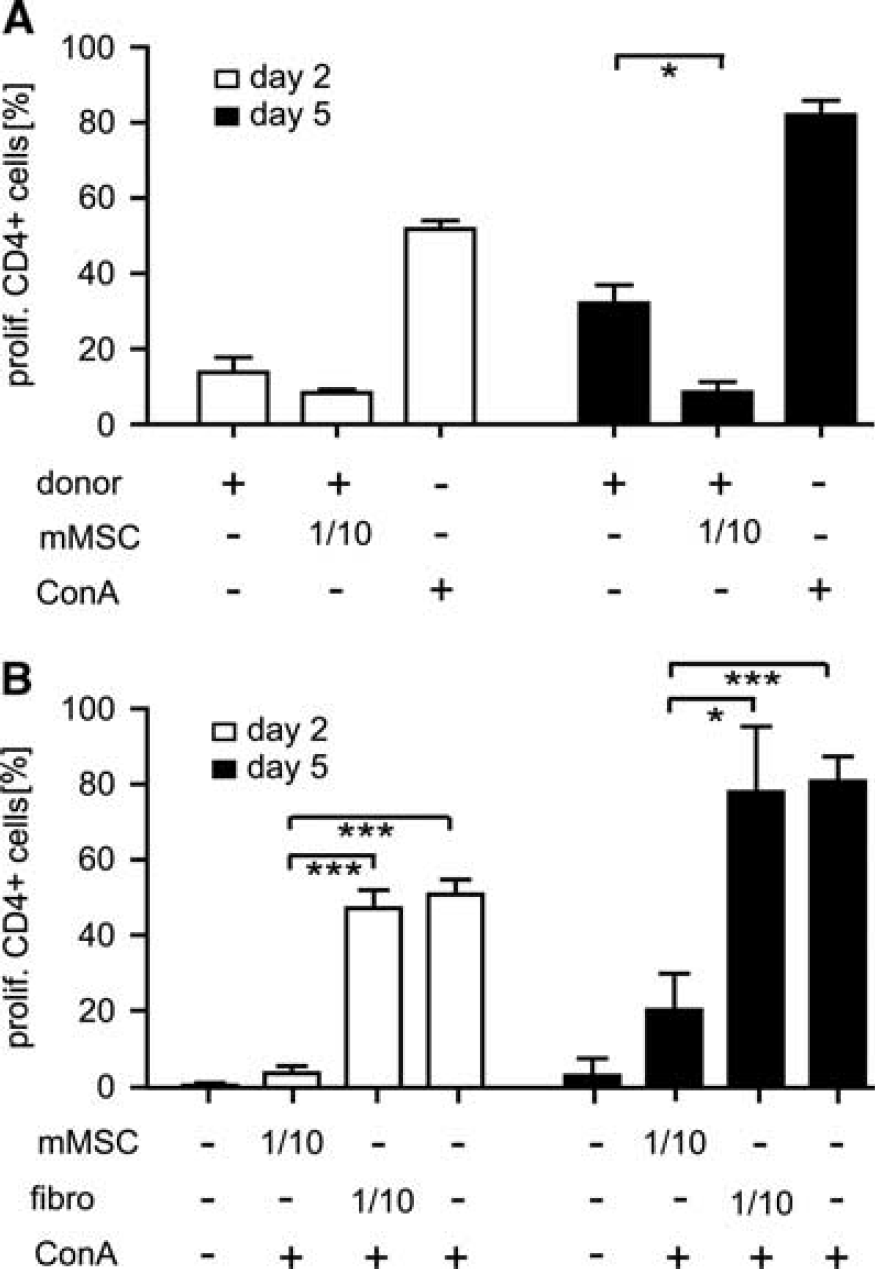
Murine mesenchymal stromal cells (mMSCs) suppress allogeneic T-cell proliferation
Transplantation of Murine Mesenchymal Stromal Cells After Focal Cerebral Ischemia in Mice
Adult C57BL/6 mice were subjected to MCAo for 60 minutes. At 6 hours after reperfusion, three groups of animals received either 1 × 106 mMSCs, 1 × 106 splenocytes (as cellular control), or PBS (as vehicle control) by tail vein injection. All MCAo animals of mMSC, splenocyte or PBS groups showed no significant differences in indirect infarct volumes at 3 days after MCAo (Figure 2A). The same was true for direct infarct volumes (mMSC group: 114.4±42.1 mm3, splenocyte group: 118.9±32.3 mm3, PBS group: 113.6±22.0 mm3). A preplanned (
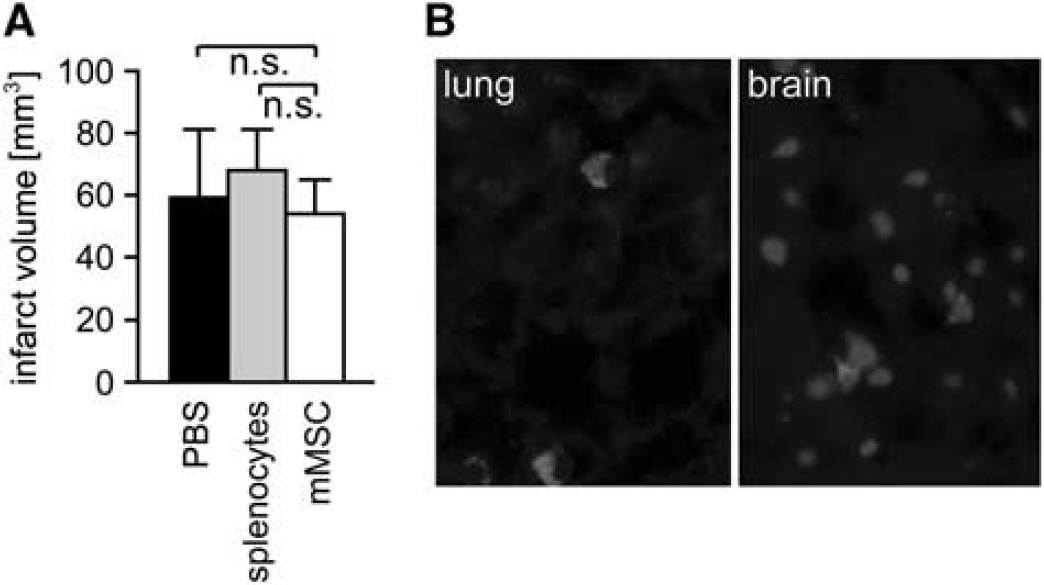
Murine mesenchymal stromal cells (mMSCs) engraft in organs, but have no impact on infarct volumes after stroke. (
Murine Mesenchymal Stromal Cells Do Not Affect Stroke-Induced Immunodepression in Mice
An immune status was obtained from PBMCs, lymph nodes, and spleens of C57BL/6 mice after MCAo and intravenous injection of mMSCs, splenocytes, or PBS. Sham-operated mice and naive mice served as controls. Absolute cell counts of different leukocyte subpopulations were quantified by flow cytometry. At 3 days after MCAo, a striking twofold to threefold reduction of T-cells (CD4+ and CD8+), B-cells (CD19+), and monocytes (CD11b+) was observed in peripheral blood (Figures 3A–3D). Moreover, we found a dramatic 5- to 15-fold loss of T- and B-cells, but also of CD11b+ macrophages in lymph nodes (Figures 4A–4D) and in spleens (Figures 5A–5D) after MCAo. Mild lymphopenia and reduction of monocytes/macrophages were also detected in sham-operated animals, but to a much lesser extent, suggesting that the observed findings were more related to ischemia rather than to surgical stress. Most importantly, mMSC administration did not induce any changes of complete cell counts and also not of defined leukocyte subsets in MCAo, sham, or PBS control groups in peripheral blood (Figure 3), lymph nodes (Figure 4), and spleens (Figure 5). In order to analyze whether mMSC effects on the immune system might occur later in the course of stroke-induced immunodepression, we also analyzed the immune status in blood, lymph nodes, and spleens at 14 days after stroke. The absolute cell counts of different leukocyte populations remained decreased in blood (Figures 3A–3D), but started to increase in lymph nodes (Figures 4A–4D) and have nearly recovered in spleens (Figures 5A–5D). Again, no effect of mMSCs on immune cell recovery after stroke was observed. Administration of splenocytes also remained without effect on poststroke immunodepression in mice (Figures 3, 4 and 5).
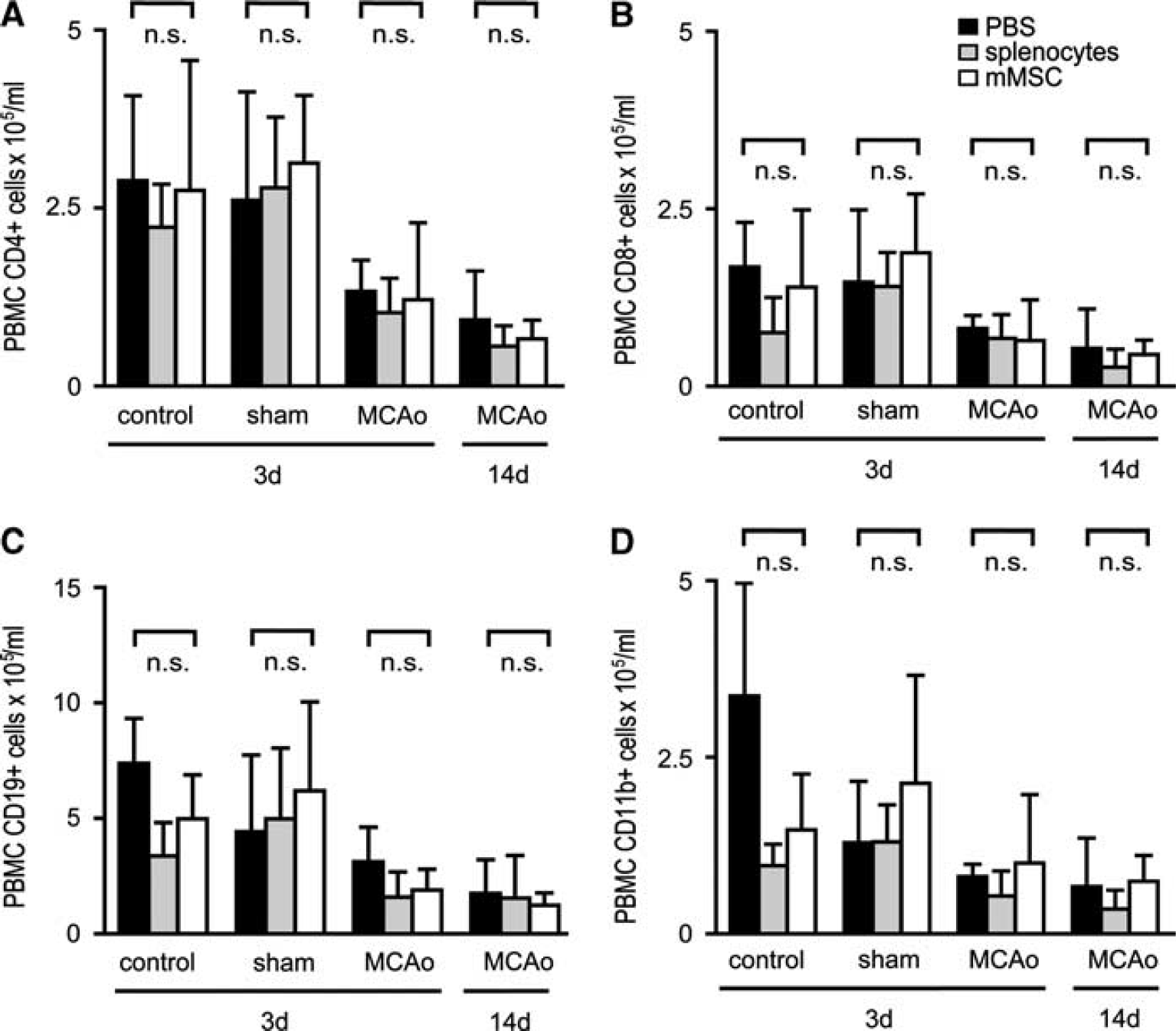
Murine mesenchymal stromal cell (mMSC) administration has no impact on stroke-induced immunodepression in peripheral blood mononuclear cells (PBMCs). Absolute cell counts of PBMC leukocyte subsets in control animals, sham-operated animals, and mice receiving 60 minutes middle cerebral artery occlusion (MCAo) were determined by flow cytometry. In sham-operated animals, reperfusion was allowed for 3 days. Mice with MCAo were analyzed after 3 days (MCAo 3d) or 14 days (MCAo 14d). All mice were injected intravenously with either phosphate-buffered saline (PBS) (black bars), splenocytes (gray bars), or mMSCs (white bars) after 6 hours. Absolute cell numbers of CD4+ T-lymphocytes (
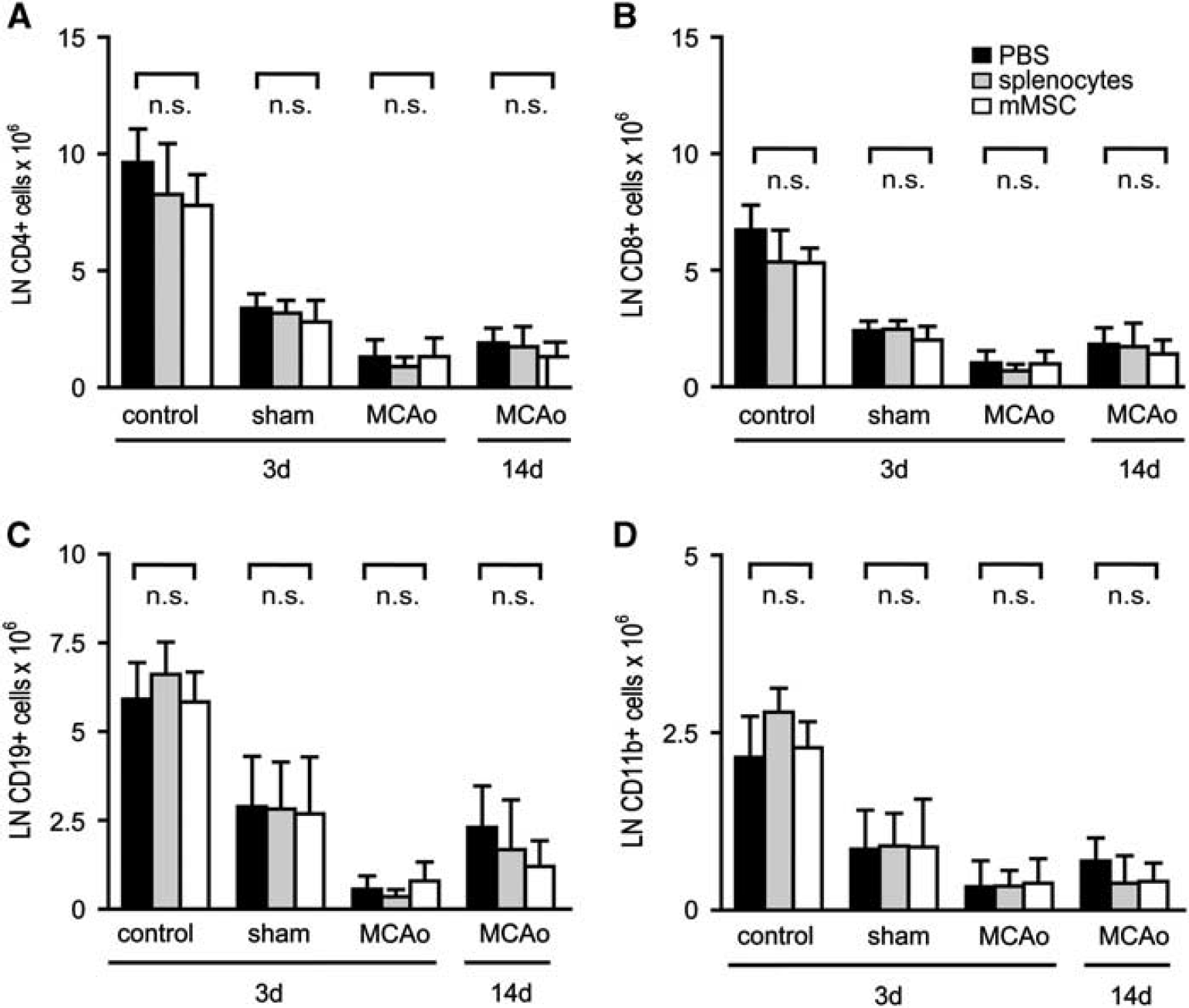
Murine mesenchymal stromal cell (mMSC) administration does not affect poststroke immunodepression in lymph nodes. Absolute cell counts of leukocyte subsets in lymph nodes (LN) of control animals, sham-operated animals, and mice receiving 60 minutes middle cerebral artery occlusion (MCAo) were analyzed by flow cytometry. After ischemia, reperfusion was allowed for 3 days (sham and MCAo 3 days) or 14 days (MCAo 14 days). All mice were injected intravenously with either phosphate-buffered saline (PBS) (black bars), splenocytes (gray bars), or mMSCs (white bars) after 6 hours. Absolute cell numbers of CD4+ T-lymphocytes (
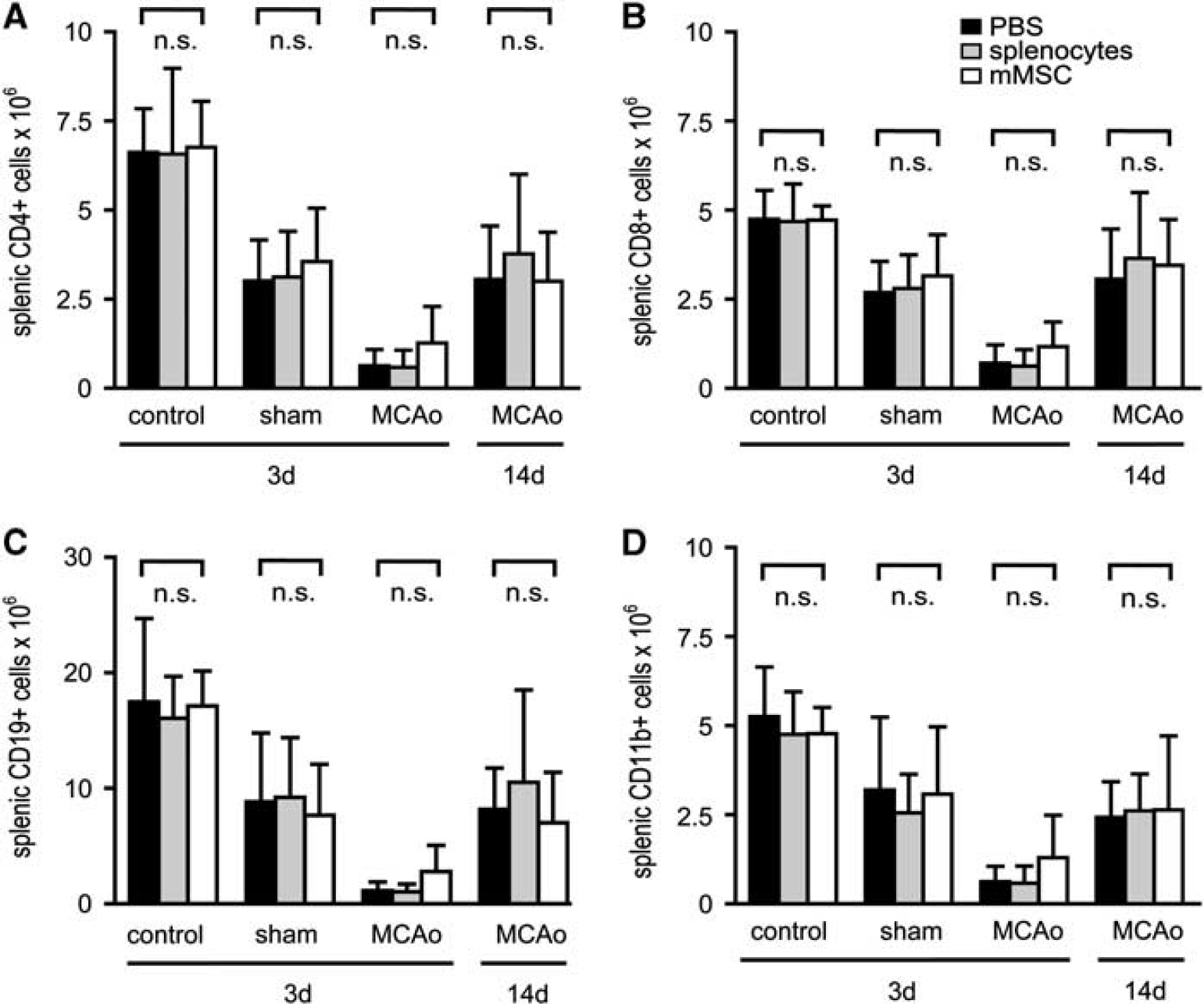
Murine mesenchymal stromal cell (mMSC) administration has no effect on stroke-induced immunodepression in mononuclear splenocytes. Absolute cell counts of mononuclear leukocyte subsets in spleens of control animals, sham-operated animals, and mice receiving 60 minutes middle cerebral artery occlusion (MCAo) were determined by flow cytometry. In sham-operated animals, reperfusion was allowed for 3 days. Mice with MCAo were analyzed after 3 days (MCAo 3 days) or 14 days (MCAo 14 days). All mice were injected intravenously with either phosphate-buffered saline (PBS) (black bars), splenocytes (gray bars), or mMSCs (white bars) after 6 hours. Absolute cell numbers of CD4+ T-lymphocytes (
In the MCAo experiments, we excluded mice with a direct infarct lesion size <35 mm3 to maintain comparability (see Materials and methods section). The excluded animals were also fully evaluated. The results are shown in Supplementary Figures S3A–S3C for the 14 days after MCAo groups. Interestingly, animals with smaller infarct volumes (<35 mm3) showed less CD8+ lymph node cells than animals with larger infarct volumes (>35 mm3) in all three experimental groups (PBS, splenocytes, MSCs).
Moreover, we quantified the following absolute cell counts of immune cell populations from PBMCs, lymph nodes, and spleens: whole leukocyte count (CD45), activated T-cell count (CD45/CD4/CD25), major histocompatibility complex class II expression on leukocytes, CD11b+ monocytes/macrophages and B-cells, and also CD86 expression on monocyte/macrophages and B-cells. Data were obtained from control, sham, and MCAo mice at 3 and 14 days after cerebral ischemia. Absolute cell counts confirmed the significant reduction of leukocyte numbers as a result of stroke-induced immunodepression in peripheral blood, lymph nodes, and spleens. No difference between the mMSC, splenocyte, and PBS groups were observed at any time point (Supplementary Figures S4–S6).
Murine Mesenchymal Stromal Cells Do Not Restore Immune Cell Function After Stroke
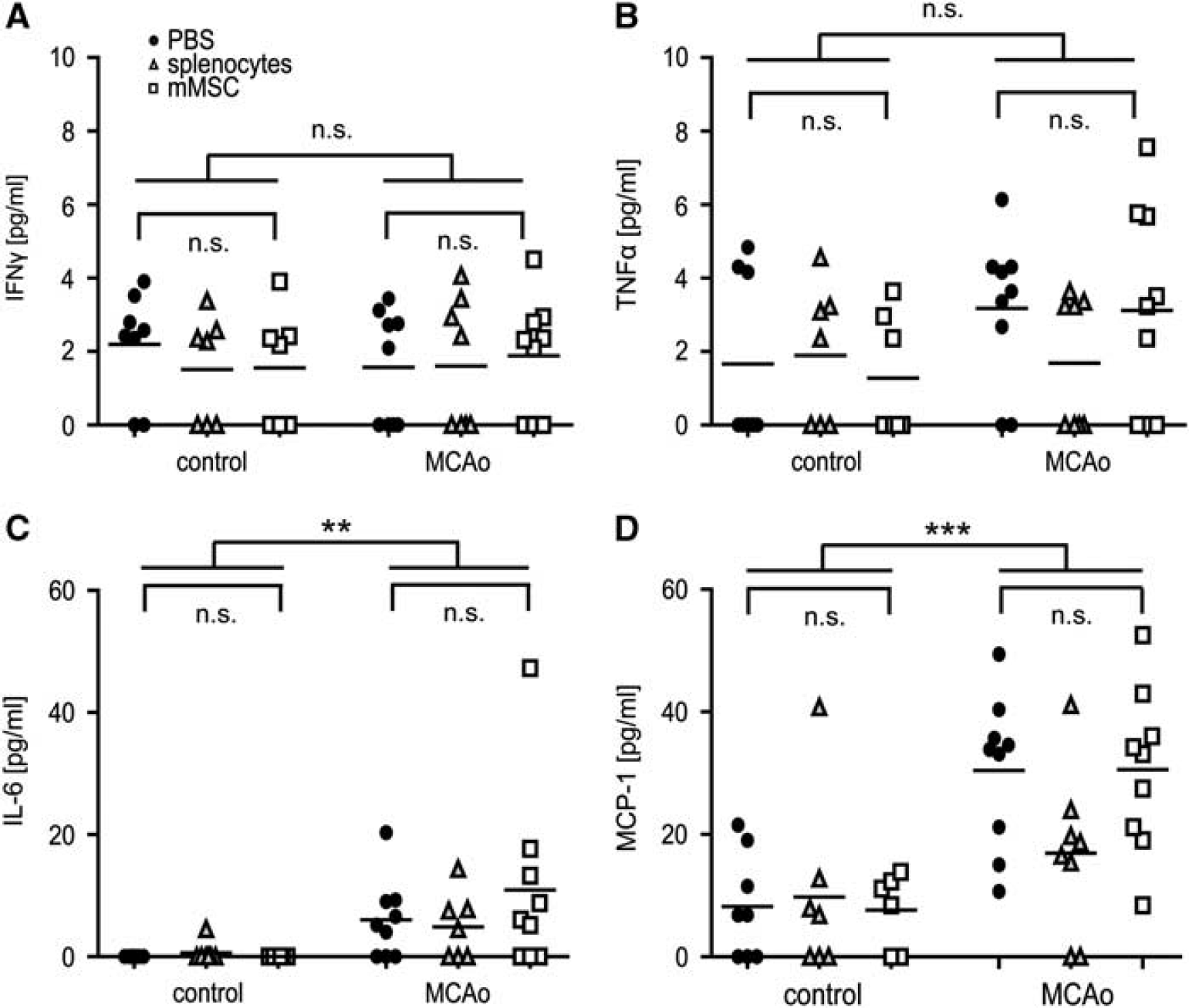
Middle cerebral artery occlusion is not only associated with dramatic apoptotic cell death of all immune cell fractions, but also with severe impairment of immune cell functions. This phenomenon is worst at 1 to 2 days after stroke and usually normalizes a couple of days later (Prass et al, 2003). Since mMSCs did not ameliorate ischemia-related cell death of immune cells, we speculated that mMSCs might have at least an impact on cellular immune functions after stroke. To test this hypothesis, blood samples, lymph node cells, splenocytes, and also CD45-positive leukocytes from lung tissue were
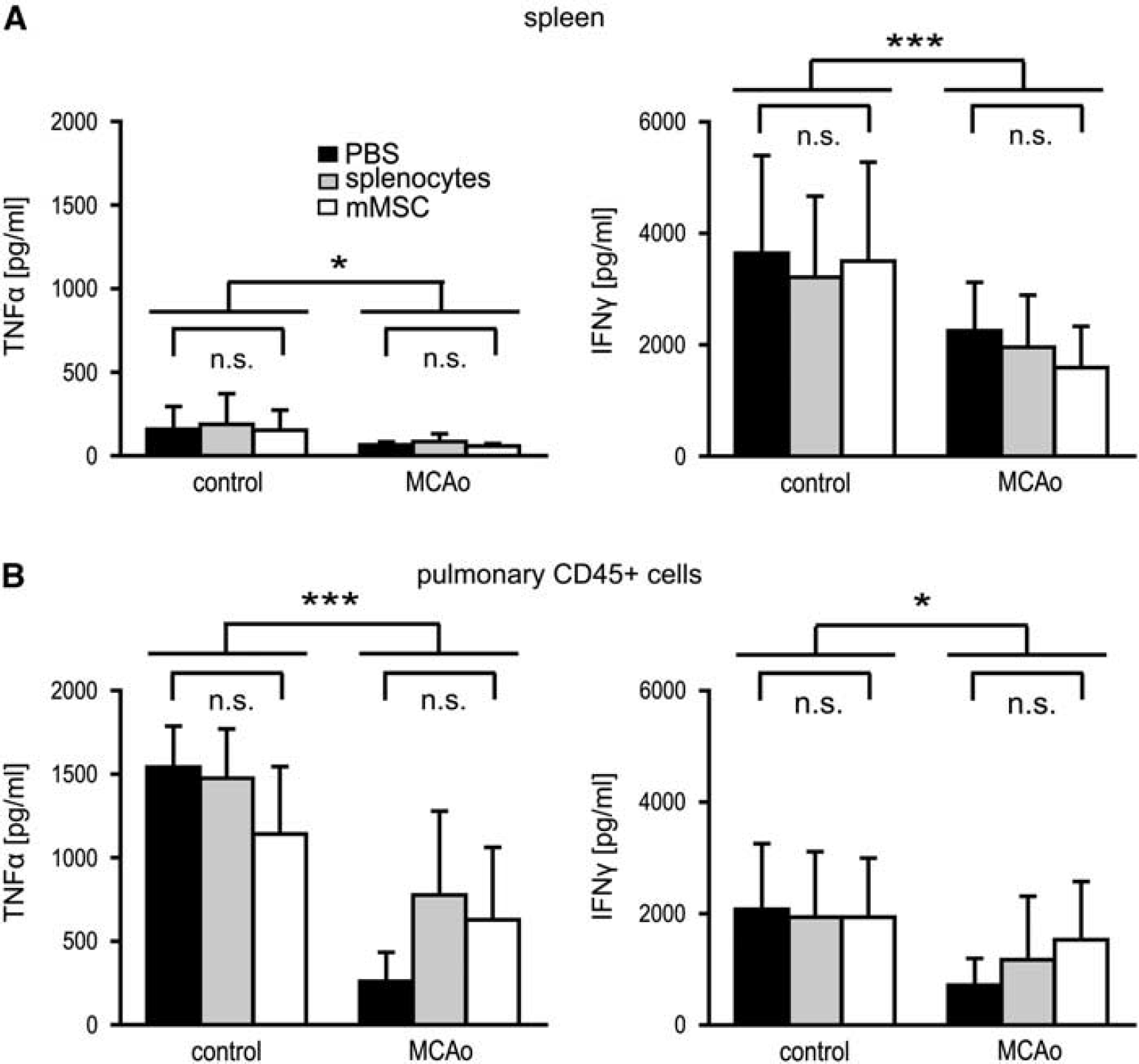
Cytokine release following
Murine Mesenchymal Stromal Cells Do Not Alter the Levels of Proinflammatory Cytokines/Chemokines in the Sera of Mice After Middle Cerebral Artery Occlusion
Cytometric bead arrays were performed with the sera of control and MCAo mice at 24 hours and 3 days after stroke to assess potential changes of proinflammatory cytokine/chemokine profiles (IL-6, IL-10, TNF
Murine mesenchymal stromal cell (mMSC) administration does not change the secretion of proinflammatory cytokines/chemokines after middle cerebral artery occlusion (MCAo). Cytokine expression levels of interferon (IFN)
Discussion
In spite of their known immunosuppressive functions, we did not observe any effects of mMSC administration in experimental stroke. Several immunological parameters, namely the immune status and the extent of immune cell death in blood and peripheral lymphoid tissues, the immune cell effector functions, and also the levels of systemic proinflammatory cytokines/chemokines remained unaltered after intravenous mMSC administration post-MCAo.
Moreover, there were no significant differences between the infarct volumes of mice that received mMSCs, splenocytes, or PBS after MCAo. Since infarct lesion sizes are highly correlated with the severity of stroke-induced immunodepression (Liesz et al, 2009), similar infarct sizes between the respective treatment groups are a prerequisite for adequate measurement of immunoregulatory mMSC functions. Our data are in line with the previous reports where the systemic transplantation of mMSCs after cerebral ischemia in mice only showed neuroprotective capacity when the cells were genetically modified and targeted to the brain by a DETA-NONOate upregulation of SDF-1/CXCR4 (Cui et al, 2007) or the cells were directly injected into brain parenchyma (Ohtaki et al, 2008). In contrast, studies using the rat MCAo model reported reduced infarct volumes after administration of 1 to 10 × 106 naïve MSCs (Nomura et al, 2005; Onda et al, 2008). The discrepancies may be related to different experimental protocols, such as application route or cell dosage. Moreover, neuroanatomical or physiological species differences (e.g., a higher intrinsic regenerative capacity of mice after MCAo) may account for the discrepancies and conceal potential tissue-protective MSC effects.
In general, the regenerative capacities of MSCs are mainly attributed to their paracrine and immunological functions (English et al, 2010). Data from autoimmune-mediated disorders like experimental autoimmune encephalomyelitis suggest that MSC administration before disease onset results in reduced disease severity with decreased inflammatory infiltrates, less demyelination, and less axonal loss in affected mice (Zappia et al, 2005). Moreover, MSCs appear to enhance regeneration by the release of prooligodendrogenic and neuroprotective factors, as well as by the induction of peripheral T-cell tolerance to the immunizing antigen (Uccelli et al, 2011) or a shift of Th2-type cytokines in T-cells (Constantin et al, 2009). Interestingly, even in a model of multiple system atrophy, intravenous MSC administration improved the survival of tyrosine hydroxylase-expressing neurons by modulation of T-cell responses and downregulation of cytokine levels, for example, IL-2, IL-17, and TNF
Similar immunosuppressive capacities of MSCs have been shown in other disease models, such as GvHD (Le Blanc et al, 2004), collagen-induced arthritis (Schurgers et al, 2010), or after organ transplantation (Crop et al, 2009). The absence of immunomodulatory effects of MSCs in stroke-induced immunodepression may result from several factors:
First, MSCs require activation by certain cytokines, for example, IFN
Second, recent reports suggest that immune functions of MSCs are highly dependent on the disease model in which MSCs are applied (Ren et al, 2008). Moreover, the effects of MSCs may also vary within a specific disease model. In collagen-induced arthritis, MSCs were found to reduce disease severity by a switch from Th1/Th17 to Th2 immune responses and by IL-6-dependent secretion of PGE2 (Bouffi et al, 2010). However, other studies failed to reproduce these encouraging
Nonetheless, the encouraging preclinical data of MSC transplantation in rat stroke models has already led to the initiation of multiple clinical stroke trials with human MSCs (http://www.clinicaltrials.gov). In this context, our results provide urgently needed experimental evidence that MSC administration after cerebral ischemia may not further deteriorate the natural course of stroke-induced immunodepression, nor raise immunological concerns on safety aspects of MSC transplantation after stroke.
Footnotes
Acknowledgements
The authors thank Peggy Mex, Monika Dopatka, and Susanne Müller for their excellent technical assistance and the BCRT FACS-core facility for providing instruments and technical assistance.
Disclosure/conflict of interest
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
