Abstract
Human liver chimeric mice are expected to be applied for drug toxicity tests and human hepatitis virus research. Human induced pluripotent stem cell-derived hepatocyte-like cells (iPSC-HLCs) are a highly attractive donor source for the generation of human liver chimeric mice because they can be produced on a large scale and established from an individual. Although these cells have been successfully used to generate human liver chimeric mice, there is still room for improvement in the repopulation efficiency. To enhance the repopulation efficacy, the human iPSC-HLCs were transduced with an adenovirus vector (Ad-FNK) expressing FNK, a hyperactive mutant gene from Bcl-xL, which was expected to inhibit apoptosis in the process of integration into liver parenchyma. We then transplanted Ad-FNK-transduced human iPSC-HLCs into urokinase-type plasminogen activator-transgenic severe combined immunodeficiency (uPA/SCID) mice (FNK mice) and evaluated the repopulation efficacy. The antiapoptotic effects of the human iPSC-HLCs were enhanced by FNK overexpression in vitro. Human albumin levels in the transplanted mice were significantly increased by transplantation of Ad-FNK-transduced human iPSC-HLCs (about 24,000 ng/ml). Immunohistochemical analysis with an anti-human αAT antibody revealed greater repopulation efficacy in the livers of FNK mice than control mice. Interestingly, the expression levels of human hepatocyte-related genes in the human iPSC-HLCs of FNK mice were much higher than those in the human iPSC-HLCs before transplantation. We succeeded in improving the repopulation efficacy of human liver chimeric mice generated by transplanting the Ad-FNK-transduced human iPSC-HLCs into uPA/SCID mice. Our method using ectopic expression of FNK was useful for generating human chimeric mice with high chimerism.
Keywords
Introduction
Human liver chimeric mice are potent tools for drug toxicity tests and hepatitis virus research. Several groups have recently reported the generation of human liver chimeric mice by transplantation of human hepatocyte-like cells (HLCs) (16,36), which were differentiated from human pluripotent stem cells, including human embryonic stem cells (ESCs) (34) and induced pluripotent stem cells (iPSCs) (26). However, to the best of our knowledge, the highest concentration of human albumin (ALB) in blood from human liver chimeric mice with human ESC/iPSC-derived HLCs (ESC/iPSC-HLCs) in previous reports was approximately 4,000 ng/ml (37). The repopulation efficacy of these mice is not sufficient for their applications. Therefore, the chimerism of human liver chimeric mice with human ESC/iPSC-HLCs will need to be enhanced before these mice can be used in medical applications.
In general, the repopulation efficacy of human ESC/iPSC-HLCs in human liver chimeric mice depends on many factors, such as the types of immunodeficient mice with liver injury and characteristics of donor cells. With regard to the recipient mice, high severity of immunodeficiency and liver injury facilitate replacing mouse hepatocytes with cryopreserved human hepatocytes (8,24). Previously, several mouse models for the generation of human liver chimeric mice were developed to meet the requirements described above [i.e., urokinase-type plasminogen activator-transgenic severe combined immunodeficiency (uPA/SCID) mice (33), fumarylacetoacetate hydrolase and recombination activating gene 2 and interleukin 2 receptor γ chain deficient (Fah-/-Rag2-/-IL2rg-/-) mice (3), and herpes simplex virus type 1 thymidine kinase-transgenic NOG (TK-NOG) mice (7)]. Among these mouse models, uPA/SCID mice are widely used to generate human liver chimeric mice with almost completely humanized livers and human liver functions through the transplantation of cryopreserved human hepatocytes (9,23,33). With regard to donor cells, human iPSC-HLCs whose characteristics are equivalent to those of cryopreserved human hepatocytes might be required for highly repopulated human liver chimeric mice. It is thus important to promote the hepatic maturation of human iPSC-HLCs. We have recently succeeded in generating highly functional human iPSC-HLCs by a combination of overexpression of hepatic transcription factors and 3D culture (21,27,28,30). The expression levels of various hepatocyte-related genes were comparable between human iPSC-HLCs and primary human hepatocytes (30). These matured human iPSC-HLCs might be appropriate as donor cells to generate human liver chimeric mice with high chimerism.
It is known that a large fraction of transplanted hepatocytes rapidly undergo apoptotic death and are removed from the recipient liver through phagocytosis by macrophages (6). Thus, it might be reasonable to presume that the engraftment efficacy would be enhanced by regulating the cell survival signals and improving the resistance to apoptotic cell death through overexpression of an antiapoptotic gene, such as B-cell lymphoma extra large (Bcl-xL). Among the Bcl-2 family proteins synthesized in the normal liver, only the Bcl-xL and myeloid cell leukemia 1 (Mcl-1) proteins were detectable at the constitutive basal level (35). In addition, only Bcl-xL expression was upregulated in the regenerated liver (35). It has also been reported that hepatocyte-specific Bcl-xL-deficient mice showed spontaneous cell death in hepatocytes (31). In contrast, Bcl-xL transgenic mice showed resistance against cell death induced using anti-Fas antibody (4,17). These reports suggest that Bcl-xL may play an important role in the survival of hepatocytes.
In this study, we first examined whether overexpression of FNK (a hyperactive mutant of Bcl-xL), which is a more powerful artificial antiapoptotic factor than Bcl-xL (1), is useful to avoid the induction of apoptosis in an in vitro culture model. Next, we attempted to generate human liver chimeric mice by transplanting the FNK-expressing adenovirus (Ad-FNK) vector-transduced human iPSC-HLCs, which were differentiated under the 3D culture condition, into uPA/SCID mice. The treatment of the human iPSC-HLCs with Ad-FNK successfully increased the concentration of human ALB in mouse blood as well as the repopulation efficacy. The transduction of FNK should be useful for the generation of human liver chimeric mice with iPSC-HLCs.
Materials and Methods
Human iPSC Culture
A human iPSC line generated from the human male embryonic lung fibroblast cell line MCR5 was provided from the JCRB Cell Bank (Dotcom, JCRB Number: JCRB1327, Osaka, Japan). This human iPSC line was maintained on a feeder layer of mitomycin C-treated mouse embryonic fibroblasts (PMEF-H; Merck Millipore, Billerica, MA, USA) with ReproStem (ReproCELL, Tokyo, Japan) supplemented with 10 ng/ml fibroblast growth factor 2 (Sigma-Aldrich, St. Louis, MO, USA). Human iPSCs were dissociated with 0.1 mg/ml dispase (Roche Diagnostics, Basel, Switzerland) into small clumps and were then subcultured every 5 or 6 days.
Ad Vectors
Ad vectors were constructed by an improved in vitro ligation method (18,19). We have constructed pHMEF5 (11), which contains the human elongation factor-1a (EF-1a) promoter and pAdHM41-K7 (12) in previous reports. The FNK gene (1) (kindly provided by Shigeo Ohta, Nippon Medical School, Tokyo, Japan) was inserted into pHMEF5 resulting in pHMEF-FNK. The pHMEF-FNK was digested with I-CeuI/PI-SceI (NEB, Ipswich, MA, USA) and ligated into I-CeuI/PI-SceI-digested pAdHM41-K7, resulting in pAd-FNK. The human EF-1a promoter-driven LacZ-, FOXA2-, or HNF1a-expressing Ad vectors (Ad-LacZ, Ad-FOXA2, or Ad-HNF1a, respectively) were construc ted previously (28,32). All of the Ad vectors contain a stretch of lysine residue (K7) peptides in the C-terminal region of the fiber knob for more efficient transduction of human iPSCs and definitive endodermal cells, in which transfection efficiency was almost 100% (10,27,29), and purified as described previously (20). The vector particle (VP) titer was determined by using a spectrophotometric method (15). We have confirmed that the Ad vector- mediated gene expression continued for 5 days and was almost disappeared at 9 days after transduction by in vitro analysis (27). Therefore, Ad vector-mediated FNK expression in the human iPSC-HLCs, which were transplanted into uPA/SCID mice, might be disappeared at about 9 days after transplantation.
In Vitro Differentiation
The hepatic differentiation protocol was based on our previous report (30). On day 34, the human iPSC- derived cells were transduced with 3,000 VP/cell of FNK- or LacZ-expressing Ad vector (Ad-FNK or Ad-LacZ).
Flow Cytometry
The human iPSCs were differentiated into HLCs as described in Figure 1A. Single-cell suspensions of human iPSC-derived cells were fixed with 2% parafor maldehyde (Wako, Osaka, Japan) at 4°C for 20 min and then incubated with the anti-asialoglycoprotein receptor 1 (ASGR1) goat antibody (1 μg/1 × 106 cells; Santa Cruz Biotechnology, Dallas, TX, USA) followed by the anti-goat IgG antibody labeled with Alexa Fluor 488 (2 μg/1 × 106 cells; Molecular Probes, Carlsbad, CA, USA). Flow cytometry analysis was performed using a FACS LSR Fortessa flow cytometer (BD Biosciences, San Jose, CA, USA).

FNK shows an enhanced antiapoptotic activity in human iPSC-HLCs against various death stimuli. (A) The procedure for differentiation of human iPSCs (Dotcom) into the HLCs via mesendoderm cells, definitive endoderm cells, and hepatoblasts is presented schematically. In the hepatic differentiation, not only the addition of growth factors but also the stage-specific transient transduction of both FOXA2- and HNF1α-expressing Ad vectors (Ad-FOXA2 and Ad-HNF1α, respectively) were performed. The cellular differentiation procedure is described in detail in the Materials and Methods section. The human iPSC-HLCs were transduced with Ad-LacZ or Ad-FNK on day 34. (B) The gene expression levels of ALB and CYP3A4 were measured by real-time RT-PCR on day 35. On the y-axis, the gene expression levels in PHs (three lots of PHs were used), which were cultured for 48 h after plating (PHs-48 h), were taken as 1.0. (C) The amount of ALB secretion was examined in Ad-LacZ-transduced human iPSC-HLCs and Ad-FNK-transduced human iPSC-HLCs, and PHs-48 h. (D) On day 35, the efficiency of hepatocyte differentiation was measured by estimating the percentage of ASGR1-positive cells using FACS analysis. (E) The cell viability was counted on day 35. (F, G) Ad-LacZ-transduced human iPSC-HLCs or Ad-FNK-transduced human iPSC-HLCs were incubated with staurosporine (an inhibitor of protein kinase C; 40 nM) (F) or calcium ionophore A23187 (2 μM) (G), and then surviving cells were counted by trypan blue exclusion. The amount of surviving cells on day 0 was taken as 1.0. All data are represented as mean ± SD (n = 3). **p < 0.01 (Staurosporine day 1 p = 0.0066, day 2 p = 0.0073, day 3 p = 0.00003, day 4 p = 0.0003; A23187 day 1 p = 0.000005, day 2 p = 0.0087, day 3 p = 0.0086, day 4 p = 0.00005).
Cell Viability Tests
The human iPSCs were differentiated into HLCs. Cell viability was assessed by using an Alamar Blue assay kit (Biosource, Carlsbad, CA, USA). The supernatants of the cells were measured at a wavelength of 570 nm with background subtraction at 600 nm in a plate reader (Wako, Osaka, Japan).
Apoptosis-Inhibiting Activity
The human HLCs were cultured in hepatocyte culture medium (HCM; Lonza, Basel, Switzerland) supplemented with 20 ng/ml oncostatin M (R&D Systems), 10−6 M dexamethasone (Sigma-Aldrich), and apoptotic inducible drug (40 nM staurosporine or 2 μM A23187; both from Merck Millipore) for 4 days. These cells were harvested by using Cell Recovery Solution (BD Biosciences), and the surviving cells were counted by the trypan blue (Nacalai Tesque, Kyoto, Japan) exclusion every day.
Transplantation of the Hepatocyte-Like Cells
The male and female uPA+/+/SCID (uPA/SCID) mice (33) at 2 to 4 weeks after birth were used as the recipient mice. These mice were anesthetized with isoflurane (Pfizer, New York, NY, USA) and injected with the 1 × 106 viable human iPSC-HLCs through a small left-flank incision into the inferior splenic pole. HCM was used as the injection vehicle. The lower reaches of the injection site were tied with string. After the injection of the cells, the mouse spleens were excised, and the superior splenic poles were tied with string. All experiments were conducted in accordance with the ethical approval of PhoenixBio Co., Ltd.
ELISA
The human iPSCs were differentiated into HLCs as described in Figure 1A. The culture supernatants, which were incubated for 24 h after fresh medium was added, were collected. Mouse blood samples were collected periodically from the tail vein. These samples were analyzed for the level of human ALB by ELISA. ELISA kits for human ALB were purchased from Bethyl Laboratories (Montgomery, AL, USA). ELISA was performed according to the manufacturer's instructions. The ALB secretion levels were calculated according to each standard.
RNA Isolation and Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Cells or whole mouse livers were homogenized in ISOGENE (Nippon Gene, Tokyo, Japan), and then total RNA was isolated according to the manufacturer's instructions. cDNA was synthesized using 500 ng of total RNA with a Superscript VILO cDNA synthesis kit (Invitrogen, Carlsbad, CA, USA). Real-time RT-PCR was performed with Taqman gene expression assays or Fast SYBR Green Master Mix using an ABI Step One Plus (all from Applied Biosystems, Carlsbad, CA, USA). Relative quantification was performed against a standard curve, and the values were normalized against the input determined for the housekeeping gene, glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Note that the human ALB, HNF4α, αA T, and CYP3A4 expression values were normalized by the other housekeeping genes: Phosphoglycerate kinase 1, HBS1-like, succinate dehydrogenase complex subunit A flavoprotein, or transferrin receptor were comparable with the gene expression values normalized by GAPDH (data not shown). The human-specific primer sequences and assay IDs used in this study are described in Tables 1 and 2. The human CYP3A4 primer for Fast SYBR Green Master Mix was used to analyze the gene expression level in the human iPSC-HLCs in vitro, and the human CYP3A4 Taqman gene expression assay was used to analyze the gene expression level in the human iPSC-HLCs, which was integrated into mouse livers. The other human-specific primers were also designed to detect only human genes but not mouse genes.
Human Primer Sequences for Real-Time RT-PCR

Taqman Assay IDs for Real-Time RT-PCR

Immunohistochemistry
Immunohistochemistry of human ALB and human CK8/18 (MP Biomedicals, Santa Ana, CA, USA) was performed at PhenixBio (Hiroshima, Japan). These analyses used anti-human ALB goat antibody (1:200; Bethyl Laboratories) and anti-human CK8/18 mouse antibody (1:50; MP Biomedicals). Immunohistochemistry of human αAT was performed as follows. Recipient mouse livers were harvested at 4 weeks after human cell transplantation. The livers were fixed with 10% neutral-buffered formalin (Wako), embedded in paraffin, and sectioned at 5 μm. Pretreatment of the paraffin tissue sections consisted of L.A.B. Solution (Polysciences, Inc., Warrington, UK) at room temperature for 15 min. After incubation with 0.1% Tween 20 (Sigma-Aldrich) and blocking with ImmunoBlock (DS Parma Biomedical, Osaka, Japan), the sections were incubated with an anti-human αAT rabbit antibody (1:50; Dako, Glostrup, Denmark) at 4°C overnight, followed by incubation with an anti-rabbit IgG-biotinylated antibody (1:100; Vector Lab, Burlingame, CA, USA) at room temperature for 1 h. Immunoreactivities for human αAT were visualized with a VECTASTAIN ABC kit and ImmPACT DAB Substrate (all from Vector Lab) according to the manufacturer's instructions. The sections were counterstained with Mayer's hematoxylin (Wako). The repopulation efficacy was calculated as the ratio of human αAT-expressing areas to the total liver section area using ImageJ (NIH, Bethesda, MD, USA). This analysis was performed using the three sections from each two lobes of mouse liver, that is, we took six sections per mouse.
Statistical Analysis
All data are represented as mean ± SD or SE from at least three independent experiments. Statistical analysis was performed using the unpaired two-tailed Student's t-test by GraphPad Prism (MDF, Tokyo, Japan).
Results
FNK Enhanced the Resistance of the Human iPSC-HLCs to Apoptotic Stimulations
To generate the human iPSC-HLCs, hepatic differentiation was performed using a stage-specific transient transduction of FOXA2 and HNF1α (28), and a 3D culture method (30) as shown in Figure 1A. On day 34, the human iPSC-HLCs were transduced with Ad-LacZ or Ad-FNK, and then hepatocyte characteristics, such as the gene expression levels of hepatocyte-related markers (Fig. 1B), the ability to secrete ALB (Fig. 1C), and the percentage of ASGR1 (a mature hepatocyte marker)-positive cells (Fig. 1D) were compared in the Ad-LacZ- and Ad-FNK-transduced human iPSC-HLCs on the next day. As a result, the hepatic characteristics of the Ad-FNK-transduced human iPSC-HLCs were similar to those of the Ad-LacZ-transduced human iPSC-HLCs, suggesting that the transient overexpression of FNK did not influence the hepatic characteristics. Moreover, the gene expression levels of ALB and CYP3A4 in the human iPSC-HLCs were comparable to those in primary human hepatocytes (PHs) that were cultured 48 h after plating the cells. Thus, these results suggested that efficient hepatic differentiation was successfully achieved. Next, we examined whether FNK overexpression had the ability to enhance the resistance to apoptotic stimulations. The number of viable human iPSC-HLCs was measured in the presence of the apoptosis-inducing drugs, staurosporine (an inhibitor of protein kinase C) and A23187 (a calcium ionophore) (Fig. 1F, G, respectively). Although the cell viabilities were not affected by the transduction of Ad-FNK (Fig. 1E), the number of surviving Ad-FNK-transduced human iPSC-HLCs was higher than the number of surviving Ad-LacZ-transduced human iPSC-HLCs in the presence of apoptosis-inducing drugs. These results suggest that FNK overexpression in the human iPSC-HLCs could enhance the tolerance to apoptotic stimulations without influence on the expression of hepatic markers.
The Human ALB Concentration Was Enhanced by the Transplantation of Ad-FNK-Transduced Human iPSC-HLCs
In order to determine whether the engraftment and repopulation efficacy of human iPSC-HLCs in immuno-deficient mice with damaged livers could be increased by Ad-FNK transduction, we transplanted Ad-LacZ- or Ad-FNK-transduced human iPSC-HLCs into uPA/SCID mice by intrasplenic injection (LacZ mice and FNK mice, respectively). Because it has been found that some intrasplenic injected hepatocytes engraft into recipient spleen (25), the recipient spleen was excised just after transplantation of human iPSC-HLCs to avoid influences of engrafted human iPSC-HLCs in the recipient spleen. The experimental design of the cell transplantation is shown in Figure 2A. Because it is known that the human ALB concentration in mouse blood correlates with the number of repopulated human hepatocytes in mice transplanted with cryopreserved human hepatocytes (3,33), we monitored the human ALB concentration in the blood of LacZ and FNK mice (Fig. 2B). Human ALB in the blood of both LacZ and FNK mice was detectable at 1 week after transplantation. The human ALB concentration in the FNK mice (approximately 73 ng/ml) was slightly higher than that in the LacZ mice (approximately 30 ng/ml). Thereafter, the human ALB concentration in these two groups was increased until 4 weeks after transplantation (to approximately 7,400 ng/ml in the LacZ mice, and approximately 24,000 ng/ml in the FNK mice). The human ALB concentration in the blood of FNK mice was significantly higher than that in the blood of LacZ mice at 2—4 weeks after transplantation. Unfortunately, both LacZ and FNK mice became weak at 4 weeks after transplantation and were therefore sacrificed at this time point. No distinct tumor was observable at the time of sacrifice. The gene expression levels of hepatocyte-specific markers in the human iPSC-HLCs in the LacZ or FNK mice were measured by using human-specific primers. First, the expression level of human ALB in the human iPSC-HLCs was analyzed by real-time RT-PCR (Fig. 2C). Because there were no significant differences between these two groups, the human ALB secretion ability of the human iPSC-HLCs in FNK mice would be similar to that of the human iPSC-HLCs in LacZ mice. Therefore, the difference in the human ALB concentration between the LacZ mice and the FNK mice would be due to the repopulation efficacy of the human iPSC-HLCs, but not due to a difference between the human ALB secretion ability of the human iPSC-HLCs in the LacZ mice and that in the FNK mice. Taken together, these results suggest that the number of the repopulated human iPSC-HLCs in the FNK mice was higher than that in the LacZ mice.

The human ALB concentration in the mouse blood was increased by the transplantation of Ad-FNK-transduced human iPSC-HLCs. Human iPSCs (Dotcom) were differentiated into the HLCs as described in Figure 1A. (A) The Ad-LacZ- or Ad-FNK-transduced human iPSC-HLCs (1 × 106 cells) were transplanted into uPA/SCID mice by intrasplenic injection. After the injection of the cells, the mouse spleens were excised. The experimental schedule is presented schematically. (B) Human ALB concentrations in the mouse blood were monitored once a week. Triangles and squares represent LacZ mice (n = 10) and FNK mice (n = 12), respectively. (C) The gene expression level of human ALB in the human iPSC-HLCs, which exist in the liver of LacZ mice and FNK mice, was measured by real-time RT-PCR at 4 weeks after transplantation (LacZ mice, n = 3, and FNK mice, n = 4, respectively). The whole livers of LacZ mice or FNK mice, not isolated human iPSC-HLCs, were used in this analysis. Undifferentiated human iPSCs (n = 3) and cryopreserved human hepatocytes (CHHs, n = 3), which were harvested just after thawing, were used as a control. All data are represented as mean ± SE. *p < 0.05 (week 2 p = 0.0323, week 3 p = 0.0369, week 4 p = 0.0306). n.s., not significant (p = 0.952).
The Gene Expression Levels of Hepatocyte-Related Genes in the Human iPSC-HLCs Were Upregulated in the Mouse Liver
To examine whether the hepatic functionalities of the human iPSC-HLCs were enhanced by transplantation into uPA/SCID mice, the gene expression levels of human hepatocyte-related markers in the human iPSC-HLCs that were engrafted into the liver of FNK mice were compared at 4 weeks after transplantation (posttransplant) with those in the human iPSC-HLCs that were not transplanted (pretransplant) (Fig. 3). Interestingly, the gene expression levels of human ALB, human αAT, human ASGR1, human HNF4α, human CYP3A4, human CYP2D6, human CYP7A1, human CYP3A5, human UGT1A1, human UGT1A3, human FXR, and human PXR were drastically upregulated after transplantation. These results might suggest that the human iPSC-HLCs were maturated in the livers of uPA/SCID mice.

The hepatocyte-related gene expression levels of the human iPSC-HLCs were enhanced in the mouse liver. Human iPSCs (Dotcom) were differentiated into the HLCs as described in Figure 1A. The Ad-FNK-transduced human iPSC-HLCs (1 × 106 cells) were transplanted into uPA/SCID mice by intrasplenic injection as described in Figure 2A. At 4 weeks after transplantation, the gene expression levels of human hepatocyte-related markers, such as human ALB, human αAT, human ASGR1, human HNF4α, human CYP3A4, human CYP2D6, human CYP7A1, human CYP3A5, human UGT1A1, human UGT1A3, human FXR, and human PXR, were measured in the human iPSC-HLCs, which were in the liver of FNK mice (posttransplantation, n = 4), by real-time RT-PCR. The whole livers of FNK mice, not isolated human iPSC-HLCs, were used in this analysis. On the y-axis, the gene expression levels in the Ad-FNK-transduced human iPSC-HLCs before transplantation (pretransplantation, n = 3) were taken as 1.0. All data are represented as mean ± SE.
FNK Could Increase the Repopulation Efficacy of the Human iPSC-HLCs in the Mouse Liver
To ascertain whether the human iPSC-HLCs could repopulate the mouse liver, immunohistochemical analyses were performed by using anti-human-specific antibodies that react with hepatocyte-specific markers. Several clusters of human ALB- (Fig. 4A), human αAT- (Fig. 4B), and human CK8/18-expressing cells (Fig. 4C) were found in the liver sections of FNK mice. The human hepatocyte-specific marker-positive clusters were also observed in the LacZ mice (data not shown). These results suggest that the human iPSC-HLCs have the potential to repopulate and express human hepatic proteins in the mouse liver. Because some nonspecific staining of human ALB antibody was observed, we calculated the repopulation efficacy of the human iPSC-HLCs in the LacZ or FNK mice by estimating the ratio of the human αAT-expressing areas to the total liver section area (Fig. 4D). The repopulation efficacy of the Ad-FNK-transduced human iPSC-HLCs was significantly higher than that of the Ad-LacZ-transduced human iPSC-HLCs. Taken together, these results reveal that the transduction of Ad-FNK into human iPSC-HLCs would be a useful tool to efficiently repopulate the mouse liver.
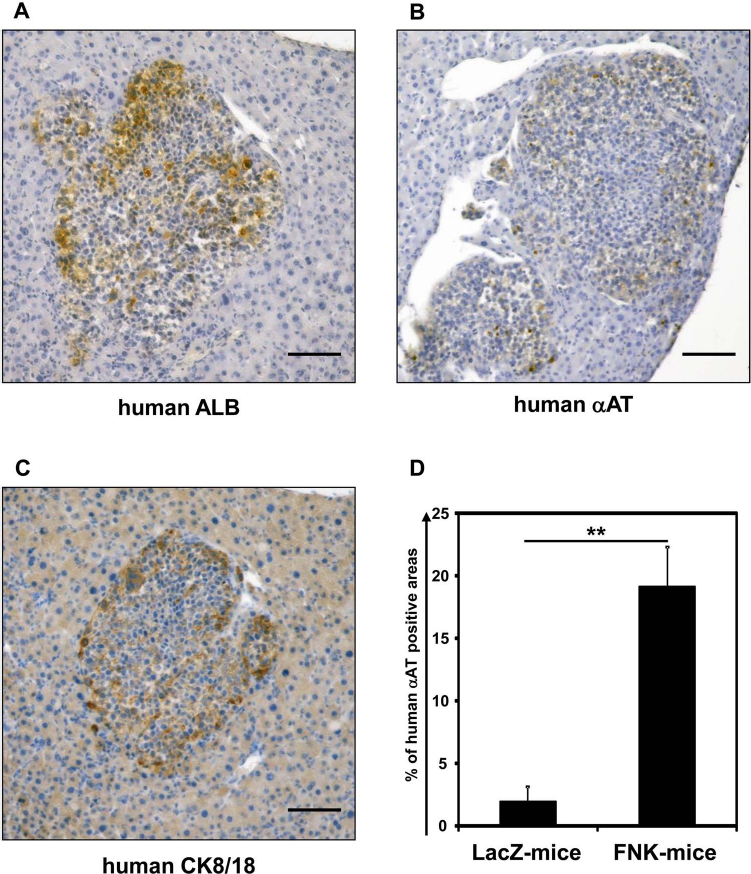
The repopulation efficiencies in the chimeric mice were upregulated by the transplantation of Ad-FNK-transduced human iPSC-HLCs. Human iPSCs (Dotcom) were differentiated into the HLCs as described in Figure 1A. The Ad-LacZ- or Ad-FNK-transduced human iPSC-HLCs (1 × 106 cells) were transplanted into uPA/SCID mice by intrasplenic injection as described in Figure 2A. (A—C) At 4 weeks after transplantation, chimeric mouse livers were analyzed by immunohistochemical staining. Clusters of human ALB- (A) and human αAT- (B) and human CK8/18-expressing cells (C) were found in liver sections. Scale bars: 100 μm. (D) The repopulation efficiencies in the LacZ mice (n = 7) and FNK mice (n = 8) were calculated by the ratio of human αAT-expressing areas to the total liver section area. All data are represented as mean ± SE. **p < 0.01 (p = 0.0004).
Discussion
This study aimed to improve the repopulation efficacy of the human iPSC-HLCs in the mouse liver for medical applications. To generate chimeric mice with a high level of human liver chimerism, the antiapoptotic gene (FNK)-transduced human iPSC-HLCs were transplanted into uPA/SCID mice. The levels of human ALB and repopulation efficacy in the FNK mice were higher than those in the LacZ mice (Figs. 2 and 4). On intrasplenic injection in hepatocyte transplantation, a major fraction of transplanted cells (more than 70%) was rapidly removed by phagocyte and macrophage responses (6). The residual transplanted hepatocytes were translocated from the liver sinusoids to the liver plate at between 16 and 20 h after transplantation. Because transplanted hepatocytes lose intimate contact with the extracellular matrix or other cells during this period, these cells might cause anoikis through the activation of caspase 8 (5,22). It is known that the ectopic expression of Bcl-xL can inhibit apoptosis by suppression of caspase 8, 7, and 3 (14). These facts suggest that the transplanted human iPSC-HLCs would be protected from apoptosis in the early period after transplantation by FNK overexpression, and then these cells could translocate from the liver sinusoids to the liver plate of recipient mice.
Although Bcl-xL is known as a cellular oncogene, no distinct tumors were observed in the FNK mice at 4 weeks after transplantation. This result suggests that the transient transduction of FNK using an Ad vector might not be involved in the tumorigenesis of transplanted human iPSC-HLCs. Consistent with this idea, a previous study showed that Ad vector-mediated FNK overexpression did not transform dendritic cells using a colony formation assay in soft agar (38). Taken together, these results indicate that FNK overexpression would be a useful method to enhance the engraftment efficacy of transplanted cells.
The concentration of human ALB in the blood of FNK mice was significantly higher than that in the blood of LacZ mice (Fig. 2A). In addition, the concentration in the blood of FNK mice (about 24,000 ng/ml) was approximately sixfold higher than that in the blood of mice in the previous study (about 4,000 ng/ml) (37). However, this concentration of human ALB in the blood of FNK mice, whose repopulation efficacy was about 19%, was approximately 42 times lower than that in the blood of human liver chimeric mice generated by transplantation of cryopreserved human hepatocytes into uPA/SCID mice (about 1 mg/ml), whose repopulation efficacy was about 20% (33). Because the repopulation efficacies were similar in these two groups, it was suggested that the lower ability of the FNK mice to secrete human ALB into the blood was due to the immaturity of the hepatic functionalities of the human iPSC-HLCs compared with cryopres erved human hepatocytes. In fact, cryopreserved human hepatocytes expressed human ALB at an approximately 50-fold higher level than the human iPSC-HLCs (Fig. 2C), even if the gene expression levels of human ALB in the human iPSC-HLCs were upregulated after transplantation (Fig. 3). Although the gene expression levels of human ALB in the human iPSC-HLCs might be upregulated over the long term, both LacZ and FNK mice became weak at 4 weeks after transplantation in the present study. These results suggest that the transplanted human iPSC-HLCs could not support the liver functions of recipient mice because the hepatic functions of the human iPSC-HLCs were lower than those of cryopreserved human hepatocytes. Therefore, more technical improvements in generating mature iPSC-HLCs will be needed in order to generate human liver chimeric mice with fully functional human-type livers. Incidentally, we sacrificed the mice at 4 weeks after transplantation in this study. However, it would be interesting to observe these mice for longer than 4 weeks.
Interestingly, we observed significant increases in the expression levels of human hepatocyte-related genes in the human iPSC-HLCs of FNK mice compared with the Ad-FNK-transduced human iPSC-HLCs before transplantation (Fig. 3). This finding may have been due to aspects of the in vivo liver environment, such as the interaction with other cells, functional polarity, and extracellular matrix. In a future study, it would be intriguing to isolate human iPSC-HLCs from a transplanted mouse liver and use them for drug toxicity screening in vitro. Because previous studies have shown that human pluripotent stem cell-derived pancreatic cells were maturated and differentiated into functional β-cells at least 3 months after transplantation into mice (13), further maturation of the human iPSC-HLCs might be expected at longer times (over 4 weeks) after transplantation. It has been shown that human hepatocytes repopulated in the livers of mice can be serially transplanted into the livers of other mice and survive (2). The repopulation efficacy of serially transplanted mice was increased compared with that in the initially transplanted mice (2). Therefore, serial transplantation of the human iPSC-HLCs might enhance the repopulation efficacy.
In this study, we succeeded in enhancing the repopulation efficiency of human liver chimeric mice generated by transplantation of the Ad-FNK-transduced human iPSC-HLCs into uPA/SCID mice. Our method using ectopic expression of FNK should be useful to generate chimeric mice with a high level of human liver chimerism. To the best our knowledge, the human ALB secretion levels and repopulation efficacy in our chimeric mice with human iPSC-HLCs were higher than those in the mice used in previous reports, although improvements are required to generate chimeric mice with fully functional human-type livers.
Footnotes
Acknowledgments
We thank Misae Nishijima, Reiko Hirabayashi, Natsumi Mimura, and Nobue Hirata for their excellent technical support. We also thank Katsuhiro Kanda (Hitachi High-Technologies Corp.) for providing nanopillar plates and Shigeo Ohta (Nippon Medical School) for providing FNK gene. H.H. and K.K. were supported by grants from the Ministry of Health, Labor, and Welfare of Japan. H.M. was also supported by the Japan Research Foundation for Clinical Pharmacology. F.S. was supported by Program for Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation. Y.N. and K.T. (K. Takayama) were supported by a Grant-in-aid for JSPS Fellows. The authors declare no conflicts of interest.
