Abstract
Promoting angiogenesis in a damaged tissue is a major challenge for tissue regeneration. Recent findings in tissue engineering suggest that fibroblasts (FBs) play an important role in orchestrating the angiogenic process. Fibroblasts maintain the structural integrity of connective tissue by continuously secreting growth factors and extracellular matrix precursors, which are essential for endothelial cell (EC) adhesion and spreading, thus playing a crucial role in angiogenesis. We hypothesized that FBs immobilized in alginate gels grafted with the RGD peptidic sequence could influence the recruitment of ECs to improve vascularization. In this work, the modulation of immobilized human FBs within the 3D synthetic extracellular matrix was assessed. Experiments using cocultures of ECs and FBs in indirect contact as well as angiogenic assays were performed to assess the influence of FBs immobilized in RGD–alginate in ECs' viability, stabilization, sprouting, and assembly into capillary-like structures. This study demonstrates the ability of FBs immobilized within RGD–alginate microspheres to modulate and support capillary-like structures' assembly. These findings indicate that the microenvironment created by these stromal cells in the scaffold modulates capillary morphogenesis, thus stimulating angiogenesis in situ and can potentially be used in regenerative medicine in clinical scenarios where vascularization is essential.
Keywords
Introduction
Strategies promoting tissue vascularization continue to represent a great challenge in regenerative medicine. A promising approach to address this problem relies on the use of implantable three-dimensional (3D) biomaterials that can provide support and protection to cells transplanted for regeneration of skin (32), nerve (3), mammary gland (23), cardiovascular tissue (48), and bone (14,17,21,38). The 3D biomaterial structure is intended to mimic the extracellular matrix (ECM), thus creating an adequate microenvironment to allow the interaction between cells and the ECM. These interactions are fundamental in cell polarization and function and crucial for vascularization of the newly formed tissue (12). Angiogenesis, or the recruitment of new vessels to the tissue formed, is a process that plays a key role in various physiological conditions, including embryonic development, wound repair, inflammation, and regeneration (7).
In tissue engineering approaches, angiogenesis has been traditionally stimulated by paracrine signals, such as vascular endothelial growth factor (VEGF) (39) and basic fibroblast growth factor (bFGF) (13), using biomaterials like hydrogels for bioactive molecule delivery. However, in the framework of tissue vascularization, the uncontrolled release of angiogenic growth factors and/or alterations of the production of natural angiogenic inhibitors could deregulate the angiogenic balance (22). Immobilization of cells in 3D structures, instead of growth factor immobilization, constitutes an alternative strategy. The immobilization of cells inside alginate microspheres is here proposed as a minimally invasive (injectable) strategy for tissue regeneration (14,37,43). Alginate is a hydrogel that has been used as a biomaterial, both in vitro and in vivo, due to its several advantages, including biocompatibility and ability to be cross-linked under harmless mild conditions to cells (12). Coupling arginine–glycine–aspartic acid (RGD) peptidic sequence to alginate promotes cell attachment, leading to improvement of tissue formation (1). The present team previously demonstrated that immobilization of osteoprogenitor cells in RGD–alginate is a valuable strategy to maintain cells viable during the period of osteoblast differentiation (5,14,21). It has also been previously shown that endothelial cells (ECs) influence osteoprogenitor cells by releasing bone morphogenetic protein-2 (BMP-2) to modulate their osteogenic differentiation in vitro and in vivo (24). Reciprocally, bone cells produce VEGF that enhances EC viability (25), thus generating a crosstalk that seems to stimulate both cell types. However, in this system, a significant decrease in EC viability was revealed upon immobilization, even in the presence of osteoblasts (21).
Recent findings suggest that fibroblasts (FBs) also play an important role in orchestrating the angiogenic process (10,16,47). FBs are mesenchymal cells with many vital functions during development and in adult organisms (8). They are responsible for the synthesis of collagen, an ECM component that provides a structure for cell migration and release of angiogenic growth factors (8). However, the potential of FBs for improving vascularization in tissue regeneration strategies remains largely unexplored. Hence, in the present study, it was hypothesized that FBs immobilized in RGD–alginate could influence ECs to improve vascularization. In this work, the modulation of human ECs and FBs immobilized within a synthetic 3D extracellular matrix was assessed. Experiments using cocultures in indirect contact and angiogenic assays were performed to assess the role of immobilized FBs in EC viability, stabilization, sprouting, and assembly into capillary-like structures.
Materials and Methods
Culture of Endothelial Cells and Fibroblasts
Human umbilical vein endothelial cells (HUVECs, ScienceCell, herein called ECs) were cultured in M199 medium (Sigma, Sintra, Portugal), supplemented with 20% (v/v) inactivated fetal bovine serum (FBS; Gibco), 80 μg/ml heparin (Sigma), 30 μg/ml endothelial cell growth supplement (ECGS, BD Science), 100 U/ml penicillin G, and 100 μg/ml streptomycin (Gibco). Cells were maintained in culture flasks coated with 0.2% gelatin (Sigma). Male neonatal human dermal foreskin fibroblasts-1 (HFF-1, ATCC, Manassas, VA, USA; herein called FBs) were cultured in Dulbecco's modified Eagle's medium (DMEM, Gibco) with high glucose and with l-glutamine, supplemented with 15% (v/v) inactivated FBS, 100 U/ml penicillin G, and 100 μg/ml streptomycin. The cells were maintained at 37°C in a humidified 5% CO2 atmosphere until 90% confluence was reached. For each experiment, ECs and FBS were used at passages 6–8, and the culture medium was changed every 2 days.
Preparation of Modified Alginate
The alginate gel for cell immobilization was prepared according to the methods previously described by Evangelista et al. with minor modifications (14). Briefly, Protanal LF 20/40 sodium alginate (gift from FMC Biopolymers, Oslo, Norway) was used as high molecular weight (high content of guluronic acid units, high MW) component to prepare alginate microspheres. To obtain the low molecular weight (low MW) alginate γ-irradiation with a cobalt-60 source (UTR – Campus Tecnológico e Nuclear, Sacavém, Portugal) at a dose of 5 Mrad was used, as previously described by Kong et al. (27). Alginate was then purified through dialysis (MWCO 3500 membrane, Spectra/Por®; Spectrum Europe B.V., DG Breda, The Netherlands) against distilled and deionized water for 3 days. After, sodium periodate (Sigma) was used for alginate oxidation, as previously described (6). Finally, oligopeptides with a sequence of (glycine)4-arginine–glycine–aspartic acid–serine–proline (abbreviated as G4-RGDSP; Peptides International, Louisville, KY, USA) were covalently grafted to the oxidized alginate (16.7 mg of peptide per gram of alginate) by using aqueous carbodiimide chemistry (37). After reaction for 24 h, the alginate was purified by dialysis (MWCO 3500 membrane, Spectra/Por®), and then filtered, lyophilized, and stored at −20°C until further use. In the present experiments, a 2 wt% RGD-modified alginate solution in 0.9 wt% NaCl (Sigma) was used, corresponding to a 50:50 mixture of oxidized and RGD-modified alginate (25 wt% high MW, 75% low MW) and unmodified high MW purified alginate (14).
Immobilization and Culture of Cells Within Alginate Microspheres
Cells were trypsinized with 0.05% (w/v) trypsin/EDTA solution (Gibco), and the cell pellet was then resuspended in 0.9 wt% NaCl (Sigma) solution and centrifuged in a 5-ml syringe (TERUMO, VWR, Carnaxide, Portugal). Afterwards, the cells were carefully homogenized with the 2 wt% RGD–alginate in 0.9 wt% NaCl solution using a dual-syringe system (5,14). A cell density of 20 × 106 cells/ml of RGD–alginate solution was used (5). Subsequently, the mixture prepared was extruded under a coaxial nitrogen flow using a Var J1 encapsulation unit (Nisco, Zurich, Switzerland) at a speed of 40 ml/h (single-syringe infusion pump, Cole Parmer, Vernon Hills, IL, USA). The RGD–alginate microspheres were allowed to form in an isotonic 0.1 M CaCl2 solution (Merck Millipore, VWR) and were kept therein for 10 min. The microspheres thus obtained were rinsed in Tris-buffered saline (TBS; VWR), followed by rinsing in incomplete culture medium (DMEM or M199). All microspheres produced were uniformly spherical (diameter of ca. 800 μm). RGD–alginate microspheres containing cells were then cultured in dynamic culture conditions using 100-ml spinner flasks (Bellco Biotechnologies, Vineland, NJ, USA) maintained at 37°C under a 5% v/v CO2 humidified atmosphere. The culture medium was changed every 3 days. RGD–alginate microspheres without cells were also prepared as a control and maintained in the same conditions. After immobilization, RGD–alginate microspheres containing ECs were cultured in the presence of 5 ng/ml of recombinant vascular endothelial growth factor (VEGF, from Biomedical Technologies, Inc., Stoughton, MA, USA) or 5 ng/ml VEGF combined with 5 ng/ml recombinant bFGF (Sigma).
Diffusional Study
The purpose of the diffusional study was to assess the diffusion of substances with distinct molecular weights into the RGD–alginate microspheres, namely, albumin (Mw 6.9 × 104) and lysozyme (Mw 14.3 × 103). Microspheres were placed in a 15-ml centrifuge tube filled with a mixture of 7.5 ml of Tris-buffered saline (TBS, pH 7.4) and 0.25% of the diffusing solute (bovine serum albumin or lysozyme, Sigma), as previously described by Orive et al. (33). At 15, 45, and 75 min, a 2-ml sample was taken from the RGD–alginate microsphere suspension and analyzed to measure the solute concentration. All diffusion experiments were performed under stirred conditions and in a 37°C incubator. Albumin and lysozyme were measured by the microbicinchoninic acid (BCA) assay (Fisher Scientific, Porto Salvo, Portugal) (41).
Uptake of Dil-ac-LDL by Endothelial Cells
Cells were incubated with 1,1×-dioctadecyl-3,3,3×3×-tetramethylindocarbocyanine perchlorate (DiI)-fluorescent acetylated low-density lipoprotein (Dil-ac-LDL, from Biomedical Technologies, Inc.), a specific marker for endothelial cells. After 4 h of incubation (10 μg/ml medium) at 37°C, the medium containing Dil-Ac-LDL was removed, and the cells were washed with probe-free medium. Immediately, the microspheres were visualized using a confocal laser scanning microscope (CLSM, Leica SP2 AOBS; Leica Microsystems) and LCS software (Leica Microsystems).
Metabolic Activity by Resazurin Assay
The resazurin assay measures the innate metabolic activity of cells by assessing the fluorescence intensity increase associated with the cellular reduction of resazurin to resorufin. Cell metabolic activity was assessed by culturing cells in 10% (v/v) resazurin solution (Sigma) for 4 h at 37°C. Afterwards, 100 μl/well was transferred to a 96-well black plate (VWR), and fluorescence was measured at the excitation and emission wavelengths of 530 nmEx and 590 nmEm in a Spectra Max Gemini XS (Molecular Devices). To allow recovery of cells immobilized in RGD–alginate, 50 mM ethylenediamine tetraacetic acid (EDTA; VWR) solution in phosphate-buffered saline (PBS, pH 7.4; VWR) for 5 min was used.
Cell Viability by Flow Cytometry
Apoptosis was determined using annexin V–fluorescein isothiocyanate (FITC) staining (BD Pharmingen™) according to the manufacturer's instructions. Propidium iodide (PI; Alfagene, Carcavelos, Portugal) is a standard flow cytometry viability probe and is used to distinguish viable from nonviable cells. Cells staining positive for annexin V–FITC and negative for PI are undergoing apoptosis. Samples of microspheres were transferred to a 24-well plate (VWR) and incubated with 50 mM EDTA solution in PBS (pH 7.4) for 5 min to dissolve alginate and recover the cells, as previously described. Cells were then washed in PBS and centrifuged at 300 × g for 5 min. Cells were then resuspended in 1× binding buffer at a concentration of 1 × 106 cells/ml. After transferring 100 μl of the solution (1 × 105 cells) to a tube, 5 μl of annexin V–FITC and 5 μl of PI were added, followed by 15-min incubation at room temperature in the dark. Afterwards, 400 μl of 1× binding buffer was added to each tube, and 1 × 104 cells were used for this analysis. Data were collected with a flow cytometer (FACS Calibur, Becton Dickinson) and analyzed with FlowJo software.
Preparation of Conditioned Medium
Conditioned culture medium was obtained by culturing ECs (CMECs) or FBs (CMFBs) during 24 h in M199 medium supplemented with 10% FBS, 80 μg/ml heparin, 30 μg/ml ECGS, penicillin G (100 U/ml), and streptomycin (100 μg/ml). The media were collected, centrifuged at 300 × g, during 5 min, and the supernatant frozen at −20°C. Both cell types were separately plated at a density of 8 × 103 cells/cm2. After 24-h incubation, media were changed: ECs were incubated in CMFBs, and FBs were incubated in CMECs. Freshly prepared media were used as control for each cell culture. The metabolic activity of cells was then assessed by the Rezasurin assay upon a 24-h incubation period.
Coculture of Endothelial Cells and Fibroblasts in Indirect Contact
FBs (3 × 103 cells) were seeded on the bottom of 24-well tissue culture plates and incubated for 24 h. Afterwards, Transwells with a membrane of 0.8 μm pores (BD Science) were coated with 50 μl of growth factor-reduced–Matrigel® (GFR–Matrigel, BD Biosciences) and incubated for 30 min at 37°C. Then, ECs (3 × 103 cells) were seeded on top of the Transwell insert, in indirect contact with the FBs in M199 medium supplemented with 10% FBS, 80 μg/ml heparin, 30 μg/ml ECGS, penicillin G (100 U/ml), and streptomycin (100 μg/ml). After 24 h of incubation, cell metabolic activity was measured by the resazurin assay, and the assembly of capillary-like structures was monitored by microscopic analysis (Axiovert 200M, Zeiss).
Assessment of Capillary-Like Structure Formation and Stability
To assess the influence of FBs immobilized within RGD–alginate microspheres on capillary-like structure formation and development, a 3D assay was performed using growth factor GFR–Matrigel-coated plates, after adaptation from Soares et al. (42). Briefly, 200 μl of GFR– Matrigel® was added to a 24-well plate and incubated at 37°C for 30 min to form a gel layer. Subsequently, RGD– alginate microspheres with or without (control) FBs were added to the well (approximately 54 ± 4 microspheres per well). Again, 200 μl of GFR–Matrigel® was added to the samples and incubated at 37°C for 30 min. Afterwards, ECs (5 × 103 cells) were cultured on top of this gel in M199 medium. Under these conditions, ECs are known to assemble into capillary-like structures after 24 h (42). A semiquantitative measurement of the structures formed was carried out as previously described (5,42). The plates were photographed after 1 and 5 days postincubation using an inverted microscope (Axiovert 200M, Zeiss). The capillary-like structures formed were then counted in each well, and the length was measured using imageJ64 (NIH, Bethesda, MD, USA).
Ex Vivo Assessment of Sprouting Using the Aortic Ring Assay
The aortic ring assay was performed as a microvessel outgrowth analysis. Thoracic aortae were removed from normal male Wistar rats and washed in PBS. After fibroadipose tissue removal, aortas were cross-sectioned (approximately 2 mm), washed in cell media, and embedded on GFR–Matrigel®. First, 200 μl of GFR–Matrigel® was added to a 24-well plate and incubated at 37°C for 30 min to form a gel layer. Afterwards, a single aorta section was placed in one side of the well. On the opposite side, RGD–alginate microspheres with or without (control) FBs were added. Again, 200 μl of GFR–Matrigel® was added to the samples and incubated at 37°C for 30 min. The aorta section and the microspheres were then cultured together in M199 supplemented with 10% FBS. Four days after incubation, vascular sprouting from aortic rings was then visualized on an inverted microscope (Axiovert 200M, Zeiss) and photographed. The composite images of aortic ring assays were created by using the photomerge tool (Photoshop CS4; Adobe, San Francisco, CA, USA) to merge all the photos taken from the same well. The area of the sprouting in the micrographs was identified using ImageJ64, and contrast was created to clearly identify the microvasculature. Animal experiments were conducted according to accepted standards of animal care (Declaration of Helsinki, European Community guidelines 86/609/EEC and Portuguese Act 129/92) for the use of experimental animals and with institutional approval.
Statistical Analyses
All experiments were performed in triplicate. Quantifications are expressed as mean ± standard deviation (SD). Statistical significance of difference between various groups was evaluated by analysis of variance (ANOVA) followed by Bonferroni test. For comparison between two groups, the Student's t test was used. A difference between experimental groups was considered significant with a confidence interval of 95%, whenever p < 0.05.
Results
Immobilization of Endothelial Cells and Fibroblasts Inside RGD–Alginate Microspheres
After immobilization of ECs (Fig. 1A) or FBs (Fig. 1B) within RGD–alginate, homogeneous and spherical microspheres were obtained. In order to investigate if immobilized cells were having access to nutrients available in the medium, RGD–alginate microspheres without cells (Fig. 1C) were used. For that purpose, diffusional studies were carried out using two different solutes: albumin and lysozyme. The results demonstrate that both of the investigated solutes were able to diffuse into the microspheres and reach their equilibrium states within 15 min (Fig. 1D). This indicates that low molecular weight compounds diffused into the microspheres.
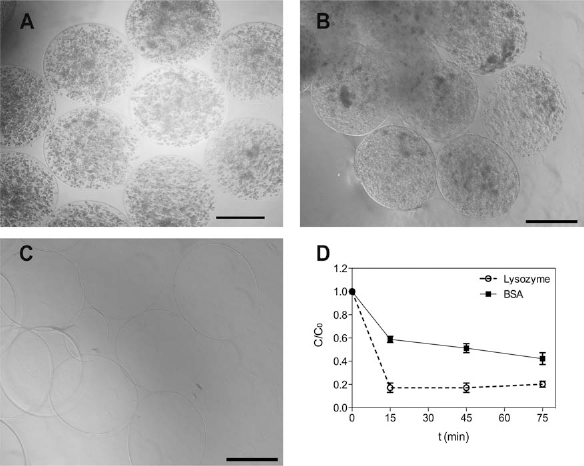
ECs and FBs immobilized in RGD–alginate microspheres. Photomicrographs of arginine–glycine–aspartic acid (RGD)-grafted alginate microspheres at day 4, illustrating the spherical and uniform shape of (A) immobilized human umbilical vein endothelial cells (ECs); (B) immobilized human dermal foreskin fibroblasts-1 (FBs); and (C) without cells (control). Scale bar: 500 μm. (D) Quantification of bovine serum albumin (BSA) and lysozyme diffusion from the Tris-buffered saline (TBS) solution into the RGD–alginate microspheres. Results are expressed as means ± SD of at least three independent experiments.
Metabolic Activity and Viability of Immobilized Cells
The immobilization of ECs within RGD–alginate may represent an in vitro model to study the effect of growth factors in the metabolic activity of these cells in a 3D microenvironment. In our previous studies, ECs immobilized alone during 1 week within RGD–alginate microspheres presented decreased metabolic activity (21). In order to assess the effect of growth factors produced by FBs in the metabolic activity of immobilized ECs, the resazurin assay was used as demonstrated in Figure 2A. ECs immobilized in RGD-grafted alginate microspheres were cultured in medium supplemented with 5 ng/ml VEGF or with the same concentration of VEGF combined with 5 ng/ml bFGF. Additionally studies of Dil-ac-LDL incorporation were performed in order to evaluate cell distribution. Results reveal that ECs immobilized within RGD–alginate microspheres, in medium supplemented with VEGF, were homogenously distributed and that viable cells indeed incorporated the Dil-ac-LDL dye, as illustrated at day 3 (Fig. 2B). For both conditions, the metabolic activity of immobilized ECs decreased during the period analyzed. However, the medium supplemented with the two growth factors provided a statistically significant improvement, which was higher at day 6, comparing to immobilized ECs cultured only in medium with VEGF (p < 0.05 vs. VEGF alone). The effect of cell apoptotic and necrotic activities was also assessed by flow cytometry analysis. Therefore, cells were labeled for annexin V–FITC (apoptosis marker) and PI (a necrosis marker). Cells that, during this period of time, were negative for both annexin V and PI corresponded to the population of viable cells. Apoptotic cells were positive for annexin V and negative for PI, while necrotic cells were positive for both annexin V and PI. Flow cytometry analysis of ECs immobilized in alginate microspheres and cultured, during 6 days, in medium supplemented with VEGF alone revealed only 12% viability (Fig. 2C). Their viability considerably increased to 85%, in the same conditions, when the medium was supplemented with VEGF in combination with bFGF (Fig. 2D).

EC survival and metabolic activity after immobilization. (A) Metabolic activity of ECs after immobilization in RGD-grafted alginate during 6 days, as assessed by the resazurin assay. Immobilized ECs were cultured in the presence of 5 ng/ml of vascular endothelial growth factor (VEGF) or 5 ng/ml of VEGF combined with 5 ng/ml basic fibroblast growth factor (bFGF). An increase in metabolic activity was observed, during 6 days in culture, upon combination of VEGF with bFGF. Bars correspond to the mean ± SD, in comparison to control (immobilized ECs at day 0), n = 3, *p < 0.05. (B) Dil-labeled acetylated low-density lipoprotein (Dil-Ac-LDL) uptake by ECs, in medium supplemented with VEGF, at day 3, visualized using a confocal laser scanning microscope (scale bar: 150 μm). (C) Flow cytometry data, at day 6, of ECs immobilized in RGD-grafted alginate and cultured in the presence of 5 ng/ml of VEGF. (D) Flow cytometry data, at day 6, of ECs immobilized in RGD-grafted alginate and cultured in the presence of 5 ng/ml of VEGF and 5 ng/ml of bFGF. The cells were labeled for annexin V–fluorescein isothiocyanate (FITC; apoptosis) and propidium iodide (necrosis). The results were expressed in terms of percentage of cells expressing these markers.
When FBs were immobilized within RGD–alginate microspheres and cultured during 14 days, their metabolic activity was also evaluated by the resazurin assay and the viability and apoptosis by flow cytometry analysis. Contrary to ECs, most of the immobilized FBs are autosufficient to maintain their metabolic activity during the whole experiment (Fig. 3A). Figure 3B illustrates that immobilized fibroblasts present still approximately 69% viability at day 14 of culture.

FB survival and metabolic activity after immobilization. (A) Metabolic activity of fibroblasts after immobilization in RDG-grafted alginate, as assessed by the resazurin assay. These cells maintained their metabolic activity during 14 days of culture. Bars correspond to the mean ± SD, in comparison to control (immobilized fibroblasts at day 0), n = 3, *p < 0.05. (B) Flow cytometry data of fibroblasts immobilized and cultured in RGD-grafted alginate during 14 days. The cells were labeled for annexin V–FITC and propidium iodide. The results were expressed in terms of percentage of cells expressing these markers.
Metabolic Activity of Cells Using Conditioned Media
In order to examine whether ECs or FBs, in 2D culture, affect each other's metabolic activity in a paracrine manner, conditioned medium of each cell culture was obtained and used in the other cell culture. This effect was compared with control ECs or FBs cultured in fresh medium. In comparison to the control, conditioned medium from FBs (CMFBs) increased (p < 0.05 vs. control) the metabolic activity of ECs (Fig. 4). An increase in metabolic activity can also be observed in Figure 4, in which FBs were cultured in the presence of conditioned medium from ECs (CMECs) (p < 0.05 vs. control).

Metabolic activity of ECs or FBs in conditioned media. ECs were incubated during 24 h in conditioned medium from fibroblasts (CMFBs) or in freshly prepared media (–, control). FBs were incubated during 24 h in conditioned medium from EC (CMECs) or in freshly prepared media (–, control). Results are expressed as percentage of metabolic activity of cells in control media. Bars represent mean ± SD (n = 3, *p < 0.05 vs. control).
Capillary-Like Structure Formation in Cocultures in Indirect Contact
A Transwell culture system was used to examine if FBs can influence the formation and organization of capillary-like structures in indirect contact with ECs. For this purpose, FBs were cultured on the lower chamber of 24-well plates, and ECs were placed on a Transwell insert coated with 50 μl of GFR–Matrigel® (Fig. 5A). ECs cocultured with FBs in indirect contact experienced a slight decrease in metabolic activity when compared to ECs in GFR–Matrigel® alone (Fig. 5B). However, the number of capillary-like structures formed by ECs in GFR–Matrigel® when in coculture with FBs was significantly higher than by ECs alone (Fig. 5C).

Cocultures of ECs with FBs in Transwells. (A) The FBs were cultured on the bottom and ECs were cultured on top of Matrigel-coated Transwell inserts. (B) Metabolic activity of ECs cultured alone or in the presence of FBs (ECs + FBs, coculture) during 24 h. (C) Semiquantification of the number of capillary-like structures formed during 24 h. Bars correspond to the mean ± SD (n = 3, *p < 0.05).
Capillary-Like Structure Formation by Influence of Immobilized FBs
The GFR–Matrigel® assay was used to assess if the ability of ECs to form capillary-like structures would be influenced by the presence of FBs. In Figure 6A, the formation of capillary-like structures can be observed with ECs attaching and spreading to form typical polygonal forms after 24 h. After 5 days of culture in GFR–Matrigel®, the ability of ECs to form capillary-like structures was not maintained (control ECs). On the contrary, the presence of fibroblasts immobilized within RGD–alginate (iFBs) maintained the assembly of ECs into capillary-like structures even after 5 days of culture (Fig. 6A). In the presence of iFBs, a significant increase in the number of capillary-like structures formed could be observed, both at days 1 and 5, compared to the control (Fig. 6B). Furthermore, after 5 days, a significant decrease in the number of capillary-like structures in the control condition could be quantified, compared to day 1. However, in the presence of iFBs, ECs maintained the capillary network even after 5 days of culture. The length of capillary-like structures formed was also quantified at day 1. When ECs were cultured in the presence of immobilized FBs, a significant increase in capillary-like structure length was observed (p < 0.05) in comparison to the control (Fig. 6C). Taken together, these data suggest that immobilized fibroblasts support and preserve the capillary network.
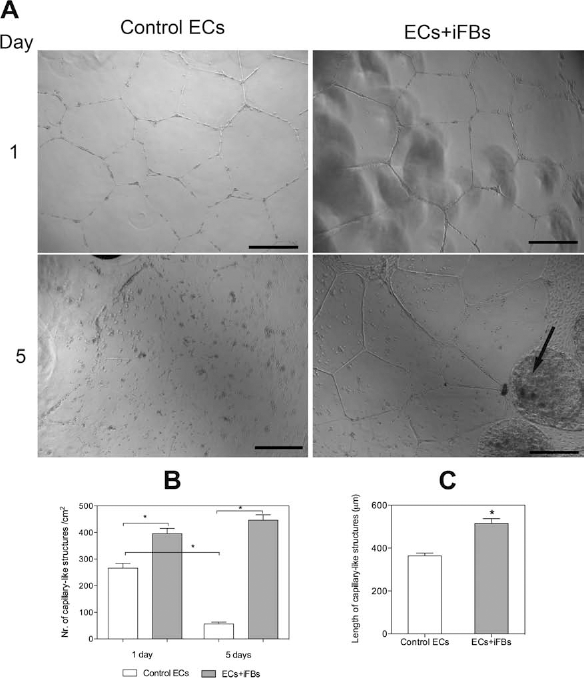
Effect of fibroblasts immobilized within RGD–alginate on formation of capillary-like structures by neighboring ECs. (A) After 1 day of culture, ECs formed capillary-like structures, and after 5 days of culture, only ECs on Matrigel in the presence of immobilized fibroblasts (ECS + iFBs, arrow) maintained the formation of capillary-like structures (scale bar: 500 μm). (B) Quantification of capillary-like structure formation. (C) Quantification of the length of capillary-like structures formed after 1 day. Results are means ± SD of independent experiments (n = 3, *p < 0.05).
Ex Vivo Sprouting by Influence of Immobilized FBs
To further examine the effect of FBs in the angiogenic process, and considering that, in vivo, it also involves other cell types, such as smooth muscle cells, an approach with increased complexity was used, namely, the aortic ring sprouting assay. Aortic rings were cultured in GFR–Matrigel® in the presence or absence of FBs immobilized in RGD–alginate (Fig. 7). The distance between the microspheres and the aortic ring was approximately 6 mm (Fig. 7C). Sprouting of vessels was observed in both cases. However, in the absence of FBs (control), the sprouting scattered throughout the whole aortic ring (Fig. 7A). In the presence of immobilized FBs, sprouting of new capillaries was preferentially oriented toward the microspheres containing immobilized FBs (Fig. 7B). The overall area of sprouting was also quantified using imageJ, showing a decrease when FBs were present and compared to the control (Fig. 7D).

Effect of fibroblasts immobilized (iFBs) within RGD–alginate in capillary-like structures' sprouting. Representative images of aortic ring at day 4 in the presence of microspheres of RGD–alginate: (A) without fibroblasts (control) and (B) with fibroblasts. (C) Scheme of well with aortic ring and RGD–alginate microspheres. (D) Quantification of the area of sprouting. Results are means ± SD (n = 3, *p < 0.05). Scale bar: 500 μm.
Discussion
In tissue regeneration research, the use of models that mimic physiological conditions to test new strategies is of paramount importance since it depends on a number of intricate interactions between distinct cell types, growth factors, and ECM components. One example is the formation of vessels, which is only possible in 3D assays of angiogenesis, a fundamental, but yet unresolved, problem in tissue regeneration. Interactions between mesenchymal and epithelial cells are known to play an important role in orchestrating the development and morphogenesis of tissues and organs (8). Bone cells of mesenchymal origin have been shown to exert a reciprocal influence on endothelial cells (4,18,21,25,45,46). Therefore, in the context of angiogenesis, it seems relevant to study how fibroblasts (FBs), which are committed mesenchymal cells, could influence the microenvironment of endothelial cells to trigger their assembly into capillary-like structures, thus contributing to the regenerative process by promoting angiogenesis. Beyond the roles usually attributed to fibroblasts, namely, contribution to pathological tumor development or fibrosis (26,29), these cells can also be advantageous in therapeutic applications since they are highly abundant, scattered throughout the organism, and can be easily isolated in a noninvasive and patient-specific manner (40).
The present study investigated the modulation of endothelial cell (EC) behavior by fibroblasts (FBs) in terms of viability, metabolic activity, and ability to form capillary-like structures in 3D models. In our study, the effect of supplementing the medium of ECs with a combination of VEGF and bFGF showed a synergistic effect of the two growth factors in improving metabolic activity of ECs immobilized in RGD–alginate microspheres. VEGF alone did not promote the same effect. FBs secrete a number of growth factors, such us bFGF and VEGF (30,36), and Presta et al. presented evidence suggesting the possibility that bFGF indirectly induces neovascularization by activation of the VEGF/VEGFR system (35). Our results are in agreement with Korff et al., who showed additive effects of VEGF and bFGF in decreasing apoptosis in spheroid endothelial cell clusters (28). The VEGF family has been described as assuming a pivotal role during embryonic development in vasculogenesis and angiogenesis (15). Soluble factors produced by FBs, such as VEGF and bFGF, are required for EC survival, differentiation, and network formation (30), although their function decreases during time in culture (9). Therefore, the transplantation of cocultures is hereby appointed as a better strategy rather than growth factor delivery.
The use of cocultures in tissue engineering is related to the fact that both cell types may support survival and growth of each other (11,21). In the present work, conditioned media from ECs and FBs demonstrated that both cell types produced soluble factors capable of influencing the metabolic activity of the other cell type, indicating the reciprocal interrelationship between these two types of cells. Nevertheless, when ECs were cultured in GFR–Matrigel® and in the same chamber with FBs but in indirect contact, the metabolic activity of ECs did not change significantly, which can be explained by the fact that ECs were at different stages of development, namely, proliferation or differentiation, thus exhibiting different levels of metabolic activity.
Proliferation or differentiation of ECs can be influenced by the 3D environment, presence of growth factors, or presence of other cell types, such as FBs. According to Kunz et al. (30), in spheroid cocultures of ECs and FBs, low viability is not found in areas where ECs can migrate and form capillary-like structures in the spheroids. Korff and Augusting (28) investigated a similar model, showing that cells at the surface monolayer of spheroids differentiated to a degree that rendered them survival factor independent, although dependence on survival factors was observed concerning the unorganized cells in the spheroid center (28).
Having this in mind, cocultures of ECs and FBs were used to study the influence of FBs immobilized within RGD–alginate in EC viability and organization. Previous work from the present team showed that other cell types, namely mouse preosteoblasts (MC3T3) (14) and human osteoprogenitor cells (5,21), can be stably immobilized in RGD–alginate and maintained viable during long periods of time (29 days). These findings were confirmed in the present study, since fibroblasts immobilized in RGD–alginate microspheres during 14 days kept a high viability of ca. 70%, which probably corresponds to an underestimation since the new matrix produced by the fibroblasts makes the recovery of all entrapped cells more difficult. The presence of the RGD sequence promoted cell adhesion to alginate and probably mediated survival signals that involve αVβ3 integrin signaling pathways (2,14,21,34). Integrins are known to play a central role in neovascularization, being αvβ3 integrin even considered a target for the development of antiangiogenic therapies in cancer (35). In EC maturation and vessel wall assembly, ECM synthesis is a critical step. Previously, the production of collagen by MC3T3 cells, fundamental in the differentiation step, has been reported as significantly higher within RGD–alginate microspheres than in unmodified alginate ones (14). Singh et al. found that FBs immobilized in alginate could produce collagen type I and, under adequate conditions, namely, combination of hypoxia, BMP-2 supplementation, and long-term intermittent application of dynamic hydrostatic pressure, could be differentiated into the chondrogenic pathway (40). Additionally, Levenberg et al. suggested a close relationship between neosynthesis of ECM and increasing levels of VEGF secreted by FBs in the formation and maintenance of microvasculature (31).
In the present work, the GFR–Matrigel® assay demonstrated that FBs immobilized within RGD–alginate were capable of influencing the formation of capillary-like structures by neighboring ECs, which is in agreement with our previous studies demonstrating similar results when using human mesenchymal stem cells (5). The length of capillary-like structures formed in the presence of immobilized FBs was higher than when ECs were cultured alone. Nevertheless, after 5 days of culture, only the network formed in the presence of immobilized FBs was maintained. The preservation of the capillary-like structures with time is a fundamental issue in tissue regeneration. These results suggest that growth factors produced by FBs could act in a paracrine manner to induce and maintain capillary-like structures. Also, the ability of FBs to contribute to EC survival and vessel assembly is probably closely related to their capacity to produce new ECM, thus creating a more adequate environment for capillary structure formation and maintenance. Grajar et al. reported that FBs stimulate ECs to form mature capillary networks and that this FB-mediated capillary morphogenesis is dependent on matrix metalloproteinase production (20).
Given the complexity of the angiogenesis process, namely, the existence of other structures and cell types besides ECs and FBs, such as pericytes and smooth muscle cells, the aortic ring assay was used to further assess the influence of immobilized FBs in the formation of capillary-like structures. The results suggest that the presence of immobilized FBs induced significant changes in the formation of capillary-like structures by ECs, namely, a decrease in the overall area of sprouting of ECs when immobilized FBs were present, although, in the presence of FBs, the new capillaries formed showed a preferential orientation toward the microspheres containing FBs. Probably, a gradient of growth factors (e.g., bFGF) was produced by the immobilized FBs, directing sprouting. Soluble factors, like bFGF and other established angiogenic factors, can promote endothelial cell scattering during the first steps of the angiogenesis process and, along with the formation of cell–cell interactions, are required phenomena for vessel maturation (35,44). For instance, bFGF has been reported to initially promote the disruption of the basal lamina by inducing protease production of various ECM components by ECs (19), thus contributing to the maturation of the vessels (35).
The current study used different angiogenesis assays to test the influence of fibroblasts in the vascularization process. This study demonstrated the ability of FBs immobilized in RGD–alginate to modulate and support capillary-like structure assembly by endothelial cells. Taken together, these findings indicate that the microenvironment created by these stromal cells in the scaffold modulates capillary morphogenesis, thus stimulating angiogenesis in situ and can potentially be used in tissue regeneration strategies, in clinical scenarios where vascularization is essential.
Footnotes
Acknowledgments
The work carried out at INEB was financed by FEDER funds through the Programa Operacional Factores de Competitividade – COMPETE and by Portuguese funds through FCT – Fundação para a Ciência e a Tecnologia in the framework of projects PEst-C/SAU/LA0002/2011, PEst-OE/ SAU/UI0038/2011, and POCI/SAU-BMA/55556/2004 and the Ph.D. scholarship SFRH/BD/40421/2007 for S.G.G. The authors would like to express their gratitude to Raquel Gonçalves and Ana Rocha for the contribution in the FACS analyses and in the aortic ring assay, respectively. The authors declare no conflicts of interest.
