Abstract
Instant blood-mediated inflammatory reaction (IBMIR) causes rapid islet loss in islet transplantation. Endothelial colony-forming cells (ECFCs) display unique abilities to promote angiogenesis and repair vascular injury compared to those of endothelial cells (ECs), which inhibits the allogeneic and xenogeneic IBMIR. We investigated the coating of pig islets with ex vivo-expanded ECFCs as a strategy to overcome xenogeneic IBMIR. Porcine islets were cocultured with human ECFCs in a specially modified culture medium for 2 days to obtain 70–90% coverage. The coating of pig islets with human ECFCs did not affect the glucose-stimulated insulin secretion capacity or diabetes reversal rate after the transplantation of a marginal islet mass under the kidney capsules of diabetic nude mice compared to that of untreated islets. Uncoated islets, PBS control without islets, and the ECFC-coated islets were examined with an in vitro tubing loop assay using human blood. After 60 min of incubation in human blood, the ECFC-coated islets showed platelet consumption inhibition and low C3a and TAT assay results compared to those of the uncoated islets. Furthermore, there was very little macroscopic or microscopic clotting in the human ECFC-coated pig islets. The protective effect was more prominent compared to that of human EC coating of pig islets in our previous study. We investigated the changes in human-specific MCP-1, IL-8, and tissue factor (TF) levels after the coating of pig islets with human ECFCs or human ECs. The IL-8 levels after coating pig islets with ECFCs were significantly lower than those after coating pig islets with ECs, but there were no significant differences in the MCP-1 or TF levels between the ECFCs and ECs. In conclusion, the coating of pig islets with ECFCs completely prevented all components of xenogeneic IBMIR. ECFCs may be a better source of protection against xenogeneic IBMIR than are mature ECs.
Introduction
Clinical islet transplantation is emerging as a viable treatment option for the restoration of β-cell function in patients with type 1 diabetes, but the shortage of human islets and poor engraftment of transplanted islets limits the application of the procedure. Pig islet cells are an attractive substitute for human islet cells (11,16), and recent studies using a pig-to-nonhuman primate islet xenotransplant model strongly support the feasibility of xenogeneic pig-to-human islet transplantation (4,9). However, these studies also confirm the occurrence of a xenogeneic instant blood-mediated inflammatory reaction (IBMIR) due to the excessive number of islets (over 25,000 IEQs/kg) needed to achieve normoglycemia (4,9). Many strategies have been developed for overcoming IBMIR. Among these, creation of a biologically active surface on the islets using heparin (3) or endothelial cells (13,15) efficiently inhibits allogeneic and xenogeneic IBMIR without increasing the bleeding risk.
Endothelial progenitor cells (EPCs) in the blood and bone marrow contribute to new blood vessel formation in ischemic tissue, and EPC-mediated neovascularization of the pancreas could, in principle, be exploited to facilitate the recovery of non-terminally injured β-cells or to improve the survival and/or function of transplanted islet grafts (6,18). Several studies have described at least two types of EPCs: so-called early EPCs or endothelial cell colony-forming units (CFU-ECs) (spindle-shaped cells with a short life span) and late EPCs or endothelial colony-forming cells (ECFCs) (cobblestone-shaped cells with a high proliferation capacity, which appear sequentially during in vitro culture) (5,10,26). Yoder et al. (26) and Case et al. (5) proposed that the term EPC be restricted to ECFCs, because CFU-ECs are unable to directly participate in postnatal vasculogenesis. Compared with mature endothelial cells (ECs), ECFCs display a unique ability to promote angiogenesis and repair vascular injury. Recent studies have shown that ECFCs are resistant to oxidative stress (8), express higher levels of urokinase (1), and are more sensitive to angiogenic factors (2) and specific thrombin receptors (PAR-1) than are ECs (25). In addition, the tissue factor levels induced by IL-1β are lower in ECFCs than in mature ECs, although ECFCs have similar procoagulant activity and tissue factor (TF) expression in their basal condition compared to those of mature ECs (7). This suggests that ECFCs might be a better source to protect against IBMIR compared to mature ECs. Therefore, further studies using ECFC coatings should be undertaken. The aim of this study was to investigate whether the coating of pig islets with ex vivo-expanded ECFCs can inhibit IBMIR in a xenograft environment.
Materials and Methods
Islet Isolation and Culture
The pancreases of adult market pigs were harvested at the local slaughterhouse. The splenic lobes of the glands were rapidly dissected ex situ (warm ischemic time <15 min), immersed in University of Wisconsin (UW) solution, and transported to the islet-processing laboratory (cold ischemic time <80 min) as previously reported (15). Pancreatic islet isolation and purification were performed as previously described (16,17). The islets were cultured free-floating in standard culture medium (M199, GIBCO BRL, Grand Island, NY, USA) supplemented with Earle's salts, l-glutamine, 2200 mg/L sodium bicarbonate, 25 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer, and 10% pig serum. Modified islet culture medium was manufactured for coculturing and was made up of M199 and endothelial basal medium-2 (EBM-2) supplemented with endothelial cell growth medium-2 (EGM-2) SingleQuots (EGM-2 medium, Lonza Inc., Walkersville, MD, USA) and 5% fetal bovine serum (FBS).
In Vitro Test of Islet Function
Glucose-stimulated insulin release was measured and expressed as the stimulation index (16,17), calculated as the ratio of stimulated (16.7 mM glucose or 16.7 mM glucose plus 5 mM theophylline) to basal (1.67 mM glucose) insulin release during 60 min of static incubation in Krebs–Ringer bicarbonate HEPES buffer. Insulin levels were determined with an insulin immunoradiometric assay kit (Biosource INS-IRMA kit; Biosource Europe SA, Nivelles, Belgium).
Endothelial Colony-Forming Cell Preparation
Human umbilical cord blood (CB) samples (70–100 ml) were obtained from the Seoul Metropolitan Public Cord Blood Bank under a protocol approved by the Institutional Review Board of Seoul National University Boramae Hospital. Total mononuclear cells (MNCs) from CB were obtained via density gradient centrifugation with a Histopaque-1077 (Sigma, St. Louis, MO, USA). MNCs were resuspended in 2 ml EBM-2 supplemented with EGM-2 SingleQuots (EGM-2 medium, Lonza Inc.) and 5% FBS and were seeded at 1 × 107 cells/well on 2% gelatin-coated (Sigma) six-well plates. After 3–4 days of culture, the medium was changed, and unbound cells were removed by washing with PBS. Thereafter, the medium was replaced every 3 days. ECFCs with a cobblestone-like morphology were monitored daily and rapidly grew from MNCs between 10 and 20 days after the initial culture. For the expansion of ECFCs, colonies were trypsinized and cells were serially passaged and cultured (20).
Flow Cytometric Analysis and Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
Surface markers of ECFCs were analyzed using a fluorescence-activated cell sorter (FACS) (FACSCalibur, Becton Dickinson). Cells (5 × 105) were stained by incubation for 20 min at 4°C with varying concentrations of the primary or isotype control antibody in 100 μl PBS with 0.5% bovine serum albumin (BSA), washed three times with PBS, and analyzed by FACS. Antibodies were fluorescein isothiocyanate (FITC)-conjugated mouse monoclonal anti-human CD31, phycoerythrin (PE)-vascular endothelial-cadherin (VE-cadherin), PE-kinase insert domain receptor (KDR, R&D Systems, Minneapolis, MN), and FITC-CD105 (Abcam, Cambridge, MA). To detect the expression of von Willebrand factor (vWF), cells were permeabilized with 9:1 methanol/PBS for 20 min at −20°C and incubated with anti-human vWF (2 μg/ml, Dako, Carpiteria, CA). The secondary antibody used for vWF was anti-mouse-PE (BD Pharmingen). After washing and fixing, at least 10,000 gated cells were acquired and analyzed using CellQuestPro software.
Total RNA was extracted from ECFCs, and RT-PCR was performed to detect the mRNA expressions of KDR, vWF, VE-cadherin, endothelial nitric oxide synthase (eNOS), fms-related tyrosine kinase 1 [Flt-1 or vascular endothelial growth factor (VEGF) receptor], CD31, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Total RNA from ECFCs was extracted with a QIAGEN RNeasy Mini Kit (QIAGEN, Hilden, Germany), and 1 μg of RNA was reverse-transcribed into first-strand cDNA using the SuperScript™ II Reverse Transcriptase (Invitrogen, Carlbad, CA). Reverse transcriptase polymerase chain reaction amplified each cDNA by AccuPower® PCR premix (BIONEER, Seoul, Korea). PCR conditions were 94°C for 5 min, 25–30 cycles of 94°C for 30 s, 54–67°C for 30 s, and 72°C for 1 min, ending with 72°C for 10 min. Products were separated on 1.5% agarose gels and stained with ethidium bromide.
To further verify that these cells were ECFCs, binding of FITC-labeled lectin from Ulex europeaus agglutinin (UEA-1; Vector Laboratories, Burlingame, CA), which is specific for human endothelial cells, and uptake of 1,1-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine-labeled acetylated low-density lipoprotein (DiI-acLDL, Molecular Probes, Eugene, OR), a function associated with endothelial cells, were assessed.
Mouse Hindlimb Ischemia
The following animal procedures applied were approved by the Institutional Animal Care and Use Committee of the Samsung Biomedical Research Institute, Laboratory Animal Research Center. Eight-week-old male, athymic BALB/c nude mice (Oriental Co. Ltd., Seoul, Korea) were anesthetized with an IP injection of a mixture of Rompun (25 mg/kg, Bayer Korea Ltd., Seoul, Korea) and Zoletil 50 (40 mg/kg, VIRBAC Laboratories, Carros, France). Unilateral hindlimb ischemia was induced in mice through ligation and excision of the femoral artery, as previously described (20), and mice were randomly administered intracardiac injections of late EPCs (1 × 106), human umbilical vein endothelial cells (HUVECs; 1 × 106), or PBS. Zero and 21 days after the induction of ischemia, the ratio of ischemic to nonischemic limb blood flow perfusion was measured with a laser Doppler perfusion image analyzer (LDPI; Moor Instruments, Wilmington, DE, USA). The perfusion ratio was calculated as the flux ratio between the ischemic and nonischemic limbs.
Histological Assessment of Hindlimb Ischemia
The mice were euthanized, and their skeletal muscles were isolated, embedded in OCT compound (Tissue-Tek; Sakura Finetek, Torrance, CA), frozen in liquid nitrogen, and stored at −80°C. Tissue was cut into 10-μm sections and stained with hematoxylin and eosin (H&E). For immunofluorescent staining, sections were fixed in acetone, blocked with 10% normal donkey serum, and incubated with primary sheep anti-human CD31 (1:100; R&D Systems, Minneapolis, MN, USA), followed by secondary donkey anti-sheep antibodies (Molecular Probes, Eugene, OR, USA). Tissue capillary density was measured via staining with FITC-conjugated murine EC-specific BS-1 lectin. Capillary density was calculated as the number of capillaries/mm2 averaged from five randomly selected fields.
Endothelial Colony-Forming Cell Coating of Pig Islets
ECFC or human aortic endothelial cell (HAEC) coating was performed as previously described (15). ECFCs or HAECs were harvested using 1x trypsin-EDTA (GIBCO BRL). ECFCs were washed with EGM-2 medium, centrifuged at 180 x g for 5 min in a 14-ml Falcon tube (Becton-Dickinson Labware, Lincoln Park, NJ, USA), and counted in a Burker chamber. After washing, ECFCs were suspended in EGM-2 medium to 3 × 106 cells/ml. Ten thousand islet equivalents (IEQ) were mixed together with 3 × 106 ECFCs in 300 μl of modified culture medium. The mixture was incubated for 1.5 h at room temperature and mixed gently via pipetting every 15 min. Coated islets were transferred to 150-mm petri dishes and cultured for 2 days in a modified culture medium. The coverage of each islet with ECFCs was examined under fluorescent microscopy and calculated in an image analysis program (i-solution version 7.3, IMT, Korea). In addition, levels of human-specific interleukin-8 (IL-8), monocyte chemotactic protein-1 (MCP-1; BD Biosciences, San Jose, CA, USA), and tissue factor (TF; Assaypro LLC, St. Charles, MO, USA) were measured using enzyme immunoassays (EIA) according to the manufacturer's instructions. Total protein was measured using a Bradford protein assay.
In Vitro Tubing Loop System
Blood donors were healthy, without specific disease, and had no medication history for the 1 month prior to donation. The protocol was approved by the Institutional Review Board of Seoul National University Hospital.
A modified tubing loop model was performed as previously described (15). Using an 18-gauge needle, fresh human blood was collected in a surface-heparinized 60-ml syringe (Corline Systems AB, Uppsala, Sweden). This blood was transferred to a heparinized polyvinyl chloride (PVC) tube (Corline Systems AB; diameter 6.3 mm, length 390 mm) via a surface-heparinized silicon tube. Each PVC tube was filled with 7 ml fresh human blood, and then 100 μl PBS was added as a negative control, 5,000 IEQ uncoated islets in 100 μl PBS were added as a positive control, or 5,000 IEQ ECFC-coated islets were added as the treatment group. The loops were then closed and placed in a 37°C incubator on a rocking apparatus. Blood was sampled at 0, 5, 15, 30, and 60 min. After a 1-h incubation, all of the loop contents were filtered through 70-μm diameter filters (Filcons, Cup-Type; DAKO, Glostrup, Denmark). The remaining filtered blood was collected in EDTA tubes for hematologic analysis (platelets, lymphocytes, monocytes, and granulocytes), assays of complement activation (C3a), and coagulation activation [thrombin–antithrombin (TAT)].
Hematologic analyses were performed with a Coulter ACT-diff analyzer (Beckman Coulter, Miami, FL, USA), and C3a and TAT were quantified using ELISA assay kits (C3a EIA kit: Quidel, San Diego, CA, USA; Enzygnost-TAT kit: Behringwerke, Marburg, Germany).
In Vivo Islet Potency
Following animal procedures applied were approved by the Institutional Animal Care and Use Committee of the Seoul National University Hospital. The in vivo tests of islet functions were assessed via islet transplantation under the kidney capsules of athymic Balb/c nude mice (Orient Co. Ltd., Seoul, Korea), which were rendered diabetic with a single IP injection of 230 mg/kg streptozotocin (Sigma Chemicals) 4–5 days prior to transplantation. Nonfasting blood glucose was measured with a glucose meter (One Touch Ultra Sensor, Lifescan, Milpitas, CA, USA). Mice with sustained hyperglycemia (>350 mg/dl) were used as islet graft recipients, into which 2,000 IEQ porcine islets with or without ECFC coating were transplanted under the kidney capsule. Blood samples were collected from the tail vein twice weekly to determine glucose levels. Islets were considered efficacious when two consecutive posttransplant blood glucose levels were below 200 mg/dl. Nephrectomies were performed to determine the graft dependence of diabetes reversal.
Morphological Evaluation of Blood Clots in the Loop Experiments
When the perfusion experiment was complete, blood clots derived from the loop experiments were collected. The clots were fixed in 4% neutral buffered formalin and embedded in paraffin. All clots were cut into 4-μm sections and stained with H&E. For immunofluorescent staining, the antigens from the deparaffinized sections were retrieved in a heated 0.1 M citrate solution (pH 6.0) and incubated with 0.5% Triton X-100 containing PBS. Nonspecific binding sites were blocked with 10% normal donkey serum and incubated with primary sheep anti-human CD31 (AF806, R&D Systems), followed by secondary donkey anti-sheep antibodies (A21436, Molecular Probes). The sections were also stained for primary goat anti-human KDR (AF357, R&D Systems) and secondary donkey anti-goat antibodies (A11058, Molecular Probes) and were additionally stained for primary mouse anti-porcine insulin (I2018, Sigma-Aldrich, MO, USA) and secondary goat anti-mouse antibodies (A11001, Molecular Probes).
Statistical Analysis
For comparison between different blood donors, platelet counts were transformed to a percentage of the zero time value, and C3a and TAT assay results were transformed to a percentage of the positive control value. Data are presented as means ± SEM. Mean values were compared using the paired Student's t-test or one-way analysis of variance followed by Tukey's multiple comparison test.
Results
Successful Isolation of CB-Derived ECFCs and Their In Vivo Neovascularization in Hindlimb Ischemia
ECFCs were successfully isolated from CB and expanded ex vivo (19). ECFCs with typical cobblestone morphologies emerged in culture after 1–3 weeks, showed DiI-ac-LDL uptake and UEA-1 binding (Fig. 1A), and expressed endothelial cell markers CD31, CD105, VE-cadherin, vWF, KDR, Flt-1, and eNOS according to FACS (Fig. 1B) and RT-PCR (data not shown). We investigated whether the isolated ECFCs improved neovascularization in a mouse model of hindlimb ischemia. Transplantation of ECFCs led to significant improvements in hindlimb blood flow recovery and limb salvage compared to those of the PBS- or HUVEC-treated control groups (Fig. 1C, D). In H&E-stained specimens, the level of fatty degeneration with necrosis was reduced in mice transplanted with ECFCs compared to that of the control mice (Fig. 1E, left panel). Capillary density measured using BS-1 lectin markedly increased in mice injected with ECFCs compared to that of the control mice (Fig. 1F). We also confirmed that ECFCs were incorporated into the capillary spaces, while HUVECs were rarely detected in ischemic muscles (Fig. 1E, right panel). These results suggest that the administered ECFCs contributed to improvements in blood perfusion and limb salvage by homing to ischemic areas.

Enhanced neovasculogenesis in hindlimb ischemia after endothelial colony-forming cell (ECFC) transplantation. (A) ECFCs had a cobblestone-shape under a light microscope (middle panel, scale bar: 100 μm) and showed endothelial-specific 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine-labled acetylated low-density lipoprotein (DiI-ac-LDL) uptake (red) and FITC-labeled lectin from Ulex europeaus agglutinin (UEA-1) binding (green) (right panel, scale bar: 100 μm). Arrows outline representative ECFC colonies appearing after seeding cord blood-derived mononuclear cells (MNCs) (left panel, scale bar: 100 μm). (B) By flow cytometry, ECFCs showed endothelial-specific surface markers. Open blue line histograms represent test antibodies, and red field histograms represent isotype IgG antibodies. (C) Quantitative graph of blood flow perfusion in the laser Doppler perfusion analysis. CB, cord blood ECFC-injected mice (n = 10); HUVEC, human umbilical vein endothelial cell-injected mice (n = 10); PBS, PBS-injected mice (n = 12), ∗p < 0.05 versus PBS or HUVEC. (D) Mean severity scores for tissue necrosis. (E) Representative photomicrographs of histological sections in ischemic hindlimb muscles after H&E staining (left panel, magnification 200x). ECFCs incorporated into the murine capillary space were stained with anti-human CD31 antibodies (red, arrows), murine vessels were stained with BS-1 lectin (green), and blue color indicates DAPI (right panel, scale bar: 50 μm). (F) Capillary density identified via staining for BS-1 lectin. Representative photographs (left panel) and quantitative graph (right panel) of capillary density. ∗p < 0.05 versus HUVEC or PBS. Scale bar: 100 μm. VE-cadherin, vascular endothelial cadherin; vWF, von Willebrand factor; KDR, kinase insert domain receptor.
ECFC Coating of Pig Islets
We used 10-fold higher numbers of human ECFCs in our two-species model (14) compared to the quantity of endothelial cells used to coat human islets in a single-species model (12). After a culture period of 2 days in the modified culture medium, 70–90% coverage of the islets was observed (Fig. 2A).

ECFC-coated islets and blood clots derived from loop experiments. (A) Pig islets coated with human ECFCs. ECFCs were labeled with the fluorescent dye PKH26. ECFC-coated islets under light (left panel, magnification 100x) and fluorescence (right panel, magnification 100x) microscopy. (B) Representative photographs of blood clots. PBS control, 100 μl PBS; uncoated islets, 5,000 IEQ (islet equivalent) uncoated islets in 100 μl PBS; coated islets, 5,000 IEQ ECFC-coated islets in 100 μl PBS. (C) Quantitative graph of clot size. ∗∗∗p < 0.001 versus ECFC-coated islets or PBS control. (D) The effects of coating pig islets with human ECFCs on platelet. Filled circles, 5,000 IEQ uncoated islets in 100 μl PBS; open circles, 100 μl PBS; filled triangles, 5,000 IEQ ECFC-coated islets in 100 μl PBS. ∗∗p < 0.01, ∗∗∗p < 0.001 versus ECFC-coated islets or PBS control. (E) Hematoxylin and eosin staining of blood clots. Immunostaining of blood clots derived from loop experiments (D, E). (F) Human CD31 (red), insulin (green), and DAPI (blue) staining. Human ECFC-coated pig islets showed less infiltration of CD31-positive cells compared with that of uncoated islets. (G) Human kinase insert domain receptor (KDR; red), insulin (green), and DAPI (blue) staining. ECFC-coated pig islets showed an intense staining of hKDR in the cells found enveloping the pig islets.
In Vitro and In Vivo Functions of ECFC-Coated Islets
The stimulation indices in the high-glucose or high-glucose plus theophylline groups of uncoated islets were 2.20 ± 0.70 (mean ± SD, n = 4) and 7.40 ± 3.25 (n = 4), respectively. The ECFC-coated islets showed similar glucose-stimulated (2.07 ± 0.38, n = 4) or glucose plus theophylline-stimulated release values (6.77 ± 1.62, n = 4) to those of the uncoated islets. The transplantation of ECFC-coated islets (2,000 IEQ) into diabetic nude mice displayed similar rates of diabetes reversal (50%) and similar blood glucose levels to those of uncoated islets (Table 1).
In Vitro and In Vivo Islet Functions

Data are means ± SD. SI, stimulation index, calculated as the ratio of stimulated (16.7 mM glucose or 16.7 mM glucose plus 5 mM theophylline) to basal (1.67 mM glucose) insulin.
Perfusion of Pig Islets with Fresh Human Blood in the Tubing Loop Model
Untreated control pig islets caused massive clotting when they came into contact with human blood (Fig. 2B, C). In parallel with coagulation activation, a considerable drop in platelet count was observed (Fig. 2D). In marked contrast, human ECFC-coated pig islets tolerated contact with human blood better than did untreated pig islets. In addition, the protective effect was more prominent with ECFCs compared to the human EC coating of pig islets in our previous study (14). Platelet counts in blood mixed with pig islets coated with human ECFCs remained in the same range as that in the control loops containing PBS alone without any islets (Fig. 2D). Furthermore, there was very little macroscopic or microscopic clotting in the human ECFC-coated pig islets or in the control loops containing PBS alone (Fig. 2B, E). Pig islets entrapped in clots from the blood-loop perfusions were examined using immunohistochemistry. In the blood clots, CD31-positive monocytes, neutrophils, platelets, and a subpopulation of T cells were found surrounding both uncoated pig islets and pig islets covered with human ECFCs. Human ECFC-coated pig islets showed less infiltration of CD31-positive cells when compared with that of uncoated islets (Fig. 2F). Pig islets covered with human ECFCs showed an intense staining of hKDR in the cells found enveloping the pig islets (Fig. 2G). The sizes of blood clots correlated to the consumption of platelets, monocytes, and granulocytes. TAT and C3a values in blood retrieved from loops containing untreated pig islets were markedly increased compared to those of the control loops without islets (Fig. 3A, B). In contrast, in loops containing pig islets covered with human ECFCs, coagulation and complement activation were reduced to background values (Fig. 3A, B). Untreated control islets caused a near complete consumption of granulocytes and monocytes; however, there was no significant decrease in lymphocyte count. Human ECFC-coated pig islets induced markedly less consumption of granulocytes and monocytes (Table 2).
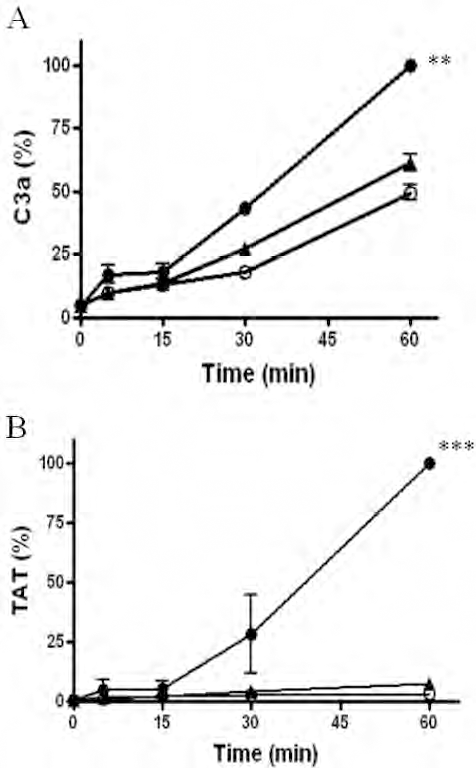
The effects of coating pig islets with human ECFCs on complements and coagulation. Samples were collected at 0, 5, 15, 30, and 60 min. (A) complements and (B) thrombin–antithrombin analyses were performed. Filled circles, 5,000 IEQ uncoated islets in 100 μl PBS; open circles, 100 μl PBS; filled triangles, 5,000 IEQ ECFC-coated islets in 100 μl PBS. ∗∗p < 0.01, ∗∗∗p < 0.001 versus ECFC-coated islets or PBS control. C3a, complement activation protein; TAT, thrombin-antithrombin.
Cell Counts of Fresh Human Blood Cells in Contact With Pig Islets Coated With Human Endothelial Progenitor Cells (EPC) or Untreated Islets After 60 Minutes

Data are means ± SEM.
Control contains blood and PBS, but no islets.
p < 0.05 compared with findings in loops without islets.
Comparisons of MCP-1, IL-8, and Tissue Factor Expressions Between ECFC Coatings of Pig Islets and EC Coatings of Pig Islets
The effect of an ECFC coating on IBMIR in this study was more noteworthy compared to that of the human EC coating in our previous study (15). Therefore, we investigated the changes in human-specific MCP-1, IL-8, and tissue factor levels, all of which have been shown to be associated with IBMIR (11,14,21,24), after coating pig islets with ECFCs or HAECs. There were no significant differences in the MCP-1 or TF levels after coating pig islets with ECFCs or HAECs (Fig. 4A, B). However, the IL-8 levels after the coating of pig islets with HAECs were significantly higher than those after the coating of pig islets with ECFCs (Fig. 4C). The levels of human-specific MCP-1, IL-8, and tissue factor were negligible in the uncoated pig islets (data not shown).

(A) Monocyte chemoattractant protein-1 (MCP-1), (B) tissue factor (TF), and (C) interleukin-8 (IL-8) levels in human aortic endothelial cells (HAECs), ECFCs, and HAEC-or ECFC-coated islets. The levels of human-specific MCP-1, IL-8, and TF did not differ between HAECs and ECFCs. IL-8 levels after the coating of pig islets with HAECs were significantly higher than those after the coating of pig islets with ECFCs; however, MCP-1 and TF levels after the coating of pig islets were not different between the ECFCs and HAECs. ∗∗∗p < 0.001 versus ECFC-coated islets.
Discussion
Pig islets constitute a possible resolution to the shortage of human islets for transplantation. However, after the intraportal infusion of porcine islets in primates, many islets are lost through IBMIR. In addition, early loss of intraportally transplanted pig islets occurred in primates (4,9), even when attempts were made to prevent IBMIR caused by cobra venom factor and heparin or low molecular weight dextran sulfate (23). Therefore, it is mandatory to develop more effective targeting of xenogeneic IBMIR to minimize the critical islet dose and possibly also the requirements for immunosuppression through prevention of IBMIR-mediated augmentation of the adaptive immune response (19). The coating of islets with ECs efficiently inhibits allogeneic and xenogeneic IBMIR without increasing the bleeding risk (13,15); however, only partial inhibition of the xenogeneic IBMIR was shown in the short-term coculture, which might be related to EC activation during the coculture of HAEC and pig islets (15).
In this study, we also found significantly increased IL-8 levels after the coating of pig islets with HAEC (Fig. 4C). Compared with mature ECs, ECFCs have a prominent ability to promote angiogenesis and repair vascular injury (Fig. 1). Therefore, ECFC-mediated neovascularization could be exploited to improve the survival and/or function of transplanted islet grafts (6,18). Furthermore, ECFCs are resistant to oxidative stress (8), are more sensitive to angiogenic factors (2), and express higher levels of CX chemokine receptor (CXCR) 4 (20) than do ECs. Furthermore, the CXCR4 expression levels vary widely in ECFCs isolated from different donors, and the in vitro and in vivo angiogenic potentials of ECFCs are linearly correlated with their CXCR4 expression levels (20). In addition, the tissue factor levels induced by IL-1β are lower in ECFCs than they are in mature ECs (7). Therefore, we investigated whether ECFCs might be a better source of protection against IBMIR compared to ECs.
In this study, there was a significant increase in IL-8 level after the coating of pig islets with HAECs. However, there was no increase in IL-8 level after the coating of pig islets with human ECFCs. In addition, compared to human EC-coating of pig islets (15), more complete inhibition of xenogeneic IBMIR was shown after human ECFC coating of pig islets in the in vitro loop model. Also, the human ECFC-coating of pig islets did not affect the glucose-stimulated insulin secretion capacity or diabetes reversal rate after the transplantation of a marginal islet mass (2,000 IEQ) under the kidney capsule of diabetic nude mice compared to that of untreated islets.
ECFCs have several advantages as islet–ECFC composites compared to mature ECs. First, ECFCs can be isolated from peripheral blood, while mature ECs require the isolation of small vessel segments. Therefore, it is relatively noninvasive to isolate ECFCs compared to that of ECs. Second, ECFCs have a robust proliferation potential, and they can be propagated in an animal serum-free system to 150 million cells from unmanipulated peripheral blood and can be cryopreserved with intact proliferation potential and function after thawing, which can be explored in terms of their clinical therapeutic applicability (22). In our previous study (15), the level of HAEC coverage was ≤50% in the subgroup cocultured for less than 5 days, while the level of ECFC coverage was higher (ranging between 70% and 90%) for a 2-day culture period in this study, which might be related to the robust proliferation potentials of ECFCs (26). Third, our previous study showed that mature ECs might be activated after EC coating of pig islets, and that endothelial cell activation might have a detrimental effect on IBMIR (15).
In this study, we also found that IL-8 levels after the coating of pig islets with HAECs were significantly higher than those after the coating of pig islets with ECFCs, although there was no significant difference in the MCP-1 or TF levels after the coating of pig islets between the ECFCs and HAECs. These results suggest that ECFCs might be a better source for the coating of pig islets compared to mature ECs. Lastly, compared to mature ECs, ECFCs have profound vessel formation in vivo (10,20), and we demonstrated this using a hindlimb ischemia model. Therefore, the coating of islets with ECFCs potentially enhances revascularization of islet grafts, although we did not find significant differences in the diabetes reversal rate of nude mice between the human ECFC-coated pig islets and uncoated pig islets. Like the mature ECs, ECFCs may be a good preventive source against allogeneic IBMIR, although we did not investigate the ECFC coating effect on allogeneic IBMIR in this study. Therefore, this topic requires further study.
Recipient ECFCs could be retrieved from peripheral blood prior to transplantation, cultured to expand their number, and cryopreserved for later use (22). Subsequently, at the time at which a suitable pig pancreas is prepared or a human pancreas donor is identified, the ECFCs could be thawed and cocultured with the isolated pig or human islets for 2 days, a clinically acceptable time frame. The coating of isolated pig or human islets with human ECFCs might abrogate xenogeneic or allogeneic IBMIR could potentially enhance revascularization, and may decrease the rejection process.
In conclusion, the coating of pig islets with ECFCs completely prevented all components of xenogeneic IBMIR. ECFCs may, therefore, be a better source of protection against xenogeneic IBMIR compared to mature ECs.
Footnotes
Acknowledgments
This work was supported by a grant from the Innovative Research Institute for Cell Therapy of the Republic of Korea (A062260). The authors declare no conflicts of interest.
