Abstract
Cold-stored arteries are biomaterials that potentially represent an abundant “off-the-shelf” source of vascular grafts for use in vascular surgery. One of the keys to reestablishing the antithrombogenic endothelial cell (EC) lining of cold-stored arterial grafts is to maximize the number of ECs that attach following seeding. In this study, the cold-stored sheep carotid artery is used as a substrate to determine the conditions that maximize EC adherence following seeding. The effect of serum concentration, duration of seeding incubation, seeding density, and period of cold storage on attachment of ECs following seeding of 4-week cold-stored sheep carotid arteries (n = 5 arteries), 8-week cold-stored sheep carotid arteries (n = 5 arteries), and 12-week cold-stored sheep carotid arteries (n = 5 arteries) was examined. Three experiments (serum concentration, time of incubation, and seeding density) were conducted to determine the conditions that maximized the number of cultured sheep carotid artery ECs that attached to cold-stored sheep carotid artery following seeding. A flat sheet model was used. Serum concentration (0%, 10%, 20%, and 30%) in the seeding suspension did not have a significant effect on overall EC adherence on 4-, 8-, and 12-week cold-stored arteries. Time of seeding incubation (30, 60, and 90 min) did not have a significant effect on overall EC adherence on 4-, 8-, and 12-week cold-stored arteries. Seeding density (500,000 cells/ml, 1,000,000 cells/ml, and 2,000,000 cells/ml) had a significant effect (p = 0.036) on overall EC adherence on 4-, 8-, and 12-week cold-stored arteries. The period of cold storage (4, 8, and 12 weeks) of the artery had a significant effect (p = 0.002, p < 0.0001, p < 0.0001 in serum, time, and seeding density experiments, respectively) on overall EC adherence following seeding. Pairwise comparisons of EC adherence revealed the following. In the serum experiment, EC adherence on 4-week cold-stored arteries was significantly greater than on 8-week cold-stored arteries (p = 0.003) and 12-week cold-stored arteries (p = 0.002). This effect was due largely to the significant difference between EC adherence on 4-week and 8-week cold-stored arteries (p = 0.0002) and between EC adherence on 4-week and 12-week cold-stored arteries (p = 0.0091) at 20% serum. In the time experiment, EC adherence on 4-week cold-stored arteries was significantly greater than on 12-week cold-stored arteries (p < 0.0001). In the seeding density experiment, EC adherence on 4-week cold-stored arteries was significantly greater than on 8-week cold-stored arteries (p < 0.0001) and 12-week cold-stored arteries (p < 0.0001). In the same experiment, EC adherence following seeding at a density of 1,000,000 cells/ml and 2,000,000 cells/ml was significantly greater (p = 0.03 and p = 0.02, respectively) than EC adherence following seeding at a density of 500,000 cells/ml. Thus, it was determined that 4-week cold-stored arteries were superior to 8- and 12-week cold-stored arteries in terms of the number of ECs that adhered. It was also determined that a seeding density of 1,000,000 or 2,000,000 cells/ml was superior to a seeding density of 500,000 cells/ml in terms of producing maximal EC attachment. The ideal conditions, from those examined, for maximizing EC attachment to cold-stored arteries were 4 weeks of cold storage, a serum concentration of 20%, a seeding density of 2,000,000 cells/ml, and a duration of incubation of 30–90 min.
Introduction
A requirement for rapid graft reendothelialization following endothelial cell (EC) seeding procedure is that the ECs adhere in large numbers across the luminal surface of the graft (5,25,28,29). One of the factors that determines adherence of ECs on a substrate is the nature of the substrate, specifically the number of binding sites it contains. The greater the number of binding sites for ECs in the substrate, the greater the number of ECs that may adhere (17,19,25,30,34).
Sheep carotid arteries that have been cold-stored for 4–16 weeks are devoid of native ECs but retain their extracellular framework. They are therefore similar in their morphology to rabbit femoral arteries that have been cold stored for at least 4 weeks (6,8). Little is known about the effect, if any, of duration of cold storage on EC attachment. The benefit of examining a range of cold storage periods, such as 4, 8, and 12 weeks, is that it helps to determine the period that is best suited to EC attachment.
The condition of the seeding suspension placed on the surface of the substrate may play an important role in determining the number of ECs that adhere to the substrate. For instance, the level of serum in the seeding suspension may be a factor. Serum, which is used in conventional cell culture technique, contains adhesive glycoproteins (9,26). Another important factor may be the density of ECs in the suspension (5,27). A high density of ECs means that a large number of ECs are available to adhere to the substrate (28,29). A contributing factor to EC adherence to the substrate may be the time of incubation of the seeding suspension on the substrate (5,27). ECs may require a period of time to settle and adhere to the substrate via the interactions that form between their membrane-bound adhesion molecules (integrins) and the binding sites in the substrate (33).
The optimal serum concentration, seeding density, and time of incubation for seeding of cold-stored arteries are not known. By “optimal,” it is meant the value, from the range of values examined, that gives maximal cell adherence in terms of the number of cells that adhere to the substrate. In the experiments of the current study, the range of values selected for serum concentration (0%, 10%, 20%, and 30%), time of incubation (30, 60, and 90 min), and seeding density (500,000, 1,000,000, and 2,000,000 cells/ml) is based on the range of values used in published studies, including optimization studies, which have examined EC seeding of substrates consisting of mechanically denuded arteries (5,23,29,31) and synthetic materials (24,32,34,35).
The current study describes the EC seeding of sheep carotid arteries that had been cold stored for 4 weeks or greater. The cold-stored sheep carotid artery differs from both the mechanically denuded artery and the synthetic materials previously used in EC seeding studies. Unlike the mechanically denuded artery, the sheep carotid artery that has been cold stored for 4 weeks or greater is acellular throughout its wall. Unlike synthetic materials, the cold-stored sheep carotid artery contains both collagen and elastin, two important components of the connective tissue of normal blood vessels.
Aims
A combination of immunohistochemistry and experimental manipulation of factors (i.e., serum concentration, time of incubation, and seeding density) will be used to determine the optimal conditions for EC adherence following EC seeding of sheets prepared from 4-, 8-, and 12-week cold-stored sheep carotid arteries. EC adherence to the sheets may depend on the concentration of serum, the time allowed for the cells to attach to the luminal surface, and the number of cells that are present in the seeding suspension. In addition, the optimal cold storage period out of the three periods examined will be determined.
Materials and Methods
Overview
Experiments 1, 2, and 3 were performed to determine the optimal conditions for EC adherence on the cold-stored sheep carotid artery. Experiment 1 was the serum concentration experiment, as the variable relating to the seeding suspension was the concentration of serum it contained. Experiment 2 was the time-of-incubation experiment, as the variable relating to the seeding suspension was the duration of its incubation on the luminal surface of the cold-stored arterial sheet. Experiment 3 was the seeding density experiment, as the variable relating to the seeding suspension was the density of ECs contained within. In each experiment, the substrate consisted of sheets prepared from three groups of cold-stored arteries. Group a consisted of 4-week cold-stored arteries (n = 5 arteries), group b consisted of 8-week cold-stored arteries (n = 5 arteries), and group c consisted of 12-week cold-stored arteries (n = 5 arteries).
Cold Storage
All experiments involving the use of animals were performed under the appropriate and ethical animal care guidelines of the National Health and Medical Research Council.
A carotid artery (15–20 cm length) was unilaterally excised aseptically from each of five outbred Merino sheep (sheep A, B, C, D, and E). Following irrigation with normal saline (Baxter, Australia) containing 10 U/ml heparin sodium (porcine mucous; Multiparin, Fisons Pty Ltd) to remove blood, the arteries were placed in storage at 4°C for 12 weeks (group c). Four and 8 weeks after these excisions, another five arteries of similar length were excised from five sheep (sheep F, G, H, I, and J at 4 weeks, and sheep K, L, M, N, and O at 8 weeks). These were irrigated and placed in storage at 4°C for 8 weeks (group b) and 4 weeks (group a), respectively.
This excision and storage protocol produced 4-, 8-, and 12-week cold-stored arteries whose cold storage periods, when elapsed, converged on a day for the performance of an experiment.
Endothelial Cell Isolation and Culture
Carotid arteries (each of 15–20 cm length) were unilaterally excised aseptically from each of four outbred Merino sheep and irrigated with heparinized Hanks' balanced salt solution-working stock (HBSS-WS; Sigma) to remove blood. Endothelial cells were isolated from the arteries using collagenase type IV (2 mg/ml; Sigma) dissolved in HBSS-WS. The isolated cells were cultured and serially passaged to P2 and P3 in 9-cm diameter petri dishes (Nunclon) containing medium 199-growth medium (M199-GM; Sigma). The P2 and P3 cells from all four sheep were pooled and cultured in M199-GM to 95-100% confluence before use in seeding procedure.
Tissue Preparation
Following storage at 4°C for the specified period, carotid arteries were equilibrated to room temperature, cannulated at one end, and flushed with HBSS-WS to remove debris. Arteries were divided transversely at 0.5-cm-length intervals to obtain pieces. Each piece was cut open with a longitudinal incision to form a sheet (0.5 × 0.5 cm). Each sheet was placed in a horizontal position on the bottom of a plastic petri dish with its luminal surface facing upwards. A drop of normal saline was added to the luminal surface and the sheets placed overnight in the cell culture incubator.
Seeding and Fixation
Petri dishes containing 95–100% confluent cultures of EC (P3 and P4) were rinsed and the cells detached and combined. An aliquot was collected and assessed using 0.4% trypan blue (Sigma) to determine the percentage of viable cells, which was found to be 95–100%. The remaining cells were used to prepare the seeding suspension.
The seeding suspension was made using M199-GM. A drop of the suspension was added to the horizontal luminal surface of each sheet and the cells allowed to adhere in a cell culture incubator. The volume of suspension added to the sheet was sufficient to cover its entire luminal surface. Following incubation, nonadherent cells were removed by adding excess nutrient medium to the petri dishes containing the sheets. The sheets were then fixed in 10% neutral buffered formalin (NBF; Confix Green, Australian Biostain Pty Ltd, Australia) for a minimum of 24 h. Following fixation of the tissue, immunohistochemical staining was performed to visualize the ECs that had adhered to the surface.
Experimental Design
Serum Concentration Experiment
M199-GM containing 0%, 10%, 20%, or 30% fetal calf serum (FCS; Commonwealth Serum Laboratories) was used to suspend the cultured ECs for seeding on the substrate. The density of ECs in the seeding suspension was constant at 1,000,000 cells/ml and the time allowed for the cells to adhere was constant at 1 h.
Seeding suspension containing 0% serum was placed on the luminal surface of a set of three sheets prepared from each of the five arteries in group a (4-week cold stored). Fifteen 4-week cold-stored arterial sheets were thus seeded using seeding suspension containing 0% serum. Seeding suspension containing 10%, 20%, or 30% serum was also placed on the luminal surface of different sets of three sheets prepared from each of the group a arteries. A total of 60 of 4-week cold-stored arterial sheets were thus seeded across the range of serum concentrations. The same procedure was performed using sheets prepared from the group b (8-week cold stored) and group c (12-week cold stored) arteries.
Time-of-Incubation Experiment
Incubation periods of 30, 60, or 90 min were allowed for the ECs in the seeding suspension to adhere to the substrate. The concentration of serum in the seeding suspension was constant at 0% and the density of cells in the seeding suspension was constant at 1,000,000 cells/ml.
Seeding suspension was allowed to incubate for 30 min on the luminal surface of a set of three sheets prepared from each of the five arteries in group a (4-week cold stored). Fifteen 4-week cold-stored arterial sheets thus underwent incubation for this period. Seeding suspension was also allowed to incubate for 60 or 90 min on the luminal surface of different sets of three sheets prepared from each of the group a arteries. A total of 45 of 4-week cold-stored arterial sheets were thus seeded across the range of incubation periods. The same procedure was performed using sheets prepared from the group b (8-week cold stored) and group c (12-week cold stored) arteries.
Seeding Density Experiment
ECs were suspended in culture medium at a density of 500,000, 1,000,000, or 2,000,000 cells/ml. The concentration of serum in the seeding suspension was constant at 0% and the time allowed for the cells to adhere was constant at 1 h.
Seeding suspension containing 500,000 cells/ml was allowed to incubate on the luminal surface of a set of three sheets prepared from each of the five arteries in group a (4-week cold stored). Fifteen 4-week cold-stored arterial sheets were thus seeded using this density of cells. Seeding suspension containing 1,000,000 or 2,000,000 cells/ml was also placed on the luminal surface of different sets of three sheets prepared from each of the group a arteries. A total of 45 of 4-week cold-stored arterial sheets were thus seeded across the range of seeding densities. The same procedure was performed using sheets prepared from the group b (8-week cold stored) and group c (12-week cold stored) arteries.
Histology, Digital Microscopy, and Assessment of Cell Adherence
Following fixation in NBF for at least 24 h, seeded sheets were rinsed in phosphate-buffered saline (PBS; Oxoid Ltd, England) and immunohistochemically labeled en face using the ovine EC-binding Datura stramonium lectin (DSL; Vector Laboratories, USA). The PBS-rinsed sheets were rinsed with distilled water (5 min), transferred to Tris buffer (5 min), and covered with biotinylated DSL (diluted 1:50 in Tris buffer) for 60 min. The biotinylated DSL-labeled specimens were subsequently rinsed with Tris buffer (5 min) and a detection system applied (Dako LSABR 2 kit, K0674). This involved covering the specimen with alkaline phosphatase-conjugated streptavidin (APCS) for 20 min. Following rinsing with Tris buffer (5 min), specimens were covered with the substrate chromogen 5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium (BCIP/NBT; Dako) for 15 min to allow for the formation of an insoluble blue reaction product at the site of lectin binding. The reaction was stopped by immersing specimens in distilled water. Adventitial tissue was carefully removed using forceps and specimens flat-mounted between two single cavity glass slides (Sail Brand, Shanghai Machinery Import and Export Co, China) in aqueous mounting medium (Gurr Aquamount improved, BDH, England).
On the luminal surface of each immunostained sheet, three areas of approximately 500,000 μm2—but could be greater or smaller—were randomly selected and outlined using image analysis software (Image-Pro Plus V4, Media Cybernetics, USA) and a Spot 2 digital camera (Diagnostic Instruments Inc., USA). The density of adherent, DSL-positive cells in each of the areas was determined by tagging and counting the cells using the software. Each adherent cell density count was expressed as number of cells/cm2 of luminal surface. Note that it was not possible to count the adherent ECs on the entire surface of each sheet due to the fact that it was not possible to visualize the entire surface in a single image.
Statistics
Statistical analysis of EC adherence data was performed using SAS version 8.2. Data were assessed for normality and found to be well approximated by a lognormal distribution, so were log-transformed prior to analysis. Analyses of the effect of period of cold storage, seeding density, serum concentration, and time of incubation on EC adherence were conducted using repeat measures analysis of variance. Pairwise multiple comparisons procedure was used to determine where the significance lies. A two-sided value of p = 0.05 was accepted as statistically significant. Results are presented as geometric means with a 95% confidence interval, and as differences in EC adherence (log-transformed data) with a 95% confidence interval for the difference.
Results
Serum Concentration Experiment Overall EC Adherence.
Serum Effect
The geometric mean values for overall EC adherence at serum levels of 0%, 10%, 20%, and 30% were, respectively: 1,825 cells/cm2 (95% CI: 1,334, 2497), 2,121 cells/cm2 (95% CI: 1,551, 2902), 1,857 cells/cm2 (95% CI: 1,357, 2540), and 1,955 cells/cm2 (95% CI: 1,429, 2,674). The effect of serum concentration on overall EC adherence was not significant (p = 0.91).
Due to the fact that the effect of serum concentration on overall EC adherence was not significant, further comparisons of EC adherence were not made.
Weeks Effect
The geometric mean values for overall EC adherence on 4-, 8-, and 12-week cold-stored arteries were, respectively: 2,854 cells/cm2 (95% CI: 2,176, 3,743), 1,609 cells/cm2 (95% CI: 1,227, 2,111), and 1,580 cells/cm2 (95% CI: 1,205, 2,073). The weeks effect on overall EC adherence was significant (p = 0.002). The geometric means and 95% CI for overall EC adherence, as well as the p-value for the interaction effect, are summarized in Table 1.
Overall Endothelial Cell Adherence Data for 4, 8, and 12 Weeks of Cold Storage

CI, confidence interval.
The weeks effect was consistent across all four serum concentrations, as demonstrated by the value p > 0.05 (p = 0.14). Thus, there is no evidence to suggest that the weeks effect differs for the different serum concentrations.
Pairwise Comparisons of EC Adherence
As the weeks effect on overall EC adherence was significant, multiple comparisons procedure was used to determine which pairs (4 vs. 8 weeks, 4 vs. 12 weeks, and 8 vs. 12 weeks) are significantly different.
Weeks Comparisons
EC adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 8-week cold-stored arteries (difference: 0.57; 95% CI: 0.19–0.96; p = 0.003) and 12-week cold-stored arteries (difference: 0.59; 95% CI: 0.21–0.97; p = 0.002). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries. The differences in EC adherence, the 95% CI for the differences, and the p-values for the comparisons are summarized in Table 2.
Pairwise Comparisons of Endothelial Cell (EC) Adherence on 4-, 8-, and 12–Week Cold-Stored Sheep Carotid Arteries in Serum Concentration Experiment

CI, confidence interval.
Log transformed data were used.
Weeks Comparisons at 0% Serum Concentration
EC adherence on 4-week cold-stored arteries was not significantly different from EC adherence on 8-week cold-stored arteries (difference: −0.19; 95% CI: −0.96–0.57; p = 0.61) or 12-week cold-stored arteries (difference: 0.06; 95% CI: −0.7–0.83; p = 0.87). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: 0.26; 95% CI: −0.51–1.02; p = 0.5).
Weeks Comparisons at 10% Serum Concentration
EC adherence on 4-week cold-stored arteries was not significantly different from EC adherence on 8-week cold-stored arteries (difference: 0.57; 95% CI: −0.2–1.33; p = 0.14) or 12-week cold-stored arteries (difference: 0.51; 95% CI: −0.26–1.27; p = 0.19). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: −0.06; 95% CI: −0.83–0.71; p = 0.88).
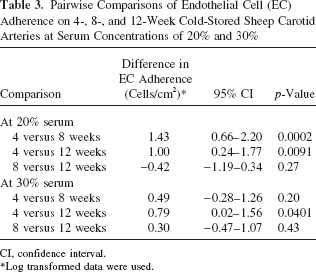
Weeks Comparisons at 20% Serum Concentration
EC adherence on 4-week cold-stored arteries was significantly greater than on 8-week cold-stored arteries (difference: 1.43; 95% CI: 0.66–2.20; p = 0.0002). EC adherence on 4-week cold-stored arteries was also significantly greater than on 12-week cold-stored arteries (difference: 1.00; 95% CI: 0.24–1.77; p = 0.0091). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: −0.42; 95% CI: −1.19–0.34; p = 0.27).
Weeks Comparisons at 30% Serum Concentration
EC adherence on 4-week cold-stored arteries was not significantly different from EC adherence on 8-week cold-stored arteries (difference: 0.49; 95% CI: −0.28–1.26; p = 0.20). EC adherence on 4-week cold-stored arteries was significantly greater than on 12-week cold-stored arteries (difference: 0.79; 95% CI: 0.02–1.56; p = 0.0401). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: 0.3; 95% CI: −0.47–1.07; p = 0.43). The differences in EC adherence, the 95% CI for the differences, and the p-values for the comparisons at 20% and 30% serum are summarized in Table 3.
Pairwise Comparisons of Endothelial Cell (EC) Adherence on 4-, 8-, and 12-Week Cold-Stored Sheep Carotid Arteries at Serum Concentrations of 20% and 30%
CI, confidence interval.
Log transformed data were used.
Time-of-Incubation Experiment Overall EC Adherence.
Time-of-Incubation Effect
The geometric mean values for overall EC adherence at times of incubation of 30, 60, and 90 min were, respectively: 1,456 cells/cm2 (95% CI: 1,070, 1,980), 1,232 cells/cm2 (95% CI: 905, 1676), and 1,344 cells/cm2 (95% CI: 988, 1828). The effect of time of incubation on overall EC adherence was not significant (p = 0.75).
Due to the fact that the effect of time of incubation on overall EC adherence was not significant, further comparisons of EC adherence were not made.
Weeks Effect
The geometric mean values for overall EC adherence on 4-, 8-, and 12-week cold-stored arteries were, respectively: 2,066 cells/cm2 (95% CI: 1,519, 2,811), 1,574 cells/cm2 (95% CI: 1,157, 2,141), and 741 cells/cm2 (95% CI: 545, 1,008). The weeks effect on overall EC adherence was significant (p < 0.0001). The geometric means and 95% CI for overall EC adherence, as well as the p-value for the interaction effect, are summarized in Table 4.
Overall Endothelial Cell Adherence Data for 4, 8, and 12 Weeks of Cold Storage

CI, confidence interval.
The weeks effect was consistent across all three times of incubation, as demonstrated by the value p > 0.05 (p = 0.19). Thus, there is no evidence to suggest that the weeks effect differs for the different times of incubation.
Pairwise Comparisons of EC Adherence
As the weeks effect on overall EC adherence was significant, multiple comparisons procedure was used to determine which pairs (4 vs. 8 weeks, 4 vs. 12 weeks, and 8 vs. 12 weeks) are significantly different.
Weeks Comparisons
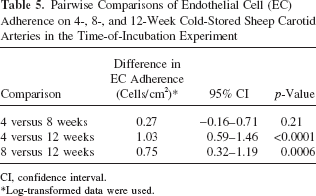
EC adherence on 4-week cold-stored arteries was not significantly different from EC adherence on 8-week cold-stored arteries (difference: 0.27; 95% CI: −0.16–0.71; p = 0.21). Endothelial cell adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 12-week cold-stored arteries (difference: 1.03; 95% CI: 0.59–1.46; p < 0.0001). EC adherence on 8-week cold-stored arteries was significantly greater than EC adherence on 12-week cold-stored arteries (difference: 0.75; 95% CI: 0.32–1.19; p = 0.0006). The differences in EC adherence, the 95% CI for the differences, and the p-values for the comparisons are summarized in Table 5.
Pairwise Comparisons of Endothelial Cell (EC) Adherence on 4-, 8-, and 12-Week Cold-Stored Sheep Carotid Arteries in the Time-of-Incubation Experiment
CI, confidence interval.
Log-transformed data were used.
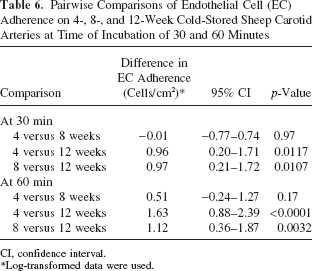
Weeks Comparisons at 30-min Incubation
EC adherence on 4-week cold-stored arteries was not significantly different from EC adherence on 8-week cold-stored arteries (difference: −0.01; 95% CI: −0.77–0.74; p = 0.97). Endothelial cell adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 12-week cold-stored arteries (difference: 0.96; 95% CI: 0.20–1.71; p = 0.0117). EC adherence on 8-week cold-stored arteries was significantly greater than EC adherence on 12-week cold-stored arteries (difference: 0.97; 95% CI: 0.21–1.72; p = 0.0107).
Weeks Comparisons at 60-min Incubation
EC adherence on 4-week cold-stored arteries was not significantly different from EC adherence on 8-week cold-stored arteries (difference: 0.51; 95% CI: −0.24–1.27; p = 0.17). EC adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 12-week cold-stored arteries (difference: 1.63; 95% CI: 0.88–2.39; p < 0.0001). EC adherence on 8-week cold-stored arteries was significantly greater than EC adherence on 12-week cold-stored arteries (difference: 1.12; 95% CI: 0.36–1.87; p = 0.0032).
Weeks Comparisons at 90-min Incubation
EC adherence on 4-week cold-stored arteries was not significantly different from EC adherence on 8-week cold-stored arteries (difference: 0.32; 95% CI: −0.44–1.07; p = 0.40) or 12-week cold-stored arteries (difference: 0.49; 95% CI: −0.26–1.24; p = 0.19). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: 0.17; 95% CI: −0.58–0.93; p = 0.65).
The differences in EC adherence, the 95% CI for the differences, and the p-values of the comparisons at 30- and 60-min incubation are summarized in Table 6.
Pairwise Comparisons of Endothelial Cell (EC) Adherence on 4-, 8-, and 12-Week Cold-Stored Sheep Carotid Arteries at Time of Incubation of 30 and 60 Minutes
CI, confidence interval.
Log-transformed data were used.
Seeding Density Experiment Overall EC Adherence.
Seeding Density Effect
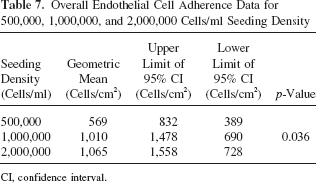
The geometric mean values for overall EC adherence at seeding densities of 500,000, 1,000,000, and 2,000,000 cells/ml were, respectively: 569 cells/cm2 (95% CI: 389, 832) 1,010 cells/cm2 (95% CI: 690, 1,478), and 1,065 cells/cm2 (95% CI: 728, 1,558). The effect of seeding density on overall EC adherence was significant (p = 0.036). The geometric means and 95% CI for overall EC adherence, as well as the p-value for the interaction effect, are summarized in Table 7.
Overall Endothelial Cell Adherence Data for 500,000, 1,000,000, and 2,000,000 Cells/ml Seeding Density
CI, confidence interval.
Weeks Effect
The geometric mean values for overall EC adherence on 4-, 8-, and 12-week cold-stored arteries were, respectively: 2,782 cells/cm2 (95% CI: 1,901, 4,070), 560 cells/cm2 (95% CI: 382, 819), and 393 cells/cm2 (95% CI: 268, 575). The weeks effect on overall EC adherence was significant (p < 0.0001). The geometric means and 95% CI for overall EC adherence, as well as the p-value for the interaction effect, are summarized in Table 8.
Overall Endothelial Cell Adherence Data for 4, 8, and 12 Weeks of Cold Storage

CI, confidence interval.
The weeks effect was consistent across all three seeding densities, as demonstrated by the value p > 0.05 (p = 0.67). Thus, there is no evidence to suggest that the weeks effect differs for the different seeding densities.
Pairwise Comparisons of EC Adherence
As the effect of seeding density on overall EC adherence was significant, multiple comparisons procedure was used to determine which pairs (500,000 vs. 1,000,000 cells/ml, 500,000 vs. 2,000,000 cells/ml, and 1,000,000 vs. 2,000,000 cells/ml) are significantly different. Moreover, as the weeks effect on overall EC adherence was significant, multiple comparisons procedure was used to determine which pairs (4 vs. 8 weeks, 4 vs. 12 weeks, and 8 vs. 12 weeks) are significantly different.
Seeding Density Comparisons
EC adherence following seeding at a density of 500,000 cells/ml was significantly lower than EC adherence following seeding at a density of 1,000,000 cells/ml (difference: −0.57; 95% CI: −1.11–0.04; p = 0.03) or 2,000,000 cells/ml (difference: −0.63; 95% CI: −1.17–0.09; p = 0.02). EC adherence following seeding at a density of 1,000,000 cells/ml was not significantly different from EC adherence following seeding at a density of 2,000,000 cells/ml (difference: −0.05; 95% CI: −0.59–0.49; p = 0.84). The differences in EC adherence, the 95% CI for the differences, and the p-values for the comparisons are summarized in Table 9.
Pairwise Comparisons of Endothelial Cell (EC) Adherence Following Seeding at a Density of 500,000, 1,000,000, and 2,000,000 Cells/ml in Seeding Density Experiment

CI, confidence interval.
Log-transformed data were used.
Seeding Density Comparisons for 4-Week Cold-Stored Arteries
EC adherence following seeding at a density of 500,000 cells/ml was not significantly different from EC adherence following seeding at a density of 1,000,000 cells/ml (difference: −0.77; 95% CI: −1.70–0.16; p = 0.10). EC adherence following seeding at a density of 500,000 cells/ml was significantly lower than EC adherence following seeding at a density of 2,000,000 cells/ml (difference: −1.14; 95% CI: −2.07–0.21; p = 0.0151). EC adherence following seeding at a density of 1,000,000 cells/ml was not significantly different from EC adherence following seeding at a density of 2,000,000 cells/ml (difference: −0.37; 95% CI: −1.3–0.57; p = 0.43). Figure 1 contains representative photomicrographs of the luminal surface following the use of each seeding density.

En face preparations showing the typical luminal surface of 4-week cold-stored sheep carotid artery sheets following seeding at a density of (A) 500,000 cells/ml, (B) 1,000,000 cells/ml, and (C) 2,000,000 cells/ml for 1 h. Note the stained, round-shaped endothelial cells. (Stain: biotinylated Datura stramonium lectin/alkaline phosphatase-conjugated streptavidin/5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium).
The differences in EC adherence, the 95% CI for the differences, and the p-values for each of the above comparisons are summarized in Table 10.
Pairwise Comparisons of Endothelial Cell (EC) Adherence Following Seeding at a Density of 500,000, 1,000,000, and 2,000,000 Cells/ml on 4-Week Cold-Stored Sheep Carotid Arteries

CI, confidence interval.
Log-transformed data were used.
Seeding Density Comparisons for 8-Week Cold-Stored Arteries
EC adherence following seeding at a density of 500,000 cells/ml was not significantly different from EC adherence following seeding at a density of 1,000,000 cells/ml (difference: −0.34; 95% CI: −1.28–0.59; p = 0.46) or 2,000,000 cells/ml (difference: −0.14; 95% CI: −1.07–0.79; p = 0.77). EC adherence following seeding at a density of 1,000,000 cells/ml was not significantly different from EC adherence following seeding at a density of 2,000,000 cells/ml (difference: 0.20; 95% CI: −0.73–1.14; p = 0.66).
Seeding Density Comparisons for 12-Week Cold-Stored Arteries
EC adherence following seeding at a density of 500,000 cells/ml was not significantly different from EC adherence following seeding at a density of 1,000,000 cells/ml (difference: −0.61; 95% CI: −1.54–0.32; p = 0.19) or 2,000,000 cells/ml (difference: −0.60; 95% CI: −1.54–0.33; p = 0.20). EC adherence following seeding at a density of 1,000,000 cells/ml was not significantly different from EC adherence following seeding at a density of 2,000,000 cells/ml (difference: 0.00; 95% CI: −0.93–0.94; p = 0.99).
Weeks Comparisons
EC adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 8-week cold-stored arteries (difference: 1.60; 95% CI: 1.07–2.14; p < 0.0001) and 12-week cold-stored arteries (difference: 1.96; 95% CI: 1.42–2.50; p < 0.0001). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: 0.35; 95% CI: −0.18–0.89; p = 0.1893).
The differences in EC adherence, the 95% CI for the differences, and the p-values for the comparisons are summarized in Table 11.
Pairwise Comparisons of Endothelial Cell (EC) Adherence on 4-, 8-, and 12-Week Cold-Stored Sheep Carotid Arteries in the Seeding Density Experiment

CI, confidence interval.
Log-transformed data were used.
Weeks Comparisons at 500,000 Cells/ml Seeding Density
EC adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 8-week cold-stored arteries (difference: 1.13; 95% CI: 0.20–2.06; p = 0.0160) and 12-week cold-stored arteries (difference: 1.73; 95% CI: 0.79–2.66; p = 0.0002). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: 0.60; 95% CI: −0.33–1.53; p = 0.20).
Weeks Comparisons at 1,000,000 Cells/ml Seeding Density
Endothelial cell adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 8-week cold-stored arteries (difference: 1.56; 95% CI: 0.62–2.49; p = 0.0009) and 12-week cold-stored arteries (difference: 1.89; 95% CI: 0.92–2.82; p = 0.0001). EC adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: 0.33; 95% CI: −0.60–1.26; p = 0.48).
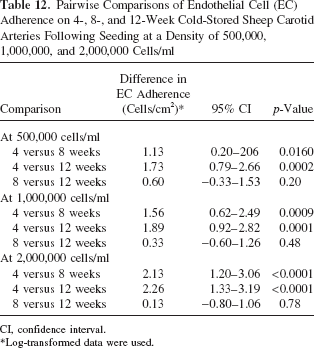
Weeks Comparisons at 2,000,000 Cells/ml Seeding Density
EC adherence on 4-week cold-stored arteries was significantly greater than EC adherence on 8-week cold-stored arteries (difference: 2.13; 95% CI: 1.20–3.06; p < 0.0001) and 12-week cold-stored arteries (difference: 2.26; 95% CI: 1.33–3.19; p < 0.0001). Endothelial cell adherence on 8-week cold-stored arteries was not significantly different from EC adherence on 12-week cold-stored arteries (difference: 0.13; 95% CI: −0.80–1.06; p = 0.78).
The differences in EC adherence, the 95% CI for the differences, and the p-values for the comparisons are summarized in Table 12.
Pairwise Comparisons of Endothelial Cell (EC) Adherence on 4-, 8-, and 12-Week Cold-Stored Sheep Carotid Arteries Following Seeding at a Density of 500,000, 1,000,000, and 2,000,000 Cells/ml
CI, confidence interval.
Log-transformed data were used.
Discussion
One of the aims of this study was to determine which of the cold-stored arteries is best suited to EC adherence (i.e., demonstrates the greatest EC adherence) following EC seeding procedure. The results of this study indicate that, within the time frames examined, 4-week cold-stored arteries are clearly the best option.
The evidence in support of 4 weeks being the best option of the three cold storage periods examined took several forms. One was the finding in each of the three experiments—the serum concentration experiment, the time-of-incubation experiment, and the seeding density experiment—that overall EC adherence was maximal on the 4-week cold-stored arteries compared with the 8–and 12-week cold-stored arteries. Moreover, the weeks effect on overall EC adherence was statistically significant in each experiment, the value for the weeks effect being as low as p < 0.0001 as overall EC adherence declined with increasing duration of cold storage. Other evidence was obtained from pairwise comparisons made in each of the three experiments. Endothelial cell adherence on 4-week cold-stored arteries was almost always significantly greater than on 8- and 12-week cold-stored arteries. Many of the differences in EC adherence were highly significant, the values being as low as p < 0.0001.
Additional evidence indicated that 4-week cold-stored arteries were the best option. In the seeding density experiment, 4-week cold-stored arteries demonstrated significantly greater EC adherence at a seeding density of 2,000,000 cells/ml than at a seeding density of 500,000 cells/ml. No significant difference in EC adherence was found following the application of identical seeding densities to 8-week cold-stored arteries. Nor was it found when these seeding densities were applied to 12-week cold-stored arteries. Thus, the advantage of increasing the seeding density was restricted to the 4-week cold-stored arteries.
What may be the explanation for the observation that the 4-week cold-stored artery is superior to the 8- and 12-week cold-stored artery in terms of EC adherence following EC seeding? It has previously been found that the cold storage process has important effects on the morphology of the sheep carotid artery. During this storage process, the native EC monolayer is lost from the luminal surface. By 4 weeks, there is a total loss of EC from the luminal surface and consequent exposure of the subendothelial basement membrane. The subendothelial basement membrane contains adhesive proteins such as fibronectin, laminin, and collagen IV, which contain sites or sequences that bind to adhesion molecules on the EC membrane (11,15,33). Between 4 and 16 weeks of cold storage, there is a progressive loss or breakdown of these adhesive proteins, and a presumed associated loss or attenuation of their binding sequences. Some of these morphological changes have also been observed in the rabbit femoral artery as a result of a similar cold storage process (6,8).
It is inferred that, after 4 weeks of cold storage, the exposed subendothelial basement membrane of the sheep carotid artery contains a greater number of binding sites for EC adherence than is the case in 8-week and, especially, 12-week cold-stored arteries.
This inference does not preclude the use of exogenous adhesive proteins, such as fibronectin, to enhance the adhesiveness of cold-stored arteries. It may be the case that arteries that have been cold-stored for 8 or 12 weeks can be coated with fibronectin and/or other adhesive proteins to increase the number of binding sites on their luminal surfaces and to enhance their ability to attach ECs. Little, if anything, is known about the feasibility of using exogenous adhesive proteins to enhance EC adherence on cold-stored arteries. However, there are numerous studies demonstrating that the application of fibronectin, collagen IV, and other adhesive polymer sequences to various synthetic substrates markedly enhances EC adherence on the substrate (12,17,22,30,32).
Another aim of this study was to determine the optimal conditions for seeding of cold-stored arteries. Since it has been determined that the 4-week cold-stored artery is the superior substrate, the following discussion of optimal seeding conditions will concentrate on the results obtained using this substrate.
Serum contains growth factors (such as fibroblast growth factor) and immunoglobulins that are known to play a role in the induction of adhesion molecules on the membrane of ECs, including those that are classified as integrins (7,14,26). Integrins are known to play an important role in the adhesion of ECs to a substrate.
In the current study, a serum concentration of 20% produced maximal EC adherence on the 4-week cold-stored arteries rather than on the 8- or 12-week cold-stored arteries. Additionally, it was at 20% serum, and not at 0%, 10%, or 30% serum, that EC adherence on 4-week cold-stored arteries was significantly greater than EC adherence on both 8- and 12-week cold-stored arteries. The apparent advantageous effect of 20% serum on EC adherence is thus restricted to the 4-week cold-stored artery. Why would the effect of 20% serum be confined to 4-week cold-stored arteries? There are two considerations here, one being the role of 20% serum and the other being the presence of binding sites for ECs in the substrate. In relation to the role of 20% serum, it is worth noting that the ECs had undergone conventional cell culture in medium containing 20% serum prior to their use in the seeding procedure. Thus, the ECs were effectively primed for expression of adhesion molecules at this serum concentration before they were placed on the substrate.
Thus, one of the main findings from the serum concentration experiment is that 20% serum in the seeding suspension appears to be optimal for EC adherence, but only on the 4-week cold-stored arteries. This concentration of serum is comparable with that which was found to produce maximal EC adherence following EC seeding of mechanically denuded rabbit femoral arteries (5).
It should be noted, however, that EC adherence on the cold-stored arteries in the current study was possible in the absence of serum. This has implications for the use of serum-free media (20) in clinical procedures. The reason as to why there is an interest in using serum-free media in clinical seeding procedures is based on the concern that in vitro use of serum may predispose the seeded graft to the development of neointimal hyperplasia in vivo, leading to graft occlusion (10,13).
The time required for seeding incubation reflects several events that must occur before ECs can attach firmly to the cold-stored arterial substrate. One is the act of EC settling, under the force of gravity, on the substrate. There is also the process in which adhesion molecules on the EC membrane locate a binding sequence or sequences in the substrate. Additionally, there is the process in which adhesion complexes form and focalize between the cell and substrate, a process that requires assembly of intracellular stress fibers (3,4,8,16).
The results from the current study indicate that the time of incubation, whether 30, 60, or 90 min, does not have a significant effect on overall EC adherence. One of the conclusions that may be drawn from this is that the optimal time of incubation for EC adherence on the 4-week cold-stored artery can be measured in terms of minutes (i.e., 30–90 min) rather than in terms of several hours. This finding is comparable with the observation that seeded ECs require only 30–60 min to adhere on the mechanically denuded rabbit femoral artery (5). The implication is that seeding of cold-stored arteries is possible within a brief time frame that is compatible with the clinical setting where time is of the essence.
It is speculated that seeding incubation times of greater than 90 min, had they been examined, may have resulted in reduced EC adherence rather than increased EC adherence. This may have occurred partly as a result of the deleterious effect on the ECs in the seeding suspension of the build-up of metabolic wastes produced by the ECs over time. This phenomenon is frequently observed in cell culture flasks in which confluent cultures of ECs are maintained in the same culture medium for longer than is desirable. The medium undergoes a change in pH and there is an accompanying increase in the number of dead, detached cells.
In the seeding density experiment, pairwise comparisons of EC adherence at the different seeding densities revealed that a seeding density of 1,000,000 or 2,000,000 cells/ml produced a significantly greater adherence than a seeding density of 500,000 cells/ml. Additionally, pairwise comparisons of EC adherence on the 4-week cold-stored arteries revealed that a seeding density of 2,000,000 cells/ml produced a significantly greater adherence than a seeding density of 500,000 cells/ml. These observations are broadly in line with observations in published seeding studies in which ECs were placed on a variety of artificial and natural substrates (5,18,21,29). Thus, the greater the number of ECs in the seeding suspension, the greater the number of ECs that should adhere on the substrate. It should be stressed that a positive and significant effect of seeding density was observed only on the 4-week cold-stored arteries. There was no significant effect of seeding density at either 8 or 12 weeks of cold storage.
The importance of seeding density in relation to EC adherence on the cold-stored artery is demonstrated by another observation. In the seeding density experiment, seeding density was found to have a significant effect on overall EC adherence, the geometric mean values for overall EC adherence being 569, 1,010, and 1,065 cells/cm2 at seeding densities of 500,000, 1,000,000, and 2,000,000 cells/ml, respectively. Neither serum concentration nor time-of-incubation demonstrated a significant effect on overall EC adherence in the respective experiments.
The advantage of a high seeding density in tissue engineering has also been observed in in vivo experiments that used different types of cells. For example, a very high density of 80,000,000 cultured autologous skin fibroblasts/ml injected into the ears of rabbits was found to be associated with enhanced collagen production (36). Sheets consisting of a high density of cultured mesenchymal cells that were wrapped around bone allografts prior to their implantation in mice and rabbits were found to increase the rate of bone healing (37).
Although it is conceivable that the use of greater EC seeding densities (e.g., 3 or 4 million cells/ml) may produce a more enhanced EC adherence than was found with 2,000,000 cells/ml, it is also possible that such higher seeding densities may not greatly enhance, and may even reduce, EC adherence. The latter might occur if the presence of a very large number of ECs in the seeding suspension increases the likelihood of formation of EC aggregates. Aggregation occurs when cells clump together and form bonds with each other through intercellular adhesion molecules (1,2). Such an event is likely to reduce the exposure of the membrane of individual ECs, and hence their membrane-bound adhesion molecules, to the substrate, thus reducing the likelihood of adherence. Moreover, a very large number of ECs may lead to increased production of cellular metabolic waste products to levels that adversely affect the viability of the ECs, and hence their ability to attach or remain attached to the substrate.
It is noteworthy that EC adherence on the 4-week cold-stored arteries represented only a proportion of the total number of ECs that were applied to this substrate in the form of the seeding suspension. Many ECs presumably did not attach at all or could not resist the shearing forces of the rinsing process that followed the seeding incubation period. The reasons for the failure of many cells to attach or remain attached to the substrate may be cell related (e.g., a lack of exposure of EC adhesion molecules to the substrate, or a lack of adhesion molecules on the EC membrane) or substrate-related (e.g., a lack of binding sites in the substrate, or a weak binding interaction between EC and substrate).
A number of experiments in our laboratory investigated the seeding of segments of complete (as opposed to sheets) 4- to 8-week cold-stored sheep carotid arteries with cultured sheep carotid artery ECs. It was found that, as in the sheets, the ECs attached and began to spread on the luminal surface during the seeding incubation period. Additionally, it was found that, following postseeding culture for up to 3 days, the ECs spread further and replicated on the luminal surface, forming islands of confluence consisting of flattened cells in a monolayer arrangement.
In addition to the sheep model of cold storage described in this article, our laboratory investigated a rabbit model. In this model, complete rabbit femoral arteries that had been cold stored for a minimum of 4 weeks were seeded with cultured rabbit jugular vein ECs and then exposed to an in vitro or in vivo regime consisting of a pulsatile flow of culture media or blood. Following exposure to flow for up to 3 days, the arteries were found to retain the seeded ECs on their luminal surfaces. This indicates that the ECs had attached to the luminal surface with a strength that was sufficient to resist the shearing forces of physiological blood flow.
A small number of trials in our laboratory examined the seeding of cultured human saphenous vein ECs on sheets prepared from 4-week cold-stored sheep carotid arteries. It was found that the ECs were able to attach and spread on the luminal surface. If human ECs can grow to confluence on the luminal surface of the xenograft, it raises the possibility of using autologous EC-seeded vascular xenografts in the clinical situation.
Summary
Using an immunohistochemical technique, it was possible to identify and count cultured ECs seeded onto sheets obtained from sheep carotid arteries that had been stored at 4°C for 4, 8, and 12 weeks. The main findings of this study are as follows. Out of the three cold storage periods examined, the best option for adherence of EC is the 4-week cold-stored artery. Out of the three seeding densities examined (500,000, 1,000,000, and 2,000,000 cells/ml), the optimal seeding density for adherence of seeded ECs to 4-week cold-stored arteries is 2,000,000 cells/ml. On the 4-week cold-stored artery, adherence of seeded EC occurs within 30 min and does not change significantly after 60 or 90 min. Out of the four serum concentrations examined (0%, 10%, 20%, and 30%), the optimal serum concentration for adherence of seeded ECs to 4-week cold-stored arteries is 20%.
Footnotes
Acknowledgments
Ian Birchall, Florey Neuroscience Institutes, University of Melbourne, Victoria, Australia contributed to advice regarding histological/immunohistochemical staining technique and digital microscopy/assessment. Yos Morsi, Swinburne University, Victoria, Australia helped to provide the facilities for the experiments. Stephen Cordner, Department of Forensic Medicine, Monash University, Victoria, Australia and Lynn Ireland, Head of Donor Tissue Bank, Victoria, Australia funded A.S.'s Ph.D. scholarship. Funding for this research was provided by Department of Forensic Medicine, Monash University, Victoria, Australia. The authors declare no conflict of interest.
